Abstract
The flammability characterization and synergistic effects of red phosphorus masterbatch (RPM) with expandable graphite (EG) in flame-retardant polypropylene (PP)/thermoplastic polyurethane (TPU) composites are investigated by limiting oxygen index, UL-94 testing, cone calorimeter tests, thermal gravimetric analysis, Fourier transform infrared (FTIR), and scanning electron microscopy. The results show that the flame retardancy of PP/TPU/EG/RPM composites is greatly influenced by RPM. The synergistic effects between RPM and EG take place in the flame-retardant composites. The presence of RPM with EG decreases significantly the heat release rates and total heat release, and UL-94 V-0 rating is achieved when suitable amount of RPM substitutes for EG in the PP/TPU/EG/RPM composites. The T onset and T 10 wt% of the composites are improved because of the presence of RPM. The FTIR spectra show that the incorporation of RPM improves the thermo-oxidative stability of PP/TPU at higher temperatures. The morphological observations indicate the reinforcement of thermal stability, and flame-retardant performance is attributed to the compact and stable char layers promoted by RPM with EG acted as an effective heat barrier and thermal insulation.
Introduction
In recent years, intumescent flame retardants (IFRs) have been widely studied due to their being halogen-free, low toxicity, low smoke production, and absence of dioxins during the combustion process. The IFRs system, generally, is made up of three parts, which are acidic source, carbonization agent, and blowing agent. 1 The intumescent system relies on the heat-induced decomposition to produce a char layer that insulates the substrate from heat and oxygen.
Graphite is a carbon source, while the gases evolved during the composition of intercalated species perform the blowing action. Expandable graphite (EG), another typical of IFRs system, is a flake graphite intercalation compound. It is a layered crystal consisting of sheets of carbon atoms tightly bound by covalent bonds to each other in the same plane. EG is a partially oxidized, intercalated form of graphite containing intercalated guest species in between the stacked graphene layers. A key property of EG is tendency to exfoliate explosively, that is, expand rapidly in a worm-like manner when heated to high temperatures. Unlike in classical intumescent systems comprising a combination of several ingredients, including char former, carbonization catalyst, and blowing agent, EG combines all functions needed for intumescence in a single compound. EG exhibits a layered structure which is similar to layered silicates and has good affinity for organic compounds and polymers. 2
When exposed to heat source, EG will expand and generate a voluminous insulative layer on the surface of materials, and thus can provide flame-retardant properties. 3,4 Some reports have shown that EG is an efficient additive which acts as a blowing agent as well as a carbonization agent and has good flame-retardant properties. 5 -9 However, the insufficient compatibility and poor interfacial adhesion between the polymer matrix and EG will worsen the mechanical properties of polymeric materials. Many modifiers are employed to enhance the interfacial compatibility. 10 -16 Our previous work shows that silane coupling agent can improve the dispersibility and compatibility of EG in polypropylene/thermoplastic polyurethane (PP/TPU) composites, and better mechanical properties and flame retardancy are obtained. 17 However, it is very difficult to achieve the V-0 in UL-94 test 17,18 in the flame-retardant composites filled with EG alone. Therefore, it is necessary to select a high efficient synergistic additive in the EG-based composites.
Red phosphorus (RP) is one of the ecologically and physically most harmless alternative fire retardants. RP has some advantages, such as high phosphorus content, low cost, and good flame-retarding effects for many polymers. Nevertheless, pure RP shows several drawbacks, such as thermal instability, strong moisture absorption, and reddish-brown color, which restrict its extensive commercial applications. 19 The encapsulation and the use of masterbatches are successfully established to eliminate problems of handling safety and stability. RP has amorphous inorganic macromolecular structure and mainly depends on the condensed phase process including liquid acid (metaphosphoric acid, phosphoric acid, and polyphosphoric acid) films covering the surface of burning materials, as well as acid-promoted dehydration accelerating the formation of a consolidated char layer. 20,21 Many systems, such as phosphorus nitrogen, halogen antimony, and metallic hydroxide RP, have been found to have synergistic effects in the flame-retardant polymers. 22 -24
However, to the best of our knowledge, no report has been published about the flammability and synergistic effects of RP with EG in the PP/TPU composites. In this present work, we introduced EG and red phosphorus masterbatch (RPM) into the flame-retardant PP/TPU blends to improve its flame-retardant properties. The combustion behaviors and synergistic effects of RPM with EG in PP/TPU composites were assessed by UL-94 test, cone calorimeter tests (CCT), thermal gravimetric analysis (TGA), Fourier transform infrared (FTIR) spectra, and scanning electron microscopy (SEM).
