Abstract
To investigate the potential of biomaterials for bone repair, Ginkgo biloba (GB)-loaded polylactic-co-glycolic acid microspheres were synthesized via solvent evaporation technique, and scanning electron microscopic (SEM) micrographs indicated a spherical and smooth surface in which the particles were distributed in the range of 1–7 µm. Chitosan–hydroxyapatite (HA) matrixes incorporated with polymeric spheres were fabricated using the freeze-drying method. SEM micrographs demonstrated an interconnected porous structure in which porosity and pore size reduced as a function of HA and microsphere addition. Fourier transform infrared spectrum illustrated the ability of sodium tripolyphosphate in cross-linking of composite scaffolds. Swelling ratio presented that higher amount of microspheres and the addition of HA reduce the ability of interaction with water molecules. The GB release behavior of hybrid scaffolds confirmed that the addition of HA significantly reduces release ratio because of a slight reduction in pore size and PBS absorption capacity, but higher amount of microspheres resulted in an increase in release level. According to the results, the synthesized hybrid scaffolds have the initial physicochemical features for further studies in terms of in vitro and in vivo assays.
Introduction
Tissue engineering is a powerful tool to treat defects, which is a complex of materials science, cellular biology, and clinical research, and provides an opportunity to regenerate injuries by mimicking the extracellular matrix (ECM). Synthetic scaffolds are required for 3-D tissue growth and reduce the risk of immune rejection, virus transfer, and solve the problem of limited resources. 1 A variety of materials depending on the application have been used to fabricate a scaffold. Bioceramics have been extensively used in hard tissue applications due to no releasing component into the human body, no foreign body reaction, compatibility of resorbable components with natural metabolism, and so on. 2 But brittleness (resulted in limitation for load-bearing applications) and high stiffness (led to the restriction of nonosseous applications) reduced the application of pure ceramics. 3 On the other hand, in hard tissue engineering applications, polymeric samples cannot supply the requirements in terms of mechanical properties and osteoconductivity. 4 Thus, fabrication of a composite structure can be a promising approach to provide the needs. 5
The bone structure is composed of a mineral phase (hydroxyapatite (HA)) and an organic phase (collagen). Therefore, fabrication of a composite scaffold with similar materials can be effective strategy due to a high degree of resemblance ECM. 6,7 Many other studies confirmed that the positive effects, such as biocompatibility, biodegradability/bioresorbaility, nonimmunogenicity, and easy availability, of collagen, gelatin, chitosan, HA, and bioglass on the regeneration of hard tissue defects 8 -10 as strong reasons for using these materials in hybrid forms. Besides, the natural polymers are able to supply the main role of structural proteins in the simulation of natural tissue. 11,12 For instance, Mohammadi et al. 13 fabricated composite of chitosan and calcium phosphate short fibers through freeze-drying technique. Their results indicated that the hybrid structure is bioactive and can support the formation of HA layers in simulated fluids. The biomimetic formation of HA may help to improve the degree of similarity to natural bones. 14 The prepared scaffolds with natural and hydrophilic polymers need further processing to improve their stability in aqueous media. Cross-linking of hydrophilic functional groups is a promising approach to control dissolution and biodegradation rate. In the field of chemical cross-linking, a wide range of cross-linkers have been used based on the type of interacted functional groups. 15 Sodium tripolyphosphate is one of the chemical compositions that has been widely used for cross-linking of chitosan. 16
Although the used materials play a critical role in supporting the needs, selecting an appropriate producing technology should not be ignored. Desirable technology to fabricate an interconnected porous structure to support cellular anchorage, growth, and proliferation is the undeniable issue in tissue engineering studies. Freeze-drying provides open pores via sublimation of frozen solvent to realize the mentioned goals. 17 Li et al. 18 fabricated chitosan–gelatin–silk fibroin scaffolds for bone tissue engineering via freeze-drying technique. Results indicated that porous microstructure of matrix support proliferation MC3T3-E1 cell on the scaffolds and osteogenesis behavior of constructs confirmed their ability for clinical application. Guo and Li 19 fabricated porous Ti6Al4 V part filled with chitosan sponge via both freeze-drying and electron beam melting. The prepared scaffolds exhibited highly porous and well-interconnected micropore architecture. The addition of Ti6Al4 V led to higher cell seeding efficiency and promote proliferation rate. Results suggested the composite matrix as useful constructs for tissue regeneration.
