Abstract
Intelligent food wrapping materials have sparked interest in preserving food from spoilage until it reaches to consumers. Sufficient moisture from the air can be picked up by the food to encourage microorganism’s growth. One of the most significant matters in food chemistry is bright packaging which enables the researchers to keep an eye on the quality of food. In the light of these factors, the researchers made long efforts to synthesize responsive-stimuli chitosan hydrogels from shrimp shell preparative chitosan which cross-linked with different weights of 1,6-diisocyanatohexane in refluxing 1% glacial acetic acid. Alternatively, the newly harvesting hydrogels were screened as meat and chicken smart packaging because they were soft, biodegradable, and flexible instead of plastic packaging that is nonbiodegradable and causes environmental pollution. The flexibility enabled hydrogels to be folded around the meat and chicken pieces. The synthesized hydrogels were confirmed by Fourier transform infrared spectroscopy, scanning electron microscopy, X-ray diffraction, and thermogravimetric analyses.
Introduction
Food safety is a scientific discipline that characterizes manufacture, storage, and food transportation in ways that prevent food-borne illness. Thus, some routines must be pursued to avoid potentially acute health risks. In this way, food safety often overlaps with food defense to protect consumers against harm. Safety between the market and the industry, then between the market and the consumer, and finally food wrapping must be tracked carefully. 1,2 Food preservation includes different methods such as canning, pickling, irradiation, pasteurization, smoking, drying, freeze-drying, and addition of chemical ingredients that prevent food from spoiling. Nowadays, food preservation has become of great importance as few people eat the food produced on their country’s land and consumers want to buy and consume food that is outside their production season. Food spoilage can occur due to one of two major reasons: (1) the contamination of food by microorganisms that causes diseases or death (pathogens) like bacteria, molds, viruses, and fungi and (2) the oxidation which destroys biochemicals, plant cells, and animal cells. Food drying is the simplest and the oldest for preserving food and requires tools of low cost. Also, it can be done indoors, outdoors, in a furnace, or in a specially built dehydrator. In the drying food technique, food is preserved by drawing moisture to a spot where the microorganisms can’t be grown. However, the quality is affected by drying because it doesn’t preserve nutrients. Thus, most efforts for preserving food quality are directed through packaging. The wrapping material must act as a barrier between the food and the air. The food will maintain its quality if it isn’t exposed to light or metal surfaces, if it is wrapped in a transparent plastic, and if it is stored in a dark space or an opaque material. 3,4 Hydrogels are generally highly biocompatible. This biocompatibility is due to the high water content and ease of biodegradation. Biodegradability or dissolution may occur by enzymatic, hydrolytic, or environmental (e.g. pH, temperature, or electric field) pathways. 5 -7 Hydrogels can familiarize themselves with the morphology of the surface onto which they are applied. Hydrogels have a wide range of applications where they are used in agriculture, 8 food packaging, 9 -11 and in the biomedical field. 12 -19 Herein, we report a novel approach to developing transparent thin films of chitosan hydrogels for food packaging using appropriate synthetic methodology to address the global ecological issue of nonbiodegradable plastic food wrapping.
Materials and methods
General remarks
Preparative chitosan from shrimp shell obtained with 80% deacetylation degree and an average molecular weight of 800,000 kDa was used. X-Ray Diffraction (XRD) was employed for the crystallinity of the polymer. XRD was performed using a Philips model PW 3710 (Philips, San Diego, CA, USA) diffractometer with copper
The feed composition and designation of the chitosan hydrogel thin films
In a round-bottomed flask equipped with a chemical stirrer, the chitosan of high molecular weight (800,000 Da) was dissolved and stirred in 1% (w/v) of acetic acid for about 1 h, then different concentrations of terminated NCO of hexamethylene 1,6-diisocyanate (HMDI) NCO/NH2 (0.025, 0.037, 0.052, 0.062, 0.075, 0.137, 0.15 mL) were added slowly with stirring, and the reaction was continued and executed at 40°C for 5 h. The reaction content was filtered off to remove the unreacted material. The film casting solution was casted onto 7 cm diameter of petri dishes and let dry at 23°C for 7 days.
