Abstract
Nanocomposite gel polymer electrolytes (NCGPEs) consisting of Zn(OTf)2 salt solution in 1-ethyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide ionic liquid (EMIMTFSI), entrapped in poly(vinyl chloride) (PVC)/poly(ethyl methacrylate) (PEMA) matrices, and dispersed with different concentrations of tin oxide (SnO2) nanofiller were prepared by simple solution casting method. The free-standing film of the composite gel polymer electrolyte (GPE) exhibited an optimum ionic conductivity value of 4.92 × 10−4 Scm−1 at ambient temperature. The gel composites developed predominant amorphous phase and porous morphologies, thus supporting the high ionic conduction as confirmed from X-ray diffraction (XRD) and scanning electron microscopic (SEM) studies. The complex formation properties of the materials were evaluated by attenuated total reflection-Fourier transform infrared (ATR-FTIR) spectroscopy technique. The dispersion of nanofillers in GPEs improved the thermal behavior of the composite system to 185°C, which was ascertained by thermogravimetric (TG) analysis. The electrochemical stability window of approximately 4.37 V with feasible plating/stripping process of zinc metal on stainless steel electrode was analyzed by voltammetric studies and all these features suggested the possibility of exploiting NCGPE films as electrolytes in batteries.
Introduction
A wide-reaching topic of importance from the last few decades is the accomplishment of sustainable energy and low-carbon society. Currently, new lithium ion batteries possessing higher energy density, longer cycle life, and elevated specific capacity are of prime importance, thus having a huge scope in the battery market for its application in hybrid vehicles, portable devices, and so on. 1,2 However, such lithium ion batteries could not be commercialized owing to their safety limitations and the association of expensive technology. 2 Hence, zinc-based battery technologies have been developed to reinstate lithium batteries and have recognized worldwide attention due to the subsequent reasons: (i) zinc metal is less toxic, cost-effective, abundantly existing, and (ii) insertion chemistry of zinc in cathodes is nearly comparable to the lithium. 1 -3
The energy storage devices, such as batteries, supercapacitors, and fuel cells, usually comprise of two electrodes and an electrolyte. The fabrication and performance of the highly flexible energy storage devices depend on the superior electrolyte materials employed, besides the selection of excellent new electrode materials. 4 Hence, the gel polymer electrolytes (GPEs) are receiving consideration nowadays, because they show promising characteristic features, such as flexibility, 5 higher ionic conductivity, 1 exceptional electrochemical properties, 5 and perform like both solid and liquid. 4 On the other hand, mostly studied GPEs are prepared based on host polymer, ionic salt, and organic plasticizers, such as ethylene carbonate, propylene carbonate, and diethyl carbonate. 4
Such organic solvent-based GPEs encountered many problems, such as volatility (restricted shelf life), flammability, leakage, corrosion with electrodes, and bulky design, thus limiting their applicability in environment-friendly and flexible energy storage devices. 2,4 Therefore, to develop safe and environment-friendly GPEs, organic solvents are replaced successfully with room temperature ionic liquids (RTILs), for example, 1-ethyl-3-methylimidazolium trifluoromethanesulfonate (EMITf), 1-butyl-3-methylimidazolium nanofluorobutanesulfonate (BMImNfO), 1-methyl-3-methylimidazolium-bis(trifluoromethyl-sulfonyl)imide (EMI-TFSI), 1-methyl-1-propylpyrrolidinium bis(trifluoromethylsulfonyl)azanide (MPPyrr-TFSA), and 1,2-dimethyl-3-propylimidazolium bis(trifluoromethylsulfonyl)imide (DMPImTFSI). 4 RTILs, consisting of large inorganic cations and relatively small organic anions, are widely studied owing to their negligible vapor pressure, nonvolatility, nonflammability, higher ionic conductivity, and better thermal and electrochemical stabilities. 6
Many polymers, such as poly(vinylidene fluoride), poly(vinylidene fluoride-hexafluoropropylene), polyacrylonitrile, polyethylene oxide, and poly(methyl methacrylate), 5 plasticized with a variety of RTILs have been adopted by many research workers to prepare GPEs exhibiting high ionic conductivity at ambient temperature. Hence, as a supporting framework 1 of GPEs, the present endeavor deals with the polymer matrices comprising of poly(vinyl chloride) (PVC) and poly(ethyl methacrylate) (PEMA) blended in the optimized ratio of 30:70. Blended polymer electrolytes augment ionic conductivity in the course of easy compositional change. 7
