Abstract
A pH-sensitive nanocomposite formed from sodium alginate (SA)/pectin (Pec)/tannic acid (TA)–silver (Ag) was developed using microwave irradiation and it was applied as a carrier for propranolol drug. TA acts as a cross-linker and a reducing agent for Ag ions. Physicochemical characteristics of the fabricated system using Fourier transform infrared spectroscopy, X-ray diffraction, field-emission scanning electron microscope, high-resolution transmission electron microscope, dynamic light scattering instrument, and energy dispersive X-ray analysis. The swelling percent and the drug release were observed to be pH-sensitive. The occurrence of Ag nanoparticles in the network enhances the drug release that is 96% at pH 7.4 within 420 min. The drug release data were adjusted into different kinetic models involved zero order, first order, Higuchi, and Ritger–Peppas models. The release mechanism is a non-Fickian character where it controls by diffusion and relaxation of polymer chains. It can be concluded that SA/Pec/TA-Ag nanocomposite is a candidate for the oral drug carrier specific for the intestinal system and has ability against the gastric fluid.
Introduction
Smart hydrogels are hydrophilic polymer three-dimensional systems that have physically or synthetically cross-linked polymer. They quickly swell and contract because of natural changes, for example, pH, temperature, and magnetic or electric field because of the presence of specific functional groups along the polymer chain. 1 Creative medication transport progress in view of perspective hydrogels has an extraordinary benefit. 2,3
The pH-responsive hydrogels are considered as engagement for the controlled release of medication, because their pH responsively moves in various locals of the body in both typical and pathological conditions. 4
Biopolymer hydrogels provide great applications in pharmaceutical area due to nonpoisonous quality, biocompatibility, and biodegradability.
5
Sodium alginate (SA) is a characteristic polysaccharide of a purified carbohydrate obtained from brown seaweeds by employing dilute alkali.
6
It is a polyanionic natural linear copolymer of (1→4) α-
The microwave irradiation is considered an advantageous system for hydrogel synthesis by utilizing a blend of polymeric reactants. 11 It has high-temperature heating process for assault the solution in a short time and restricted the side reactions. 12 Xu et al. reviewed the application of microwave irradiation in polymer synthesis, and found the microwave radiation has advantages over the conventional thermal method in improving properties of the polymer synthesis. 13
Nanoparticles (NPs) have been broadly examined particularly noble metals in account of their novel functions and exceptional properties. 14 Silver (Ag) NPs are non-harmful and environmentally eco-friendly. 15 Among various noble metal NPs, Ag shows extraordinary consideration because of their prevalent antibacterial properties. 16 They have high favoritism toward phosphorus- or sulfur-containing proteins inside or outside bacterial cell layers actuating assistant changes, which impacts bacterial cell feasibility. 17,18
Herein, the examination of various properties of inorganic nanomaterials incorporated biopolymer hydrogels is uncommon; however, the readiness of SA/Pec/TA biopolymer hydrogel consolidate with Ag NP as a drug carrier is new in the literature. In this study, a pH-responsive hydrogel of SA/Pec/TA-Ag nanocomposite was synthesized using microwave irradiation to act as a drug carrier. TA here has a dual impact, and it acts as a cross-linker and a reducing agent for Ag ions more over it rich in carboxylic acid groups. The nanocomposite was characterized by Fourier transform infrared spectroscopy (FTIR), X-ray diffraction (XRD), field emission scanning electron microscope (FESEM), high-resolution transmission electron microscope (HRTEM), and dynamic light scattering instrument (DLS). The obtained nanocomposite was examined to study its pH-responsive swelling and the controlled drug releasing by using propranolol drug as a model of drug.
Experimental
Materials
Pec (from apples) was acquired from (Sigma-Aldrich, Saint Louis, MO, USA). TA was supplied from Qualikems (India). Propranolol drug was obtained from El Qahera for Pharmaceutical & Chemical Industries (Egypt). Other chemicals, for example, ammonium persulfate (APS), silver nitrate, phosphate, and acetate buffers were purchased from (Sigma-Aldrich) and utilized without assist refinement.
