Abstract
The chiral poly(amide-imide) (PAI) was synthesized by the direct polycondensation reaction of imide-dicarboxylic acid, N-trimellitylimido-
Introduction
Ijima first discovered carbon nanotubes (CNTs) in 1991. 1 Since then, attention to this class of one-dimensional nanomaterials has been growing in various disciplines because of their unique electrical, mechanical, and thermal properties. 2,3 It is well known that CNTs have found in a wide range of applications such as electronic devices, 4 biosensors, 5 field emission displays, 6 hydrogen storage, 7 and composites. 8 Furthermore, CNTs are exciting ultra-high-strength reinforcements in polymer matrix composites. 9 –12 CNTs with higher flexibility and lower density can enhance the mechanical, thermal, and conductive properties of the polymers as compared to metal nanofillers. Nanotube–polymer composites have applications in ultrafast all-optical switches, 13 electromagnetic interference shielding, 14 photovoltaic devices, 15 gas sensors, 16 and biocatalytic films. 17
Aromatic polyimides are well known as a class of high-performance polymers because of their excellent thermal, mechanical, chemical, and physical properties. 18,19 However, their poor processability has been major problems as a result of high melting or glass transition temperatures and limited solubility in most organic solvents. To overcome such a difficulty, different copolyimides have been developed. Therefore, poly(amide-imide)s (PAI)s have been developed as one of the most successful classes of high-performance polymers that offer a concession between the superior mechanical properties associated with amide units and the high thermal stability gritty by imide structures. 20 –22
The design and development of synthetic optically active polymers have received considerable attention due to their sophisticated functions related to their structures. Synthetic chiral polymers are applied in various uses, for instance, the separation of racemic compounds, 23 electrodes for enantioselective recognition for performing bioelectro synthesis, 24 microwave absorbents, 25 liquid crystals, 26 nonlinear optics, 27 and the catalysis of asymmetric syntheses. 28 Amino acid–based chiral polymers are likely to possess crystallinity with the ability to form higher ordered structures that show enhanced solubility characteristics. 29
In this study, we report the fabrication and characterization of composite films by the dispersion of acid-functionalized multiwalled carbon nanotubes (MWCNTs) in a chiral PAI containing amino acid,
Experimental
Materials
All chemicals were purchased from Fluka Chemical Co. (Buchs, Switzerland), Aldrich Chemical Co. (Milwaukee, WI, USA), Riedel-deHaen AG (Seelze, Germany), and Merck Chemical Co (Darmstadt, Germany). 1,2,4-Benzene tricarboxylic anhydride,
Techniques
Proton nuclear magnetic resonance (1H NMR) spectra were recorded on Bruker Avance 400 MHz spectrometer (Rheinstetten, Germany) in dimethyl sulfoxide-d 6 (DMSO-d 6). Inherent viscosity was measured by a standard procedure using a Cannon-Fenske routine viscometer (Mainz, Germany) at the concentration of 0.5 g/dL at 25°C. FTIR spectra were recorded with a Jasco-680 spectrometer (Tokyo, Japan) in the range of 400–4000 cm−1. Vibration bands were reported as wave number (cm−1). FTIR spectra of all samples were collected by making their pellets in KBr as a medium. The XRD pattern of prepared materials was recorded in the reflection mode using a Bruker, D8 Advance diffractometer (Rheinstetten, Germany). Nickel-filtered CuKα radiation (radiation wavelength, λ = 0.154 nm) was produced at an operating voltage of 45 kV and a current of 100 mA. The redox behavior was investigated with cyclic voltammetry (CV) on a potentiostat (EG&G Co., PARSTAT 2273, Tennessee, USA) instrument. It was conducted for the dipped polymer film on the working electrode in dry acetonitrile containing tetrabutylammonium hexafluorophosphate (0.1 M) as an electrolyte. The morphology and dispersity analysis were performed on TEM analyzer on Philips CM 120 (Eindhoven, The Netherlands) operating at 100 kV. The reaction was carried out on a Misonix ultrasonic liquid processor (Eindhoven, The Netherlands), XL-2000 Series (Eindhoven, The Netherlands). Ultrasound was a wave of frequency 2.25 × 104 Hz and power 100 W. TGA is performed with a STA503 (Eindhoven, The Netherlands) win TA at a heating rate of 10°C/min from 25°C to 800°C under a nitrogen atmosphere.
Monomer synthesis
N-Trimellitylimido-
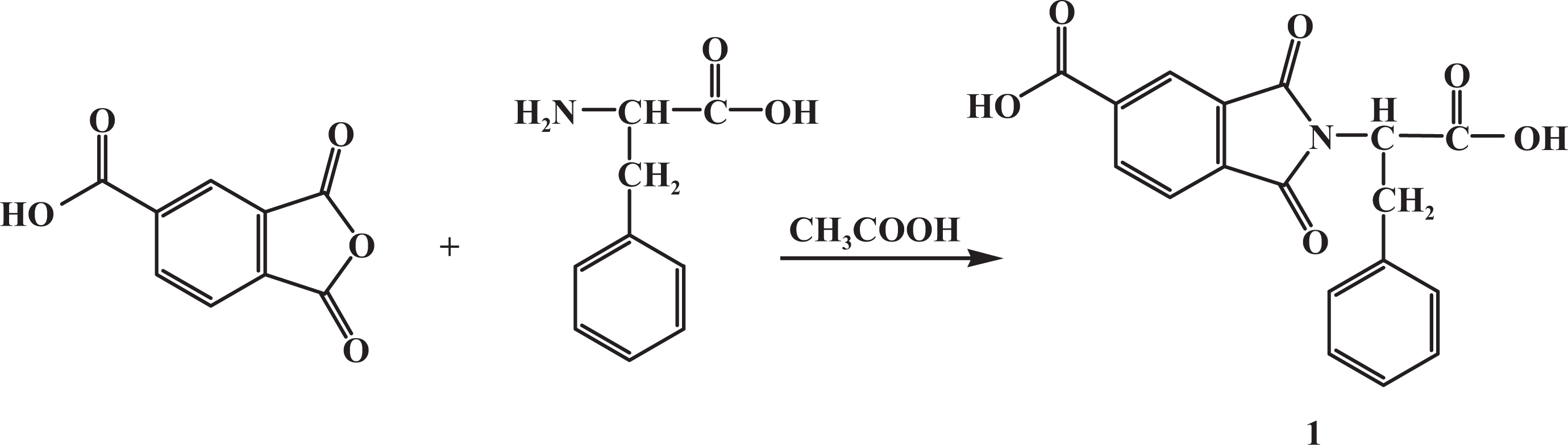
Synthesis of imide-dicarboxylic acid monomer 1.