The widespread application of PVC is ascribed to the subsistence of pair of electrons at the chlorine atom where inorganic salts can be solvated. 8 However, the stiffening of the polymer backbone via dipole–dipole interaction between the hydrogen and chlorine atoms, 8 low impact strength, 9 and poor thermal stability of PVC confines its applications. 10 These particular intricacies might be evaded by blending PVC with another polymer, namely PEMA. 9,10 PEMA is a methacrylic ester polymer, having admirable thermal stability 10 and chemical and surface resistance, and presents high optical transparency. 11 PEMA is extremely appropriate for use as a polymer host for the conduction of ions in electrochromic devices because of its varied properties, such as superior mechanical strength, elasticity, and good adhesion onto substrates. The pair of electrons at the oxygen atoms of C=O and C−O−C2H5 groups of PEMA could easily coordinate with the cations from inorganic salt resulting in the formation of polymer–salt complexes. 11 The large pendant group in PEMA amplifies the production of free volume by restraining the close packing of the chains, thereby developing the interconnecting pathways and thus enhancing the conductivity. 12
Areas which still need more attention in GPEs are the capability of liquid retention, interfacial stability with the metal electrode, 13 and a low value of cationic transport number. 2 The dispersion of ceramic fillers, such as TiO2, Al2O3, SiO2, and ZrO2 in GPEs and referred to as “nanocomposite gel polymer electrolytes (NCGPEs),” is a useful approach to achieve increased transference number, improved mechanical, thermal, and electrode/electrolyte interfacial properties, and good electrochemical stability. 2,13 NCGPEs are a heterogeneous or homogeneous mixture of four component materials comprising of host polymer, salt, plasticizer, and nanofillers. 7
The presence of ceramic fillers in GPEs changes the dipole orientation of the polymer, 7 thus encouraging chain flexibility as well as ion dissociation owing to the availability of surface charge on the filler. Interaction of nano-sized fillers with anions and cations in the electrolytic species provides additional conducting pathways in the surrounding area of fillers for the transportation of ions. The concentration of space charge in the subinterface could be greatly enhanced by the dispersion of ceramic fillers into GPEs. 7 Further, the addition of nanofillers in GPEs reduces the degree of crystallinity and improves the ion transport in the system by slowing down the recrystallization kinetics of the polymer chain. 7 The increase in the transference number could be achieved by the presence of Lewis acid–base centers on the surface of the fillers, which efficiently increases the number of free cations. 14
In general, the ionic conductivity and surface area are inversely proportional to the particle size, 7 and hence, many existing factors, such as high concentration of ions, large surface area of the fillers, enhanced amorphous phase of the host polymers, Lewis acid–base interaction between the filler and polymer, and availability of space charge regions between GPEs and fillers, play an efficient role in enhancing various properties of NCGPEs. 7 In the light of all the above mentioned facts, the objective of the present endeavor was to investigate the influence of the addition of different concentration (i.e. 1, 2, 3, and 4 wt%) of tin oxide (SnO2) nanofiller on the structural, morphological, electrical, thermal, and electrochemical properties of GPEs comprising of optimized polymer blend, namely [PVC (30 wt%)/PEMA (70 wt%)] doped with 30 wt% zinc triflate [Zn(OTf)2] salt and plasticized with 80 wt% 1-ethyl-3-methylimidazoilium bis(trifluoromethylsulfonyl)imide (EMIMTFSI) ionic liquid (IL).
Experimental facts
Starting materials
Poly(vinyl chloride) (PVC, average molecular weight ∼233,000 gmol−1), poly(ethyl methacrylate) (PEMA, average molecular weight ∼515,000 gmol−1), Zinc triflate [Zn(OTf)2] with a molecular weight 363.53 gmol−1, ionic liquid (IL) 1-ethyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide (EMIMTFSI), nano-sized tin oxide (SnO2) with the particle size < 100 nm were procured from Sigma-Aldrich, St Louis, Missouri, USA. The Zn(OTf)2 salt and SnO2 were vacuum dried at 100°C for 1 h prior to use to remove the residual moisture content while the polymers PVC, PEMA, EMIMTFSI and solvent N,N-dimethyl formamide (DMF) [purchased from Sisco Research Laboratories (SRL), Mumbai, India] were used as received.
