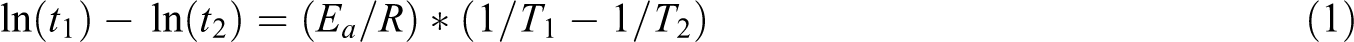Abstract
The discovery of oil fields in deepwater over the last decades led the oil and gas industry to the necessity of replacing the steel wire cables of the mooring systems of offshore platforms by polymeric ropes. These systems must be designed to work for at least 20 years without showing substantial loss in tensile strength or in their mechanical behavior along this period. However, some polymers present degradation by seawater through the process of hydrolysis, and the question whether it affects significantly the materials’ ultimate tensile strength arises. Accelerated hydrolysis tests were conducted in yarn samples of aramid at high temperature in order to use the Arrhenius correlation to predict their lifetime under service conditions. In order to decouple the total degradation into a thermal and a purely hydrolytic part, separate aging experiments were performed into a dry chamber and the conclusion was that thermal degradation does not play a significant role in the total degradation of aramid due to the water submersion at the temperatures tested.
Introduction
In the course of the past decades, offshore oil fields have been discovered in increasingly deeper waters, which pushed the oil and gas industry into replacing the traditional anchoring systems of the platforms based on steel cables and chains, since the water depth in which they are installed would tend to make the whole system potentially too heavy, to the point where the cables’ and chains’ self-weight could exceed the platform floating forces. In order to overcome this issue, the industry was motivated to search for lighter mooring systems, and one approach was to invest in the development of ropes made of lighter, polymeric materials. 1
Offshore structures and systems must be designed to a service life of at least 20 years 2 and, in the case of their anchoring ropes, the severe environment in which they operate (i.e. seawater, marine life, wave, wind loading acting on the platform etc.) might speed up their deterioration, which might lead to substantial change in their mechanical properties. One of the major aging phenomena that takes place in some polymers in oilfield service is hydrolysis, 3 by which the polymer chains are split when in contact with water, resulting in a rapid decrease in the molecular weight and posterior deterioration in some mechanical properties, in special the ultimate tensile strength. 4
For that purpose, the goal of this article is to assess the change in the ultimate tensile strength due to hydrolytic degradation of the aramid fiber Kevlar® 29 3300 dtex, a material that may be considered as an alternative to polyester considering mooring ropes of offshore platforms. Due to the fact that this kind of aging for this material is a long-term process, an accelerating technique must be employed in the experiments, and the use of high temperature water was chosen in order to speed up the hydrolysis reaction. In this article, care was taken to check whether it is necessary to uncouple the total degradation into a thermal and a purely hydrolytic part, so as to prevent spurious degradation mechanisms when calculating the lifetime of the materials.
Some results concerning durability of commercial aramid fibers in marine environments might be found in the literature, 5 -8 including a few tests regarding hydrolytic aging. In Derombise et al., 5 the authors studied degradation in the Twaron® and Technora® grades of aramid, using both seawater and deionized water, considering different temperatures, pH, and tensile load along the samples. In Morgan et al., 6 the aramid studied was Kevlar® 49, although the total degradation was not decoupled into hydrolytic and thermal. Both in Derombise et al. 5 and Morgan et al., 6 their approaches are different than the one presented here, where long-term behavior is extrapolated from short-term results using the Arrhenius equation. 9 Throughout the experiments of, 7 the Arrhenius relation was employed; however there was also no decoupling of the total aging phenomenon. In Hall and Knoff, 8 the total degradation was purely thermal, so no hydrolysis was considered. Results for the assessment of the aging of liquid crystal polymer (LCP) are found in Komatsu et al. 10 ; however their results are not based in seawater submersion and studies regarding this specific type of aging in LCP are rare in the literature. Among the most commonly used polymers used in mooring ropes, polyethylene terephthalate is found to be the most frequently tested to hydrolysis, both in pure or seawater. As examples of such studies one can cite. 11 -13
It is important to point out that this study aims to assess the macro effects of hydrolysis on each material and the evaluation of microstructural or chemical reactions goes beyond the scope of the article.
Accelerated aging
At the seabed, the water temperature in ultra-deep regions is assumed to be in the order of 4°C, which imposes serious restrictions to the development of real-time research in the field of hydrolysis, basically because of its long-term effects. For that reason, degradation experiments of this kind have to be conducted making use of accelerating techniques, and one stress variable that can be used in the case of hydrolysis is the water temperature. However, temperature itself imposes its own degradation mechanism, and for that reason parallel experiments concerning this type of deterioration exclusively must be performed, aiming to determine the necessity of uncoupling the overall process into a thermal and a purely hydrolytic part.
Performing such accelerated aging tests through the use of a stress variable can reduce drastically the overall time of the experiments, obtaining long-term parameters and results in a significantly shorter period of time. The Arrhenius equation (equation (1)) can be used to extrapolate the results obtained in the accelerated tests to predict the behavior of a chosen parameter in the long term, when the physical mechanism to be modeled is known to show an exponential nature. Here, based on two distinct experiment conditions, performed at two different water temperature, it is possible to calculate the activation energy of the degradation reaction using
where t 1 and t 2 are the time (days) to reach a limiting situation in conditions 1 and 2, respectively, Ea is the activation energy of the reaction (kJ mol−1), R is the gas constant and equal to 8.314 J mol K−1, and T 1 and T 2 are the temperatures (K) in which the limiting situation was achieved in conditions 1 and 2, respectively.
In equation (1), the limiting situation is typically when a chosen parameter reaches a determined intensity that will lead to the end of the material’s lifetime.
In this article, attention is focused on the residual ultimate tensile strength (or yarn break load (YBL)) of yarn specimens submitted to hydrolysis. From a safety point of view, 90% YBL is adopted as a threshold for degradation considering Arrhenius approach. Equation (1) is used to extrapolate the results at 80°C and 90°C down to the service temperature of mooring ropes (4°C) and at the sea surface (20°C). It is important to point out that these testing temperatures are considerably lower than the material’s glass transition, which is of approximately 600°C. 14,15
Materials and methods
Materials
This article conducted experiments in yarn samples of the para-aramid fiber Kevlar® 29 3300 dtex, which is a specific grade with titer of 3300 dtex specially designed to be used in ropes applied to marine operations.
Equipment
Hydrolysis
In order to accelerate the hydrolytic degradation of the materials, two different water temperatures were adopted as levels of the stress variable: 80°C and 90°C. Two tanks (Figure 1), each one for each water temperature, were designed and built by Policab—Stress Analysis Laboratory, at the Universidade Federal do Rio Grande (FURG), in the city of Rio Grande, Brazil.