Algae collection
Brown alga, Sargassum asperifolium which related to family Sargassaceae, genus Sargassum, was collected from coast of Jazan, Kingdom of Saudi Arabia, and used as a source of SA. The algal samples were washed several times with tap water then with distilled water, and dried overnight at 40–45°C in an oven.
Extraction and purification of SA
Grinded algal sample (10 g) was suspended in 2% calcium chloride for 2 h, and then it was washed with deionized water. 19 SA was extracted by the addition of 1 M of sodium carbonate and 0.5 g of ethylenediaminetetraacetic acid. The pH of the suspension was adjusted to be 11 for 48 h. This was then filtrated through muslin cloth. SA was precipitated by addition of two volumes of ethanol under stirring. SA was purified according to the method of Gomez et al. 20 The insoluble polymer was separated, and then it was exhaustively washed with acetone by Soxhlet for 48 h. Finally, the biopolymer was dried at room temperature under vacuum until a constant mass.
Synthesis of SA/Pec/TA hydrogel
Different ratios of SA, Pec, and TA were tested to choose a ratio to give hydrogel with the best properties. SA:Pec:TA in the ratio of 10:4:3 was dissolved in hot water with stirring. After cooling, 0.5 wt% of APS (initiator) was added. The solution was briefly mixed by a prop sonication for 10 min to form a homogeneous solution. After that, the mixture was irradiated in a domestic microwave oven (Thai Samsung Electronics Company) at 300 W for 10 min. The formed hydrogel was extricated for 2 h in hot water to evacuate the homopolymer, and it was dried under vacuum at 40°C for 12 h. 21
Synthesis of SA/Pec/TA-Ag nanocomposites
A 100 mg of dried SA/Pec/TA hydrogel was placed in 50 mL of Ag ions solution of concentration 250 mg L−1 for 24 h to dope metal ions in the blend matrix. Blended matrix with loaded metal ions was placed in distilled water for 24 h to remove the unbound metal ions. Then, they were reduced by transferring them into 50 mL of 5% sodium hydroxide for 6 h and then in 50 mL of 0.5 M sodium borohydride for another 6 h to complete reduction of the metal ions, soaking them in deionized water for 12 h and drying at 40°C.
Characterization
FTIR spectroscopy analysis was done utilizing Nicolet iS-10 FTIR, Thermo Fisher Scientific (Waltham, MA, USA) in the range of 400–4000 cm−1. HRTEM estimations were done by using JEOL 2100-Lab6 (Akishima, Tokyo, Japan). Its magnification is from 2000× to 1,500,000×. The accelerating voltage is 80–200 kV. Morphological examinations were done using FESEM, Quanta 250 FEG; the accelerating voltage is 30 kV (FEI Company, Netherlands). It was attached with energy dispersive X-ray analysis (EDX) unit (energy dispersive X-beam analysis). The XRD was done using XD-DI Series, Shimadzu apparatus with a copper target (λ = 1.542 Å). All the diffraction patterns were accomplished at an operating voltage of 40 kV, an electric current at 30 mA, and a room temperature over a range of 2θ (10–90°) at a scan speed of 8° min−1. DLS (Zeta Sizer Nano Series HT; Nano ZS) is obtained from Malvern Instruments Ltd, Worcestershire WR14 1XZ, UK. Absorbance estimation was done utilizing ultraviolet–visible spectrometer (UV-Analytic Jena AG, Germany), with a quartz cell of 1.0-cm optical length. The synthesis and characterization are shown in Figure 1.

Preparation and characterization of SA/Pec/TA-Ag nanocomposites.
Swelling measurement
The clean, dried, weighed hydrogel sample was soaked in bi-distilled water or buffer solution at room temperature for different time intervals. The sample was removed and the excess water on the surface was removed by blotting quickly with filter paper and reweighed. The swelling percent was calculated as follows:
where Wd and Ws are the masses of dry and swelled samples, respectively.