Experimental
Materials
PP (075, melt flow index = 6.1–10.0 g/10 min; density = 0.38–0.43 g cm−3) was purchased from Maoming Petrochemical, China. TPU (481, polyester grade; density = 1.2 g cm−3) was provided by Bayer Company, Germany. EG (average particle size: 45 μm; expansion ratio of EG: 300 ml g−1) was supplied by Qingdao Kangboer Graphite Company, China. RPM with 60 wt% RP and 40 wt% PP was supplied by Chenguang Chemical Research and Design Institute Co., Ltd, China. Maleic anhydride-grafted PP (PP-g-MAH), supplied by Nanjing Deba High Polymer Material Co., Ltd, China, with melt flow index of 35 g/10 min (190°C/2.16 kg) and with 1.0% MAH grafting level, was used as a compatibilizer. Silane coupling agent KH-570 (γ-methacryloxypropyl trimethoxy silane) was supplied by Nanjing Shuguang Chemical Company, China.
Preparation of samples
Modification of EG particles was performed according to the following steps. Firstly, the EG particles were dried at 80°C for 4 h to eliminate possible absorbed water on the surface of the particles. Secondly, a certain amount (1 g) of silane coupling agent was dissolved in enough amount of ethanol at 80°C under mechanical stirring for 3 min. Thirdly, desired amounts of (50 g) dried EG particles were put into the above solution under stirring at 1500 r min−1 for 30 min at 80°C. The surface-treated EG particles were dried at 80°C for 4 h.
PP/TPU blends with PP, TPU, and PP-g-MAH were performed on a twin-screw extruder (type TSE-20A/600-4-40, Nanjing Ruiya Polymer Equipment Co., Ltd., Nanjing, China) at 180–230°C and screw speeds were 150 r min−1. The PP/TPU blends and EG were dried at 80°C for 4 h, and then PP/TPU composites filled with the desired amounts of treated EG and RPM were blended in a twin-screw extruder at 160–190°C and screw speeds were 150 r min−1. All samples were hot-pressed at about 190°C under 10 MPa for 10 min and cut the sheets into the suitable dimensions for measurements and characterization. The formulations of the flame-retardant PP/TPU composites were presented in Table 1.
Formulation and flame retardancy of PP/TPU flame-retardant composites.

PP: polypropylene; TPU: thermoplastic polyurethane; PP-g-MAH: maleic anhydride-grafted PP; EG: expandable graphite; RPM: red phosphorus masterbatch; LOI: limiting oxygen index.
a PP/TPU = 80/20 (wt/wt) blends in this work.
Measurements and characterization
UL-94 test
The UL-94 vertical burning test was performed with a CTF-2 type instrument (Nanjing Jiangning Analytical Instrument Factory, China). The specimens for the test were of dimensions of 127 × 12.7 × 3 mm3 according to the ASTM D635-77 standard. The three ratings (V-2, V-1, and V-0) are defined. The V-0 rating stands for the highest requirements.
Limiting oxygen index
The limiting oxygen index (LOI) value was measured using a JF-4 type instrument (manufactured by Jiangning Analysis Instrument Factory, Nanjing, China) on sheets 120 × 6.5 × 3 mm3 according to the standard oxygen index test (ISO 4589).
The LOI value is calculated according to the following equation:

where [O2] and [N2] are the concentration of O2 and N2, respectively.