Circular transparent films are flexible, more folded, strong with heating and the IR of chitosan hydrogel at 0.025 of cross-linker: ύ (cm−1) = (3141.71 broadband, OH and traces of NH2), (2922.10, CH), (2140, CH2), 1651, traces of CO), (1338, –C–O–C–). IR of chitosan hydrogel at 0.037 of cross-linker: ύ (cm−1) = (3342.71 broadband, OH and traces of NH2), (2922.22, CH), (2140.21, CH2), 1650, traces of CO), (1331, –C–O–C–). IR of chitosan hydrogel at 0.052 of cross-linker: ύ (cm−1) = (3443.04 broadband, OH and traces of NH2), (2922.37, CH), (2150.16, CH2), 1558, –C–N–), (1327.16, –C–O–C–). The IR spectrum of chitosan hydrogel at 0.062 of cross-linker showed strong absorption bands at ύ (cm−1) = (3141.71 broad band, OH and traces of NH2), (2921.71, CH), (2150, CH2), 1527.59, –C–N–), (1338.89, –C–O–C–). Also, the IR of chitosan hydrogel at 0.073 of cross-linker displayed a stretching absorption band at ύ (cm−1) = (3422.74 broadband, OH and traces of NH2), (2922.10, CH), (2146.44, CH2), 1559, traces of CO), (1381.56, –C–O–C). IR of chitosan hydrogel at 0.15 of cross-linker ύ (cm−1) = (3442.74 broadband, OH and traces of NH2), (2923.10, CH), (2146.44, CH2), 1559, –C–N–), (1381.56, –C–O–C).
Preparation of food packaging samples
In five petri dishes, 5 g for each piece of the chicken and meat cubes was weighed, then the samples were washed, and signed as A, B, C, D, and E. Piece A was wrapped with chitosan hydrogel film (0.062 mL), piece B was wrapped with chitosan hydrogel film (0.075 mL), piece C was wrapped with chitosan-based film, piece D was wrapped with plastic cover, and piece E left without any wrapping. All were incubated at room temperature and the physical changes were observed. Observations were recorded after 3 days and 7 days. The piece wrapped with plastic cover and without wrapper were showed a thin layer of yellow- and black-colored molds on the surface, unpleasant odor, and formation of crawling insects such as maggots, while the pieces covered with films showed no changes.
Swelling of designed hydrogel films method
In a 150-mL beaker, a specific weight of each sample (0.08 g) of each concentration of cross-linker (0.025, 0.037, 0.052, 0.062, 0.075, 0.137, 0.15 mL) was taken, followed by adding them to 50 mL distilled water at different temperatures (0°, 18°, 50°, and 100°C), acidic solutions at pH = 1, 2, and 3, and basic media at pH = 11.9, 12.4, and 13.9. Next, each sample wass left in different aqueous solutions to allow complete swelling. After that, each swollen sample was weighed. Swelling equilibrium percentage (S%) for each one was calculated. Graph relationship between the concentration of cross-linker at different environment and swelling equilibrium percentage (S%) was calculated.
SEM method
Specimens of flexible hydrogel thin films are mounted on stubs appropriate for the particular scanning electron microscope being used. A convenient adhesive is a double-stick, electrically conductive carbon tape. To assure electrical continuity of the specimen with the stub, a small drop of silver paint may be added by the critical point drying technique. Critical point drier (CPD) is used to replace all of the ethanol with liquid carbon dioxide under pressure. The pressure and temperature are raised in the CPD until the specimen is above the triple point at which time it is safe to decrease the temperature and release the pressure. The volume of liquid carbon dioxide is replaced several times until ethanol is no longer present in the purge line. One or two additional changes of liquid carbon dioxide are typically used to assure that no ethanol is present during the drying stage. Once the dried material is removed, it needs to be stored in a desiccated environment until viewing.
Thermal gravimetric analysis method:
Samples were subjected to a controlled heating/cooling program (non-isothermal or isothermal) and their weights were measured over time at a range of temperatures.