Hydrolysis tanks for immersion in seawater.
Both tanks were filled with seawater collected at the Marine Station of Aquaculture, in the beach of Cassino, Brazil. Its salinity and pH were monitored along the course of the experiments and were kept constant at 29,500 ± 500 mg L−1 of NaCl and 8.5 ± 0.1, respectively. Salinity was measured using a Kasvi K52-100 refractometer (Kasvi, São José dos Pinhais, Brazil) and pH was monitored by a Kasvi K39-0014P pH meter (Kasvi, São José dos Pinhais, Brazil). Being sure that these parameters do not vary during the experiments, we exclude spurious modes of degradation from the final results. 16,17
The tank at 80°C (TK-80) was expected to degrade the materials in a slower rate, so it had 105 one meter long aramid yarn specimens submerged, of which 15 were also taken out every 7 days. This tank had a total of seven specimens withdrawn (7, 14, 21, 28, 35, 42, and 49 days).
Inside the tank with water at 90°C (TK-90) 60 aramid yarn specimens one meter long were inserted, of which 15 were taken out of the tank every 7-days interval in order to be tested for the residual YBL. That way, this tank had a total of four specimens withdrawn (7, 14, 21, and 28 days).
Temperature chamber
In order to assess whether the hot water of the hydrolysis tanks induces an undesirable, parallel aging mechanism in the polymers due to the high temperature, other sets of samples were placed into a dry temperature chamber (TC-80 and TC-90) (Figure 2) and tested in parallel to those immersed in seawater, considering the same withdrawn times.