Thermal gravimetric analysis
TGA of the flame-retarded samples was examined under nitrogen flow with a flow rate of 30 ml min−1 in a temperature range of ambient to 700°C with a heating rate of 10°C min−1 by a NETZSCH TG 209F1 thermogravimetric analyzer, Germany. About 8–10 mg samples were used in this test.
FTIR spectroscopy
The FTIR spectra were recorded between 400 cm−1 and 4000 cm−1 with a resolution of 2 cm−1 using a Nicolet 560 spectrophotometer (Nicolet Instrument Co., Newburyport, USA). The residues left after degradation of PP/TPU/EG (PTE1) and PP/TPU/EG/MRP (PTE2) composites degraded at different temperatures for 20 min were mixed with KBr powders, and then the mixture was compressed into plates for FTIR spectra analysis.
Cone calorimeter test
The CCTs (Stanton Redcroft, UK) were carried out according to ISO 5660 standard procedures. Each sample of dimensions 100 × 100 × 3 mm3 was wrapped in aluminium foil and exposed horizontally to an external heat flux of 35 kW m−2.
Morphology observation
The char formed surfaces were investigated by an SEM (Model Fei Quanta 200, FEI Ltd, Eindhoven, Netherlands). Gold sputter-coated samples were examined using a Cambridge Stereoscan 250 (Cambridge Instrument Co., Cambridge, UK), with an accelerating voltage of 10 kV.
Results and discussion
Flammability: LOI and UL-94 tests
The LOI and UL-94 tests are widely used to evaluate the flame retardancy of materials, especially for screening flame-retardant formulations of materials. 25 Table 1 lists the LOI values and UL-94 ratings obtained from the flame-retardant PP/TPU composites with different contents of EG and RPM additives. The LOI value of PP/TPU is only 18.1%, which indicates its flammable nature. However, the LOI value of the composites increases with the addition of RPM. It reaches a maximum and then decreases when the content of RPM is higher than that of EG. For example, the LOI value of the PP/TPU/EG composites (PTE1 sample) is 21.6%; however, the LOI values of the PTE2 and PTE4 samples with 5 and 15 phr RPM are 23.8% and 22.2%, respectively. The data suggest that the increase of LOI of the composites is attributed to the better synergistic effects of suitable amount of RPM with EG in the flame-retardant PP/TPU composites. But excess RPM will cause an anti-synergistic effect for RPM and EG in the flame-retardant PP/TPU composites. It is observed that PP/TPU and the PP/TPU/EG composites with individual 20 phr EG do not pass the UL-94 rating test. To solve this problem, RPM is selected as a synergist of EG to improve the flame retardancy of the PP/TPU/EG composites, where the total content of additives including both EG and RPM are kept at 20 phr. The data listed in Table 1 show that the UL-94 flammability rating of the above composites is remarkably improved because RPM is selected instead of partial EG particles. For example, after combining RPM with EG in PP/TPU, UL-94 V-0 rating is achieved for the PTE2 and PTE3 samples. This indicates that the RPM with EG can exhibit higher efficiency and better synergistic effects in the flame-retardant PP/TPU composites. However, there is no rating with further increasing the content of RPM to 15 phr (PTE4 sample). This is because few chars will be induced from resin by RPM to connect the brittle EG layers and the gaps between EG that will affect the fire performance, especially for the UL-94 rating. This indicates that the suitable amount of RPM acts as a synergist of EG in the flame-retardant PP/TPU/EG/RPM composites.
Figure 1 presents the photos of the char residues of PP/TPU, PP/TPU/EG, and PP/TPU/EG/RPM with different amounts of RPM after UL-94 tests. It is clearly observed that there is no char residue left for the PTE0 sample. The char residues of the PTE1 sample with only 20 phr EG are loose and brittle, whereas the char residues of the PP/TPU/EG/RPM samples are more compact and integrated, especially for the PTE3 sample, as shown in Figure 1(d). However, the char residues become to be loose again with further increasing the RPM content for the PTE4 sample, as presented in Figure 1(e). This indicates the suitable amount of RPM with EG can improve the structures of the char layers and enhance the stability of char layers. The stable and compact char residues formed the surface of underlying polymer materials act as excellent barriers of heat transfer and insulation.