Preparation of NCGPEs
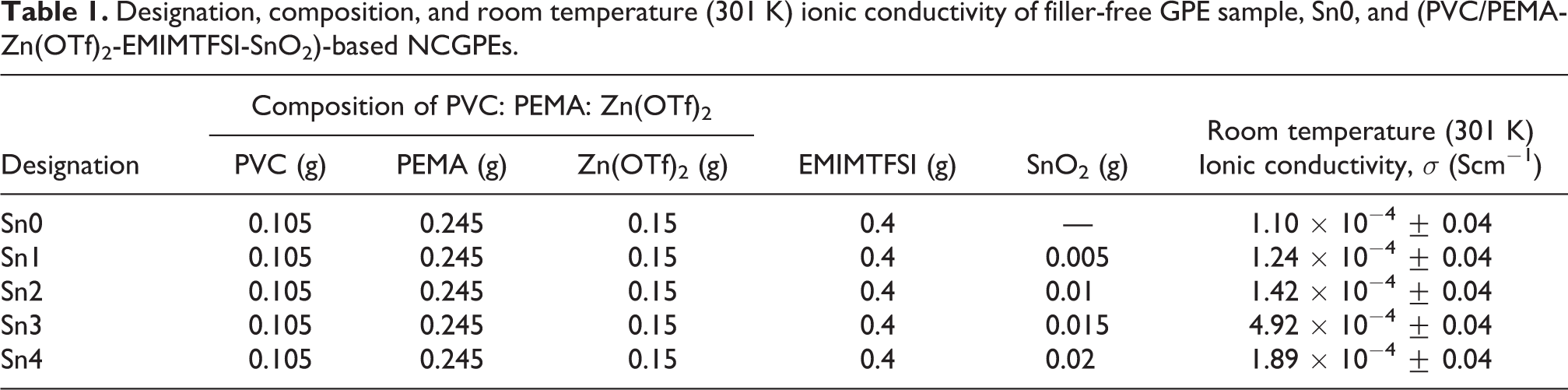
To achieve a series of highly conducting thin film NCGPEs, dried and ultrasonicated SnO2 of four different concentrations, namely 1, 2, 3, and 4 wt% ranging from 0.005 g to 0.02 g, were dispersed into the solutions containing the optimized amounts of polymer blend, salt, and RTIL (0.105 g of PVC, 0.245 g of PEMA, 0.15 g of Zn(OTf)2, and 0.4 g of EMIMTFSI) in 25 ml of a common solvent, namely DMF. The resulting mixtures were magnetically stirred continuously for several hours at room temperature until the formation of a visibly homogeneous solution. The resulting homogeneous solution was then cast onto glass petri dishes of uniform size and dried in a vacuum oven at 60°C for 24 h (pressure of 0.133 Pascal) to obtain free-standing composite gel films which were devoid of traces of solvent and moisture. After subsequent vacuum drying, the films were further left overnight in the glove box and stored in a highly evacuated desiccator for 2 days to remove any trace of residual moisture. This procedure yielded visually homogeneous free-standing NCGPE thin films of average thickness of 250 µm. The series of NCGPE system corresponding to 1, 2, 3, and 4 wt% dispersion of SnO2 filler having the typical configurations, PVC/PEMA-Zn(OTf)2-EMIMTFSI-SnO2, are designated as Sn1, Sn2, Sn3, and Sn4, respectively, whereas Sn0 denotes the filler-free GPE sample.
Characterization techniques
The X-ray diffraction (XRD) pattern of the NCGPEs was recorded over the Bragg angle (2
The electrical conductivity of all the prepared NCGPEs was determined to employ four-probe method with a computer-controlled Hewlett-Packard Model HP4284A Precision LCR Meter (Tokyo, Japan) over the frequency range from 1 MHz to 20 Hz with an excitation signal of 50 mV at ambient temperature (301 K). The NCGPE films were sandwiched between a pair of smoothly polished stainless steel blocking electrodes. The conductivity (σ) of the samples was calculated using the following equation:

where
The electrochemical stability and the reasonable reversibility of zinc plating/stripping mechanism of the best conducting NCGPE film on stainless steel electrode were determined by linear sweep voltammetry (LSV) and cyclic voltammetric (CV) studies at a scan rate of 10 mVs−1 at room temperature. The analyses were performed by an electrochemical analyzer (model CHI 1100 A, Lianelli, United Kingdom) in three-electrode cell configuration employing stainless steel disc as a working electrode and zinc metal (Zn) acting as both reference and counter electrodes.
The thermal stability of pure SnO2 and the best conducting specimen of the gel nanocomposite system were determined by thermogravimetric analysis (TGA) by means of TGA Q500 V20.10 Build 36 instrument (Bangalore, India) in the temperature profile 35–750°C at a heating rate of 10°C min−1 under inert (nitrogen) atmosphere. The surface morphology of the preferred samples was examined by S-3400 N scanning electron microscopic (SEM) instrument (Hitachi, Iowa City, IA, USA) with an electron beam energy of 11 kV at ambient temperature. An ultrathin layer of gold was coated on the surface of all the samples to prevent the accumulation of static electric charges on their nonconducting faces.