One of the significant problems in conventional pharmaceutical agent usage is the lack of delivering these therapeutic compounds to the target site resulted in reduced effectiveness. To drawback the limitations, drug delivery vehicles have been developed. In the drug delivery system, the pharmaceutical or biological agents transfer to the exact place of action without any influence on the undesirable place and unpleasant side effects. 20 Besides, by controlling the biodegradation of vehicle, the drug release level can be controlled, therefore, the polymeric constructs can be the most well-known and extensively studied structures to fulfilling the aim. 21 Polylactic acid-co-glycolic acid (PLGA) microspheres are common synthetic vehicles in drug delivery systems because of excellent biocompatibility, nontoxicity, controllable biodegradability, and outstanding interaction with biomolecules. 22 Ginkgo biloba (GB) is a native Chinese tree that contained 22–27% flavonoids (kaempferol, quercetin, isorhamnetin, rutin, luteolin, delphidenon, and myricetin), 5–7% terpenes (diterpenes: ginkgolides A, B, C, J (M is found in the root), sesquiterpene: bilobalide, and triterpenes: sterols), and 5–10% organic acids (acetic, shikimic, p-hydroxybenzoic, vanillic, kynurenic, and ascorbic acid). The standardized extract of GB is called EGB761, which is free of allergic or immunotoxic component and promoted because of its health properties including antioxidant effects, an anti-inflammatory reaction, inhibition of platelet-activating factor, signal transduction, and inhibition of glucocorticoid synthesis. 23 -25 GB is one of the effective agents that protect osteoblast-like bone cells against free-radical damages and stimulate the proliferation of these cells as Brayboy et al. 26 studies.
In this study, the PLGA microspheres were synthesized using the solvent evaporation technique. Then, chitosan-HA scaffolds were fabricated via the freeze-drying method and incorporated with GB-loaded PLGA microsphere. The physicochemical features were then evaluated. The samples were frozen to characterize the effect of adding HA and a varying amount of microspheres. The optimum structure which indicated the necessary properties for the regeneration process was determined and introduced for further investigation.
Materials and methods
Materials
Chitosan (medium molecular weight), PLGA (M w 38,000–54,000 g mol−1), polyvinyl alcohol (PVA, M w 31,000–50,000 g mol−1), and sodium tripolyphosphate (M w 367.86) were purchased from Sigma Co. Ltd (Missouri, USA). HA (M w 502.32 g mol−1) and acetic acid (M w 60.05 g mol−1) was purchased from Merck Co. Ltd (Germany). GB was purchased from Beijing Herbal health biotech Co. Ltd (China). Phosphate-buffered saline (PBS, powder, pH 7.2–7.4) was purchased from Aprin Advanced Technologies Development Co. Ltd (Iran).
Preparation of GB-loaded PLGA microspheres
GB-loaded PLGA microspheres were synthesized via solvent evaporation of a double emulsion (water-in-oil-in-water) technique such as previous study. 4 Briefly, PLGA was dissolved in chloroform with a concentration of 20 wt%. Then, the PLGA solution was added dropwise to the 2% (w/v) of the homogenized PVA solution, which contained 1 wt% GB. The prepared emulsion was added to the PVA solution with a concentration of 0.2% (w/v) under gentle stirring. After 4 h, the solution was centrifuged at 11,000 r min−1 for 10 min and the collected spheres were lyophilized (freeze-dryer, FD-10, Pishtaz Engineering Co., Iran) for 48 h.
Fabrication of chitosan-HA-PLGA microspheres scaffolds
Chitosan-HA matrixes were prepared by the freeze-drying method. 22 So, chitosan was dissolved in 1% (v/v) acetic acid to prepare a solution with a concentration of 2% (w/v). Then, 1% (w/v) HA, 10 and 30% (wt) PLGA microspheres were dispersed in the polymeric solution. The final solution was stirred for 24 h at ambient temperature. After that, the solution was poured into molds with 20 mm height and was frozen at −20°C. Finally, the samples were transferred to freeze-dryer for 48 h. The prepared scaffolds were cross-linked via immersion of the samples in 3% (w/v) of sodium tripolyphosphate solution for 2 h. Table 1 shows the details about synthesizing scaffolds.