The char morphology of the burned bars of PP/TPU and flame-retardant composites: (a) PTE0, (b) PTE1, (c) PTE2, (d) PTE3, and (e) PTE4.
Flammability: CCT test
CCT is a bench-scale test to simulate real fire conditions. The CCT based on the oxygen consumption principle has been widely used to evaluate the combustion behaviors of materials. 26 -29 Some parameters, such as heat release rate (HRR), total heat release (THR), and mass loss, can be obtained from CCT, among which the value of peak HRR (PHRR) is an important parameter that can be used to evaluate the intensity of fires. 30 The detailed data obtained by CCT from the aforementioned series of composites are listed in Table 2.
Cone calorimeter data of the flame-retarded PP/TPU composites.

PP: polypropylene; TPU: thermoplastic polyurethane; PHRR: peak heat release rate; THR: total heat release.
Figure 2 shows the dynamic HRR curves versus time for PP/TPU, PP/TPU/EG, and PP/TPU/EG/RPM with different amount of RPM. It is observed from Figure 2 that the flammability behaviors of the flame-retarded PP/TPU composites are influenced by the additives, especially for the addition of RPM with EG. Pure PP/TPU matrix quickly burns with a high PHRR value of 485.5 kW m−2 and burns out within 500 s after ignition. This indicates that the combustion process of the PP/TPU sample is very sharp and complete. However, the values of PHRR of the composites decrease remarkably with the addition of the flame retardants. In addition, the further decrease is observed from the synergistic effects of RPM with EG in flame-retardant PP/TPU systems. For example, the PHRR value of the PTE1 sample with only 20 phr EG is 111.1 kW m−2, which is only 22.9% of that of the PP/TPU sample. For the PTE2 and PTE3 samples, the values of PHRR are dramatically reduced to 73.6 and 91.7 kW m−2, which are only 15.6% and 18.9% of that of the PTE0 sample, respectively. It is reasonable that these reductions result from the synergistic effects of RPM with EG to form the stable and compact char layers on the surface of the substrate polymer during the combustion. This char layer effectively separates the matrix from heat and oxygen, resulting in a lower PHRR value. At the same time, it is found from Figure 2 that the combustion time of all the flame-retardant composites is prolonged in comparison with that of PP/TPU sample, which is due to decomposition of the transient char. Because the transient char quality of the PTE2 sample is higher than that of the PTE3 sample, it shows a slower decomposition rate, resulting in a better fire resistance.

HRR curves of PP/TPU and its flame-retardant composites: (a) PTE0, (b) PTE1, (c) PTE2, and (d) PTE3.
The THR curves versus time of the above four samples are shown in Figure 3. Clearly, the addition of the flame retardants greatly affects the THR values of PP/TPU. It is obviously noted from Figure 3 that the THR value of PTE0 sample increases fast at the beginning of combustion and then levels off after above 500 s. This indicates that the flammability of PP/TPU sample is quite quick and violent. And PP/TPU has a high THR value of about 70.3 MJ m−2. However, for the flame-retardant PP/TPU composites, the THR values increase slowly during the combustion and then level off at the end of burning due to the addition of EG and RPM. At the same time, it is also found that the increasing trend and THR values of PP/TPU composites with EG and RPM decrease further compared with PTE1 sample with only 20 EG. The value of THR of PTE1 sample is about 68.9 MJ m−2 after burning; however, the THR values of the PTE2 and PTE3 samples are reduced to 48.4 and 55.5 MJ m−2, respectively. The heat generation reduces with the char formation on the surface of the flame-retarded materials due to the synergistic effects between RPM and EG. In this process, fire retardant forms char residues, which can isolate heat from the outside and combustible gases from the inside and shows a better fire resistance. The results are consistent with UL-94 tests and further confirm the synergistic effects of RPM with EG on improving the flame retardancy of PP/TPU matrix.

THR curves of PP/TPU and its flame-retardant composites: (a) PTE0, (b) PTE1, (c) PTE2, and (d) PTE3.