Drug loading
The stacking of propranolol drug onto SA/Pec/TA-Ag nanocomposite and SA/Pec/TA hydrogel as a control was completed by swelling technique. The samples were allowed to swell in the drug solution of different concentrations and pH for 24 h and then it was dried at room temperature. The concentration of the rejected solution was estimated to determine the percent of drug caught in the lattice at the wavelength of 194 nm
where Ci (mg L−1) is the initial drug concentration and Ce (mg L−1) is the equilibrium drug concentrations in the solution.
In vitro propranolol drug release studies
In vitro release studies of the drug were carried out by suspending 100 mg of the drug-loaded sample in 10 mL of the buffer-releasing medium (pH 2.1 and 7.4) at 37°C. The amount of drug released was assayed spectrophotometrically. All the studies were carried out in triplicate and the total uncertainty range was 2–4%.
Drug release kinetic
To investigate the propranolol release mechanism from SA/Pec/TA-Ag nanocomposite and SA/Pec/TA hydrogel as a control, different kinetic models were considered to fit the experimental data. 22,23
Zero-order drug release kinetic model is given as follows:
The first-order drug release kinetic model is given as follows:
The Higuchi square root model is given as follows:
The Ritger–Peppas model is given as follows:
where Mt and Minf are the drug release at time t and equilibrium, respectively. k0 is zero-order release constant. k1 is the first-order release constant. kH is Higuchi release constant. k is the rate constant. n is the release exponent.
Results and discussion
Physicochemical characteristics of SA/Pec/TA and SA/Pec/TA-Ag
The FTIR spectra of SA, SA/Pec/TA hydrogel, and SA/Pec/TA-Ag nanocomposite appear in Figure 2. Clearly, SA demonstrates an expansive band at 3456 cm−1 ascribed to O–H groups. The band at 2925 cm−1 identified with C–H stretching vibration. The C=O stretching vibration showed up at 1543 cm−1. SA has a characteristic band showed up at 982 cm−1 because of Na–O. 24 For SA/Pec/TA hydrogel, the bands of O–H, C–H, and C=O stretching vibrations showed up at 3376, 2973, and 1635 cm−1, respectively. The increase of the intensity of the O–H band and the sharpness of the C=O band compared with SA because Pec and TA present in the hydrogel network. Smaller bands were seen at 915 and 587 cm−1 related to the substituted benzene ring in TA. 25 For SA/Pec/TA-Ag nanocomposite, it could be noticed that no new bands were seen; however, slight shifts of the bands were observed. This is because the electrostatic attraction between the Ag and the electron donating groups presents in SA, Pec, and TA.

FTIR spectra of SA, SA/Pec/TA hydrogel, and SA/Pec/TA-Ag nanocomposite.
The surface morphology of SA/Pec/TA hydrogel and SA/Pec/TA-Ag nanocomposite was inspected by utilizing FESEM, as shown in Figure 3. For SA/Pec/TA hydrogel (Figure 3(a)), the surface shows up as a smooth which demonstrated good compatibility and effectively fused polymeric segments in the system structure of the hydrogel. For SA/Pec/TA-Ag nanocomposite (Figure 3(c)), the surface totally changed to a gruff surface with some sporadic pores as a result of joining of Ag NPs inside the polymeric structure. The EDX micrographs of SA/Pec/TA hydrogel and SA/Pec/TA-Ag nanocomposite are shown in Figure 3(b) and (d), respectively. It can be observed that carbon (C) and oxygen (O) are the main backbone elements in the SA/Pec/TA network (Figure 3(b)). The distribution of C, O, and sodium in the weight percent is 40.53, 49.32, and 6.35, respectively. The presence of traces 3.8wt% of calcium and chloride elements pointed to an elemental residue amid in SA extract. For SA/Pec/TA-Ag nanocomposite, the percentage of the weight of C, O, Na, and Ag is 18.02, 21.60, 2.73, and 57.65, respectively. The presence of Ag signals exhibited the presence of Ag NPs.