Details about chitosan-HA scaffolds incorporated with PLGA microspheres.

HA: hydroxyapatite; PLGA: polylactic acid-co-glycolic acid.
Characterization
Morphology observation
The morphology of the PLGA microspheres and composite scaffolds was observed by scanning electron microscopy (SEM, Stereoscan S 360-Leica, UK) at an accelerating voltage of 20 kV. Nonconductive matrixes were coated with a layer of gold.
Porosity and size distribution
The average diameters of microspheres and scaffold’s pores were measured using image analysis software (KLONK Image Measurement Light, Edition 11.2.0.0) through 25 measurements at each micrograph. The porosity of the scaffolds was calculated using equations (1) to (4) 27 :




where V p is the volume of the scaffold pores; V S is the volume of the matrix; ρ s is the density; and ε is the porosity. 28
FTIR spectroscopy
The determination of interaction and chemical bonds in the raw materials and hybrid scaffolds were performed by Fourier transform infrared (FTIR) spectrophotometer (Nicolet Is10, USA) at a wave number of 400 and 4000 cm−1.
Swelling behavior
The ability of absorption PBS solution by the chitosan-HA scaffolds was determined using equation (5). 29 Thus, the samples were weighed (W 0) and immersed into 50 ml PBS solution at 37 ± 0.5°C and rotational speed of 30 r min−1 for 0.25, 0.5, 1, 2, 3, and 24 h. At each time point, the wet samples were weighed (W) and the absorption capacity was calculated according to the following equation:
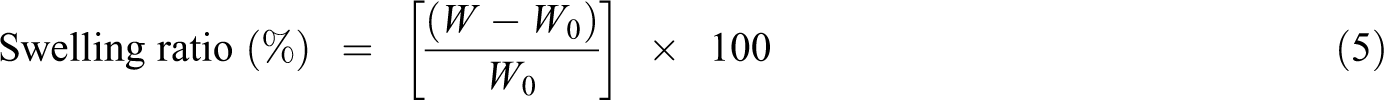
In vitro release rate
To measure the release behavior of samples, drug-loaded scaffolds and microspheres were immersed in 5 ml PBS solution at 37 ± 0.5°C and rotational speed of 30 r min−1 for 1, 3, 5, 7, 9, 11, 13, and 15 days. The concentration of GB in the PBS was specified at each time point using a ultraviolet–visible spectrophotometer at a wavelength of 273 nm. The media contained samples free of GB were introduced as a blank solution.
Statistical analysis
The porosity, pore size, density, and release values in different groups were calculated and reported as mean, standard deviation, minimum, and maximum. To compare different groups, one-way analysis of variance was used. The p value of <0.05 was considered significant.
Results and discussion
Morphology observation
The microstructure of the synthesized PLGA microspheres, hybrid scaffolds, and the average size of particles and pores was observed by SEM micrographs. Figure 1(a) and (b) presented SEM images of the GB-loaded PLGA particles. According to the images, the spherical particles with a smooth surface and without any wrinkles were synthesized using double emulsion technique (W/O/W) and an average diameter of 1–7 µm (Figure 1(c)). The similar results were obtained in the study of Gu et al. 30

(a) and (b) SEM micrographs and (c) size distribution of the GB-loaded PLGA microspheres.
SEM micrographs of freeze-dried C, CH, CP10, CP30, CPH10, and CPH30 hybrid matrixes (Figure 2(a) to (f)) indicated highly porous microstructure with interconnected and randomly oriented pores were formed during the freeze-drying process. The same results were obtained in Baniasadi et al. 31 investigation. Figure 3(a) and (b) confirmed the presence of HA and GB-loaded PLGA microspheres in the pore wall.

