Abstract
Antistatic packagings are very important for proper transportation and storage of electronic components since these components can be damaged with electric overloads generated through rubbing. Therefore, the purpose of this study was to develop an innovative material for antistatic packaging with a sustainable character. For this, a blend of recycled poly(ethylene terephthalate) (PET), polycarbonate (PC), and polyaniline (PAni) was prepared by melt extrusion. Samples have a final content of 68 wt% of PET, 29 wt% of PC, and 3 wt% of PAni doped with cobalt (PAni-Co). The properties were determined by differential scanning calorimetry, thermogravimetric analysis, tensile testing, electrical resistivity analysis, and Fourier transform infrared spectroscopy. To the best of our knowledge, no previous work studied the incorporation of PAni-Co in PET/PC blend.
Keywords
Introduction
Plastic packagings are widely used to preserve the quality and integrity of different products. Electronic components are commonly transported and stored inside thermoplastic packagings, which can undergo electric overloads when rubbed during transportation. 1,2 Therefore, the use of antistatic packagings in these cases is very important, since electrostatic discharge can damage semiconductor devices and electronic components, causing total failure, reduction of life expectancy, or malfunction. 3
In the present work, an innovative material for antistatic packaging through the combination of recycled poly(ethylene terephthalate) (PET) and polycarbonate (PC) was developed. PET is a semicrystalline thermoplastic polyester widely used in several applications due to its low relative cost, good mechanical properties, and easy processing. 4 -6 Nonetheless, PET is one of the most consumed plastics, and it is used in numerous applications with short shelf life, such as disposable bottles and other packaging, generating a large volume of waste that is often inappropriately disposed in the environment. 7 Therefore, considering the importance of looking for applications that stimulate the recycling of the PET residue, the present study was developed using recycled PET water bottles. PC was added to the recycled PET matrix to improve the thermal stability and the impact resistance of the final product without compromising transparency. 8,9
For application as antistatic packaging, it is necessary to convert the insulating thermoplastic material formed by the PET/PC blend into a product that can dissipate the static charge with a resistivity range of 108–1010 Ω·m. 10 This property can be reached with the use of polyaniline (PAni). 11,12 Nevertheless, to reach the state of conduction of PAni, it is necessary to dope it, what was done using a cobalt complex, forming PAni-Co, as presented in the sequence. 13
In this study, a blend of PET/PC with PAni-Co was processed by single-screw extrusion and characterized aiming applications as antistatic packaging. To the best of our knowledge, no previous work studied the incorporation of PAni-Co in a PET/PC blend.
Materials and methods
Polyaniline synthesis and doping
PAni was synthesized by chemical oxidation of aniline (Vetec, p.a. Sigma-Aldrich, Brazil), as shown in Figure 1, using ammonium persulfate (Alphatec, p.a., Brazil) as the oxidant. For this, 40 mL of aniline was dissolved in 600 mL of a solution of hydrochloric acid (HCl) in water (1.0 mol L−1) (Alphatec, p.a.). Concomitantly, 23.00 g of ammonium persulfate was dissolved in 400 mL of a solution of HCl in water (1.0 mol L−1). The aniline and the ammonium persulphate were dissolved in the separate acid solutions, under constant stirring, for 2 h, at approximately 0°C. In sequence, these solutions were mixed for two more hours at 0°C. The reaction medium underwent changes of coloring, going through purple and blue until it reaches the dark green tone. The resulting emeraldine base polyaniline (EB-PAni) precipitate was collected on a filter and washed with water and acetone solution (5: 2) under vacuum.

Scheme of synthesis of PAni-Co using chemical oxidation method.
Although synthesized EB-PAni has already been doped to a conductive state, it would not be possible to use it in an extrusion and injection methods because of its acidic character that can damage the equipments. Thus, a 1.0-mol L−1 aqueous solution of ammonium hydroxide (Alphatec, p.a.) was carried out under stirring for 24 h, for dedoping the polymer. It was then filtered and dried under vacuum for about 72 h. Subsequently, the doping process was carried out with cobalt for the achievement of a nonacid state of conductive PAni. For this, PAni was suspended in a 0.05-mol L−1 cobalt chloride (Synth, 98%) solution in acetonitrile (Vetec, p.a.) for 48 h. Then, it was filtered, washed, and dried under vacuum for 24 h at 80°C resulting in PAni-Co.
