Abstract
Aramid III is a high-performance fiber obtained by bulk structure modification based on the molecular structure of Kevlar fiber. Unlike the intermittent heat treatment method of Armos, Aramid III is prepared by continuous heat treatment technology. In this paper, the effects of heat treatment temperature, tension, and speed on fiber properties are studied by orthogonal experimental design. It is concluded that temperature is the most critical factor affecting fiber tensile strength and elastic modulus. The effects of different heat treatment temperatures on the chemical structure, surface morphology, aggregation structure, and the fiber’s mechanical properties and thermal properties are studied. Results show that: the chemical structure of the fiber keeps unchanged and surface morphology of the fibers are similar under the different heat treatment temperatures; the crystallinity of the fiber increases with the increase of temperature; the orientation degree of the fiber increases slightly after heat treatment; with the temperature increasing, the elastic modulus of the fiber increases, the tensile strength and elongation at break increase to a peak and then gradually decrease; the thermal decomposition temperature of the fiber is 533°C, and the decomposition rate is slightly different at different heat treatment temperature.
Introduction
There are two kinds of para-aramid products, the para-aramid homopolymer fiber represented by Kevlar1,2 and the para-heterocyclic aramid copolymer fiber represented by Russia Armos3,4 and Chinese Aramid III. 5 Kevlar is a kind of binary polymer that is polymerized by monomer p-phenylenediamine(PPDA) and terephthaloyl chloride(TPC). Armos and Aramid III is a ternary polymer polymerized by p-phenylenediamine(PPDA), terephthaloyl chloride(TPC), and 5(6)-amino-2-(4-aminophenyl)-benzimidazole(PABZ).
Armos and Aramid III are modified aramid fibers based on Kevlar molecular structure by adding PABZ. Compared with Kevlar fiber, Armos and Aramid III has higher mechanical properties, wear resistance, interfacial properties, and UV resistance, and its comprehensive properties are excellent.6–9
Both Armos and Aramid III are prepared by polycondensation of the same monomer, but they have two main differences: ① different proportion of PPDA and PABZ in molecular structures; ② different heat treatment methods and conditions (Armos is produced by intermittent while Aramid III is produced by continuous heat treatment).
In this paper, the influence of process factors, such as heat treatment temperature, tension, and speed on the properties of Aramid III are studied by online continuous heat treatment experiment, and the effects of temperature on the chemical structure, surface morphology, crystallinity, orientation degree, mechanical properties and thermal decomposition properties of aramid III are further explored. In this research, we mainly focus on revealing the mechanism of continuous heat treatment of Aramid III, which may provide help for the preparation of high-performance organic fibers.
Materials and methods
Materials
Aramid III precursor fiber (linear density: 75 tex; tensile strength: 8 cN/dtex; elastic modulus: 360 cN/dtex; elongation at break: 3.41%) is obtained from China Bluestar Chengrand Co., Ltd.
Sample preparation of Aramid III
50 L of compound solvent DMAC/LiCl is added into the reactor, and then 296 g of PPDA and 919 g of PABZ are added respectively and are completely dissolved in a compound solvent. When the temperature is controlled to less than 10°C, 1387 g TPC is added into the reactor for condensation polymerization to obtain homogeneous polymerization solution after 1.5 h reaction time (Figure 1①). Aramid III precursor is obtained by wet spinning (Figure 1②). The continuous heat treatment experiment is carried out by using the precursor fiber.
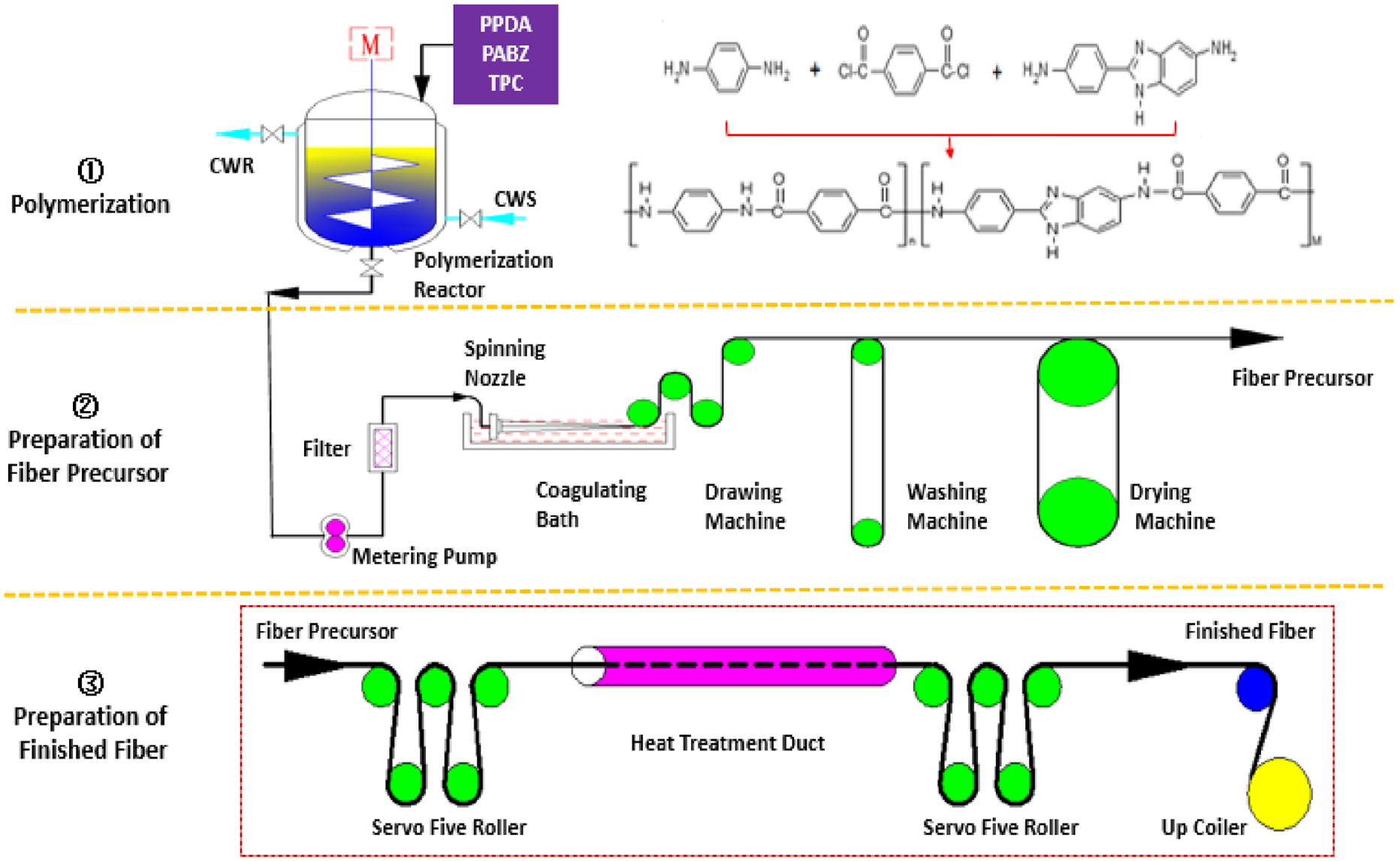
Experiment process: ① polymerization process, ② spinning process, and ③ continuous heat treatment process.
The guide roller leads the Aramid III precursor fiber into the high-temperature duct for heat treatment continuously and stably, and then pulled out by another set of guide rollers and wound by winder (Figure 1③). The orthogonal experiment method is used to study heat treatment process factors such as temperature, tension, and speed on fiber properties by using the L25 (53) orthogonal table. After finding out the most critical factors which affect fiber properties, further research and discussion are carried out. The factors and levels of the orthogonal experiment are shown in Table 1.
Factors and levels of orthogonal experiment.