Mass is used to evaluate the thermal oxidation decomposition, volatile, and burning degree in a certain fire environment. 27 The curves of mass versus time for the above samples are shown in Figure 4. PP/TPU blend loses its mass quickly and mass reduces to nearly zero in a short burning time, further suggesting the combustion of PP/TPU sample is very sharp and complete. However, the mass loss of the flame-retarded PP/TPU composites with EG and RPM is lower than that of PP/TPU during the whole process, and the composites also show a higher char residue. As shown the curves b–d in Figure 4, the mass loss of the composites is slow, especially for the PTE2 sample. The possible reason is that metaphosphoric acid and polyphosphoric acid are generated during the flammability of PP/TPU/EG/RPM samples, which can connect the carbon layer formed by EG and substrate after burning, isolating heat from outside and combustible gases from inside. In addition, the PO· radical is generated, and the PO can react with H· or HO· radical, which holds back the combustion chain reaction. This shows that the synergistic effect occurs between RPM and EG. So, the flammability and decomposition of polymer are prolonged and prevented, and then a better fire performance is obtained for the PTE2 sample.

Mass curves of PP/TPU and its flame-retardant composites: (a) PTE0, (b) PTE1, (c) PTE2, and (d) PTE3.
Thermal stability
TGA is widely used to investigate the thermal stability of polymer. 31,32 Figures 5 and 6 display that the TGA and derivative thermogravimetric (DTG) curves of PP/TPU, PP/TPU/EG, and PP/TPU/EG/RPM with different amounts of RPM under a flow of nitrogen at a heating rate of 10°C min−1, respectively. The TGA and DTG data for PP/TPU and its flame-retardant composites, including decomposition temperatures of 10% and 50% mass loss, onset temperature of TGA, and peak temperature of DTG, are listed in Table 3. The thermal stability of PP/TPU/EG and PP/TPU/EG/RPM composites is visibly improved compared with that of PP/TPU. It is obviously noted that both PP/TPU and its flame-retardant composites experienced one-step process. PP/TPU underwent complete and rapid thermal degradation at temperatures up to 450°C, and there is hardly any residue at the end of degradation. The peak temperature (T peak) from DTG curve is about 435.9°C. The thermal stability of the PP/TPU/EG composites is improved due to the addition of EG compared with that of PP/TPU, and there are about 13.0% char residues left after the degradation. In addition, it is found from Figure 5 that the PP/TPU/EG/RPM composites with different contents of RPM also show only one-step decomposition process as that of PP/TPU/EG composites. Moreover, PP/TPU/EG/RPM composites show better thermal stability than PP/TPU/EG composites when the temperature is below 420°C. The presence of RPM has a little effect on the degradation of the composites when the temperature is over 420°C, as shown in Figure 5 (samples PTE0–4). Although the thermal stability of PP/TPU/EG/RPM composites is hardly influenced by the change of the RPM content, the char residues after degradation show a difference, and the value decreases with increasing the content of RPM. The data listed Table 3 show that the T onset value for the PTE1 sample is lower than that of PTE0 due to the addition of EG, which is attributed to the expansion of EG because the reaction between H2SO4 and graphite produces blowing gases above 190°C. 33 However, the suitable amount of RPM in the PP/TPU/EG/RPM samples can increase the initial decomposition temperature compared with PTE1 with 20 phr EG alone.