Polymer synthesis
A mixture of 0.10 g (2.95 × 10−4 mol) of imide-dicarboxylic acid 1, 0.0466 g (2.95 × 10−4 mol) of diamine, 1,5-naphthalenediamine (2), 0.06 g of calcium chloride, 0.31 mL (1.18 × 10−3 mol) of triphenyl phosphite (TPP), 0.15 mL of pyridine, and 0.4 mL of NMP was refluxed for 3 h. After cooling, the reaction mixture was poured into 30 mL of methanol with constant stirring, and the precipitate was washed thoroughly with methanol and hot water, collected on a filter, and dried under vacuum to give 0.114 g (78%) of PAI.
PAI: FTIR (KBr, cm−1): υ = 3276 (m), 3027 (m), 2927 (m), 1776 (m), 1718 (s), 1675 (s), 1617 (s), 1600 (s), 1485 (m), 1380 (m), 1327 (m). 1H NMR (DMSO-d 6, ppm): δ = 10.87 (d, 1H, N–H), 10.29 (d, 1H, N–H), 8.56–7.10 (m, Ar), 5.40 (m, 1H, CH), 3.64 (m, 2H).
Functionalization of MWCNT
The chemical treatment on MWCNTs was done as suggested by Esumi et al. 31 The required sample of MWCNTs was suspended in the mixture of concentrated nitric acid (65%) and sulfuric acid (98%) by the volume ratio of 1:3 and refluxed at 140°C for 30 min. After washing the MWCNTs with deionized water until the supernatant attained a pH around 7, the samples were dried at 100°C. Thus, the chemically treated sample was obtained.
Preparation of the PAI/MWCNT-COOH composite films
PAI/MWCNT-COOH composite films were prepared with different weight percentages of MWCNT-COOH (5, 10, and 15 wt%) as follows:
First, PAI was dissolved and MWCNT-COOH was separately dispersed in DMAc with stirring at 40°C for 1 day. Then, two stock solutions were mixed to achieve the desired weight percentages of MWCNT-COOH (5, 10, and 15 wt%). The PAI/MWCNT-COOH mixture was stirred at 40°C for 1 day and then ultrasonicated in a water bath for 2 h. The PAI/MWCNT-COOH mixture was spread on a glass plate, and the solvent was removed at 60°C for 1 day; then, the semidried film was further dried in vacuum at 160°C for 8 h in order to remove the residue solvent and a solid film was formed.
PAI/MWCNT-COOH 10 wt%: FTIR (KBr, cm−1): υ = 3434 (m), 2923 (m), 1778 (m), 1716 (m), 1639 (m), 1378 (m).
Results and discussion
Polymer synthesis
The PAI was prepared via the direct polycondensation of optically active imide-dicarboxylic acid 1 with aromatic diamine 2 through Yamazaki phosphorylation reaction, using TPP/Py/NMP/CaCl2 as a condensing agent (Figure 2). The polymerization in NMP proceeded homogeneously, indicating that the polymer showed good solubility in the polymerization media. The PAI was obtained almost in good yield (78%) and had inherent viscosity value 0.41 dL g−1. The structure of PAI was confirmed by FTIR and 1H NMR spectroscopic analyses. FTIR spectrum of PAI (Figure 3(a)) revealed the characteristic absorptions of imide groups occurred at 1776, 1718, and 1380 cm−1, and those of the amide group occurred around 3276 and 1675 cm−1. The assignments of the 1H NMR spectrum of PAI agree well with the proposed polymer structure (Figure 4).
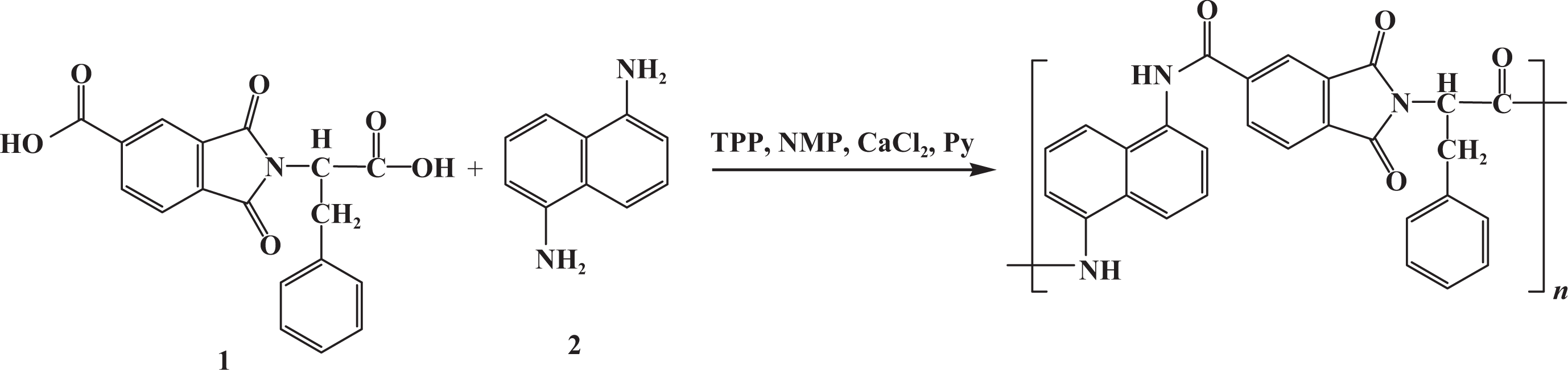
Preparation of PAI. PAI: poly(amide-imide).
