Abstract
Terephthaldehyde–acetone polymer was synthesized in an attempt to study its different structural and spectroscopic aspects along with thermal stability. The effect of modification was studied by introducing 4,5-dihydro-1H-pyrazole ring in the polymer chain and silver nano particles (Ag NPs). The structural features of the polymer are supported by scanning electron microscope (SEM), Fourier transform infrared, and X-ray diffraction (XRD) studies. The modification of the polymer is indicated by the appearance of new absorption bands in infrared spectra, due to the formation of dihydropyrazole ring and the interaction of Ag NPs with the main chain of the polymer. XRD revealed the amorphous polymer with an improved degree of crystallinity in the case of nanocomposites. The polymer was found thermally stable up to 210°C, and the stability improved by the addition of 4,5-dihydro-1H-pyrazole (325°C) and Ag NPs (up to 360°C). The polymer looks fibrous in SEM micrographs and a decrease in this character is observed on modification. The optical and fluorescence properties warrant its use in LEDs and as an ultraviolet absorber.
Keywords
Introduction
Terephthaldehyde is an important monomer for the synthesis of conjugated polymers, mostly through condensation reactions, and are known as polyarylidenes. Several polymers containing this monomer include polyarylidene with acetylacetone moiety in the main chain, 1 polyarylidene cycloalkanone, 2 poly(2,6-dibenzylidene)-4-phenylcyclohexanone, 3 and terephthaldehyde–acetone (TA). These polymers are thermally stable and semicrystalline. They exhibit conducting behavior, 4,5 light-emitting properties, 6 and are used as catalyst support. 7 An earlier known polymer of terephthaldehyde is TA, which was first synthesized back in 1885 by Loew, 8 whose structural features, on the basis of oligomers synthesized, were studied by Kaul and Fernandez. 9 The latter study revealed the mechanism of polymer synthesis from the oligomers, whose structures were supported by the spectroscopic techniques. However, there was no study regarding the properties and behavior of the TA polymers and its possible applications.
We synthesized TA polymer and modified it to explore its properties such as crystallinity, morphology, thermal stability, and optical behavior. Thus, a simple study reported by Loew and others is further enriched to find suitable technological applications. The polymer possesses repeated electrophilic centers and nitrogen atoms, which make it environment friendly due to self-degradable nature. The expected improvement in the thermal stability of the polymer on modification is observed. Furthermore, fluorescent properties are also investigated to foresee the energy-storage properties of the polymer and its modified forms. Thus, the study is highlighting the scope of technological work for TA and its modified forms.
Experimental
Materials and methods
The infrared (IR) spectra of terephthaldehyde–acetone polymer (TAP) and its heterocyclic derivative (HCD) were recorded on an IR Affinity-1 Fourier transform infrared (FTIR) spectrophotometer MIRacle 10 (Shimadzu, Japan). X’Pert PRO X-ray diffraction (XRD) spectrometer (UK), was used to determine the amorphous or crystalline nature. Morphology of the synthesized and modified polymer was analyzed by field-emission scanning electron microscopy (TESCAN MIRA3 MU, France), a scanning electron microscope (SEM) coupled with energy-dispersive X-ray (EDX). Optical properties of the TAP and its modified form were studied by employing Shimadzu 1800 ultraviolet–visible (UV-Vis) spectrophotometer and photoluminescence by Shimadzu RF-6000 spectroflourophotometer. TGA Q50 V6 Build 187, USA, was used for thermogravimetric analysis (TGA) under nitrogen atmosphere at a heating rate of 10°C min−1.
Chemicals
For experimental purpose, terephthaldehyde (Merck, Germany) 95%, sodium hydroxide (NaOH, BDH chemicals LTD, UK), silver nitrate (AgNO3; Sigma-Aldrich, Germany), zinc acetate dihydrate (Zn(CH3COO)2·2H2O; Merck, Germany), copper nitrate trihydrate (Cu(NO3)2·3H2O; Merck, Germany), ethylene glycol (EG; 99%, Sigma-Aldrich, Germany), poly(vinylpyrrolidone) Sigma-Aldrich, Germany, hydrazine monohydrate (80%, Daejung, Korea), acetone, ethanol, and methanol (analytical reagent grade, Lab Scan, Spain) were used. All these chemicals were of high purity grade and used without any purification.