Samples preparation
Waste PET bottles were collected, washed, dried, and cut in a propeller mill. Polymer blends were prepared in a single-screw extruder from Seibt model ES25. First of all, it was prepared a blend of 70 wt% of recycled PET and 30 wt% of virgin PC, from Lexan LS2111, through single-screw extrusion. The pellets of this blend were then manually mixed with 3 wt% of dried PAni-Co powder and fed in the extruder again. The temperature profile used in extrusion, from the feed zone to the matrix, was 230°C, 235°C, 240°C, and 245°C. The final material was submitted to injection molding using a temperature profile of 225°C, 230°C, 235°C and a mold temperature of 40°C. Dog-bone samples were injection molded for tensile tests (type IV of ASTM-638). Samples of PET/PC blend (70:30) without PAni were also processed through the same route for comparison. Figure 2 illustrates the processes performed to produce the specimens.

Characterization
The thermal stabilities of the extruded blends were studied by thermogravimetric analysis (TGA) in a DTG-60 from Shimadzu, Brazil, from 25°C to 750°C with a heating rate of 10°C min−1 in air atmosphere. Differential scanning calorimetry (DSC) of the extruded blends was performed in a DSC 8000 from Perkin-Elmer, Brazil, under nitrogen atmosphere, with N2 flow of 20 mL min−1. All samples were submitted to a first heating (from 40°C to 300°C), followed by an isotherm at 300°C for 2 min, and cooling from 300°C to 20°C, using heating and cooling rates of 20°C min−1. The crystallinities (%C) from the blends were calculated using equation (1), considering the melting enthalpy of PET 100% crystalline (ΔH PET100%) as 140 J g−1 and the real PET content in the blend 16 :
Fourier transform infrared (FTIR) spectroscopy was performed using a Perkin-Elmer 2000 equipment with attenuated total reflectance method (ATR), with a resolution of 4 cm−1, 32 scans, and wave number range of 400–4000 cm−1. Tensile properties were measured in an Instron EMIC 23-100 machine, Brazil, according to ASTM D-638. The electrical conductivities (σ) of PAni-Co, PET/PC, and PET/PC/PAni-Co were measured, at room temperature, using a megohmmeter from Megabras, model MD 5060, Brazil. For this, the samples of each composition were cut into a cylindrical volume of 1.5 cm3. The resistivity (R) was calculated from the electrical conductivity according to equation (2), where L is the length and A is the area:
Results and discussion
Thermal properties
Figure 3 shows the heating and cooling DSC curves of samples. From the heating curve of the reference PET/PC blend, it was possible to observe the melting temperature of the recycled PET (peak temperature = 237.6°C) and also to calculate the crystallinity resulting from the PET presence (%C = 16.9%). Moreover, it was possible to observe one glass transition (T g) around 91.3°C. The typical T g from PC, an amorphous polymer, is around 140°C, while the T g expected for PET is around 70°C. 17 The fact that we observed only one T g in the DSC curve, between the expected values of both polymers from the blend, can be an indicative of the miscibility between PET and PC, as commented by Wang et al. 18 This miscibility may have been favored by transesterification reactions between PET and PC, induced through the extrusion process, as commented by Garcia et al. 9 A crystallization temperature of PET was not observed in the cooling curve of the reference blend, probably because of the fast cooling rate used in DSC (20°C min−1).

DSC curves: PET/PC/PAni-Co, cooling (a) and heating (b); PET/PC, cooling (c) and heating (d).
For the PET/PC/PAni-Co blend, the DSC heating curve showed the PET melting temperature of 255°C, related to a crystallinity degree of 22.6%. In the cooling curve, there was an appearance of an exothermic peak at 150°C, indicating the crystallization of PET. Therefore, the presence of PAni-Co in the blend increased the melting temperature and the crystallinity of the blend (heating curve), and also favored the PET crystallization during the cooling process. These results indicate that PAni-Co acted as a nucleating agent of PET crystallization.
In the heating curve of PET/PC/PAni-Co blend (Figure 3(b)), it was also possible to observe an exothermic peak with a maximum around 120°C. This peak could be associated with a recrystallization or cross-linking reaction of PAni-Co, as verified by Mendes et al. 19 since there was no corresponding weight loss at this stage according to the TGA curve (Figure 4).