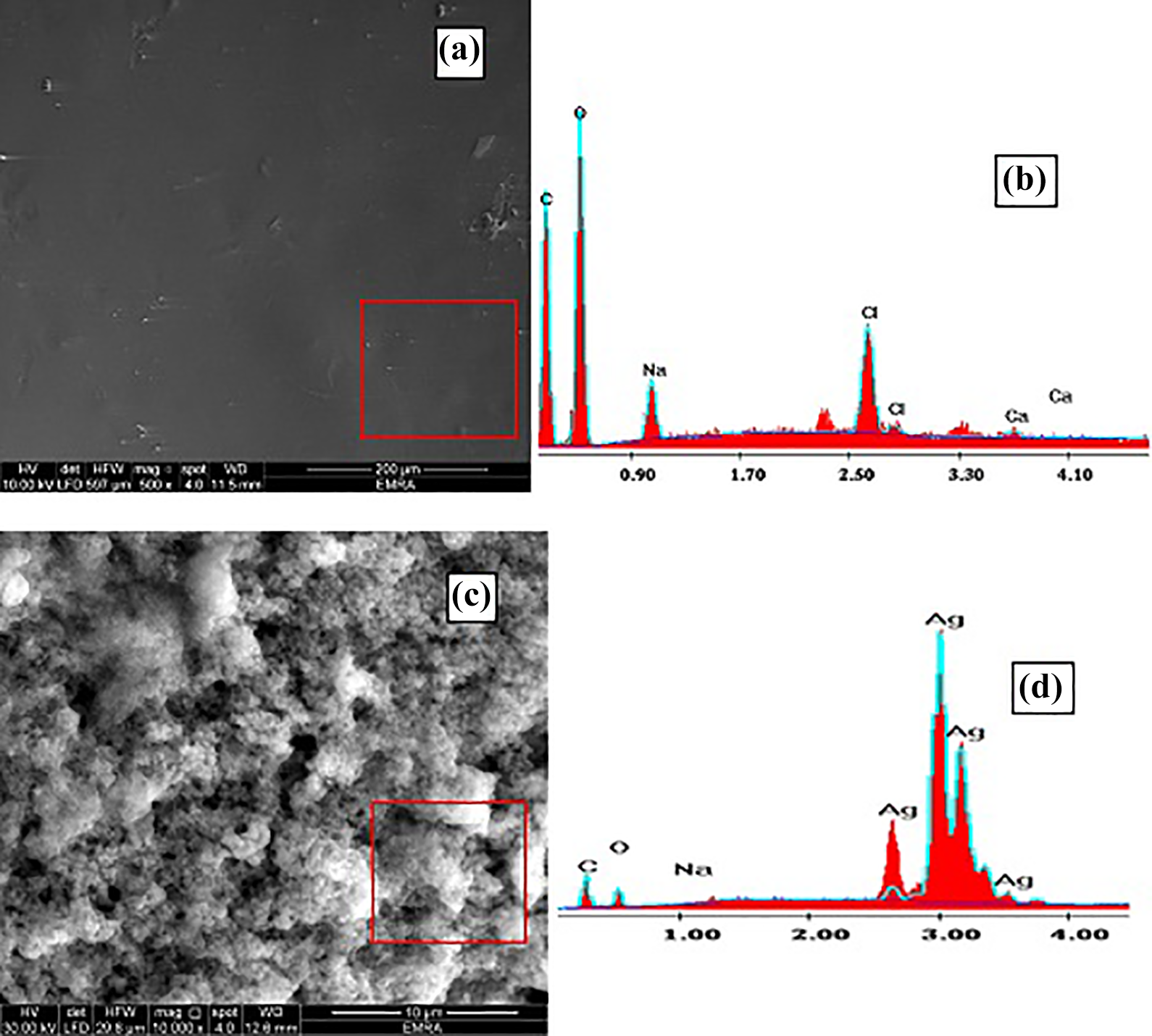
(a) FESEM and (b) EDX of SA/Pec/TA hydrogel. (c) FESEM and (d) EDX of SA/Pec/TA-Ag nanocomposite.
Figure 4 demonstrated the HRTEM images of SA/Pec/TA-Ag nanocomposite. As found in Figure 4, an arbitrary of Ag NPs showed up as dark circles of various particle sizes. DLS measure examination for SA/Pec/TA-Ag nanocomposite showed that the Ag NP readiness and distribution are ordinarily with noteworthy peaks of average size between 21.91 nm and 34.04 nm.