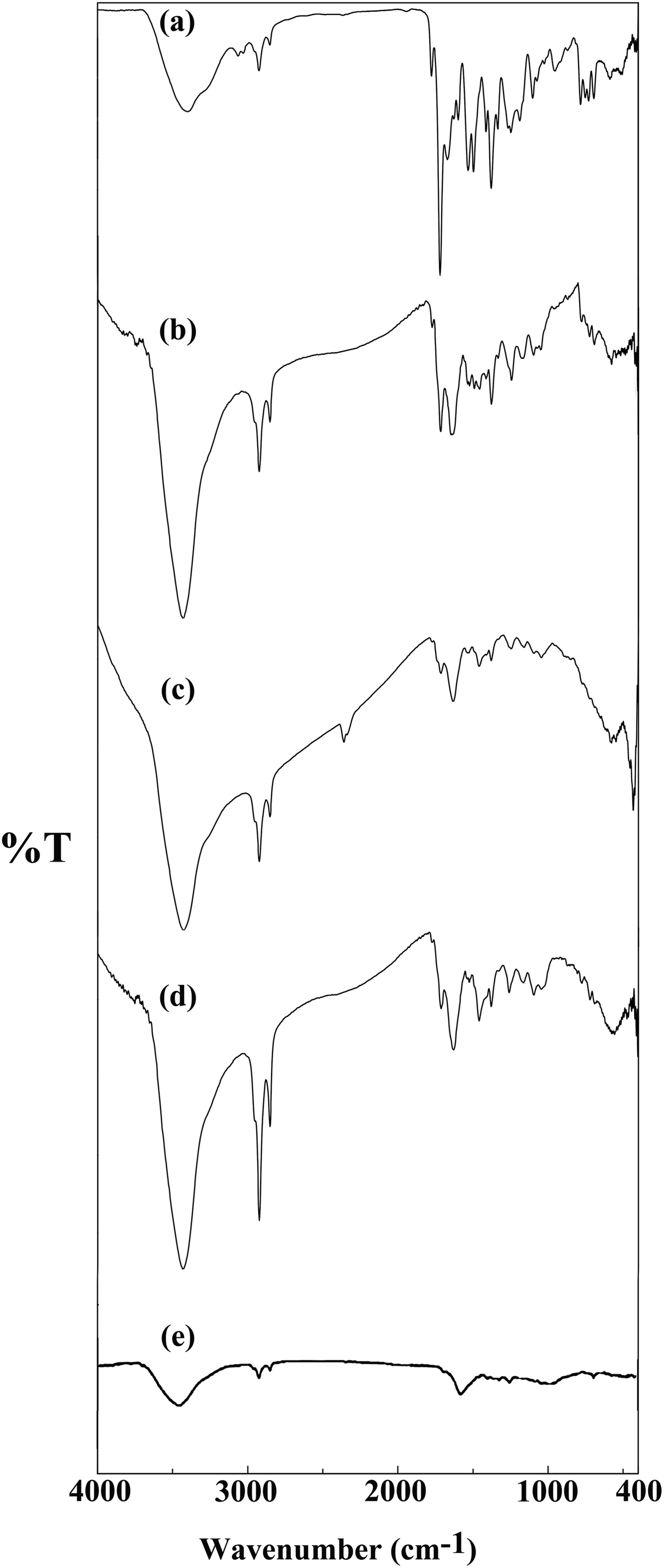
FTIR spectra of PAI and the PAI/MWCNT composites: (a) PAI, (b) MWCNT/PAI 5 wt%, (c) MWCNT/PAI 10 wt%, (d) MWCNT/PAI 15 wt%, (e) acid-functionalized MWCNT. PAI: poly(amide-imide); MWCNT: multiwalled carbon nanotube.
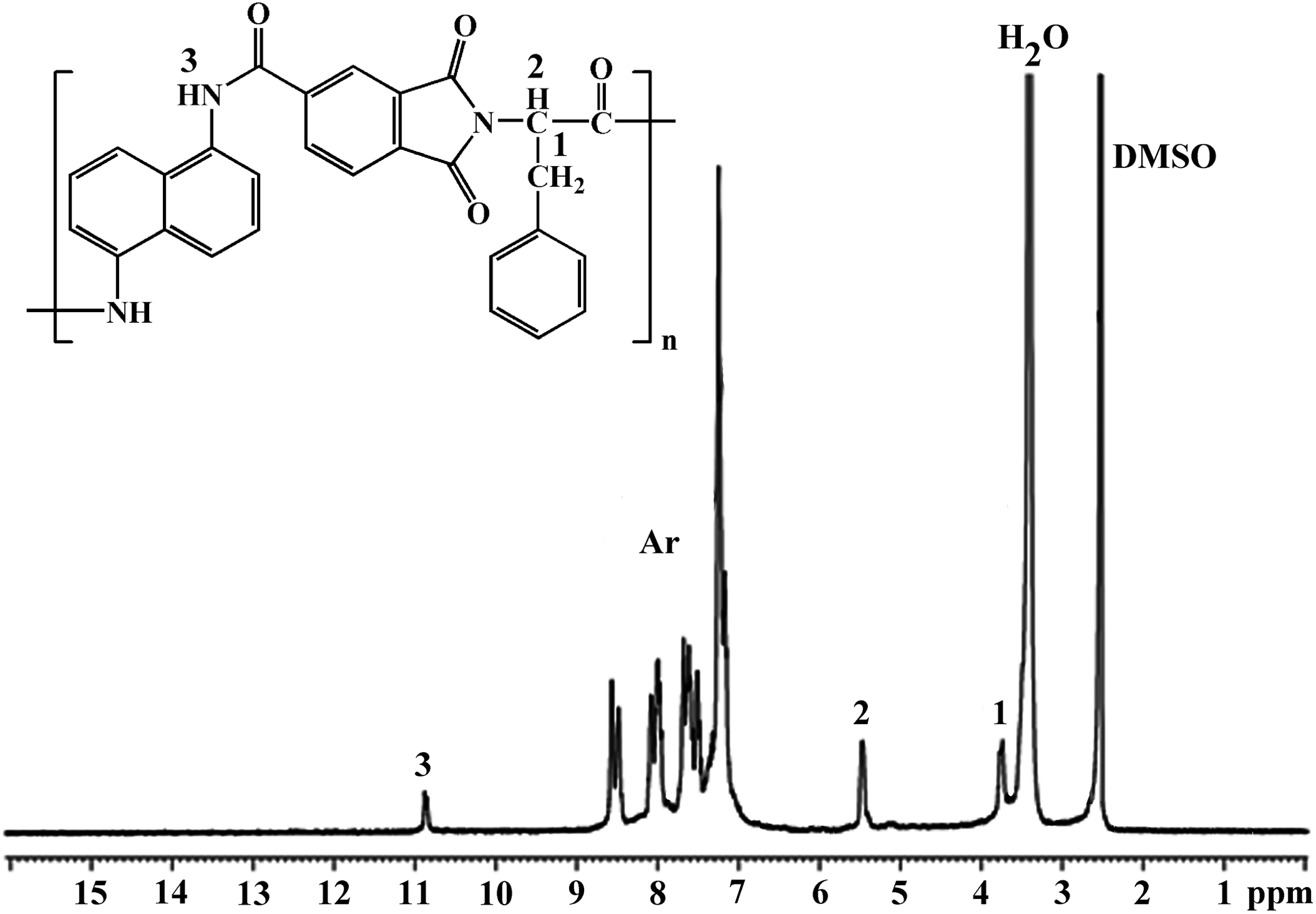
1H NMR spectrum of PAI in DMSO-d 6 solution.
Preparation of PAI/MWCNT-COOH composite films
The effective utilization of nanotubes in composites depends on the ability to disperse CNTs homogeneously throughout the matrix without destroying their integrity. Also, prior to the mixing of CNTs with a matrix, it is required to perform a chemical treatment on CNTs, since a better interfacial bonding between CNT and polymer and also the enhancement of load transfer are believed to occur. So, for better dispersion of MWCNTs into the polymer matrix, carboxylated MWCNTs were applied in this work. PAI/MWCNT-COOH composite films were prepared by dispersion of the different amounts of acid-functionalized MWCNT (5, 10, and 15 wt%) solutions of PAI in DMAc via a vigorous stirring for 1 day, using a homogenizer, followed by utrasonication for 2 h.