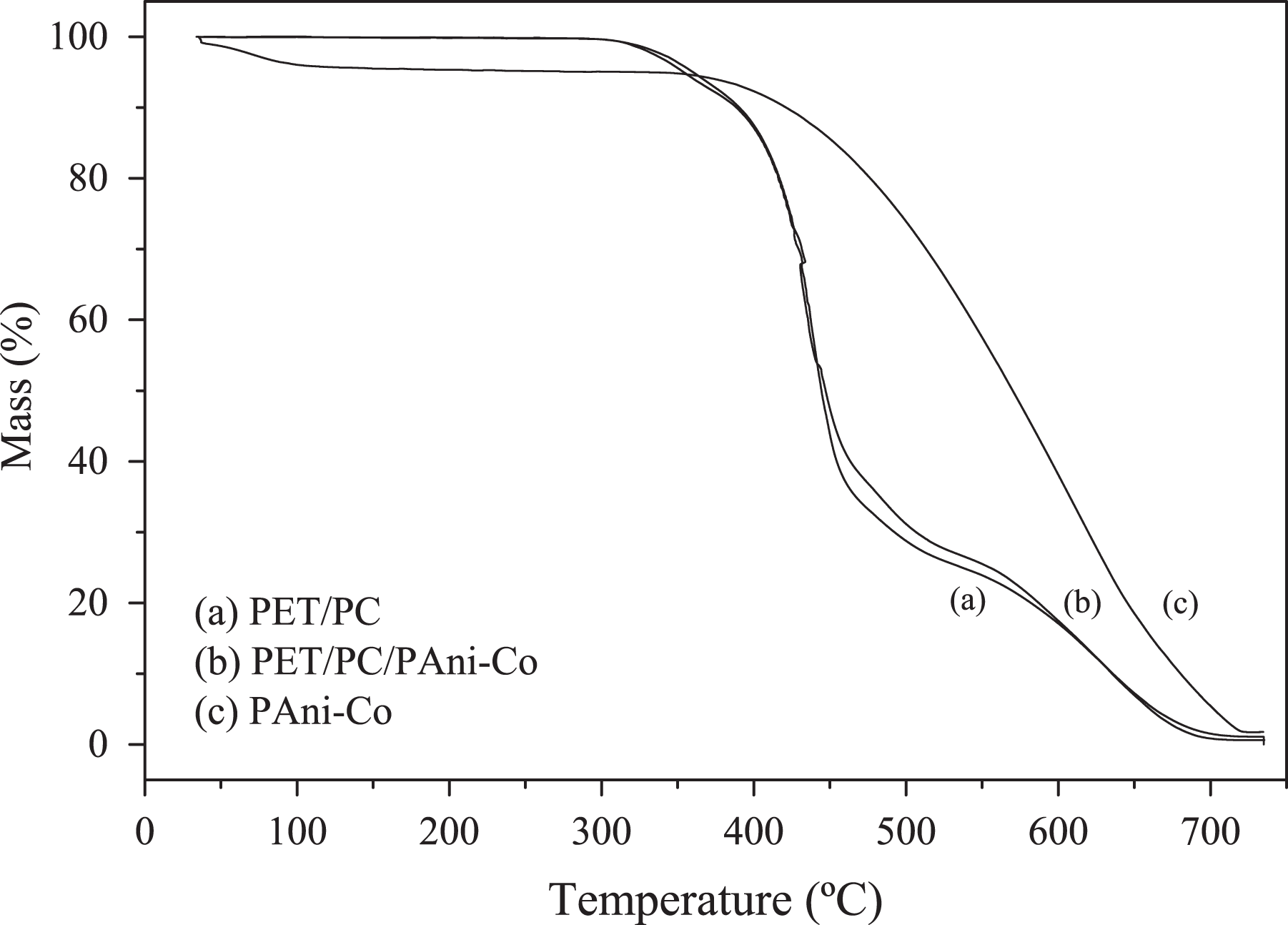
TGA curves: PET/PC (a), PET/PC/PAni-Co (b), and PAni-Co (c).
The TGA curves of the blends (Figure 4) showed three distinct events, which are related to the phases present. The first mass loss with onset temperature around 300°C was attributed to the degradation of PC homopolymer, while the second mass loss, more intense, represents the thermal degradation of the PET, with onset at 375°C. 20 The mass loss around 500°C is correlated to the carbonization of ashes because the analysis was performed in air atmosphere. The results indicated that the addition of PAni-Co did not alter the thermal stability of the PET/PC blend.