SEM micrographs of freeze-dried scaffolds. (a) C, (b) CH, (c) CP10, (d) CHP10, (e) CP30, and (f) CHP30.

(a) and (b) SEM micrographs of distribution of HA and PLGA microspheres in the composite scaffolds.
As shown in Figure 4(a), the porous matrix has more than 60% porosity in which the porosity reduces by the addition of HA and increasing PLGA microspheres content. Besides, the average pore size (Figure 4(b)) adapts to the porosity reduction process. Distribution of pores in test samples between approximately 100 µm and 160 µm confirmed the proper structure for supporting biological behavior such as cell anchorage and proliferation. Karageorgiou and Kaplan 32 illustrated in their study that the minimum size of pores should be 100 µm due to accordance with cell size, besides larger pores help to vascularization, direct osteogenesis, and bone formation. Moreover, they explained that the gradient of pore size may support multiple tissue formation.

(a) Porosity, (b) pore size distribution, and (c) density of scaffolds.
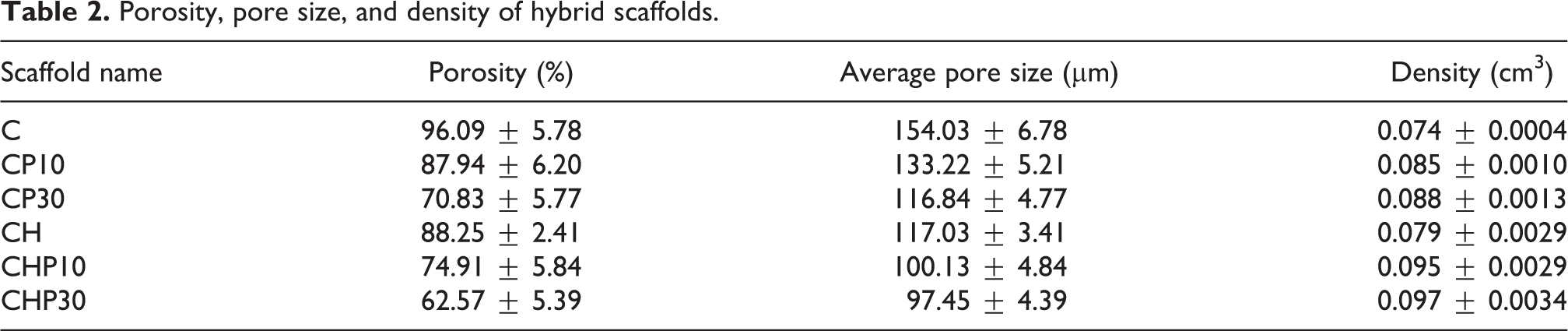
The porosity of scaffolds was measured using density bottle methods and the results of density measurements indicated inversely between porosity and density in which scaffolds with lower density presented higher porosity. Moreover, the addition of HA and microspheres can affect the changes in density significantly. The porous microstructure of scaffolds allows absorption of nutrients and excretion of waste products and finally, proper cell spreading and vascularization. 33 Table 2 represents the porosity, pore size, and density of chitosan-based scaffolds. Based on measured data in Table 2, at a fixed amount of chitosan, porosity and pore size decrease by the addition of HA. Furthermore, at a constant chitosan or HA content, mentioned parameters reduce by increasing GB-loaded PLGA microspheres amount.
Porosity, pore size, and density of hybrid scaffolds.