Characterization of the composites
The acid-functionalized MWCNTs were characterized by FTIR. The FTIR spectrum of carboxylated MWCNTs (Figure 3(e)) shows a broad absorption band centered at 3424 cm−1, which is attributed to the O–H stretching bands of carboxylic acid moieties from the surface of MWCNTs. The peak around 2923 cm−1 is ascribed to aliphatic sp3 C–H of defects. 32 The presence of MWCNTs in the polymer matrix showed very little changes in the FTIR spectrum, probably due to the low MWCNT composition and the weak vibration signals of MWCNTs. The FTIR spectra of PAI/MWCNT-COOH composites with different contents of MWCNT-COOH are shown in Figure 3(b) to (d). From these data, it is clear that with increasing the amount of MWCNT-COOH, the intensity of absorption related to aliphatic sp3 C–H bonds was enhanced.
XRD patterns of PAI (a), PAI/MWCNT-COOH composite 10 wt% (b), and MWCNT-COOH (c) are given in Figure 5. The diffraction pattern for the pure PAI shown in Figure 5(a) demonstrates that the PAI is completely amorphous. For the MWCNTs, two peaks appear at 2θ =26° and 43° which correspond to the interlayer spacing d (002) and d (100) reflection of the CNTs (Figure 5(b)). 33
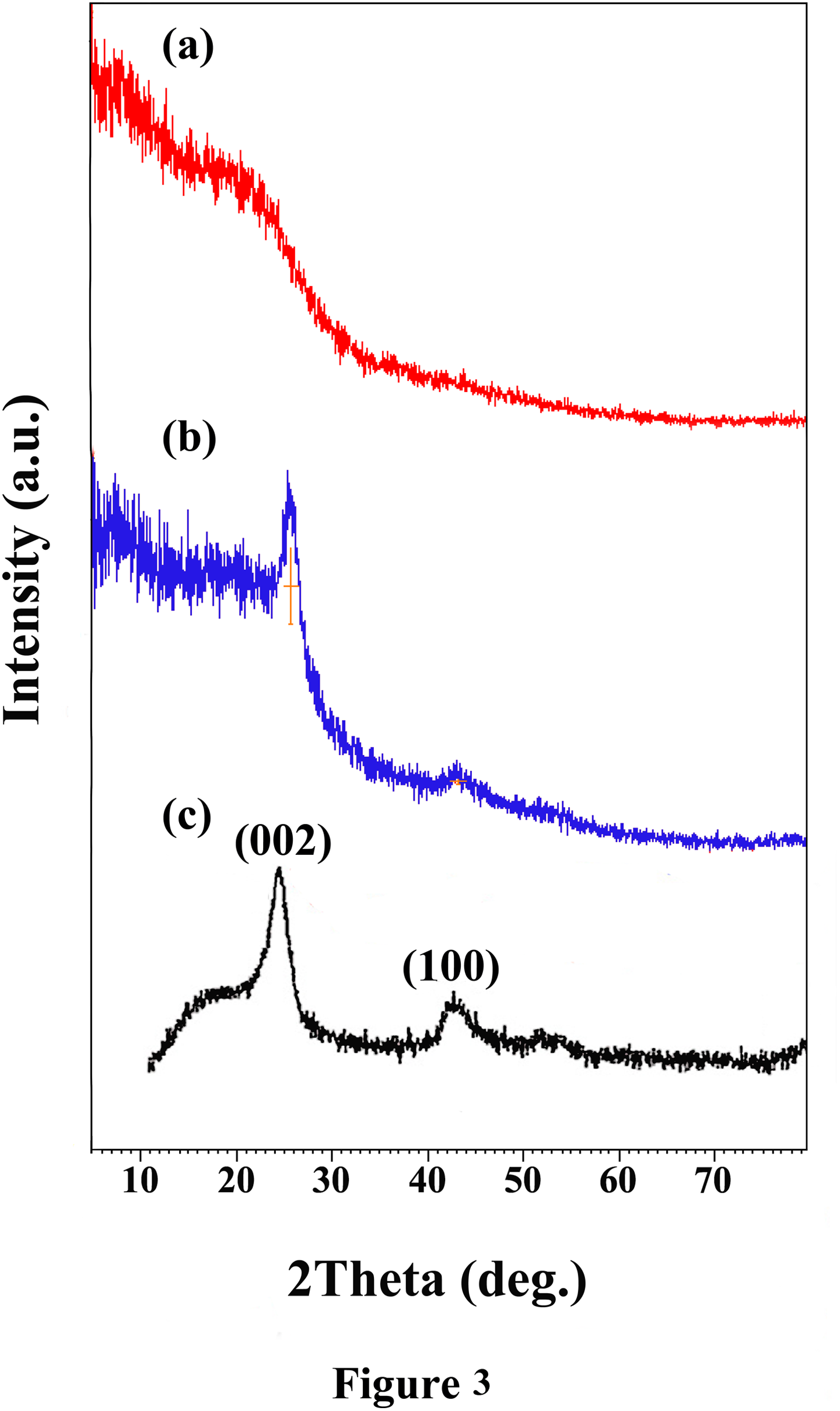
XRD patterns for (a) neat PAI, (b) MWCNT/PAI composite containing 10 wt% MWCNT, and (c) MWCNT-COOH.
TEM analysis was performed for PAI/MWCNT-COOH composite 10 wt% to identify the distribution of MWCNT within polymer matrix. Figure 6 shows the TEM image of the composite 10 wt%. As can be seen, the MWCNTs were well dispersed in the polymer matrix.

TEM image of PAI/MWCNT composite containing 10 wt% MWCNT. PAI: poly(amide-imide); MWCNT: multiwalled carbon nanotube.
The ultraviolet–visible (UV–vis) spectra of the PAI and composites were recorded in DMF. It is apparent that the wavelength of maximum absorption is related to the π → π* transition resulting from the conjugation between the aromatic rings and nitrogen atoms in the aforementioned compounds. All of these compounds show almost similar UV–vis spectra pattern. The PAI showed maximum absorption at 304 nm, while the composite films exhibited maximum absorption around 298 nm in DMF solution. In UV–vis spectra of the composite films with different MWCNT contents with the increasing CNT, the absorption band moves toward shorter wavelength.
Charge transport in organic materials is believed to be governed by the hopping process involving redox reaction of charge transport molecules. CV is a preliminary characterization method to determine the redox properties of polymeric materials. Figure 7 shows the representative cyclic voltammogram of PAI/MWCNT-COOH composite 15 wt% in acetonitrile with 0.1 M Bu4NPF6. One pair of redox waves was observed in these composites. As shown in this figure, composite showed an oxidation wave of which a peak top is at 1.7 V (versus Ag/AgCl).