Results and discussion
Chemistry
Advances in polymer science have led to the development of hydrated and biodegradable biopolymer such as a superabsorbent, eco-friendly, and intelligent polymer (hydrogel) to improve food packaging implementations via utilizing the ecological features of biopolymers. In this article, a novel biodegradable hydrogel films that are able to respond to environmental changes as pH, solvent composition by changing their dimensions and temperature were destined. Conventionally, transparent chitosan hydrogel thin films were synthesized by solution casting method, the acidic solution of chitosan and different concentrations of HMDI as cross-linker (0.025, 0.037, 0.052, 0.062, 0.075, 0.137 and 0.15 mL) were refluxed with stirring at 40° C for 5 h (Figure 1). The resulted thin films were confirmed by Fourier transform infrared (FTIR) spectra, XRD, thermogravimetric analysis (TGA), and SEM analyses. IR spectroscopy (FTIR) showed a characteristic joint peak at ύ (cm−1) 3141.71–3443.04 corresponding to a hydroxyl group and an amino group stretching, respectively. On the other hand, the FTIR spectrum displayed absorption bands at ύ (cm−1) 2922.10–2922.43, 1651–1648, and 1338–1327. These bands corresponded to CH, an amidic carbonyl group –NHCONH– and –C–O–C– linkage. The morphological SEM analysis shows that softness, smoothness, and porous surface through the entire hydrogel structures of higher activity incorporating cross-linker of weights 0.052, 0.062, and 0.075 mL (Figure 2). This promoted the efficiency of films in absorbing the moisture and their activity in packaging applications via screening them by SEM after swelling in a water medium and revealed the bounded water inside the porous positions on hydrogel surfaces of concentrations (0.052, 0.062, and 0.075 mL) (Figure 3).

Schematic representation of cross-linking between chitosan and cross-linkers (hexmethylene-1,6-diisocyanate).

Scanning electron micrographs of hydrogels at (a) 0.025, (b) 0.037, (c) 0.052, (d) 0.062, (e) 0.075, and (f) 0.137 mL cross-linker before swelling.