Synthesis of TAP
Methanol (15 mL), NaOH (10%, 0.006 mol), and acetone (0.25 mL, 0.003 mol) were stirred at room temperature for 20 min. Then, terephthaldehyde (0.4 g, 0.003 mol) was added into the reaction mixture, while stirring. After the addition of terephthaldehyde, the color of the reaction mixture turned yellow within a few minutes. The stirring was continued for about 3 h. The precipitation during reaction indicated the formation of polymer. After the completion, the reaction mixture was poured into ice-cold water, neutralized, and washed with water and ethanol, repeatedly. The resulting precipitates were dried under vacuum overnight.
Modification of TAP with hydrazine
TAP (0.5 g, 0.003 mol) was added to methanol (15 mL) with stirring. Hydrazine monohydrate (0.009 mol, 3 eq.) was added to the stirring reaction mixture. The reaction mixture was heated under reflux for about 5 h. The reaction mixture was poured into ice-cold water, neutralized, and washed thoroughly with water and ethanol. There was a visible change in the color of the polymer from yellow to orange.
Modification of TAP with Ag NPs
AgNO3 solutions (0.2%, 0.6%, and 1%) were prepared in EG (5 mL) and heated till the appearance of yellow color. During polymerization, the AgNO3 solution was added dropwise into the reaction mixture. The yellow color of the reaction mixture turned green. After complete addition of AgNO3 solution, the reaction mixture was sonicated for about 2 h to ensure the homogeneous dispersion of silver nanoparticles (Ag NPs) on the polymer surface. The green precipitates obtained after 8 h were washed with distilled water and ethanol. The modified polymer was dried at room temperature. The optimized method by Ahmad et al. is applied. 10
Results and discussion
The polymer was synthesized by polycondensation method and modified by reaction with hydrazine leading to the formation of 4,5-dihydro-1H-pyrazole in the polymer backbone (Figure 1). The modification of the polymer by in situ method using Ag NPs was also performed. The synthesis and modification of polymer were initially established by its physical properties (Table 1).

Synthesis of TAP and its HCD.
Physical data of TAP.

TAP: terephthaldehyde–acetone polymer; HCD: heterocyclic derivative; Ag: silver.
Morphology of TAP and modified forms
The synthesis of the polymer was indicated by the precipitation of yellow solid during the reaction and was established by microscopic and spectroscopic techniques. The morphology of the polymer as observed by SEM indicates the fibrous nature of TAP (Figure 2(a)) which is possibly due to π–π stacking. However, a clear difference in the morphology can be seen in the SEM image (Figure 2(b)) upon modification of the TAP, as the introduction of dihydropyrazole leads to saturation in the main chain and reducing the π–π stacking and hence the corresponding fibrous nature. The SEM images for Ag-doped TAP also showed a similar character (Figure 2(c)).

SEM images of (a) TAP, (b) HCD, and (c) TAP/Ag nanocomposites.
The introduction of Ag NPs decreased the fibrous nature of TAP. The morphology of Ag-doped TAP in Figure 2(c) illustrates that Ag NPs are uniformly distributed in the polymer matrix. SEM image shows good dispersion of Ag NPs which may be due to the rolling of TAP fibers around NPs. The presence of Ag NPs and dihydropyrazole nucleus in the main chain of the polymer is further indicated by the EDX (Table 2). The peak for silver and nitrogen in the EDX spectrum in addition to carbon and oxygen confirmed the presence of nitrogen in the modified polymer. There are also peaks for Si and oxygen which are due to the glass substrate used for sample preparation.
EDX data of doped TAP.