Chemical characterization
The FTIR spectra and schematic of the cross-linking reaction are shown in Figure 5. For pure chitosan, the absorption peaks around 846 and 1150 cm−1 are related to the saccharide structure. 34 Also, the peaks at 1740, 1480, and 1346 cm−1 correspond with the vibration of amide I, II, and III peaks, respectively. 35,36 The CH stretching vibration is shown at 2899 cm−1. The broad peak observed at 3447 cm−1 is a sign of amine N–H symmetrical vibration and hydroxyl groups. In the case of HA, the O–H bond is presented at 3553 cm−1. The CO3 2 − peak is detected at 1602 cm−1. The symmetric stretching and bending mode of PO4 3 − are observed at 977.74, 626.05, and 558 cm−1. 37,38 About the PLGA, absorption peaks at about 1755 and 1130 cm−1 can be allocated to the stretching vibration of C–O. C–O–C ether group vibration is observed at 1184 cm−1. Moreover, the peaks at 1450 cm−1 are related to the methyl group C–H bond. 39 Related to the GB, NH and OH stretching vibration is illustrated at 3411 cm−1. C=O, C=C, and C=N bonds can be observed at 1706 and 1609 cm−1. The peaks at 1512 and 1264 cm−1 can be related to the vibration of aromatic rings. The peaks at 1070 cm−1 are assigned to the CN and CO stretching vibration. 40 About the composite structure, the hydrogen bonding of cross-linker and polymer can be observed at 3444 cm−1. Besides, the NH3 + group of chitosan interacts with phosphate groups of tripolyphosphate and its characteristic peaks appear at 1561 cm−1. 41
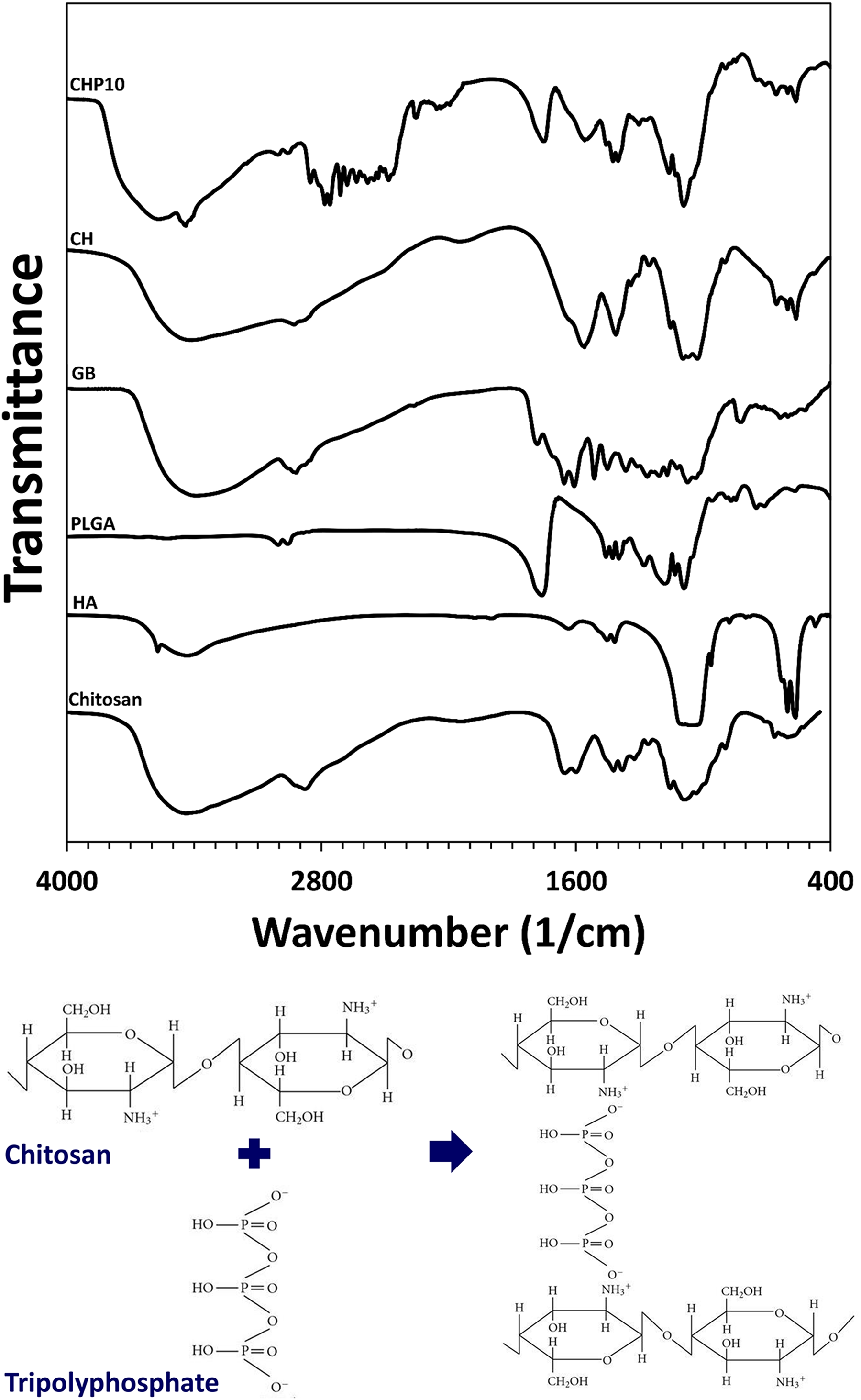
(a) Fourier transform infrared spectroscopy of raw materials and polymeric scaffolds and (b) schematic of cross-linking reaction.