SEM micrographs after swelling of the most effective chitosan films: (a) 0.052, (b) 0.062 and (c) 0.075.
XRD exhibits a crystallinity of chitosan hydrogels ranging from 0° to 80° (2

XRD of chitosan hydrogel: (a) 0.052, (b) 0.062, and (c) 0.075 mL.
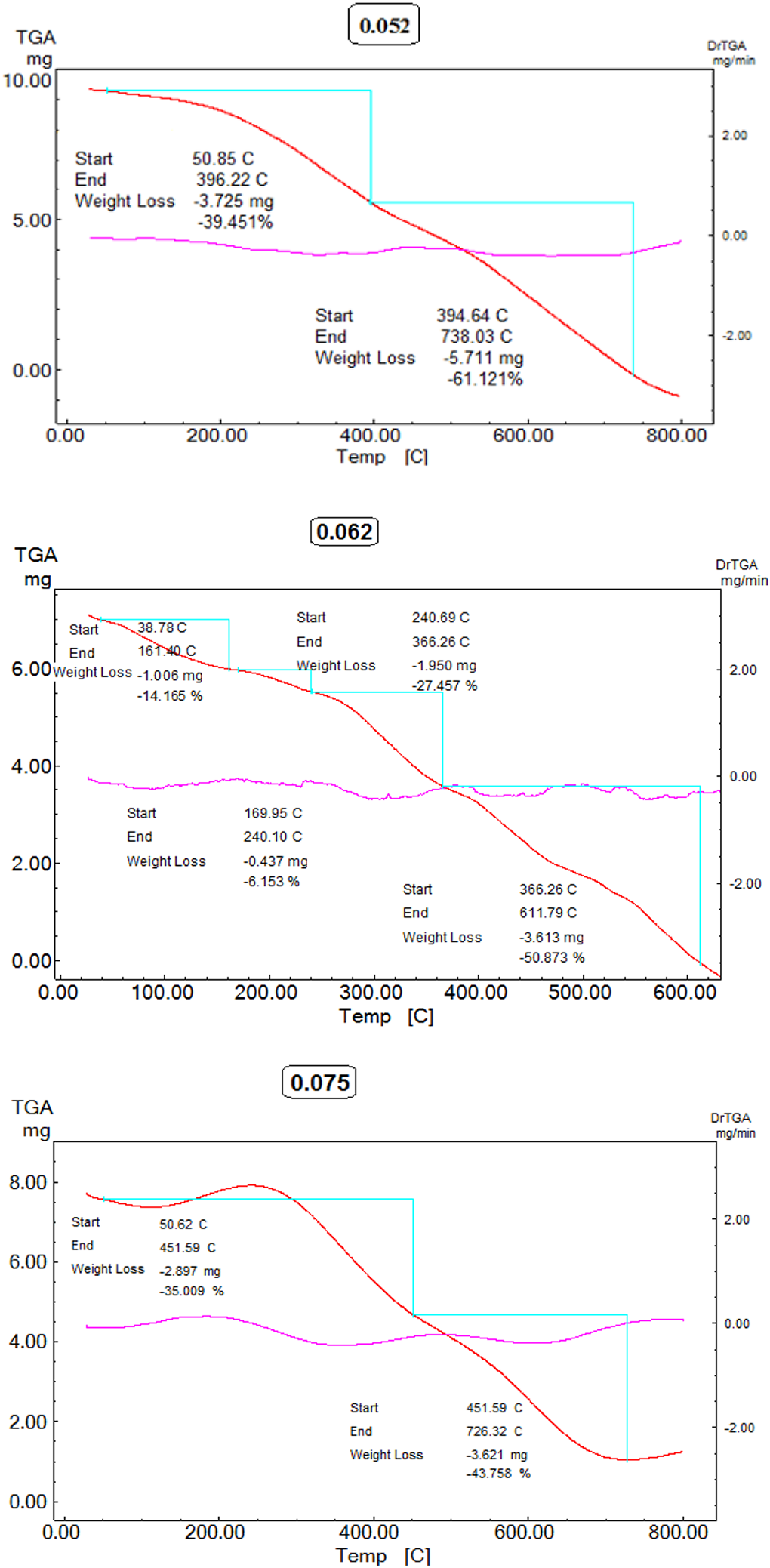
TGA of chitosan hydrogel: (a) 0.052, (b) 0.062, and (c) 0.075 mL.
Kinetic study of smart transparent chitosan hydrogels
Stimuli-sensitive chitosan hydrogels and swelling studies of water-absorbing hydrogels at different temperatures, in acidic and basic medium at different pH
Gels are highly hydrated hydrophilic polymeric network gels that swell owing to the capability of absorbing large amounts of water or acidic and basic liquids but do not dissolve in water due to their thermal stability, so they are widely used in food wrapping and in the medical industry as dressings and even in tissue regeneration and tissue engineering. 20 As a result, water uptake of dried chitosan hydrogel samples was investigated in different conditions of aqueous solutions and the swelling equilibrium age % of these intelligent hydrogels was calculated as follows (Figures 6 to 9):

Images of stretching transparent hydrogel film before and after swelling.
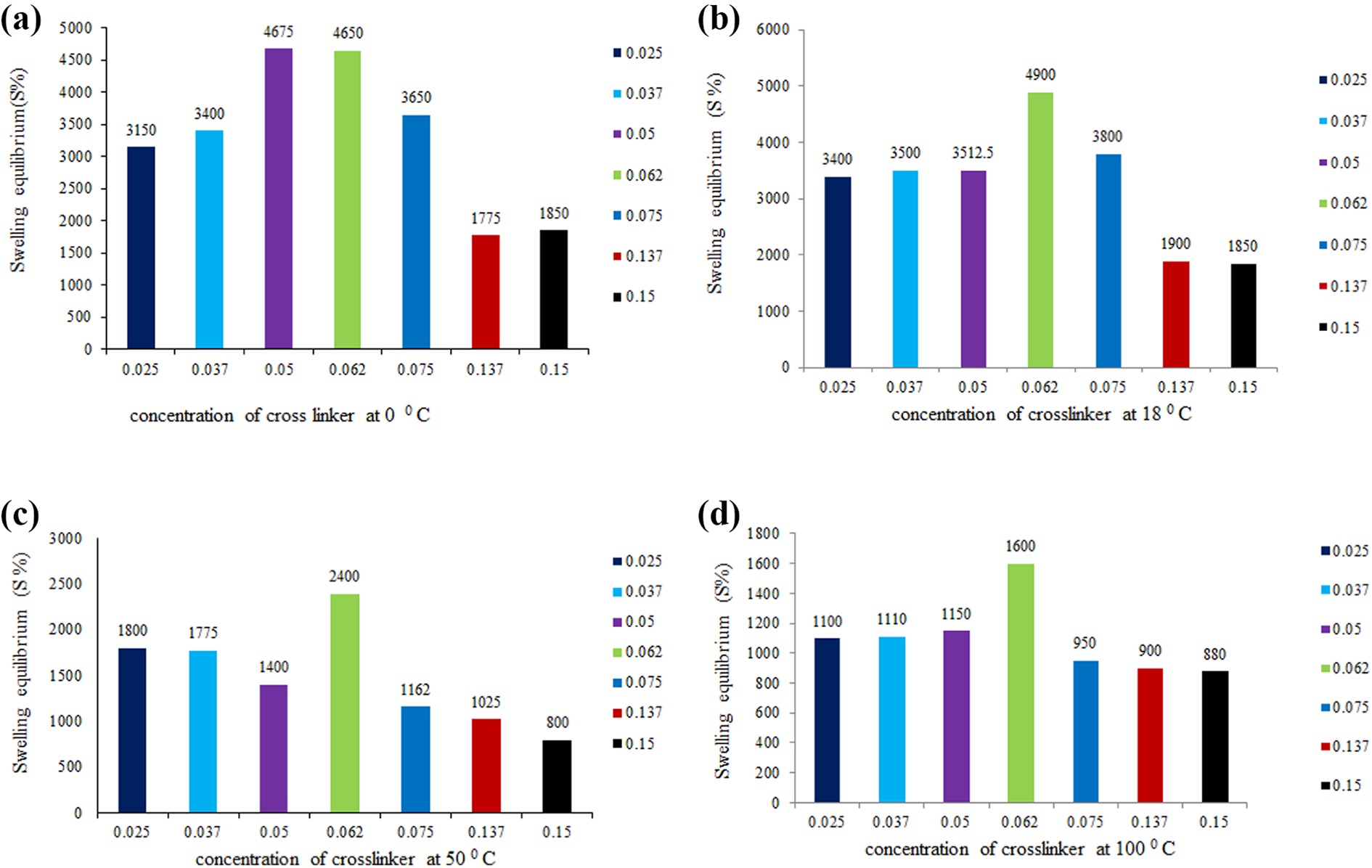
Effect of water medium on hydrogel at (a) 0, (b) 18, (c) 50, and (d) 100°C.

Effect of acidic medium on hydrogel at (a) pH = 1, (b) pH = 2, and (c) pH = 3.

Effect of alkaline medium on hydrogel at (a) pH = 11.9, (b) pH = 12.4, and (c) pH = 13.4.

where
From swelling test overview, chitosan hydrogels own high capacity of water absorption at 0.052-, 0.062- and 0.075-mL concentration of a cross-linker. This is essential since several nourishment materials release humidity that should be absorbed by the packaging materials to memorize food from rapid deterioration. To be folded as desired during packaging, it must be elastic and non-fragile. The most essential parameter is the packaging material biodegradability to avert ecological risks. The scenario of degradation of the prepared gel films was studied beneath planned environment (30°C, pH 