(a to d)HRTEM images of different magnification of SA/Pec/TA-Ag nanocomposite and DLS.
XRD diffractograms of SA/Pec/TA hydrogel and SA/Pec/TA-Ag nanocomposite are shown in Figure 5. The diffractogram of SA/Pec/TA hydrogel demonstrates a wide peak at 2θ of 19.6°, showing the amorphous phase of the hydrogel. The diffractogram of SA/Pec/TA-Ag nanocomposite indicates sharp diffraction peaks at 2θ of 37.03°, 44.46°, 64.62°, and 77.54° corresponding to (111), (200), (220), and (311) planes of the cubic Ag, respectively. 26 These peaks confirmed the presence of Ag in the nanocomposite.

XRD diffractograms of SA/Pec/TA hydrogel and SA/Pec/TA-Ag nanocomposite.
Swelling behavior
The effect of time on the swelling of SA/Pec/TA hydrogel and SA/Pec/TA-Ag nanocomposite at various pH values is shown in Figure 6. It can be seen that SA/Pec/TA hydrogel and SA/Pec/TA-Ag nanocomposite exhibited higher swelling response which increased with time until equilibrium attained within 500 min. The higher swelling response is due to the high hydrophilicity of polymeric chains. The –COOH groups in SA, Pec, and TA perform a significant role in swelling. When carboxylate bunches are protonated, the particles have a tendency to assimilate water to fill the pores in the polymer network, until the point that balance is accomplished.
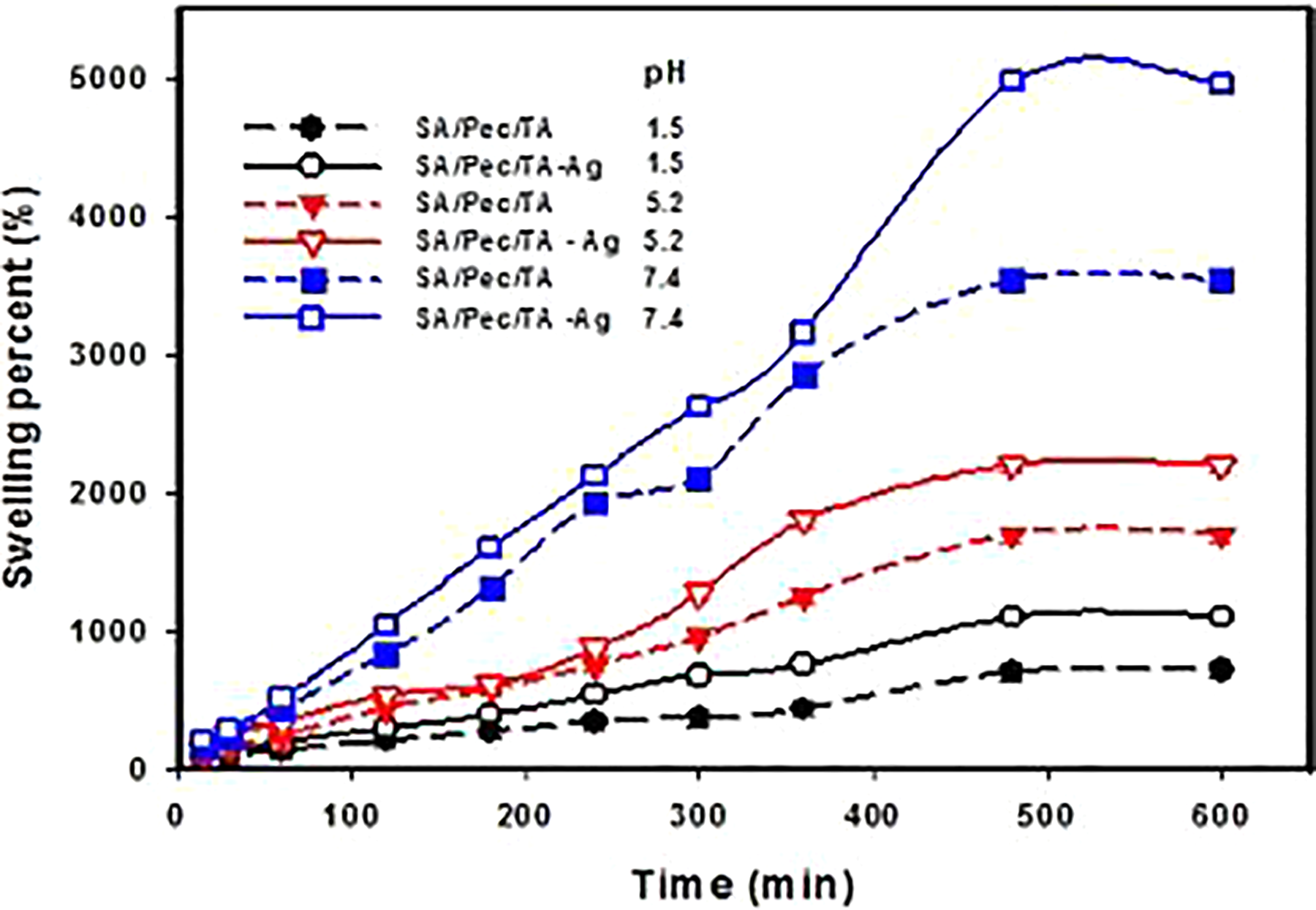
The swelling percent of SA/Pec/TA hydrogel and SA/Pec/TA-Ag nanocomposite as a function of time at pHs 1.5, 5.2, and 7.4.