FTIR
In this work, the FTIR was used to verify the transesterification reactions of the PET/PC/PAni-Co and PET/PC blends as well as the doping of the PAni (Figure 5). According to Wang et al., 21 if transesterification reactions occur during melt processing of PET/PC blends, absorption bands at 1070 and 1720 cm−1 should be observed, which can be attributed to characteristic peaks of ester aromatic groups present in the copolymer formed. By analyzing the spectrum obtained for PET/PC blends, it was only observed the presence of the characteristic absorption bands of PET and PC homopolymers. Even evaluating the magnification of the region between 1500 cm−1 and 1000 cm−1 (Figure 5(b)), it is difficult to identify new peaks, different from the original homopolymers peaks, that could confirm the occurrence of transesterification reactions and the formation of the copolymer. However, this no-observation of the transesterification peaks can be a consequence of an overlapping effect due to the proximity of the wavenumbers related to these peaks and the ones from homopolymers.
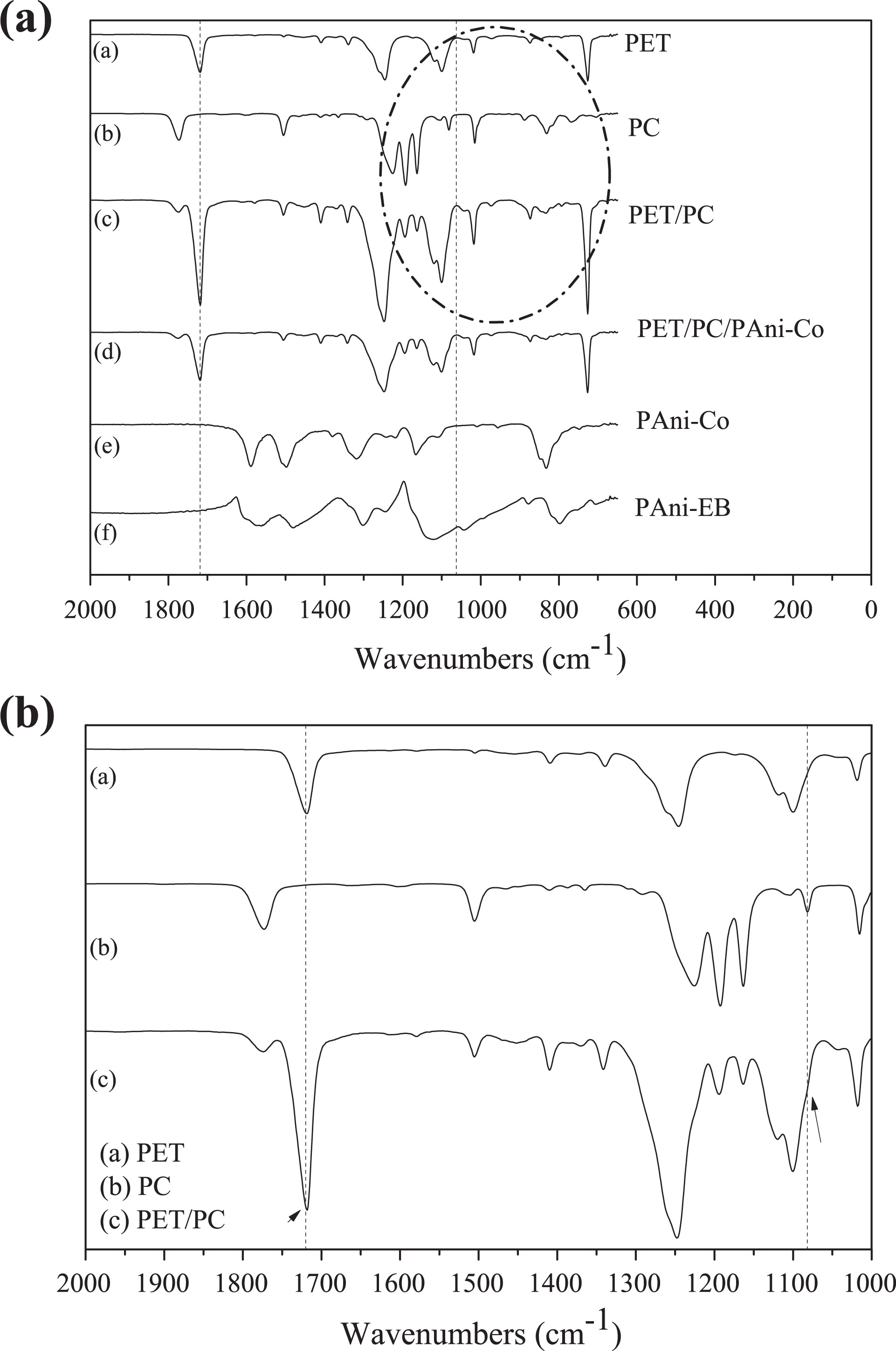
(a) IR spectra of samples: PET (a), PC, (b) PET/PC, (c) PET/PC/PAni-Co (d), PAni-Co (e), PAni-EB (f); (b) Magnification of region between 1500 cm−1 and 1000 cm−1.
