Abstract
The use of conductive polymers as a substitute for metallic conductors and semiconductors has attracted much attention in the literature. In particular, aromatic heterocyclic polymers constitute an important class since they possess chemical and electrical stability in both the oxidized (doped) and neutral (undoped) state. A series of poly(pyrrole-co-2-nitrocinnamaldehyde) were obtained via the condensation of pyrrole and 2-nitrocinnamaldehyde in chloroform using acid exchanged montmorillonite clay called maghnite-H+ as an efficient catalyst. The conjugated copolymer was characterized using proton nuclear magnetic resonance, ultraviolet–visible spectroscopy, Fourier transform infrared spectroscopy, and scanning electron microscopy.
Introduction
As a class of materials, conducting polymers are of great interest in scientific research due to their various physical, chemical properties, and large applications. 1,2 Conducting polymers 3 have attracted much attention because of their potential applications in different fields such as super-capacitors, 4 sensors, 5 photovoltaic cells, 6 electrochromic devices, 7 organic light-emitting diodes, 8 and actuators. 9 Significant progress has been achieved in increasing the conductivity, stability, and processability of conducting polymers. Polypyrrole has been widely employed as a conducting polymer and extensively characterized due to its high electrical conductivity, long-term environmental stability, and ease of synthesis by chemical or electrochemical means. 10,11
A wide range of conjugated polymer and especially those based on pyrrole attracted our interest to investigate their synthesis for application as active layers in plastic solar cells. 12 These polymers possess good stability at environmental conditions, solution and melt processability, good synthetic versatility, and ability of modification of their electronic and optical properties which make them useful in many various optoelectronic devices.
As an example, the copolymerization of pyrrole with benzaldehydes substituted are supposed to be processable material and eventually can be dissolved in common solvents, which could simplify their elaboration in thin layers through simple techniques commonly used in the fabrication of cost-effective optoelectronic devices. 13
The purpose of the present study is to synthesize a soluble copolymer of pyrrole with 2-nitrocinnamaldehyde in common organic solvents including chloroform, dimethylformamide, N-methylpyrrolidone, and dichloromethane. The resulting copolymer is characterized by different techniques, such as Fourier transform infrared (FTIR) spectroscopy, ultraviolet (UV)–visible spectroscopy, proton nuclear magnetic resonance (1H NMR), and scanning electron microscopy (SEM).
Experimental
Reagents
Pyrrole, 2-nitrocinnamaldehyde, and all commercial solvents were used without any additional purification. The synthetic method at different stages of the formation poly(pyrrole-co-2-nitrocinnamaldehyde) (PPNC), at ambient room temperature and atmospheric pressure, is presented in Figure 1.

Synthetic method of PPNC by Mag-H+ catalyst.
Copolymer preparation
The copolymer was prepared by dissolving 8 millimole of pyrrole and 8 millimole of 2-nitrocinnamaldehyde in 10 ml of chloroform and then maghnite-H+ (Mag-H+) were added with amount presenting 10% of mixture weight.
The reaction was carried out at 20°C for 6 h resulting in a mixture which was filtered to remove the clay, and then it was slowly added to a stirred cold methanol.
Finally, the copolymer was dried under vacuum at room temperature for 24 h. The yield obtained in these conditions is around 83%. The experimental conditions are summarized in Table 1.
The experimental conditions.

Copolymer characterization
To characterize the copolymer structure, we used a Bruker Vector 33 FTIR spectrometer (Bruker Biospin Corporation, Billerica, MA, USA), in the spectral range of 400–4000 cm−1 in the transmission mode. To understand well the morphology of the surface, we undertook SEM measurements. To determine the optical constants (refractive index and optical bandgap) of the copolymer, the optical measurements were carried out on a Shimadzu spectrometer (UV 3600) (Shimadzu France Le Luzard II - Bât B - Deuxième étage 9, Grande Allée du 12 Février), in the range of 250–750 nm.