The pH exceedingly influenced the swelling percent as obtained from Figure 6. The swelling capacity expanded with expanding pH where the lowest pH obtained at 1.5 and the highest one obtained at pH 7.4. At pH 1.5, the carboxylic group is associated and hydrogen bonds formed between the polymeric chains, which reduce the free space in the hydrogel network; therefore, the swelling percent decreases. 27 At pH 7.4, the –COOH groups are dissociated to –COO–, thus a repulsion of the negatively charged carboxylate ions prompts improving the free spacing in the hydrogel whatever it enhances the swelling capacity. It tends to be additionally seen that the swelling percent of SA/Pec/TA-Ag nanocomposite is higher than SA/Pec/TA hydrogel. The nearness of Ag NPs brings down the cross-linking density in the network structure and more free spaces that manage the opportunity for the rapid diffusion of water molecules into the matrix as well as enhances the swelling behavior.
Propranolol drug loading
Impact of propranolol initial concentration on the drug stacking of SA/Pec/TA hydrogel, and hydrogel and SA/Pec/TA-Ag nanocomposite was learned at various pHs (1.5, 5.2, and 7.4) and results are presented in Figure 7. It tends to be noticed that the drug-loaded amount expanded with expanding the drug concentration because the free space accessible destination allows the diffusion of more drug molecules. It must be noticed that the –COOH on SA, Pec, and TA of the backbone are responsive to pH changes. Therefore, the drug loading at pH 7.4 is higher than at pHs 1.5 and 5.2. At pH 1.5, the –COOH are protonated which contracted the polymer chains by formation of hydrogen bonds and they obstruct the arrival of drug molecules into the networks. At pH 7.4, the –COOH are deprotonated. The repulsion of the adversely charged ion among the –COO– anions results the extension of polymer chains. It allows to more drug to enter into the network.