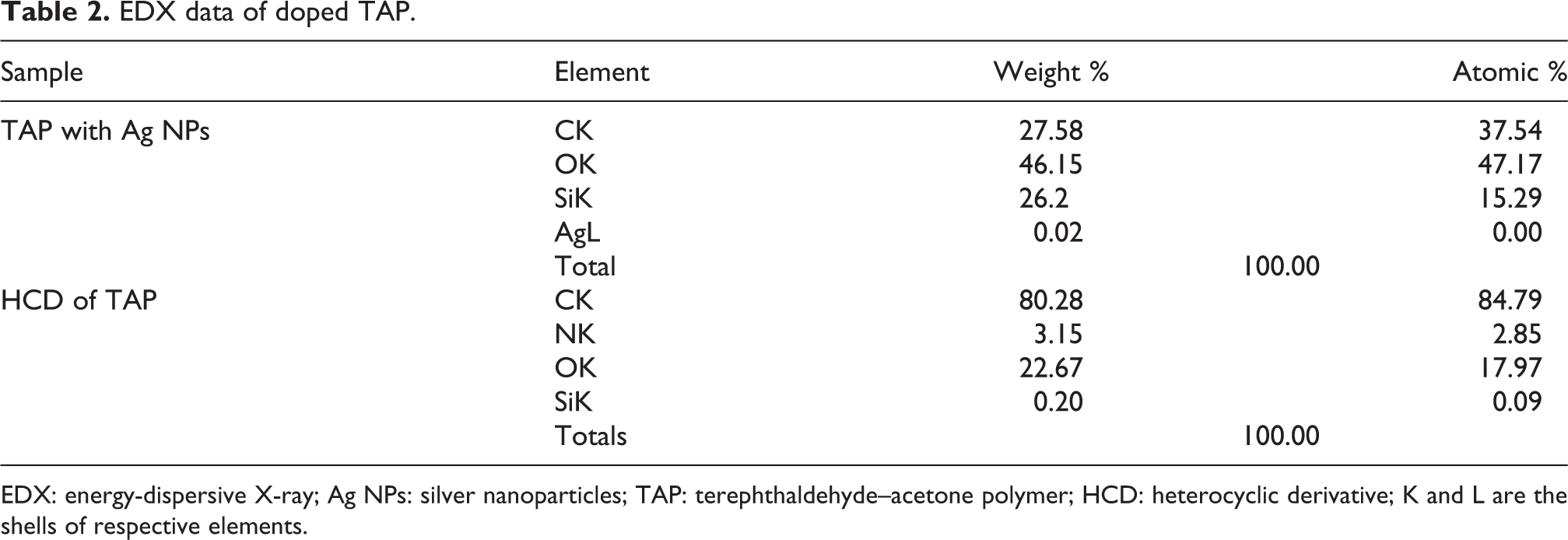
EDX: energy-dispersive X-ray; Ag NPs: silver nanoparticles; TAP: terephthaldehyde–acetone polymer; HCD: heterocyclic derivative; K and L are the shells of respective elements.
FTIR characterization
The functional groups in the polymer were identified by FTIR spectrum shown in Figure 3. The spectrum depicts the appearance of absorption band at 1624 cm−1 for C=O, and the shifting of C=O stretching to lower waver number is due to the conjugation of the carbonyl with arylidene moiety. The bands in the regions of 1600 cm−1 and 1520 cm−1 are due to the olefinic and aromatic C=C stretching, respectively. 11 The reaction of polymer with hydrazine leads to the formation of 4,5-dihydro-1H-pyrazole ring and is indicated by the shifting of absorptions in the FTIR spectrum. Besides the disappearance of C=O stretching, new absorption band appeared at 2860 cm−1 which is attributed to the aliphatic C–H stretching frequency, and this partial saturation is generated as a result of the heterocyclic ring formation. Another important absorption at 1641 cm−1 was assigned to C=N of the pyrazole ring and is in accordance with the reported C=N stretching for the dihydropyrazole ring. 12 The spectra of both polymers are shown in Figure 3 for a comparison, where a clear difference can be seen in going from TAP to its HCD.