6.0–8.5, and humidity content ∼61%) using commercially obtainable manure, and it was performed by soil burial test in a fertilizing environment up to 6 weeks. Structural deformation of the hydrogel films with time was detected through SEM analysis. The hydrogel films indicate acute weight lack until 5 weeks. After that, there were no meaningful residues of thin films that existed in the manure bed. The SEM images present a favorable proof of the biodegradation of chitosan gel film having 0.062 cross-linker. The hydrogel surface was nearly soft before degradation. After 4 weeks, irregularities of the surface of destined films can be observed. This indicates that the interior framework of chitosan hydrogel films has initiated to degrade. The destined film’s surface became more irregular with some sedimentations after 6 weeks (Figure 10). The hydrogel films were biodegraded to CO2, H2O, and glucosamine moieties as shown in TGA.Recently, there has been a great evolution of innovative and new approaches in the technology of food packaging as a result of the request for perfect quality and safe nourishments other than changes in customer inclination. This technology has helped to observe and communicate data about food fineness. Generally, the master signal of food damage in packed foods is the occurrence of elevated chitosan hydrogels. Thus, to avert deterioration of food is necessary. The biodegradable polymers produce several problems in their execution and processing such as brittleness and non-flexibility. 21,22 To avoid this problem, researchers exerted significant efforts to design flexible and non-brittle packaging material to be folded as desired during packaging. In fact, the benefits of hydrogels expanded successfully to meat and chicken packaging. We will extend our work on these hydrogels in the next article in the near future to use it as fertilizers for agriculture. This decision relies on their biodegradability to eco-friendly components such as CO2, H2O, and glucosamine moiety and also use biodegradability testing standards like EN 13432 to check their safety.