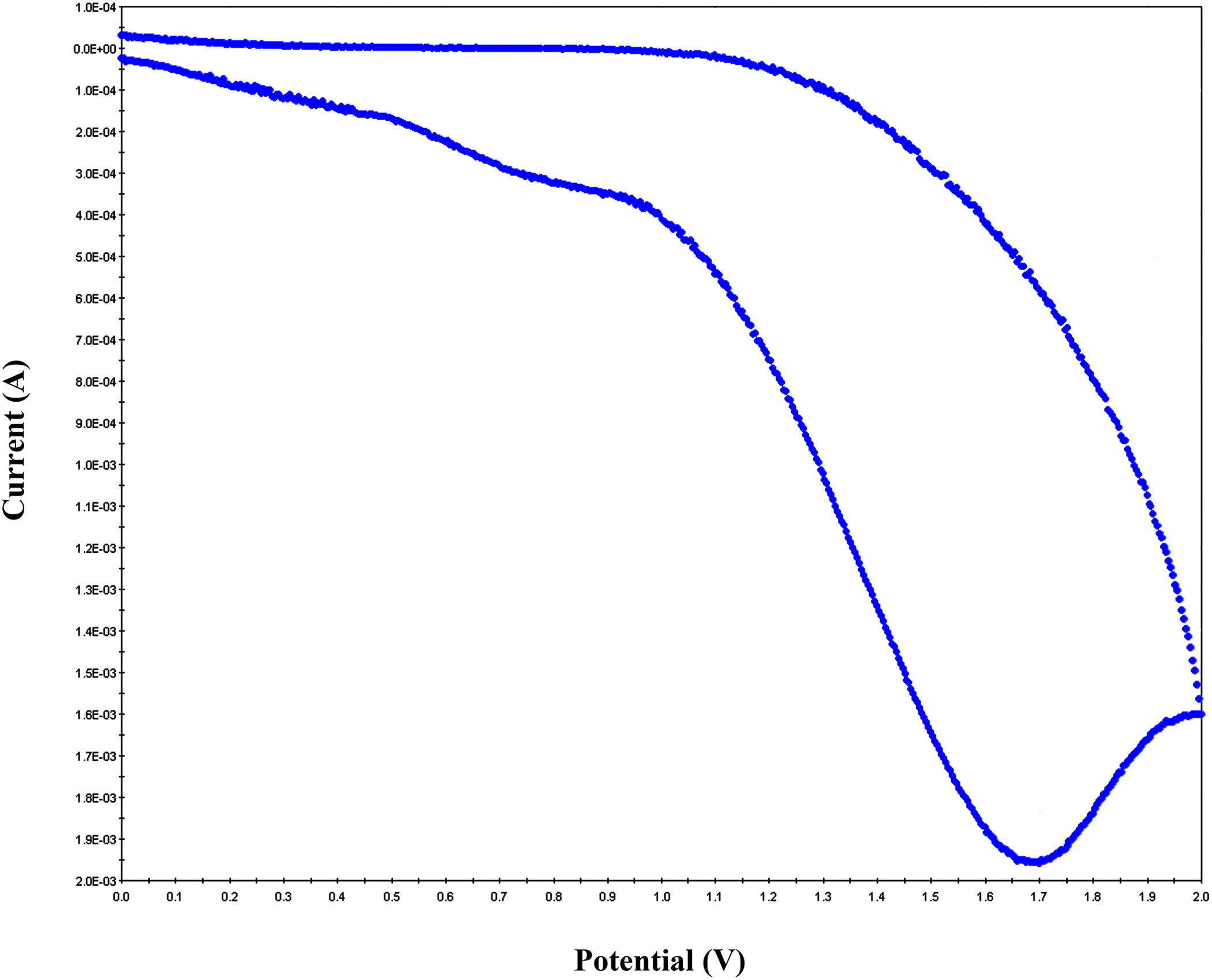
Cyclic voltammogram of PAI/MWCNT-COOH composite containing 10 wt% MWCNT in acetonitrile with 0.1 M Bu4NPF6.
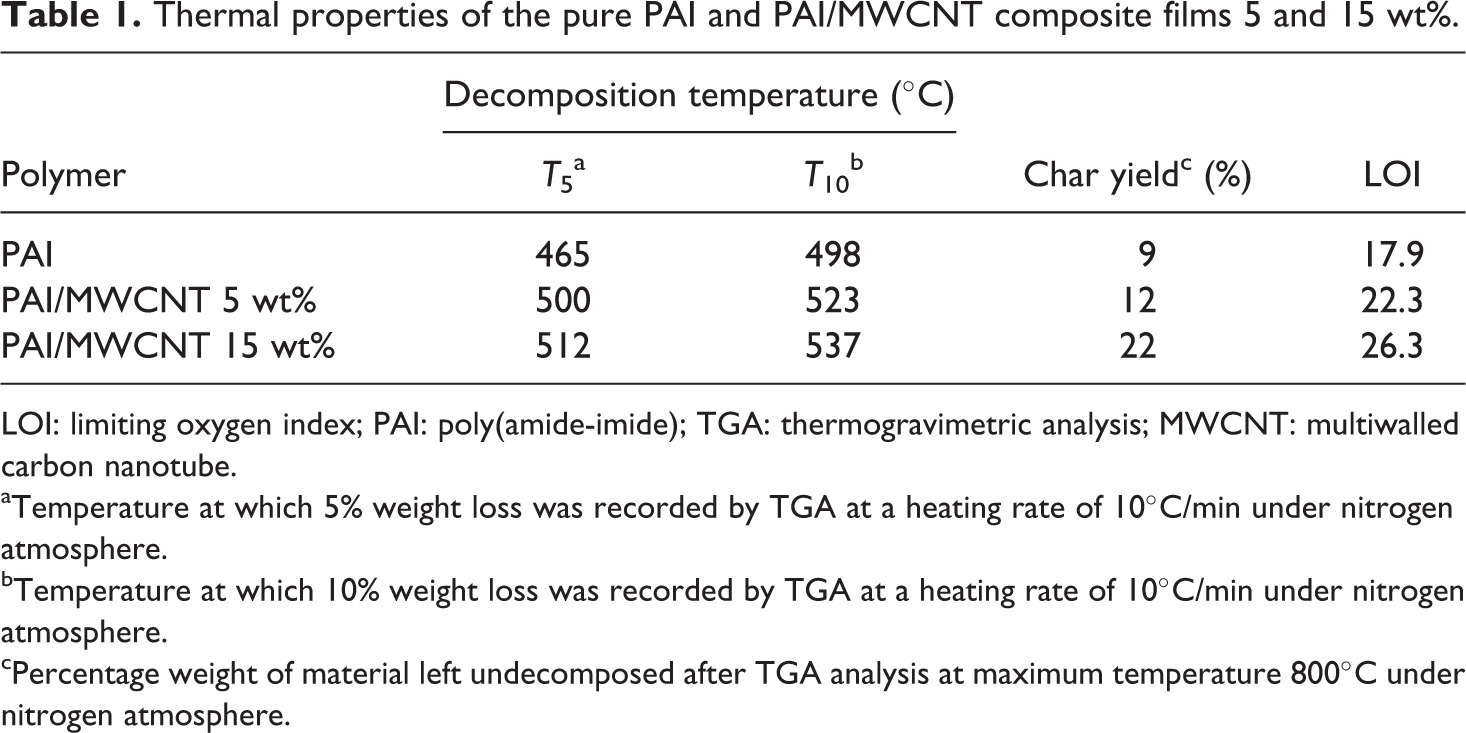
The thermal properties of pure PAI and PAI/MWCNT-COOH composites 5 and 15 wt% were studied using TGA at a heating rate of 10°C min−1, under a nitrogen atmosphere. The TGA curves of pure PAI and PAI/MWCNT-COOH composites 5 and 15 wt% are shown in Figures 8 to 10. The thermoanalysis data of these samples are summarized in Table 1. These studies show that the composites are thermally stable up to 450°C. The 10% weight loss temperatures of the pure PAI and composites were recorded in 498, 523, and 537°C for PAI and PAI/MWCNT-COOH composites 5 and 15 wt%, respectively. The amount of residue (char yield (CR)) of these composites was more than 12% at 800°C. As shown in Figures 9 and 10, the thermal stability of composites is higher than pure PAI that is due to the good compatibility of carboxylated MWCNTs with polymer matrix. CNTs have high thermal stability due to their larger surface area, so the incorporation of MWCNTs can improve the thermal resistance of composites.