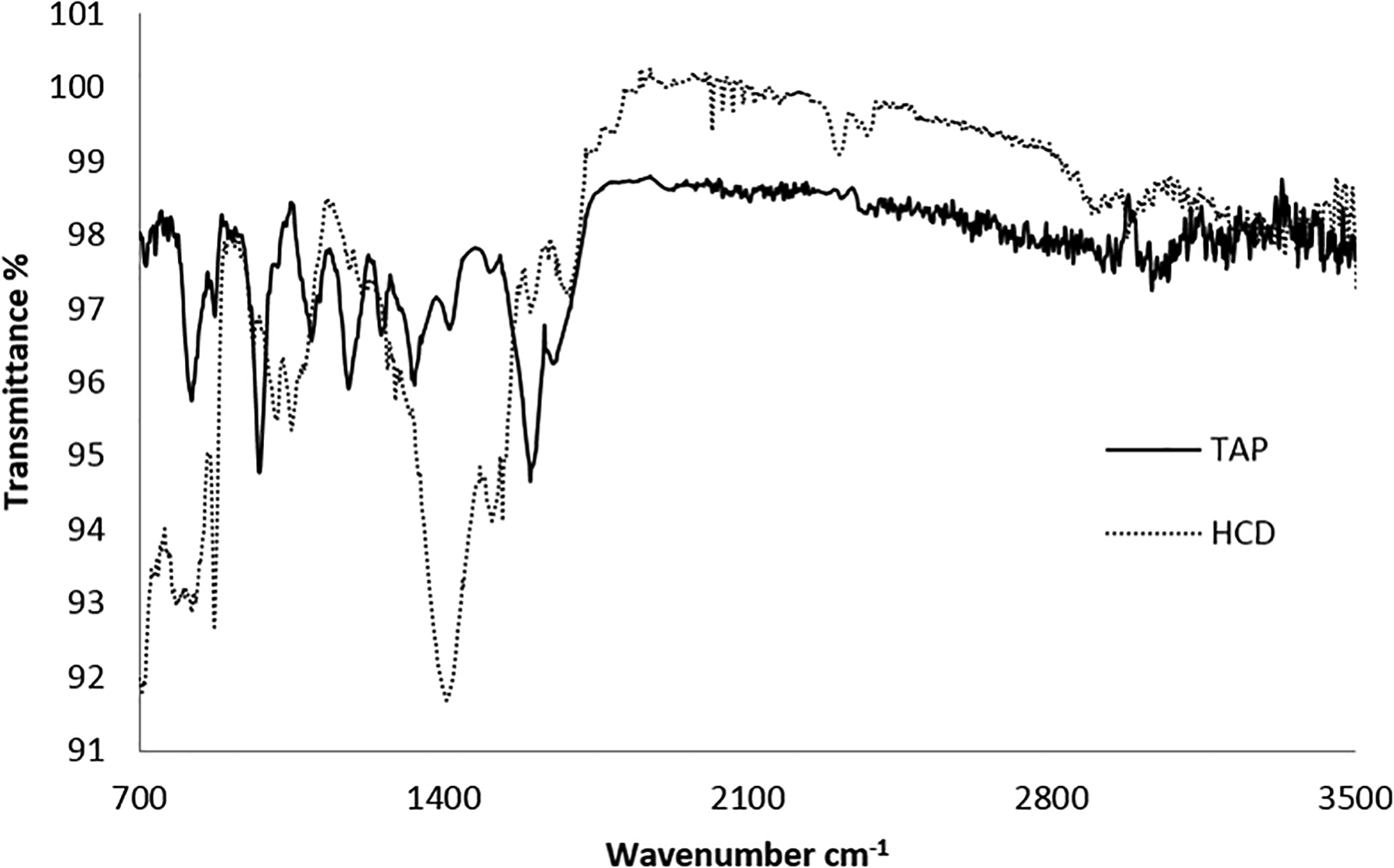
FTIR spectra of TAP and HCD.
The nature of the polymer whether amorphous or crystalline was studied by XRD analysis. Figure 4 shows the XRD pattern in which a broad diffraction peak centered on about 19.62°, ranging from 10° to 26° (2θ), depicting the amorphous structure of TAP. The peak height is reduced in case of modification, with a slight shift in peak (18.89°) is also observed. The appearance of the sharp peak at 2θ = 29. 38° indicates the semicrystalline nature of TAP. The appearance of diffraction peaks at 2θ = 29.59–47.69° in the modified polymer shows that the degree of crystallinity increases in the modified polymer as compared to TAP. This increase is attributed to the presence of C=N and C–N as polar groups and NH is also present which arises the possibility of hydrogen bonding. The effect of Ag NPs doping on the structure of TAP was also studied. XRD pattern for Ag-doped TAP by the in situ reduction method is shown in Figure 4(b). The change in the pattern as compared to that of pure TAP indicates Ag/TAP composite formation. The broad peak valley at 2θ = 19.62° for TAP decreases, which indicates an increase in the degree of crystallinity of TAP after doping.