Swelling ratio
The ability of absorption water in one of the critical features of tissue engineering constructs to support cellular interactions and supply their needs. Figure 6 indicates the PBS absorption rate of freeze-dried hybrid matrixes for 0.25–24 h immersion in the PBS solution. After the first 0.25 h, the scaffolds can absorb high values of PBS. This phenomenon may happen because of a high degree of porosity. 4 The amount of absorbed PBS increased within 24 h but with a steady rate compared with initial hours and finally achieved balanced swelling. It could be happened owing to the water binding of chitosan functional groups. The cross-linking reaction between chitosan and sodium tripolyphosphate prevents the rapid dissolution of constructs. In addition, the addition of HA resulted in decreased PBS absorption of constructs. In fact, higher chitosan content improves the interaction of scaffolds with water molecules because of amine and hydroxyl functional groups in the chemical structure of the chitosan. 42 According to the results, increasing the PLGA microspheres reduces the swelling ratio due to semihydrophobic nature of polymer. 43 PBS uptake percent is significantly lower in CP30 and CHP30 groups (p value < 0.05). Also, owing to the reduction of pore size and porosity by increasing HA- and GB-loaded PLGA microsphere, the amount of PBS absorption decreased slightly such as Pourhaghgouy et al. 44 studies. But it is in the acceptable range for supporting cellular interactions. 22

The swelling ratio of hybrid scaffolds at 0.25, 0.5, 1, 2, 3, and 24 h.
In vitro release behavior
Drug delivery vehicles help to control and target release of agents. A mixture of tissue engineering and drug delivery carriers led to the diffusion of therapeutic or biological agents through the pores of scaffolds. 45 Figure 7 indicates the cumulative release rate of GB from PLGA microspheres (Figure 7(a)) and microsphere incorporated in chitosan-based scaffolds (Figure 7(b)). Results indicated that increasing the microspheres content enhances the level of releasing GB. On the other hand, release behavior influenced by the absorption rate in which the addition of HA significantly reduces the release rate as a reduction of swelling through decreasing the average pore size and porosity (p value < 0.05). In fact, diffusion of water in scaffolds or microspheres resulted in rapid release of the GB during the first 24 h. After that until day 7, the GB release level follows a steady enhancement due to balanced swelling. Then, the release rate increases suddenly because of fastening biodegradation of microspheres. Release ratio is affected by both diffusion and biodegradation mechanism until collapsing the structure and complete release of GB. The fast release rate of GB may happen because of degrading amorphous sections of the substrate (PLGA); in contrast, remaining crystalline parts can control and reduce the release rate in the following. 46

(a) and (b) GB releases behavior of both PLGA microspheres and composite scaffolds for 15 days.
Conclusion
In short explanation, in the present study, GB-loaded PLGA microspheres were fabricated using solvent evaporation technique and incorporated in the freeze-dried chitosan-HA scaffolds. The prepared hybrid scaffolds represented interconnected porous microstructure in which the addition of HA and PLGA microspheres resulted in a reduction in the diameter of pores and porosity. The cross-linked samples via sodium tripolyphosphate showed the capability to interact with water molecules but the rate of absorption reduced as a function of HA and microsphere enhancement. The GB release level confirmed the strong effect of swelling and pore size on release ratio. The required physicochemical performance of scaffolds led to further preclinical and clinical studies that will be published in the future.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