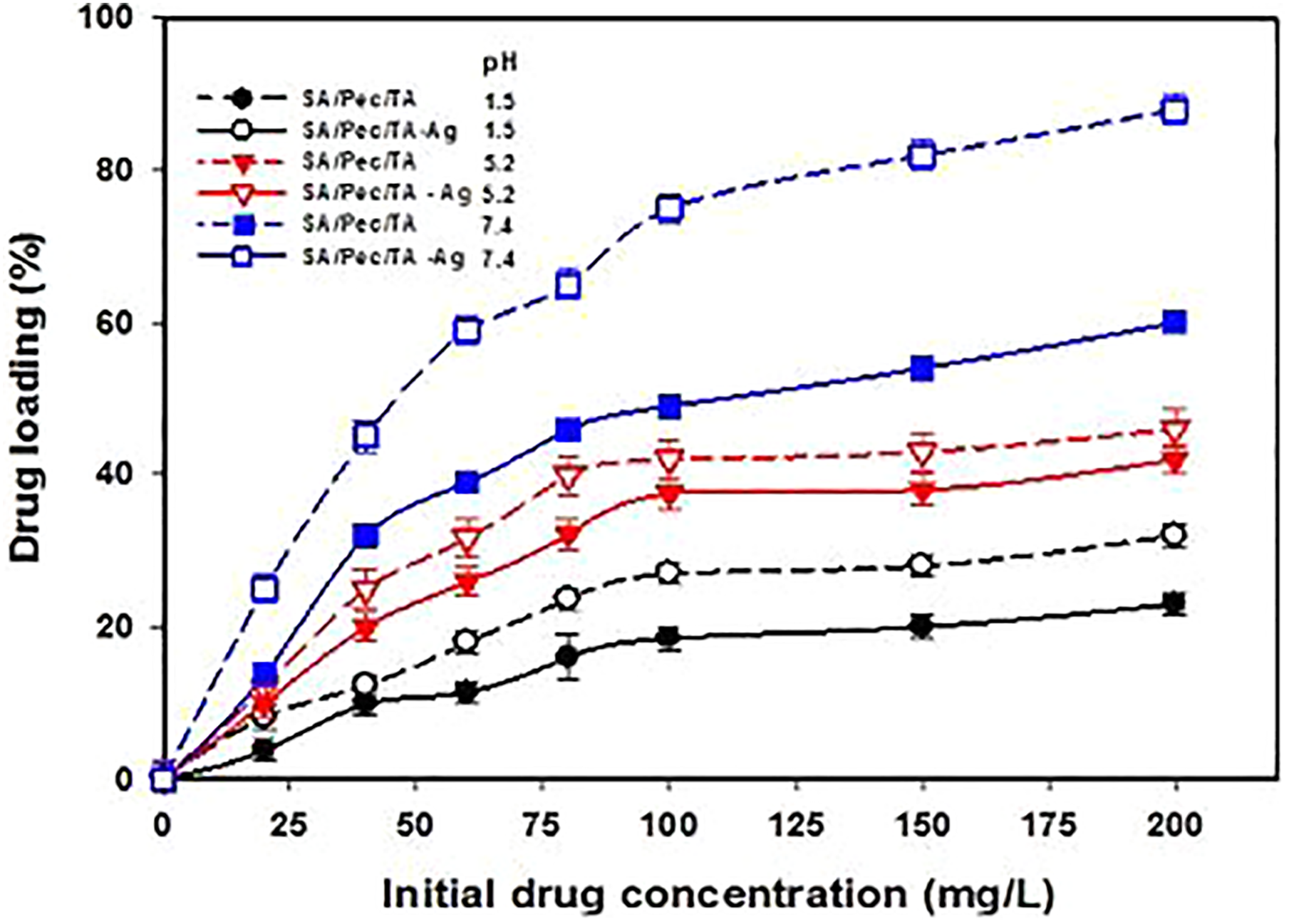
Drug loading of SA/Pec/TA hydrogel and SA/Pec/TA-Ag nanocomposite at different pHs.
In vitro propranolol drug release studies
The cumulative percentage of propranolol release from SA/Pec/TA hydrogel and SA/Pec/TA-Ag nanocomposite as a function of time at 37°C is shown in Figure 8. It is clear that the drug release at pH 7.4 is much higher than that at pH 2.1. The drug release from SA/Pec/TA-Ag nanocomposite is 46% at pH 2.1 and it is 96% at pH 7.4 within 420 min. Drug-releasing behavior relies upon the properties and behavior of matrix stacked this drug. The release of water dissolvable drug from a system happens simply after permeation of water into the matrix, which swells and dissolves the drug trailed by diffusing the drug. 28 In other meaning, the external medium penetrated into the matrix by the osmotic pressure as well as the drug dissolved and released into the medium. Based on the consideration, both SA/Pec/TA hydrogel and SA/Pec/TA-Ag nanocomposite dissociated in basic medium (7.4) and associated in acidic medium (2.1) as discussed in the swelling behavior. So the pH-sensitive hydrogels are effective drug transporters for a specific site where drug release can be controlled by pH changing. On the other hand, it can be observed that the drug release of SA/Pec/TA-Ag nanocomposite is higher than SA/Pec/TA hydrogel. This means the chemical formulation is highly affected the drug release. The presence of Ag NPs in the network structure of SA/Pec/TA-Ag nanocomposite enhances the hydrophilicity as well as the drug release. For all of these, the investigated systems particularly SA/Pec/TA-Ag nanocomposite are candidate for the oral drug carrier. It could keep the drug from destroying by acidic gastric fluid.