Temperature chamber to assess thermal degradation.
Firstly, the chamber was set to 90°C and 60 aramid yarn specimens were placed into it. Fifteen samples were taken out every 7 days and tested for their YBL. After the four withdrawals, the temperature was lowered to 80°C and 105 new yarn specimen were inserted into the chamber. Every 7 days, 15 samples were taken out and tested for their YBL. That way, the same temperatures and times between the tanks and the temperature chamber experiments were preserved.
Tensile tests
The mechanical property assessed throughout the degradation processes was the materials’ ultimate tensile strength, so an Instron 3365 testing machine with a ±1 kN load cell was used for the tension tests. They were performed according to ASTM D7269, 18 a specific Standard regarding aramid yarn specimens. The samples tested were 500 mm long and the tests were conducted with displacement control of 250 mm min−1. The yarns were twisted around their axis with 60 rounds per meter. All tension tests were performed in dry samples that were left at room temperature for at least 24 h after their withdrawal from the tanks and the chambers.
In order to evaluate the evolution of the YBL of the material, it was first necessary to find that property of an unaged, unhydrolyzed specimen. Series of 30 tension tests were performed and the average break load was determined as the control group.
Results and discussion
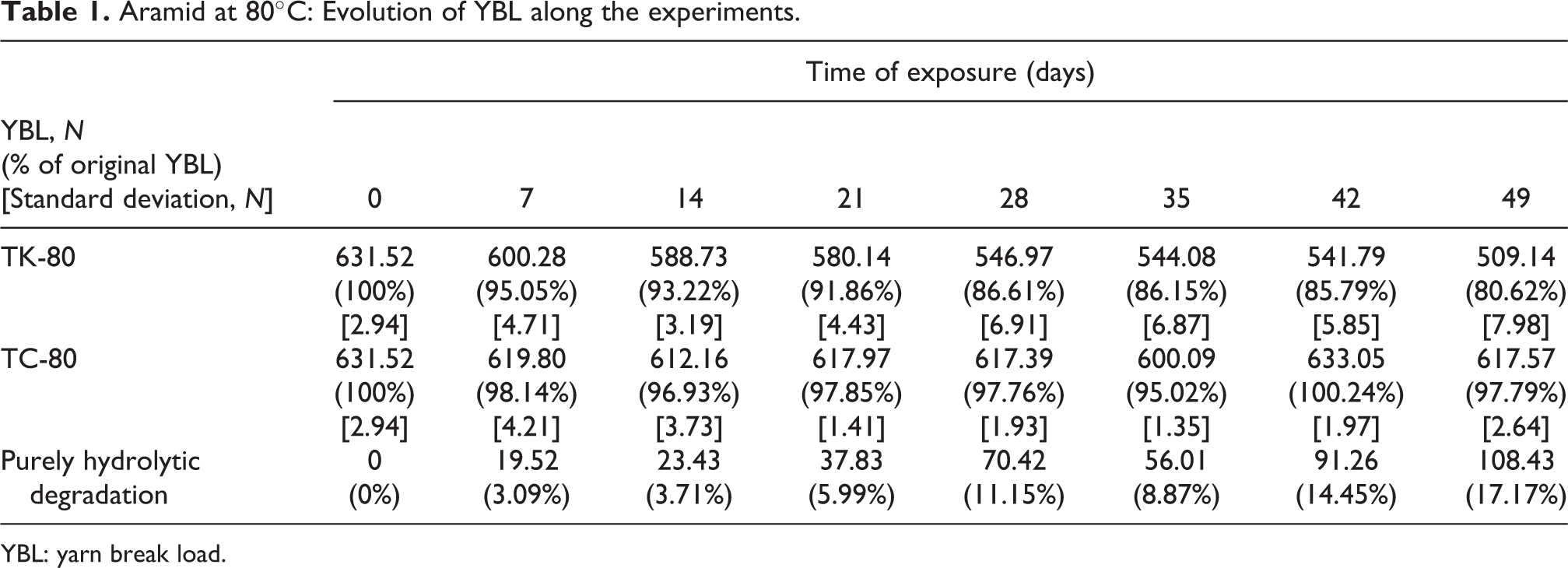
Figure 3 shows the evolution of the residual tensile strength of aramid, in terms of a residual YBL, throughout the experiments (tanks and chamber) for each temperature (80°C and 90°C). The total degradation (hydrolytic + thermal) obtained from the specimens placed into the TK-80 and TK-90 is depicted along with the thermal degradation due to temperature alone (TC-80 and TC-90), found by the exposure into the temperature chamber. Tables 1 to 2 resume the data of the graphs in Figure 3.