Results and discussion
Structural analyses: XRD and ATR-FTIR
To explore the perceptible changes in the structural characteristics of GPEs after the dispersion of nano-sized SnO2 filler, XRD patterns of pure Zn(OTf)2 salt,
15
filler-free gel sample Sn0,
15
pure SnO2 nanofiller, and various specimens of PVC/PEMA-Zn(OTf)2-EMIMTFSI-

Room temperature XRD of (a) Pure Zn(OTf)2 and (b) Sn0 gel sample.

Room temperature XRD pattern of (a) pure SnO2 and (b)–(e) NCGPEs, i.e. PVC/PEMA-Zn(OTf)2-EMIMTFSI-
On closer inspection, it may be clearly noted that the diffraction hump observed for filler-free gel polymer sample (Sn0) 15 in Figure 1(b) becomes relatively weaker, broader, as well as less significant 18 in all the gel composite complexes with the addition of ceramic nanofiller up to 3 wt%. This feature suggests the existence of possible Lewis acid–base interaction between the ether oxygen of the polymeric chain and Lewis acid sites of SnO2. 19,20 The filler owing to its large surface area prevents the polymer chain reorganization, thereby increasing the degree of disorder and amorphous phase of the resultant NCGPEs. 18 However, further increase of SnO2 content (>3 wt%) to Sn0 increases the intensity of the diffraction hump to a little extent 21 and the sample Sn4 exhibiting this kind of change is anticipated to be the one having least ionic conductivity among the gel composites. It is also quite obvious that the characteristic peaks of pure zinc triflate salt and SnO2 filler completely disappear in all the NCGPE samples, thus confirming the absolute dissolution and complexation of the salt and filler in gel composite complexes. On the whole, XRD spectra disclose the enhancement of the amorphous phase of the NCGPEs. 19