Results and discussion
Characterization
FTIR spectra
The infrared measurements gave more information about the C–H and C–C bonding in the structure of our copolymer.
The typical FTIR spectra of the copolymer pyrrole/2-nitrocinnamaldehyde with molar ratio of 50:50 are shown in Figure 2. These spectra present two main absorption regions located at: Low frequencies (below 1800 cm−1) corresponding to the carbon bonds in different modes (wagging, bending, etc.). This range area is more complicated for quantitative analysis due to the possible overlapping of numerous vibrations coming from C–H
x
bands. Nevertheless, we can distinguish the peak located at 753 cm−1 of PPNC which is characteristic of α-linkage in pyrrole ring and it is ascribed to the Cβ–H out-of-plane vibration and also the peak around 820 cm−1 generally attributed to the out of plane C–H vibration mode. The compound containing the NO2 group has two strong absorption bands corresponding to symmetric and asymmetric elongations at 1341 and 1517 cm−1, respectively. Similar results, concerning the positions of these bands which depend on the substitutions and the rate of saturation within the NO2 group, are reported in the work.
14,15
An important signature of the PPNC absorption band appears in one single group at 678 cm−1, which can be assigned to the C=C stretching vibration of the quinoid ring and methine conjugated with the quinoid ring. High wave numbers (above 3000 cm−1) characterized by broadband centered at 3416 cm−1 and attributed to N–H stretching vibration of pyrrole rings.

IR spectra of PPNC.
UV/visible spectra