XRD of (a) TAP, HCD, and (b) TAP nanocomposites.
Four diffraction peaks at 2θ = 37.46°, 41.9°, 64.48°, and 75.58° resemble the silver planes (111), (200), (220), and (311), respectively. The strong peak at 2θ = 29.38° for TAP also reduces which indicates the incorporation of Ag NPs in the polymer matrix. All the peaks are well indexed for the face-centered cubic structure of Ag NPs (JCPDS card no. 011164). 13,14 The observation of XRD depicts a possible change in the morphology of the polymer matrix.
Optical and fluorescent properties
The polymer was further investigated by the absorption spectroscopy. The UV-Vis spectra are shown in Figure 5, recorded in ethanol (suspension by sonication) at room temperature. A broad absorption band at 366 nm is due to the π–π* transitions of benzylidene ketone which is blueshifted (300 nm) by the incorporation of pyrazole nucleus, as the conjugation is reduced in the modified polymer.
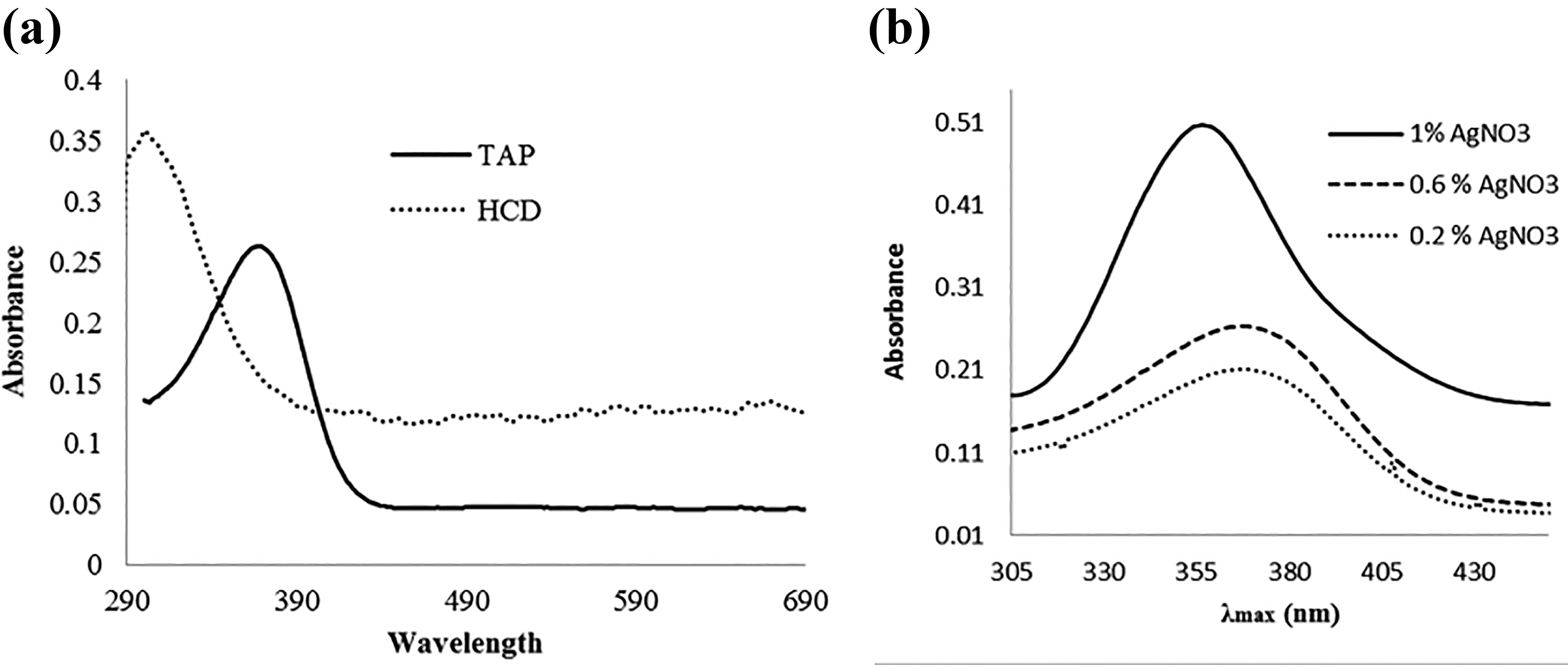
UV-Vis spectra of (a) TAP, HCD, and (b) TAP nanocomposites.