Figure 3(a) to (e) shows the ATR-FTIR spectra of pure SnO2 nanofiller and nanocomposite gel complexes recorded at ambient temperature, whereas the ATR-FTIR spectra of pure Zn(OTf)2, pure EMIMTFSI, and filler-free gel polymer sample (Sn0), which were already reported in our previous endeavor, 15 are presented in Figure 4(a) to (c) and considered here as reference spectra. The ATR-FTIR spectrum of pure SnO2 shows a broad band centered at 3470 cm−1, which is attributed to the presence of vibration of the hydroxyl group since the filler has the ability to retain some moisture content from the ambient atmosphere. The band at 1648 cm−1 is due to the bending mode of O−H bonds. 16,17 The role of nano-sized filler in GPE is to interrupt only the initial order of the polymer electrolyte complexes owing to its fine particle size (<100 nm). 22
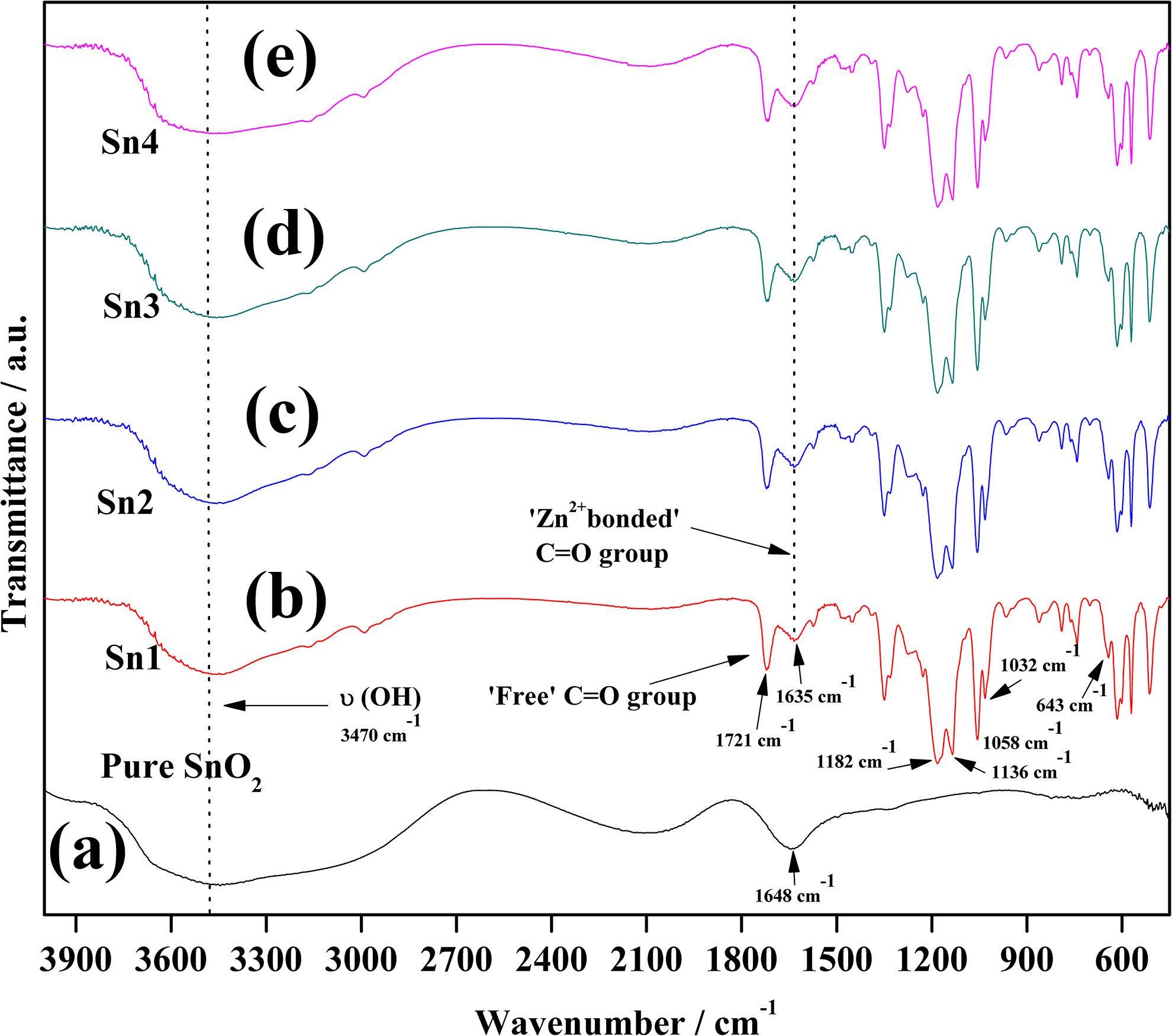
ATR-FTIR spectra of (a) pure SnO2 and (b)–(e) NCGPEs with varying concentrations of SnO2 in the wavenumber range from 4000 to 400 cm−1 at ambient temperature.

ATR-FTIR spectra of (a) pure Zn(OTf)2, (b) pure EMIMTFSI, and (c) Sn0 gel sample in the wavenumber range from 4000 to 400 cm−1 at ambient temperature.
The subsistence of the peak at 1721 cm−1, which is ascribed to the C=O group of PEMA, shows evidence of somewhat increased intensity upon addition of 1 wt% SnO2 nanofiller to Sn0. Usually, the oxygen atom of C=O group in PEMA acts as an electron donor resulting in the configuration of a coordinate bond with zinc ions; however, after the incorporation of filler in different weight percentage (1–4 wt%) to the gel polymer sample, the intensity of the this band increases divulging the configuration of hydrogen bonding between carbonyl oxygen (C=O) and the hydroxyl surface group (Sn−OH) of SnO2, thus resulting in the formation of −Sn−O…H…O=C− species. The improved coordination of Zn2+ cations and ceramic phase with C=O group of PEMA could be identified by the higher intensity of this peak. Furthermore, the occurrence of interactions between the nano-sized fillers and water molecules