Figure 3 shows the UV–visible absorbance spectra of the PPNC copolymer. The spectrum presents three major absorption bands: The first one at 280 nm is assigned to the chromophore (C=C) of the benzene rings, the second one at 345 nm is attributed to the π-π* transition of pyrrole rings, and the third one at 455 nm corresponding to 2.72 eV of gap energy is due to the π-π* bandgap transition. 16,17
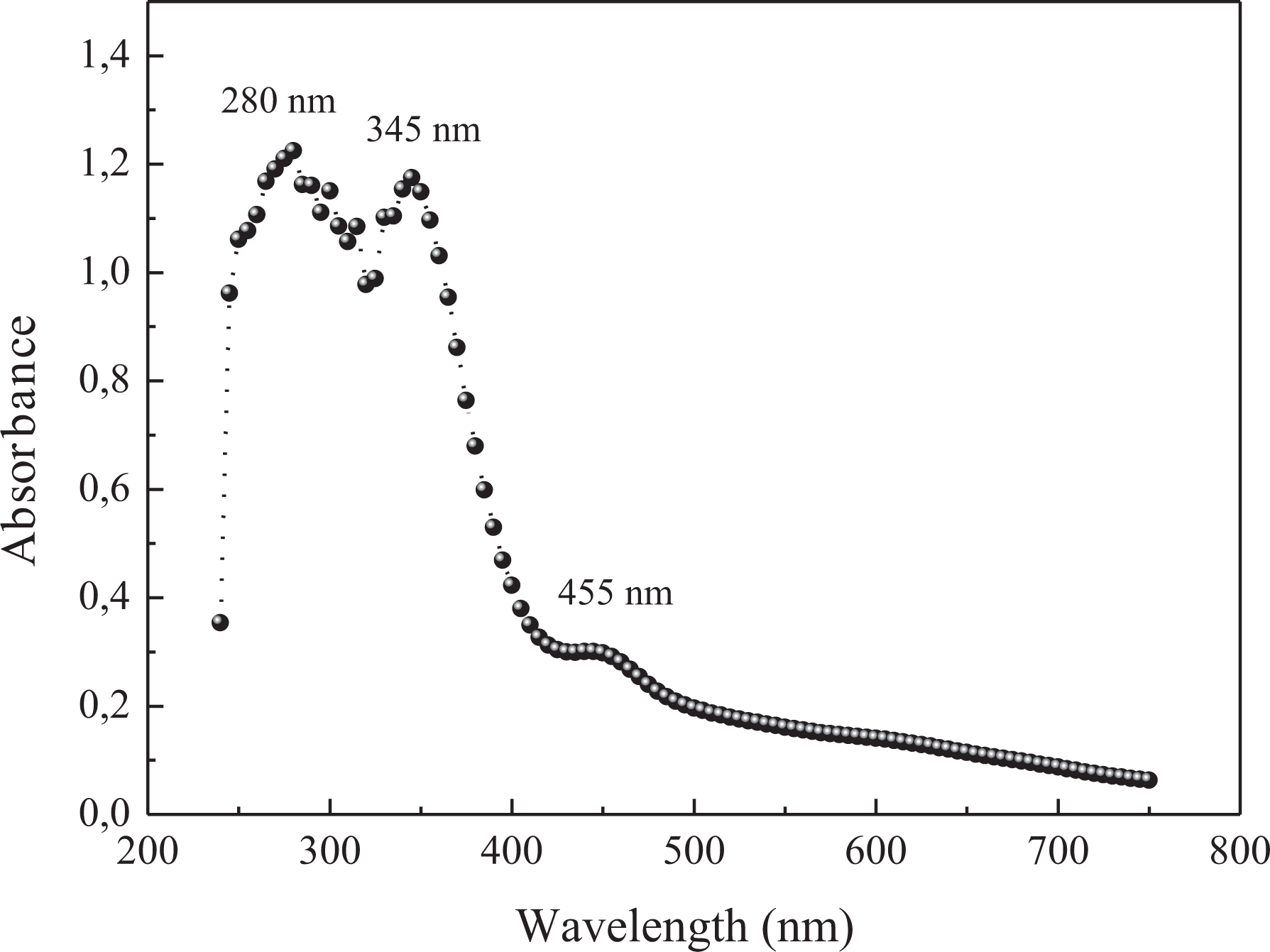
UV-vis spectra of PPNC in CHCl3.
Electrical results
The electrical measurements of our copolymer PPNC were performed with Ecopia-HMS3000 Hall effect equipment (Bridge Technology, Chandler Heights, AZ, USA) at room temperature conditions. The results are summarized in Table 2. These results show that our material exhibits a character of a semiconductor material when examining the values of the conductivity and the mobility which are equal to 6.78 S cm−1 and 39.3 cm2 V−1 S−1, respectively.
The electrical results.
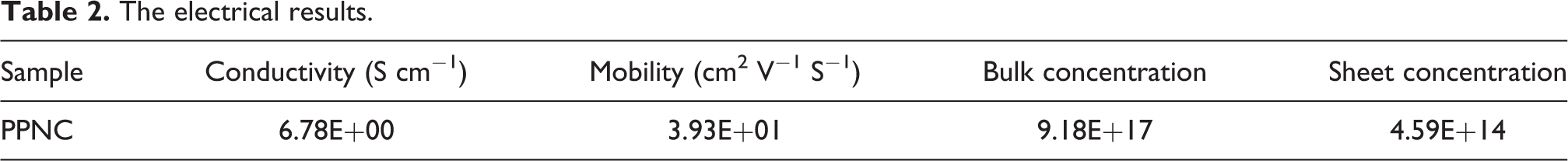
Analysis of 1H NMR spectra
The 1H NMR dimethyl sulfoxyde (DMSO-d 6) spectrum of PPNC is shown in Figure 4. The solvent peak at 2.51 ppm is marked with x as is impure water in DMSO-d 6 at 3.38 ppm. The measurements reveal that the peaks between 5.73 and 5.93 ppm can be ascribed to protons at the positions number 1 and 2 of the pyrrole rings, the peaks between 6.70 and 6.90 ppm can be assigned to protons at the positions number 3 and 4, and the peaks between 7.47 and 8.13 ppm can be attributed to protons at the positions number 5, 6, 7, 8, and 9. The same results are reported by Zhang et al. 18 1H NMR spectra of poly[(3-octanoylpyrrole-2, 5-diyl)-P-nitrobenzylidene].