Drug release of SA/Pec/TA hydrogel and SA/Pec/TA-Ag nanocomposite at 37°C.
Release kinetics
The learning of release kinetics is fundamental for the efficient utilization of the drug carriers. The information was obtained by in vitro release experimental data at pH 7.4 and was fitted with different observational kinetic models. These models included zero order, first order, Higuchi square root, and Ritger–Peppas models, as shown in Figure 9, and the examined information is abridged in Table 1. By contrasting the correlation coefficients, the R2 of the Higuchi model is higher than the zero-order and first-order models. This implies the kinetic of propranolol releasing from SA/Pec/TA hydrogel and SA/Pec/TA-Ag nanocomposite pursues the Higuchi square root model.

Propranolol release from SA/Pec/TA hydrogel and SA/Pec/TA-Ag nanocomposite fitting curves of different models (a) zero order, (b) first order, (c) Higuchi square root, and (d) Ritger–Peppas models at 37°C and pH 7.4.
Kinetic model parameters of different models for propranolol release from SA/Pec/TA hydrogel and SA/Pec/TA-Ag nanocomposite at 37°C and pH 7.4.

SA: sodium alginate; Pec: pectin; TA: tannic acid; Ag: silver.
For the Ritger–Peppas model, the estimation of n is utilized to describe the release mechanism, if n in the range of 0.43–0.85 demonstrates non-Fickian release where both diffusion and relaxation control the release, if n ≤ 0.43 shows Fickian release where the release is diffusion control, and if n ≥ 0.85 demonstrates case-II transport where the release is relaxation control. Plainly n values for SA/Pec/TA hydrogel and SA/Pec/TA-Ag nanocomposite are in the range of 0.43–0.85. This means that the propranolol release is non-Fickian anomalous transport in which it controlled by both diffusion and relaxation of polymer chains. The release rate of propranolol from SA/Pec/TA-Ag nanocomposite is higher than SA/Pec/TA hydrogel because of expanding the porosity of the lattice as appeared by surface morphology examination and additionally the swelling capacity thus encourages the diffusion of the drug.
Conclusion
A nanocomposite based on SA/Pec/TA incorporated with Ag NPs is synthesized by green method using microwave irradiation. The presence of Ag in the nanocomposite was confirmed with XRD. The surface morphology totally changed from smooth to a gruff surface by joining Ag NPs inside the polymeric structure. HRTEM examination explained an arbitrary dispersion of Ag NPs which showed up as an about circular dark of various particle size average between 21.91 nm and 34.04 nm by DLS. The swelling studies attained that a higher swelling response of the hydrogel and the nanocomposite attributed to the high hydrophilicity of polymeric chains. pH sensitivity was confirmed due to the presence of –COOH group and limited swelling obtained at pH 1.5 and higher swelling is done at pH 7.4. The drug release curve demonstrated that the presence of Ag NPs in the network structure of the nanocomposite enhances the drug release that is 96% at pH 7.4 within 420 min. The drug release was fitted well by Higuchi model and the mechanism is found to be non-Fickian anomalous transport. It can be concluded that SA/Pec/TA-Ag nanocomposite is candidate for the oral drug carrier specific for intestinal system.
Footnotes
Acknowledgements
The authors gratefully acknowledge the Deanship of Scientific Research, Jazan University and also thank for the continuous technical support provided for completing this work.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Deanship of Scientific Research, Jazan University under project no. JUP8//000322.