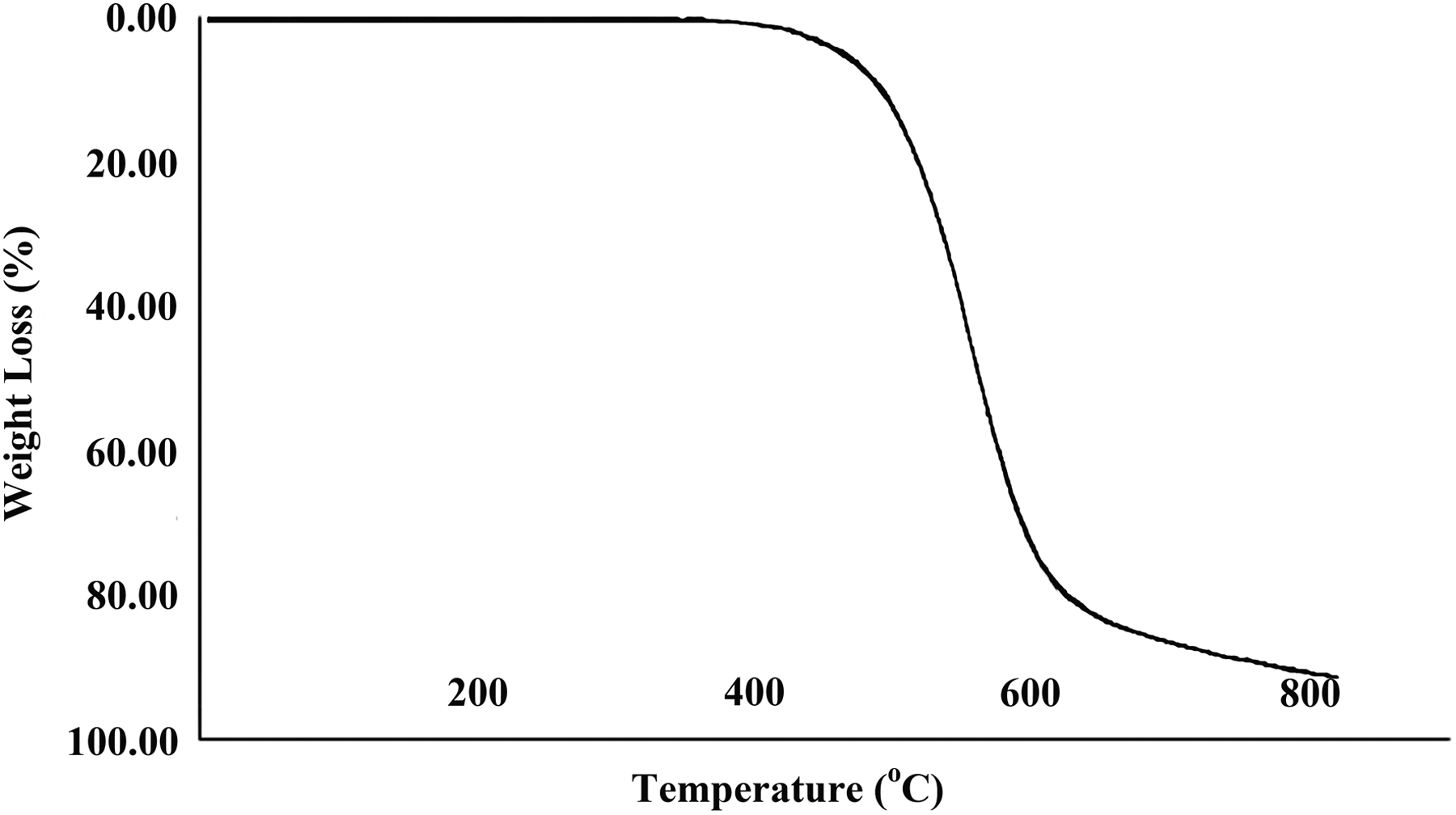
TGA thermogram of neat PAI. PAI: poly(amide-imide); TGA: thermogravimetric analysis.