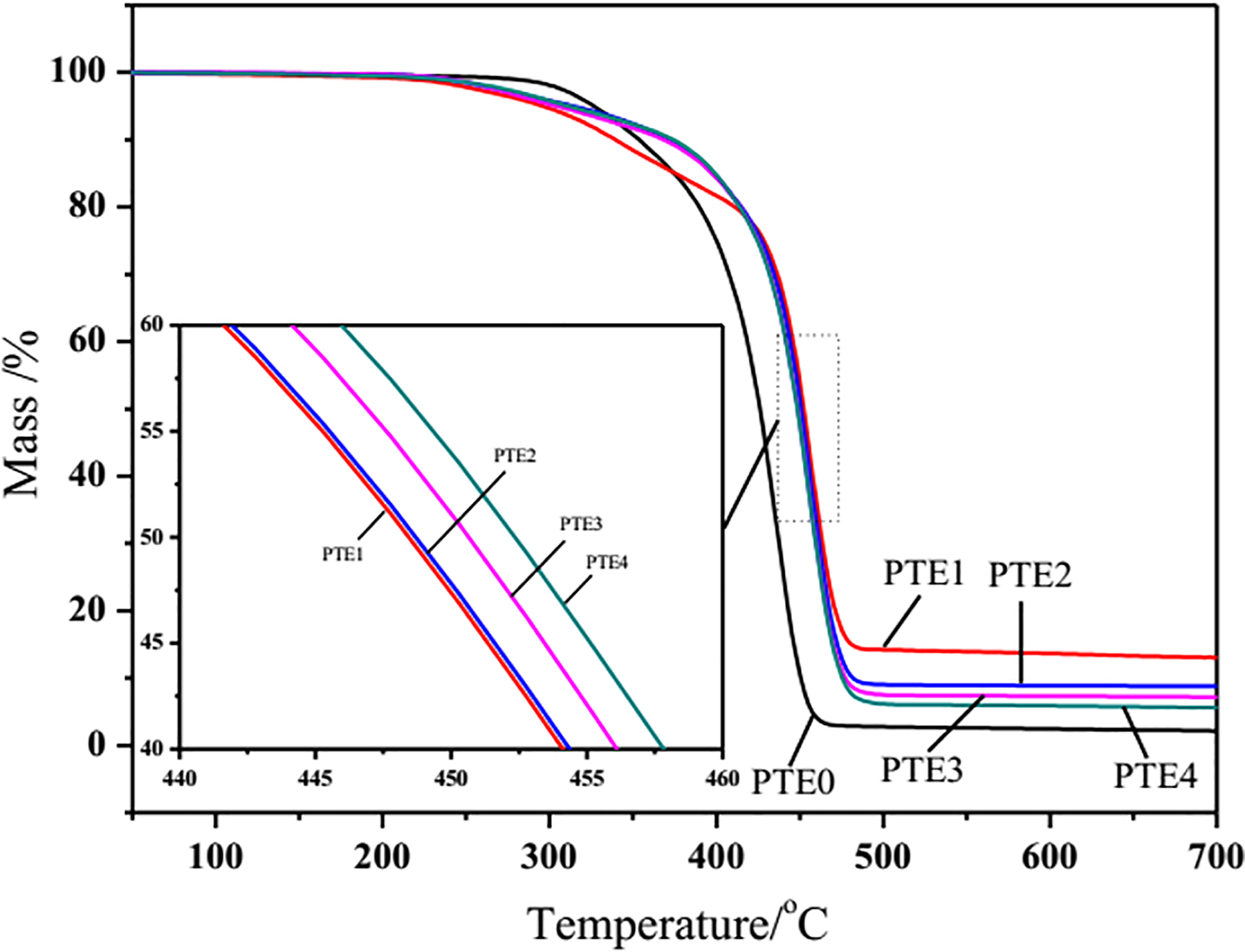
TG curves of PP/TPU and its flame-retardant composites.

DTG curves of PP/TPU and its flame-retardant composites.
Thermal properties of PP/TPU and flame-retardant composites.