1H NMR spectrum of PPNC.
Morphological analysis of PPNC film
To understand the structure of our copolymer and to get more information about the morphology and the surface roughness of its layer, we have undertaken SEM measurements. Figure 5 shows the texture of the layer and illustrates the heterogeneous and the compact structure of the PPNC.

SEM image of PPNC.
Effect of the amount of Mag-H+
Figure 6 shows the effect of the amount of Mag-H+, using various weight ratios Mag-H+/monomer, on the polymerization rate. The time of the polymerization was 24 h at room temperature, and the other experimental parameters are summarized in Table 2. The yield seems not to be affected by the percentage of the amount of Mag-H+. The maximum yield of 83.33 wt% is reached for 10 wt% of Mag-H+, and a higher addition of clay causes a relative decrease of the reaction yield. This trend is probably the result of the number of “initiating active sites” responsible for inducing polymerization; this number is proportional to the catalyst amount used in the reaction.

Effect of the amount of Mag-H+ on the yield % of the PPNC.
Effect of time on polymerization
The yield of polymer versus the time for polymerization (by condensation) of pyrrole and 2-nitrocinnamaldehyde with the presence of 10% of Mag-H+ is presented in Figure 7. The curve shows two stages of polymerization with time. During the first stage (from 2 to 6 h), the yield % increases linearly with time with a rate of 16.66% h−1, then the curve exhibits a plateau throughout the second stage (from 6 to 24 h). This is probably due to the increase of the liquid viscosity. Therefore, the time polymerization was limited to 6 h for the rest of this study.

The yield % of the PPNC copolymer versus time for polymerization of pyrrole and 2-nitrocinnamaldehyde.
Effect of the temperature on the polymerization rate
Figure 8 illustrates a significant effect of the polymerization temperature on the yield % of PPNC. Four experiments were carried out at different temperatures. The yield % of polymerization decreases from 83.33% to around 60% when the temperature rises from 20°C to 60°C. This behavior indicates that the optimum polymerization reaction temperature is around 20°C.

Variation of the yield % of PPNC with polymerization reaction temperature.
Effect of the molar ratio on polymerization
The copolymer was synthesized at different molar ratios of pyrrole and 2-nitrocinnamaldehyde (Figure 9). The result indicates clearly that the copolymer of pyrrole with 2-nitrocinnamaldehyde was significantly influenced by the monomer ratio. The copolymer exhibits the highest yield at a ratio of (5:5) of the [pyrrole]/[2-nitrocinnamaldehyde] mixture. This result is explained by the fact that in polymerization by condensation, one mole of pyrrole monomer reacts with one mole of 2-nitrocinnamaldehyde monomer, so if the amount of any of these two monomers increases, the rate of polymerization will decline.