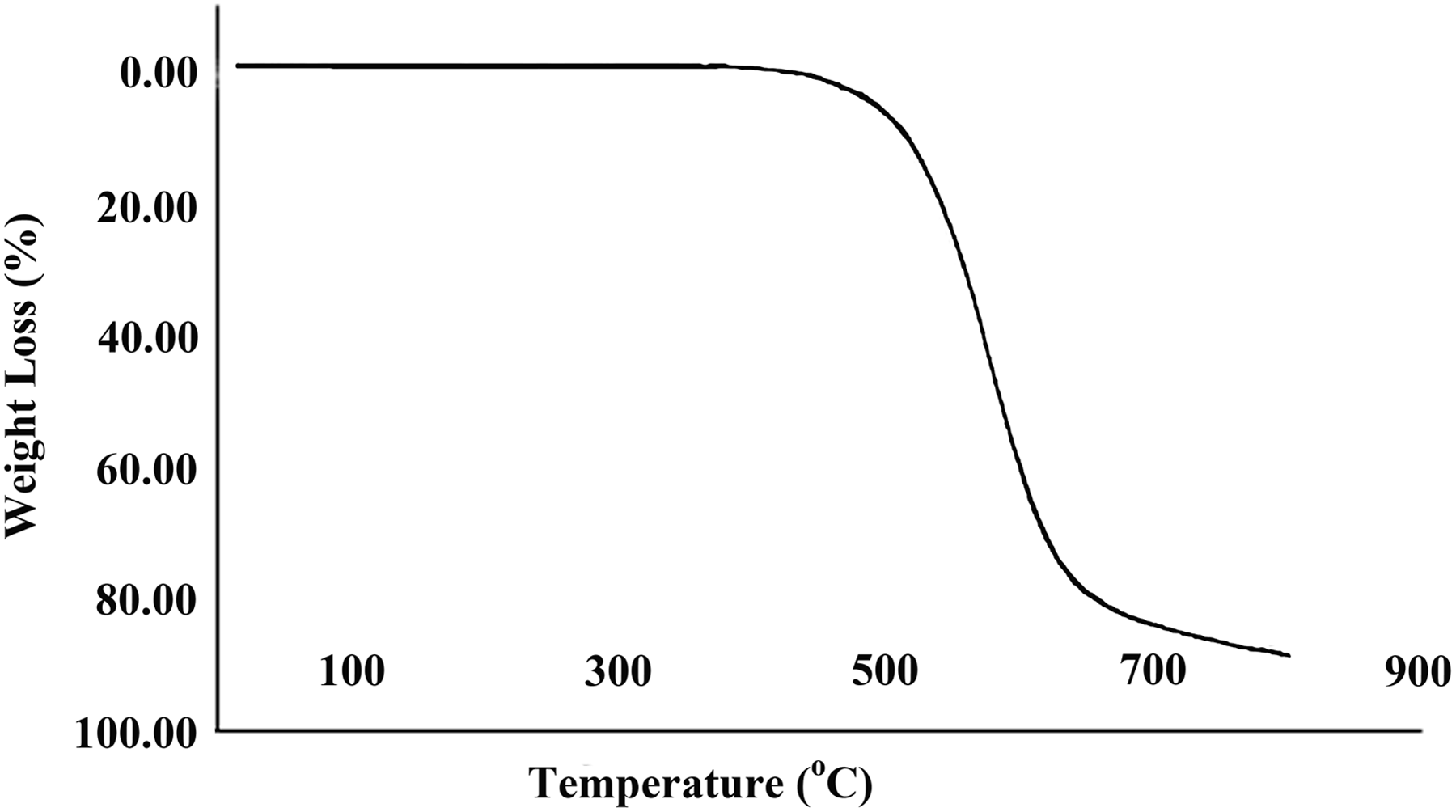
TGA thermogram of PAI/MWCNT composite containing 5 wt% MWCNT. PAI: poly(amide-imide); TGA: thermogravimetric analysis; MWCNT: multiwalled carbon nanotube.
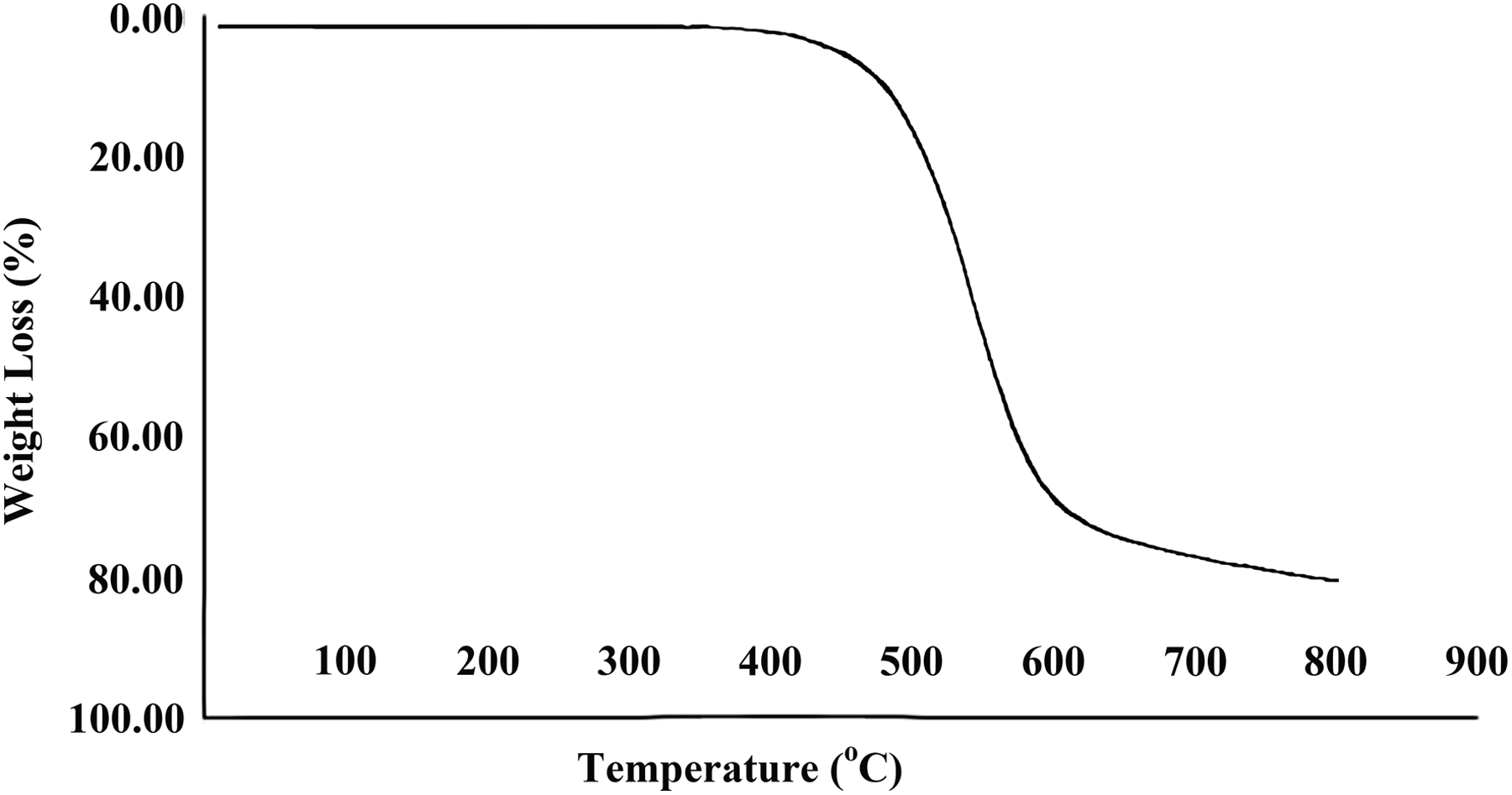
TGA thermogram of PAI/MWCNT composite containing 15 wt% MWCNT. PAI: poly(amide-imide); TGA: thermogravimetric; MWCNT: multiwalled carbon nanotube.