present in the gel system is substantiated by the appearance of a shoulder peak at 1635 cm−1. The position of C=O group and the shoulder peak remains unchanged beyond the addition of 1 wt% SnO2 concentration to the gel polymer sample Sn0, whereas the peak profile changes noticeably by an augment in the intensity of the peak. This feature of increased intensity pattern for the C=O group and the shoulder peak is more obvious up to 3 wt% filler loading. Further addition of nanofiller (4 wt%) results in an unaltered intensity pattern for those peaks and such characteristics might be accredited to the aggregation of excessive filler particles in the polymer matrices leading to the saturation state of coordination of filler and Zn2+ with C=O group rather than forming complexation. 15,23 The characteristic bands of NCGPEs between the region 3700 cm−1 and 3000 cm−1 gets broadened and become more intense with the addition of SnO2 filler. This might be due to the overlapping of characteristic absorption peaks of Zn(OTf)2 and nanofiller centered at 3500 cm−1 and 3470 cm−1, thus authenticating the contribution of hydroxyl groups in band interactions. 15 Even though the observed changes are up to a little extent in NCGPEs, this kind of optimistic suggestion of local structural changes sustained by the addition of nanofiller creates the path for Zn2+ ions to move freely and faster, thereby boosting the conductivity. 24
Electrical and electrochemical properties: Ionic conductivity, LSV, and CV
The value of ionic conductivity of NCGPE samples has been evaluated from the complex impedance diagram in the form of Nyquist plot. Figure 5 displays the Nyquist plot observed for four different compositions of SnO2 filler in GPE system (PVC/PEMA-Zn(OTf)2-EMIMTFSI-
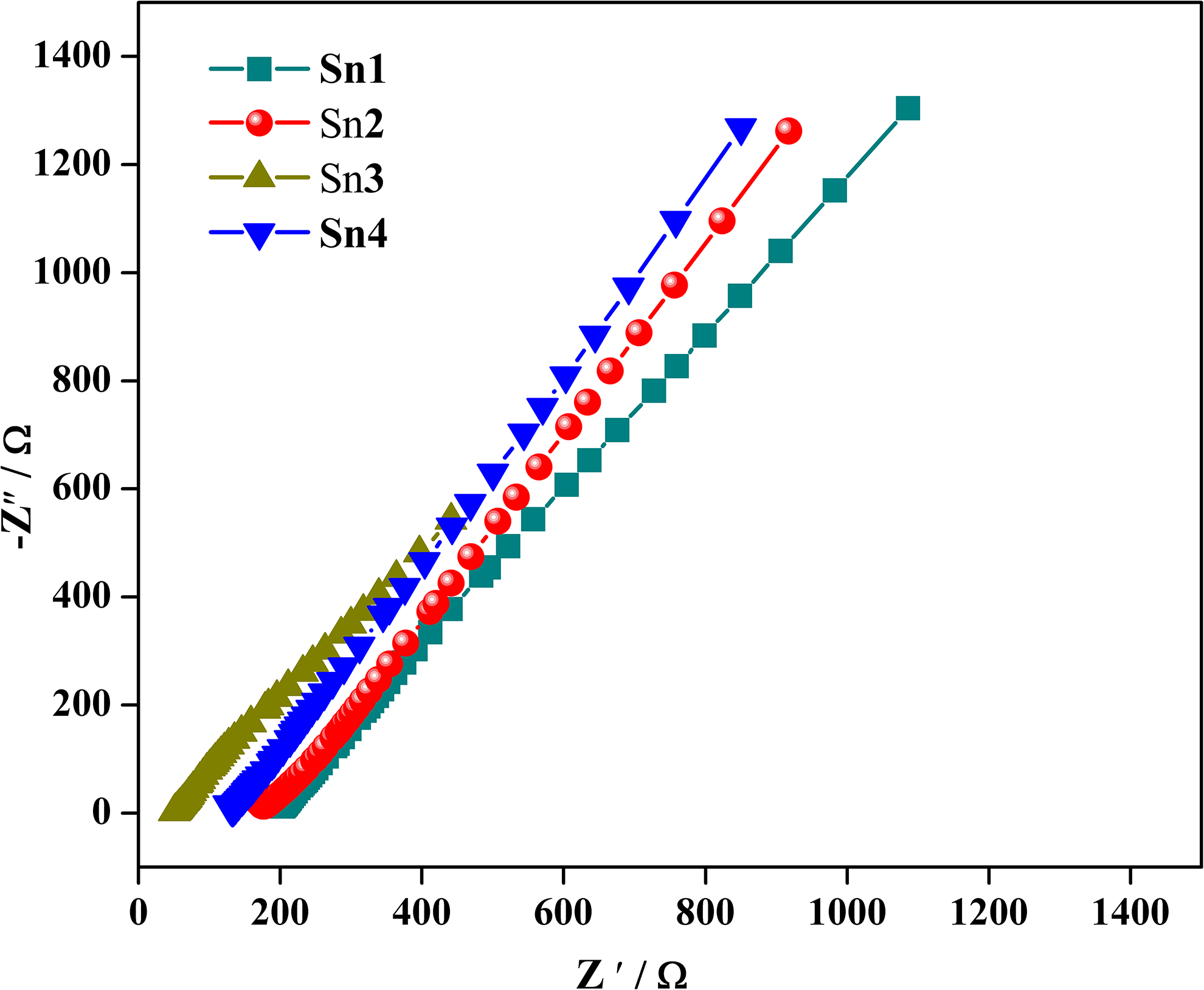
Complex impedance plot of NCGPEs for various concentrations of SnO2 filler at ambient temperature.