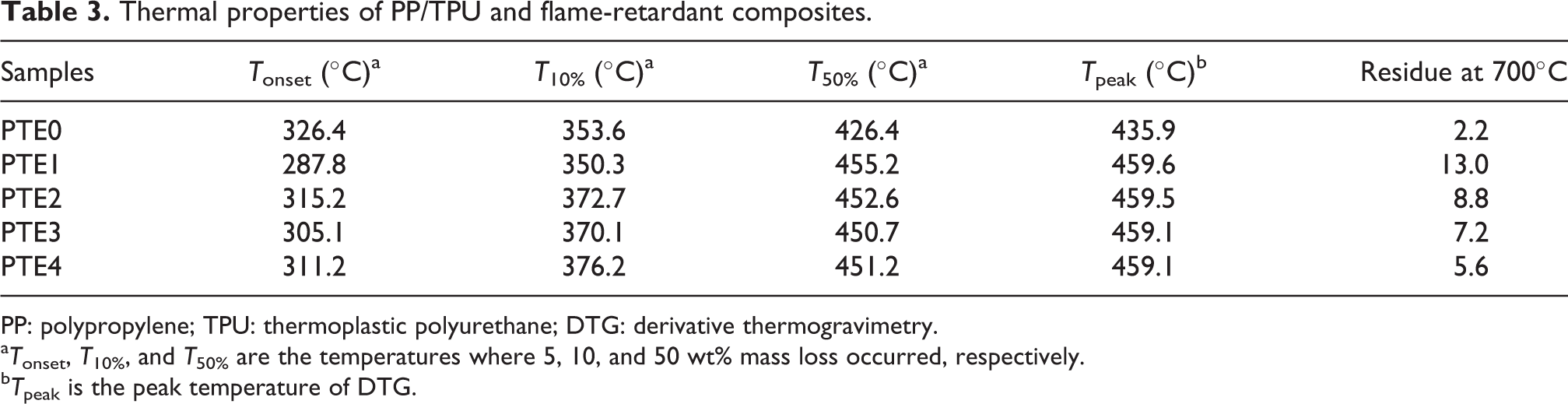
PP: polypropylene; TPU: thermoplastic polyurethane; DTG: derivative thermogravimetry.
a T onset, T 10%, and T 50% are the temperatures where 5, 10, and 50 wt% mass loss occurred, respectively.
b T peak is the peak temperature of DTG.
FTIR analysis of the charred residues
Dynamic FTIR was employed to evaluate the solid pyrolysis products of PP/TPU/EG (PTE1) and PP/TPU/EG/RPM (PTE2) composites. The changes of FTIR spectra of the PTE1 and PTE2 samples with increasing the pyrolysis temperature are given in Figures 7 and 8, respectively. The assignment of dynamic FTIR spectra of PET1 and PET2 is presented in Table 4. It is seen that the peaks at 3444, 2958, 2919, 2871, 2838, 1731, and 1166 cm−1 are the characteristic absorptions of PP/TPU. 34,35 The band at 3444 cm−1 nearly disappears at the temperature of 350°C, and this can be explained by the release of water. The relative intensities of other characteristic peaks do not change below 350°C. However, while the temperature rises to 400°C, it is found that the absorption peaks at 2958, 2919, 2871, 2838, 1535, 1459, 1376, and 1166 cm−1 disappear, indicating that the main decomposition happens at this stage. This is consistent with the TGA results.

Changes in the charred residue left FTIR spectra of PET1 sample at different pyrolysis temperatures.

Changes in the charred residue left FTIR spectra of PET2 sample at different pyrolysis temperatures.
Assignment of FTIR spectra of PET1 and PET2.

FTIR: Fourier transform infrared.
The FTIR spectra of PET2 show very similar features to that of PET1. It is observed from Figure 8 that the characteristic peaks of RPM are not obvious because its characteristic bands probably coincide with the characteristic bands of PP/TPU. It is worth noting that the intensities of most peaks decrease sharply at 450°C, which means that the presence of RPM delays the thermal degradation of PP/TPU. This conclusion is also suggested by the TGA analysis above. Therefore, it is concluded that the better flame-retardant performances can be achieved for PET2.