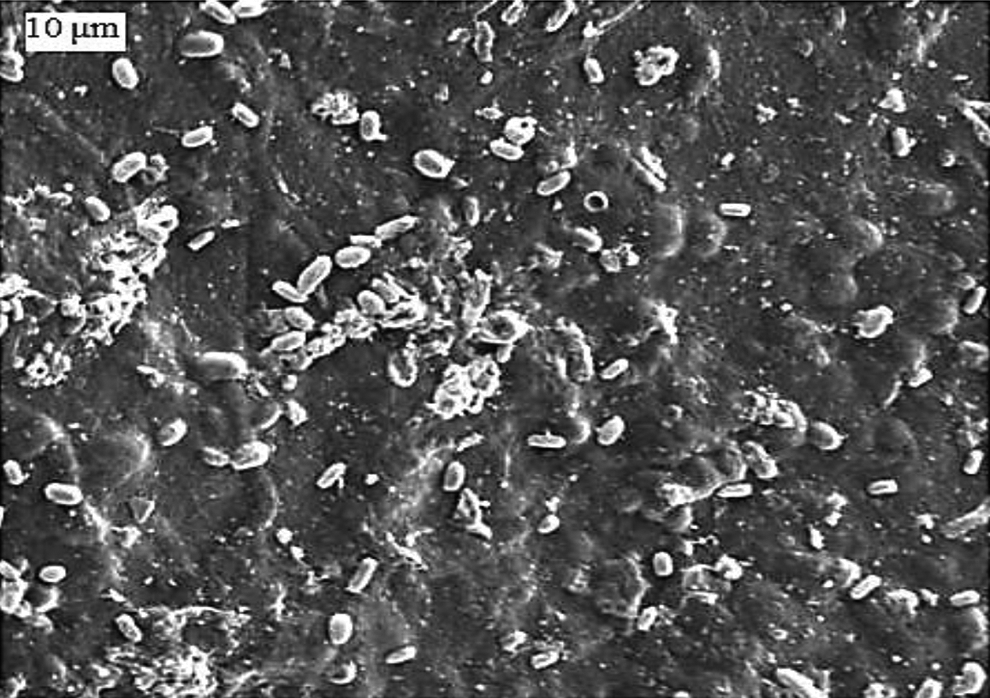
Chitosan–hydrogel film with 0.062 cross-linker after biodegradation (after 4 weeks’ degradation).
Application of chitosan, chitosan film, chitosan hydrogel 0.062 and 0.075 based film in chicken and meat packaging
Successfully, the five pieces of the chicken and meat cubes (5 g each) were washed and marked as A, B, C, D, and E. Piece A was wrapped with chitosan hydrogel film (0.062 mL), piece B was wrapped in chitosan hydrogel film (0.075 mL), piece C was wrapped with chitosan-based film, piece D was wrapped with plastic cover, and piece E was left without any wrappers. All were incubated at room temperature, and the physical changes were observed. But we notice that after few days, the piece wrapped with plastic cover and without wrapper generate a thin layer of yellow- and black-colored molds on the surface, unpleasant odor, and formation of crawling insects such as maggots as a result of the oxidation of food tissues by moisture, while the pieces covered with films showed no changes (Figures 11 and 12).

Meat and chicken packaging with chitosan–hydrogel at 0.062 cross-linker (A), packaged with chitosan–hydrogel at 0.075 cross-linker (B), samples packaged with chitosan film (C), samples packaged with plastic cover (D), and without packaging (E) at the moment (a) and after 3 days (b).

(a) Meat and chicken packaging with chitosan–hydrogel at 0.062 cross-linker (A), packaged with chitosan–hydrogel at 0.075 cross-linker (B), samples packaged with chitosan film (C), samples packaged with plastic cover (D), and without packaging (E) after 7 days showing formation of maggots (b).
Conclusion
Today, hydrogels still mesmerize material researchers and scientists, and enormous strides have been done in terms of their formularization and enforcements. Finally, the article concludes that the flexible and smart super porous chitosan hydrogel thin films are tested in the packaging food area. This concept turned out to be a very fruitful biodegradable hydrogel because of the rich performance of films in meat and chicken wrapping until reaching to consumer regarding with respect to water sensitivity, mechanical properties with high flexibility, and biodegradability of them. Researchers achieved high success to face the environmental pollutants caused by other packaging materials such as plastic due to nonbiodegradability.
Footnotes
Acknowledgement
Authors would like to express their sincere thanks to their institutions.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