It has been observed from Figure 5 that the extent of intercept of the spike on the real axis at the high-frequency region of the impedance plot (representing the electrolyte bulk resistance,
The compositional dependence of the ionic conductivity of the nanocomposite GPE is displayed in Figure 6 with their corresponding ionic conductivity values exposed in Table 1. The ionic conductivity value of the gel sample Sn0 15 increases initially from 1.10 × 10−4 Scm−1 and reaches a maximum of 4.92 × 10−4 Scm−1 at 3 wt% of SnO2 content, and upon further increase of SnO2, the conductivity value drops to 1.89 × 10−4 Scm−1. The elucidation for an increase in the ionic conductivity value is envisaged on the fact that the Lewis acid–base interaction between the dispersed filler surface and anions compete with the interactions existing between the cations and anions, thus encouraging the dissociation of the salt by means of the formation of “ion-filler” complex. The establishment of the space charge layer between the GPEs and filler interface contributes to more conducting pathways for the transportation of Zn2+ ions, thus dominating over ion pairing effect. 28,29 In addition, the mutual effect of the weakening of polymer–cation association and increase in the uptake of the liquid electrolyte 30 by the nanocomposite films induced by the addition of fillers also attributes to the improvement of ionic conductivity.

Variation of the ionic conductivity of (PVC/PEMA-Zn(OTf)2-EMIMTFSI) gel electrolyte system for various concentration of SnO2 at ambient temperature.
Designation, composition, and room temperature (301 K) ionic conductivity of filler-free GPE sample, Sn0, and (PVC/PEMA-Zn(OTf)2-EMIMTFSI-SnO2)-based NCGPEs.
As filler content increases to 4 wt% in gel sample, the blocking effect of the existing conducting pathways owing to the increased conglomeration (pairing effect/aggregation) of excessive SnO2 filler reduces the conductivity value significantly and this feature is in perfect accordance with ATR-FTIR study. 28
The electrochemical stability window and the feasibility of stripping/platting mechanism of the gel composite film Sn3 are determined by LSV and CV measurements employing stainless steel as a working electrode and zinc (Zn) metal as reference and counter electrodes and are displayed in Figures 7 and 8. The LSV measurement is carried out from 0 V to 6.5 V at a scan rate of 10 mVs−1 at ambient temperature. The decomposition voltage of the gel composite film is identified by the onset of current in the anodic region and it commences at about 4.37 V for the composite film dispersed with 3 wt% of SnO2 content. The voltage stability factor of the NCGPE film has greatly improved when compared to the filler-free GPE sample and this may due to the interaction of Lewis acid sites on the anionic surface of the filler with Lewis base sites of electrolytic active species, thus retarding the decomposition of the zinc salt anion. Thus, the measured working voltage range of Sn3 appears to be adequate for the appliance of such nanocomposite GPE film as a solid-state separator in batteries. 30

Linear sweep voltammogram (LSV) of composite GPE sample Sn3 at ambient temperature.

Cyclic voltammogram (CV) obtained for the representative sample consisting of cell configuration, SS/Sn3/Zn, at a scan rate of 10 mVs−1 at ambient temperature.
On the other hand, the CV measurement is recorded over the potential range from −1.5 to +1.5 V at a scan rate of 10 mVs−1 for 10 consecutive voltammetric cycles at room temperature. The anodic peak centered at +1.12 V represents the stripping mechanism (oxidation) of the zinc metal, whereas a sharp cathodic current beginning at around −0.21 V symbolizes the plating process (reduction) of zinc metal on the stainless steel electrode. Hence, the occurrence of anodic oxidation and cathodic reduction occurring at +1.12 and −0.21 V versus Zn/Zn2+ suggests the feasibility of the system to be chemically reversible. Moreover, the peak currents decrease with increasing cycle numbers and this aspect may be due to the passivation phenomenon associated with the zinc metal surface. 26,31,32
Thermal investigation: TGA
To characterize the thermal stability of the sample, TGA is carried out on the GPE specimen Sn3 and is based on the measurement of mass loss of the membrane as a function of increasing temperature under nitrogen atmosphere. 33 The TGA curves of pure SnO2 nanofiller and Sn3 are shown in Figure 9. The TGA curve of pure SnO2 does not illustrate any significant weight loss up to 750°C and is owed to its high melting point. Thermogravimetric plot of Sn3 shows multistep weight loss regions varying from the temperature range 35–750°C, which may be elucidated as follows. The initial weight loss of 2.3% is due to absorption of moisture while loading the sample for analysis, loss of DMF solvent from the electrolyte, hygroscopic nature of zinc triflate salt, or due to the presence of any other volatile impurities in the nanocomposite gel polymer complexes. 25

TGA data for the specimen of pure SnO2 and the NCGPE sample Sn3 in the temperature range 35–750°C.