Morphology of char residues
In order to elucidate how the formation of chars affected the flammability of the flame-retardant PP/TPU composites, the residues left after CCTs are examined for changes in char appearance by SEM. Figure 9 shows the SEM micrographs of char residues of the PTE1 and PTE2 samples. RPM, as a synergistic flame retardant, has a great effect on the char structure of the composites after combustion. A typical of worm-like expanded graphite agglomerates and char residues are found in the burned layer; among the flakes are gaps to allow further the sample to burn, as shown in Figure 9(a). A fractured char layer is shown for the PTE1 sample due to brittle char formation during combustion, resulting in a poor flame-retardant performance. The magnification SEM micrographs, as shown in Figure 9(c), indicates that EG particles in the exterior of the burned composites expand many times and the char residue is mainly composed of expanded graphite. Nevertheless, compared with the PTE1 sample with only 20 phr EG, uniform and coherent char layer structure with less holes and crack can be formed from the burning of the PTE2 sample, as shown in Figure 9(b). In addition, the char residue of the PTE2 sample is more compact and denser than that of the PTE1 sample. The better charring performance effectively insulates heat and oxygen and exhibits excellent flame retardancy. The possible reason is that the RPM is oxidized into phosphorus pentoxide during the combustion of PP/TPU/EG/RPM composites. On the one hand, phosphorus pentoxide rapidly diffuses on polymer surface, diluting oxygen, and slacking the burning of composites. On the other hand, the reaction between phosphorus pentoxide and water generates phosphoric acid that will react with phosphate further, which generates polyphosphoric acid and metaphosphoric acid. Polyphosphoric acid is a kind of viscous vitreous material, which is tightly wrapped on the surface of the underlying polymer. This can prevent heat transfer between the flame zone and the substrate, and thus protect the underlying polymers from further burning and retard the pyrolysis of materials.

SEM photographs of residue surface of low magnification of (a) PTE1 sample and (b) PTE2 sample and high magnification of (c) PTE1 sample and (d) PTE2 sample.
Combined with the above discussion, the synergistic flame-retardant mechanism is proposed for RPM with EG in the flame-retardant PP/TPU/EG/RPM composites. In the gas phase, the nonflammable gases including CO2, SO2, and H2O release during the decomposition of the composites can dilute the combustible gases. In the condensed phase, RPM and EG play an important role in different periods. Initially, the “worm-like” char structure is formed by the inflation of EG at the range of 180–250°C, but the charred layers cannot effectively endure heat flux for a long time at a high temperature. Then phosphoric derivatives generated from RPM cover the surface of the underlying polymer, which can strengthen the char barrier for its strong adhesion effect. Therefore, the flame-retardant performances of polymer materials are improved greatly due to the presence of RPM.
Conclusions
In this work, we focus on studying the flammability behaviors and synergistic effects of RPM with EG in the flame-retardant PP/TPU composites by LOI, UL-94, CCT, TGA, and morphological observations. The results obtained from the LOI, UL-94, and CCT show the good synergistic effects between RPM and EG in the flame-retardant PP/TPU/EG/RPM composites, and the UL-94 V-0 rating is achieved when the suitable amount of RPM substituted for EG. The CCT results show the suitable amount of RPM considerably decreases the HRR, PHRR, and THR values of PP/TPU/EG/RPM composites. The PHRR and THR values of the PTE1 sample with only 20 phr EG are 111.1 kW m−2 and 68.8 MJ m−2, whereas that of the PTE2 sample decreases to 73.6 kW m−2 and 48.4 MJ m−2, respectively. The data from TGA indicate the addition of the flame retardants considerably enhances the thermal stability and char residues of PP/TPU blends. Compared with the PTE1 sample with 20 phr EG alone, the final char residues of the PTE2∼PTE4 samples decrease due to the addition of RPM, and decrease gradually with increasing the amount of EG. However, the T onset and T 10 wt% are improved because of the presence of RPM. The morphological observations exhibit the synergistic effects and mechanism of RPM with EG in the flame-retardant PP/TPU/EG/RPM composites. The reinforcement of thermal stability and flame retardancy is attributed to the compact and stable char layers promoted by RPM with EG acted as an effective heat barrier and thermal insulation.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was financially supported by the National Natural Science Foundation of China (51763002), the International Science and Technology Cooperation Project of Sichuan (2019YFH0047), the Opening Project of the State Key Laboratory of Polymer Materials Engineering (Sichuan University) (2017-4-02), the International Science and Technology Cooperation Project of Chengdu(2017-GH02-00051-HZ), and High-Level Innovative Talent-Training Program in Guizhou ([2016]5667).