The doping of the PAni-EB with the cobalt complex can be proved through the presence of the band at 1137 cm−1 in the curve of PAni-Co, characteristic of the Co-doped polymer, according to Hasik et al. 13
Mechanical characterization
The PET/PC/PAni-Co injected samples were submitted to tensile tests. It was observed that the material obtained presents high fragility since it presented an average tensile strength of 6.6 ± 1.6 MPa. These values are much lower than the expected ones for the pure PET (between 48.3 and 72.4 MPa 22 ) and the pure PC (around 63.8 MPa 17 ). This can be associated with the addition of PAni in the blend and its intrinsic behavior of low mechanical properties, besides the low miscibility of the single-screw extruder. 10,11 In previous studies, 23 PET/PAni blends also resulted in hard and brittle material due to the hydrolysis of PET ester bonds induced by the PAni acid dopants. PAni needs to be doped for conducting state and, for this, functionalized protonic acids are usually used, as dodecylbenzene sulfonic acid, forming a complex with the polymer and at the same time promoting the doping of PAni. The use of these acids allows the processing of PAni with good mechanical properties. 24 However, in this work, an alternative doping route with cobalt was used, following the procedure of Hasik et al. 13 According to Mendes and Pereira, 25 cobalt can promote the insertion of PC in PET, contribute to miscibility, but high contents can cause degradative effect on the properties of the material and cause shear effect of the homopolymer chains if they prevail over the exchange reactions.
Nevertheless, the elastic modulus of samples was not affected by the PAni addition. The value obtained for PET/PC/PAni-Co, of 2.7 ± 0.4 MPa, is within the expected range of 2.76–4.14 MPa for the pure PET and 2.38 MPa for the pure PC. 22
Electrical properties
According to Martins and de Paoli, 10 an antistatic material has the ability to dissipate energy and exhibits resistivity in the range of 108–1010 Ω·m. By means of Table 1, it was possible to prove the antistatic character of the PET/PEC/PAni-Co blend, showing that the proposed objective was reached and this blend can be used to protect electronic components.
Resistivity values of the samples of PET/PC, PET/PC/PAni-Co, and PAni-Co.
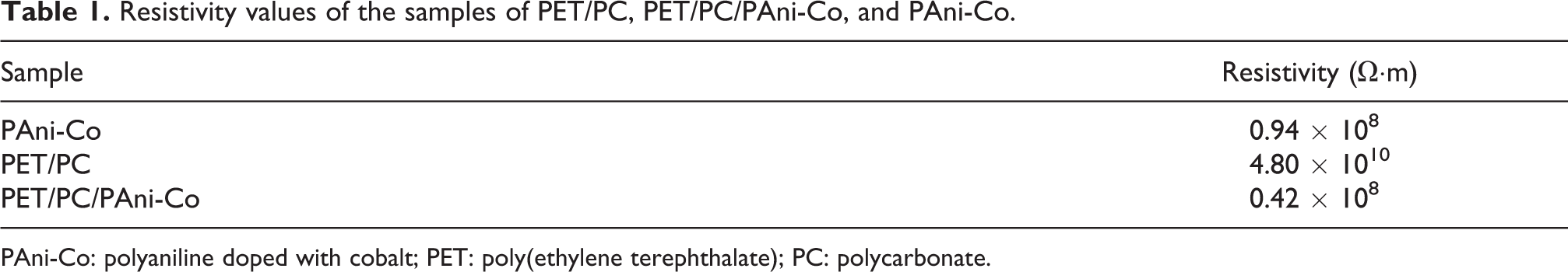
PAni-Co: polyaniline doped with cobalt; PET: poly(ethylene terephthalate); PC: polycarbonate.
Conclusions
In this article, a blend of recycled PET, PC, and PAni was prepared by melt extrusion. The PET/PC/PAni-Co blend presented low tensile strength attributed mainly to PAni-Co and its doping route. Nonetheless, the elastic modulus and the thermal stability were not impaired by PAni addition. PAni-Co acted as a nucleation agent of PET crystallization. The electrical properties confirmed the obtainment of a material for antistatic applications, being an alternative for the reuse of waste PET bottles.
Footnotes
Acknowledgement
Our sincere thanks to the Empresa de Desenvolvimento de Itabira Ltda (Itaurb) for donating the waste PET bottles.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.