The second thermal degradation step starting from 185.5°C to 239.7°C with a weight loss of 26.4% is due to the result of degradation of blended polymer together with the considerable reduction in the molecular weight of the polymer chain owing to many degradation reactions, such as random chain scission reaction, depolymerization, dehydrochlorination, and intra- and intermolecular chain transfer reactions. 25 The third prominent weight loss of 52.5% in the temperature range from 286.2°C to 485.1°C is ascribed to the decomposition of zinc triflate salt and IL followed by the residual mass of about 18.8% above 485°C. The residual mass could be due to the presence of filler and carbonization enthused by the nitrogen and sulfur atoms during the thermal decomposition of the films. 25,34 The dispersal of nanofillers into the optimized GPEs works as an insulating surface thus averting the heat from expanding swiftly and increases the thermal stability of NCGPE sample noticeably to approximately 185°C. 35
Morphological study: SEM
The swollen pore-free surface morphology of the gel polymer sample Sn0, which was already reported in our earlier endeavor 15 and presented in Figure 10, has been changed into a porous network-like structure (as evidenced by the darker regions) by the addition of 1 wt% of SnO2 nanofiller as shown in Figure 11(a) to (c). The development of such porous morphology is one of the imperative factors in determining the ionic properties of an electrolyte in conventional batteries. The pores are highly responsible for the entrapment of fillers along with the large volume of plasticizer (salt + IL) in the cavities. 36,37 The enhanced ionic conductivity in composite GPEs may be attributed to the hopping of Zn2+ with the aid of pores 36 . The pore density increases and widens with an increase in the concentration of the filler and this might be due to the occupancy of more fillers in the pores and this observable fact reaches its maximum at 3 wt%. 38 High porous surface morphology of the gel composite film is accounted towards the interaction of dispersed filler with the polymer matrices and affinity with the solvent. 38 However, beyond 3 wt% addition of filler content to the filler-free gel sample, the density of the pores in the sample Sn4 appears to decrease and this sample exhibits the least ionic conductivity value among the gel composite system. Overall, the morphology of the electrolyte film typically depends on the nature of host polymer, salt, plasticizer, nanofiller, and the preparative method adopted. 36

SEM image of the filler-free gel polymer sample Sn0.

SEM image of the composite gel film dispersed with (a) 1 wt% SnO2 (Sn1), (b) 3 wt% SnO2 (Sn3), and (c) 4 wt% SnO2 (Sn4).
Conclusions
The synthesis and characterization of PVC/PEMA-based zinc ion conducting NCGPEs plasticized with EMIMTFSI and dispersed with nano-sized passive filler; SnO2 was reported in the present endeavor. The enhanced amorphous nature of the blended polymer matrices owing to the dispersion of nanofiller and the occurrence of complexation among the constituents of NCGPEs were evidenced from XRD, SEM, and ATR-FTIR studies. Gel electrolyte film incorporated with 3 wt% of SnO2 exhibited a maximum room temperature ionic conductivity value of 4.92 × 10−4 Scm−1 and better thermal stability up to 185°C. A good electrochemical stability window of 4.37 V and feasible zinc plating/stripping process of the gel composite sample signifies the potential applicability of such films as electrolytes.
Footnotes
Acknowledgments
One of the authors (C.M.S) of the present work gratefully acknowledges the financial support received in the form of WOS-A programme from the Department of Science and Technology (DST), New Delhi. The author (C.M.S) would also like to express gratefulness to Ms.R.Pavithra Marthandam and Ms.S.Kamatchi, St. Peters University for their kind help and support in carrying out ATR-FTIR measurements, Mr.R.Ramesh Kumar, Crystal Growth Centre, Anna University for his invaluable aid in XRD analysis, Central Leather Research Institute (CLRI), Chennai for their kind help in carrying out TGA measurement and would also like to thank NCNSNT, University of Madras for SEM analyses.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work received financial support from the Department of Science and Technology (DST), New Delhi, under the DST sanction no. SR/WOS-A/PS-32/2013 dated April 23, 2014.