Residual YBL versus time of exposure for aramid samples at: (a) 80°C and (b) 90°C.
Aramid at 80°C: Evolution of YBL along the experiments.
YBL: yarn break load.
Aramid at 90°C: Evolution of YBL along the experiments.

YBL: yarn break load.
Analyzing the data in Tables 1 to 2, it is possible to see that, within the time-frame considered, the experiments regarding purely thermal degradation (TC-80 and TC-90) led to negligible aging, comparing to that of the specimen submerged in TK-80 and TK-90. Exponential fits were performed to the data of the residual YBL in the tanks and can be seen in Figure 3.
As mentioned, we seek the answer to the question: How long would it take until the material reaches a residual YBL of 90%, if it were immersed in seawater at 4°C and 20°C? Equation (1) is used to extrapolate results at 80°C and 90°C down to these conditions, but it is first necessary to find the activation energy Ea of the hydrolysis reaction, whose results are found in Table 3. There, the rows named as theoretical times represent t 1 and t 2 in equation (1) and refer to the times necessary to reach 90% YBL calculated by the exponential regression plotted in Figure 3 at temperatures T 1 = 353 K (80°C) and T 2 = 363 K (90°C), respectively. These relationships between immersion time and residual YBL are shown in detail in Figure 4.
Activation energy for the hydrolysis of aramid.

YBL: yarn break load.

Theoretical evolution of the residual YBL over time.
By using the results in Table 3 and equation (1), it is possible to determine the lifetime of aramid yarn specimens at a given temperature. Here, we seek for a 10% drop in YBL at 4°C and 20°C, which gives us the results in Table 4.
Lifetime prediction for aramid fiber at 4°C and 20°C

Conclusions
It is noticeable that even though aramid shows a tendency to degrade when submerged in seawater, it would take approximately 90 centuries until its tensile strength decreases by 10% at 4°C. In practice, it means that failure due to hydrolysis should not be a concern for the designers of mooring systems of offshore platforms considering the grade evaluated of aramid yarns.
ISO 17920 19 suggests, for aramid, that the samples must retain a residual strength of at least 90% YBL after immersion for 21 days in 80°C seawater, which is very close to the results obtained in the present study: 91.86% YBL (see Table 1). Analyzing the results in Table 4, it is possible to conclude that this recommendation can be considered as quite conservative.
The Arrhenius extrapolation methodology followed here is robust and can be applied to find the lifetime for any other percentage of YBL, any other service temperature or any other material. It is important to point out that performing separate tests with specimens subjected only to high temperatures is essential to determine whether the thermal aging plays or not an important role in the overall degradation of the material, so as not to overestimate the hydrolytic aging of the water baths. In the case studied, it was found negligible, so the calculations were performed using data of TK-80 and TK-90, not taking into account purely thermal degradation.
It is also important to make sure that a few basic parameters are kept constant along the baths, such as the water’s pH and salinity. That way one assures that no degradation due to spurious mechanisms takes place.
Footnotes
Authors’ note
Felipe Tempel Stumpfis also affiliated with Departamento de Engenharia Mecânica, Universidade Federal do Rio Grande do Sul, Porto Alegre, Brazil.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Petrobras in the framework of the cooperation agreement 0050.0086975.13.9. All the equipment used was acquired through this funding and is located at Policab—Stress Analysis Laboratory, in the Federal University of Rio Grande (FURG), Brazil.