A blueshift in the UV-Vis spectrum of Ag-doped TAP by in situ reduction method was observed in the range 360–350 nm. This change in shift is attributed to a decrease in the resonance within the polymer chain due to the presence of Ag+1 ions which may interact with π electrons of TAP. By increasing the concentration of AgNO3 (0.2–1%) solution, blueshift increases. The bandgap of TAP and its modified form is 3.39 and 4.00 eV, respectively. Bandgap indicates that TAP doped with Ag NPs by in situ reduction method may act as an insulator.
The polymer was also investigated for its expected fluorescence properties. TAP and its modified form were excited at their respective λmax obtained in the UV-Vis spectroscopy. The optical excitation of TAP gives a strong orange–red fluorescent peak at 680 nm (Figure 6(a)), which is blueshifted to 600 nm in the modified polymer containing dihydropyrazole in the main chain of polymer attributed to a decrease in conjugation. The broad emission peaks centered on approximately 685 nm for Ag-doped TAP are shown in Figure 6(b). There is an increase in peak intensity in the range of 680–685 nm. The intensity of emission peaks increases by increasing the concentration of AgNO3 solution (0.2–1%).

PL spectra of (a) TAP, HCD, and (b) TAP nanocomposites.
Thermal studies of TAP and its modified forms
Besides the fluorescent properties, the polymer was also studied for its thermal stability. TGA was carried out at a rate of 10°C min−1 under nitrogen atmosphere. The TGA curves in Figure 7 exhibit that TAP is stable up to 210°C, while modification of the polymer by dihydropyrazole nucleus increased its stability up to 325°C. The thermogram for TAP shows that the degradation of polymer occurs in two steps. The first step is rapid and involves 32% weight loss up to 400°C. The expected degradation of polymer is –CO–C=C– linkage, and the expected loss is carbon monoxide. In the second step, a gradual weight loss of another 26% up to 565°C might be due to cleavage of alkenyl–phenyl bond, with possible losses being ethylene and acetylene. The thermograph of the modified polymer shows the initial weight loss of 1–5% at 10–50°C, which is possibly the entrapped solvent or water molecules hydrogen bonded to the dihydropyrazole nucleus. From 325°C to 800°C, the degradation leads to possible losses of small molecule like nitrogen, ethylene, and acetylene. The relative increase in stability is attributed to the dihydropyrazole nucleus and its ability to have interchain hydrogen bonding, which leads to stabilization.

TGA curve of TAP and HCD.
The effect of Ag NPs on the thermal stability of TAP was also studied. The TGA curves in Figure 8 indicate that composite-based modified TAP (0.2–1% AgNO3) has gained more thermal stability. The stability of Ag-doped TAP increases by increasing the concentration of AgNO3 solution (0.2–1%). The initial weight loss in each case is possibly due to the entrapped solvent.

TGA curves of TAP nanocomposites.
The greater stability is attributed to the change in the morphology of the polymer upon doping as indicated by SEM. The decrease in the fibrous character is a possible reason for the increase in thermal stability due to the rolling of polymer fibers on the Ag NPs.
Conclusions
TAP and its modification were carried out in excellent yields. TAP was found fibrous by SEM, the fibrous nature was reduced by introducing a dihydropyrazole ring in the main chain and by doping with Ag NPs. XRD showed the semicrystalline nature of TAP which was improved by modification. The thermal stability of TAP was enhanced by HCD and Ag NPs. The fluorescence spectra of these polymers exhibit emission in the orange–red region. These results show that the polymers can be employed in the solar cell as well as in organic light emitting diodes (OLEDs). The polymers may be employed in UV absorber devices and in sunscreen lotions.
Footnotes
Acknowledgements
The authors are grateful to Dr M Rafique, GM, PMO, NESCOM Pakistan, for the thermal analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