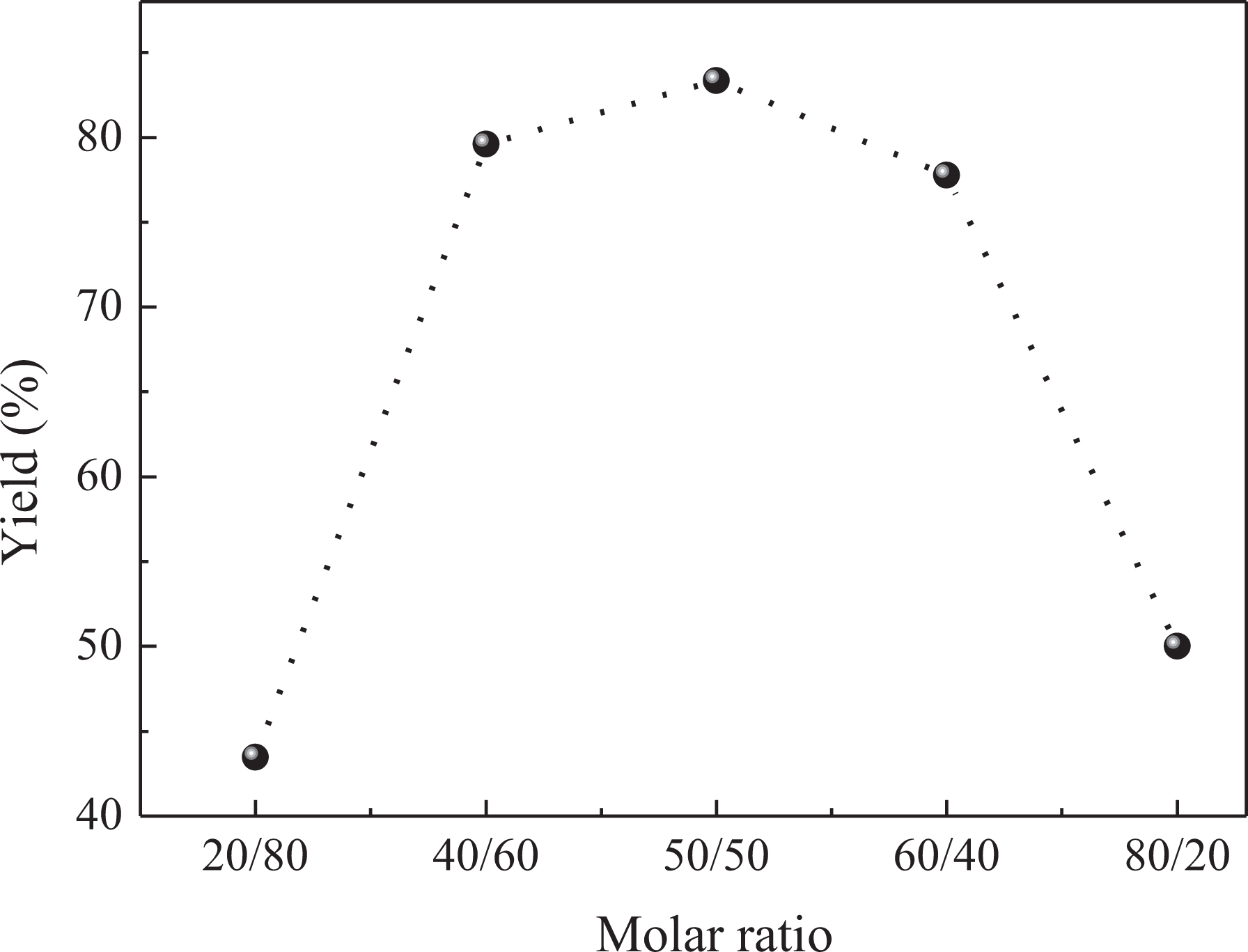
Variation of PPNC yield % with molar ratio.
Effect of the solvent on polymerization
The polymerization of PPNC was carried out in toluene (dielectric constant = 2.38), chloroform (dielectric constant = 4.8), and dichloromethane (dielectric constant = 8.93): the effect of solvent on the polymerization of PPNC was studied. The rate of polymerization in dichloromethane was found to be much higher than that in chloroform and toluene (Figure 10). Regarding the dielectric constant of the solvent (toluene C6H5CH3, chloroform CHCl3, and dichloromethane CH2Cl2), it affects the yield of monomer: the higher polar solvent leads to the higher conversion yield.