Thermal properties of the pure PAI and PAI/MWCNT composite films 5 and 15 wt%.
LOI: limiting oxygen index; PAI: poly(amide-imide); TGA: thermogravimetric analysis; MWCNT: multiwalled carbon nanotube.
aTemperature at which 5% weight loss was recorded by TGA at a heating rate of 10°C/min under nitrogen atmosphere.
bTemperature at which 10% weight loss was recorded by TGA at a heating rate of 10°C/min under nitrogen atmosphere.
cPercentage weight of material left undecomposed after TGA analysis at maximum temperature 800°C under nitrogen atmosphere.
CR can be applied as decisive factor for estimating limiting oxygen index (LOI) of the polymers based on Van Krevelen and Hoftyzer equation 34 :
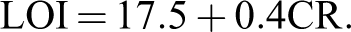
The composites had LOI values calculated derived from their CR was higher than 22. On the basis of LOI values, such composites can be classified as self-extinguishing materials.
Removal of MG from aqueous solution by PAI/MWCNT-COOH composite 15 wt% adsorbent
PAI/MWCNT 15 wt% was used as a novel and effective adsorbent for ultrasonic-assisted removal of MG dye from aqueous solution. The sonochemical adsorption experiment was carried out in a batch method as follows: 0.02 g of adsorbent was thoroughly dispersed in 50 mL of MG solution (5 mg L−1) at pH 6, and Erlenmeyer flask was immersed in an ultrasonic bath for 30, 45, and 60 min at room temperature. In later stage, the sample was immediately centrifuged for 5 min at 3500–4000 r min−1 and MG final concentration was quantified by UV–vis spectrophotometer. Figure 11 shows the UV–vis absorption spectra obtained at different times of ultrasonication exposed to reaction mixture. The quantitative amount of MG removed by PAI/MWCNT-COOH composite 15 wt% was obtained higher than 62%.
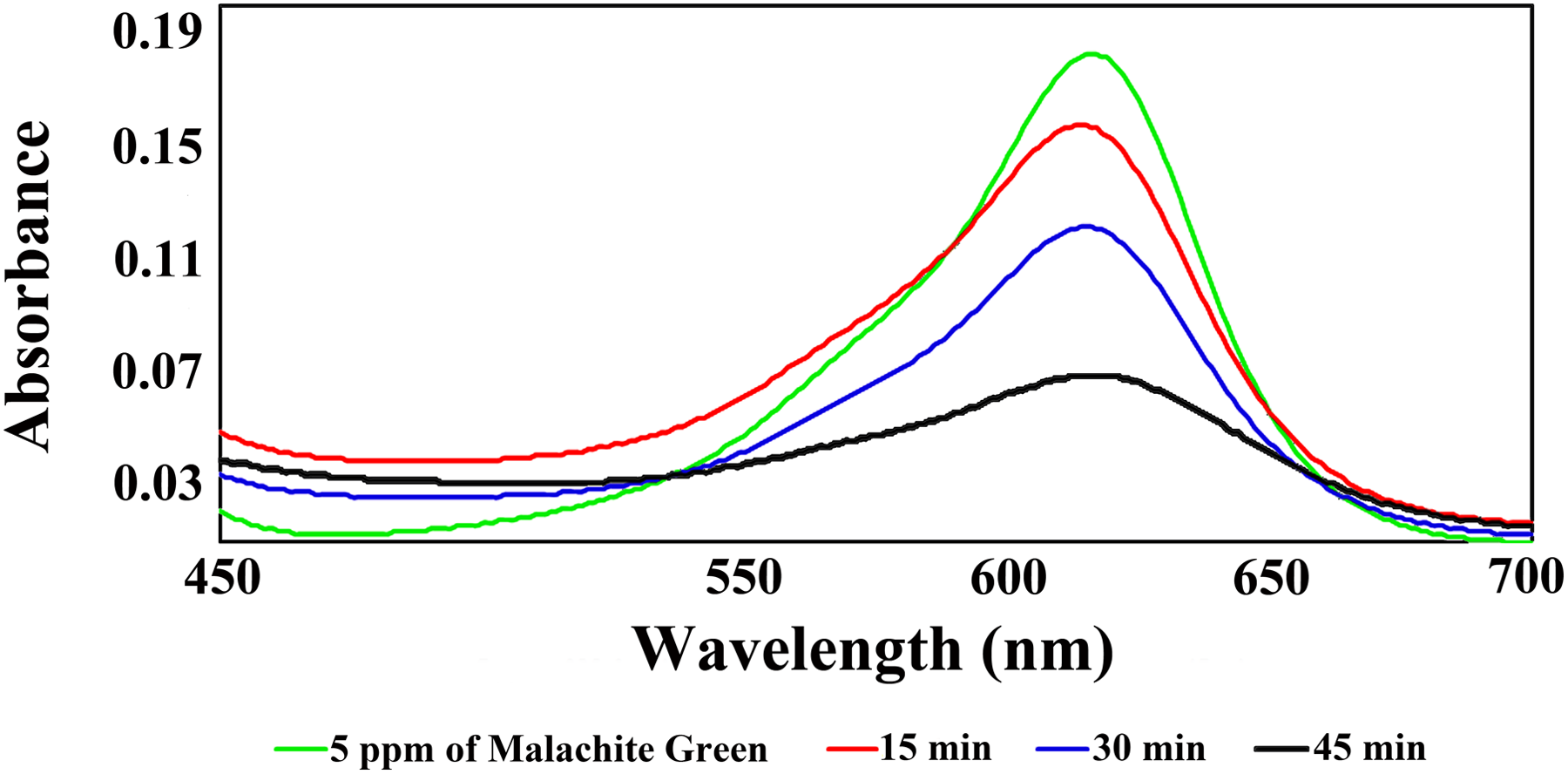
The change in UV–Vis spectra after the addition of 0.02 g PAI/MWCNT composite containing 15 wt% MWCNT to 5 mg L−1 MG at pH 6) at different times (0–45 min.)
Conclusions
Novel optically active amino acid–based PAI/MWCNT-COOH composite films were successfully prepared and characterized by different methods. For prevention of aggregation, the carboxyl-modified MWCNTs were used. TEM image confirmed nanoscale and homogeneous dispersion of MWCNTs in the PAI matrix. The TGA results indicate that the resulting PAI/MWCNT-COOH composite films are thermally stable. The use of PAI/MWCNT-COOH composite film as eco-friendly adsorbent was investigated as an ideal alternative material for toxic adsorbents to remove MG from wastewater.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors wish to express gratitude to the Yasouj University for financial assistance.