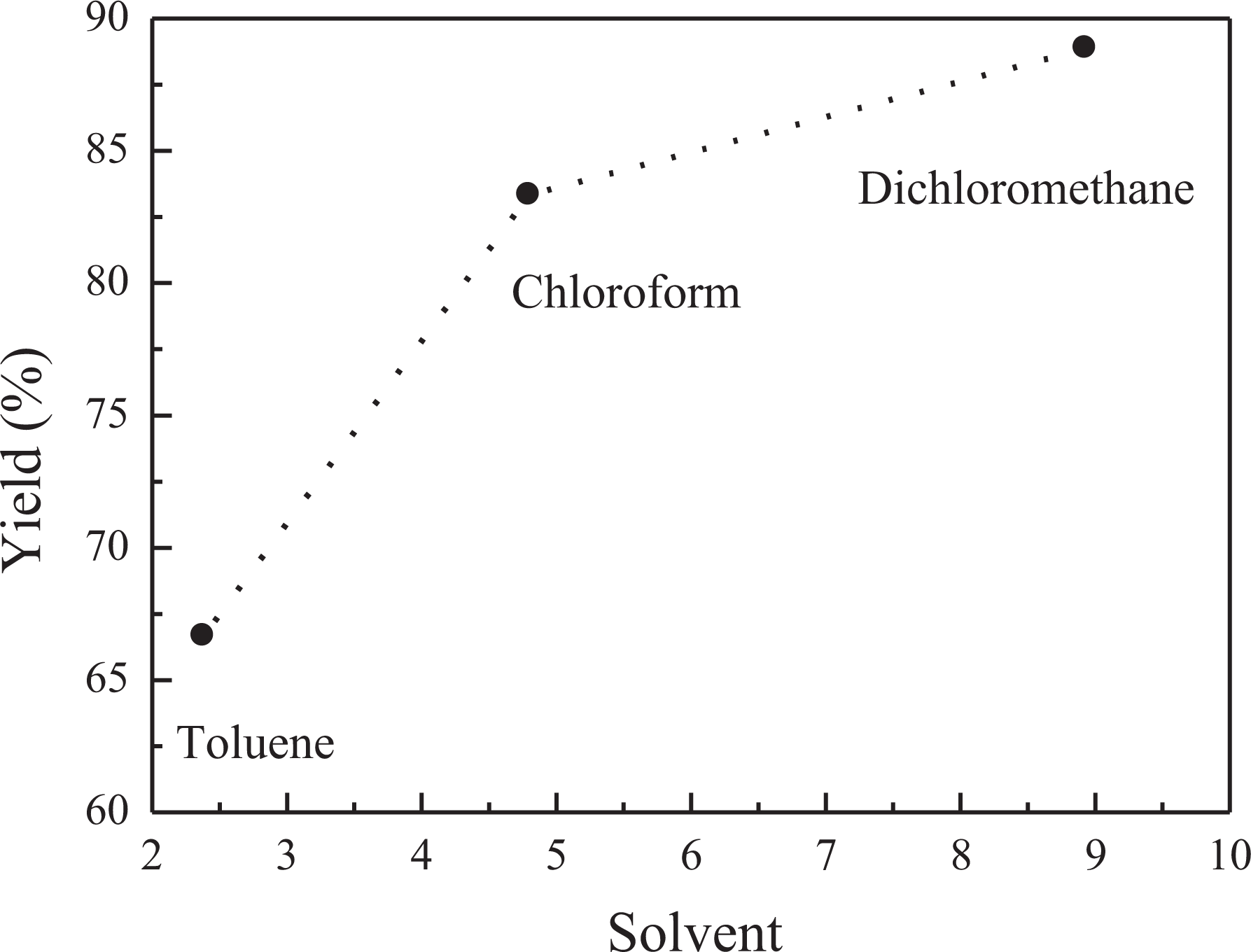
Effect of the solvent on the polymerization of PPNC copolymer.
Conclusion
The synthesis of a novel soluble copolymer, PPNC, which has a π- the conjugated chain was synthesized by polycondensation of pyrrole and 2-nitrocinnamaldehyde catalyzed by Mag-H+. The effect of different parameters on the polymerization of PPNC copolymer yield was investigated. The optimum values of the reaction parameters found were 10 wt% of Mag-H+, 6 h polymerization times at room temperature, 50/50 molar ratio. A yield of 83.33 wt% was achieved using theses optimum parameters. Finally, the optical results obtained from the absorption spectra show that the produced copolymer has a lower energy bandgap (Eg = 2.72 eV) which corresponds to better semiconductors.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