Testing and characterization
After heat treatment, the chemical structure, morphology, crystallinity, and orientation degree of Aramid III fiber are analyzed by Fourier transform infrared spectrometer (FTIR), scanning electron microscope (SEM), X-Ray Diffractometer (XRD), and the sound velocity orientation meter, respectively. An electronic universal testing machine tests the fiber’s mechanical properties, and the fiber’s thermal decomposition property is tested by a thermogravimetric analyzer (TGA).
The FTIR test is conducted with the Nicolet 6700 Fourier infrared spectrometer of Thermo Electron Corporation. The samples are tested by ATR method, with infrared scanning wavenumber range of 4000–400 cm−1, wavenumber accuracy of 0.01 cm−1, resolution of 0.09 cm−1, scanning times of 16.
The SEM is conducted with a scanning electron microscope of JSM-5900LV type of JEOL. The test conditions are acceleration voltage 20 kV, SE mode, working distance 11 mm, magnification 5000 times, samples sprayed with gold.
Philips X’pert Pro MPD diffractometer is used to analyze the crystallinity of the samples. The test conditions are tube voltage 40 kV, tube current 40 mA, Cu target, Ka ray, wavelength λ = 1.5406 × 10−10 m, scanning speed: 2°/min, 2θ scanning range: 5°–50°, wide-angle diffraction scanning.
X-ray diffraction is recognized as one of the most widely used methods with a definite meaning among many methods for determining crystallinity. 10 The crystallinity (Xc) of polymer determined by the X-ray diffraction method is defined 11 as:


Where:
According to the theory of X-ray diffraction, the scattering intensity of unit mass of crystalline and amorphous samples in the whole reciprocal space is the same, and the value of K should be equal to 1. 12 Therefore, the formula for determining the crystallinity (Xc) of the polymer by X-ray diffraction method is as follows:

The relative integral intensities of crystalline and amorphous peaks in different samples are calculated. The crystallinity Xc can be calculated according to formula (3).
The crystallinity of fiber is calculated by the computer peak division method. The fitting function of peak division is the Gauss function. The crystallinity measured by X-ray in this paper is not the absolute crystallinity of the fibers. However, these relative values can be used to compare the crystallinity difference of the fibers laterally.
SCY-III sound velocity orientation instrument of Donghua University is used to measure the sound velocity figure of fiber samples by the multi-point test method; the sound velocity orientation factor (f) of fiber is calculated using the Moseley formula.13–15
The sound velocity method 16 calculates the orientation degree of materials by measuring the propagation velocity of sound waves in fiber materials. The principle is based on the anisotropy of acoustic wave propagation caused by the orientation of macromolecular chains in fiber materials. Any deformation of the fiber during sound wave transmission is the sum of intramolecular and intermolecular deformation. The formula of orientation degree is as follows:

Where:
CMT6503 electronic universal testing machine is used, the tensile speed is 25.4 mm/min, the pre-tension is 0.02 cn/dex, 18 and the clamping distance is 170 mm.
Q500 thermogravimetric analyzer of TA company is used. The test temperature range is 50°C −900°C, the heating rate is 10°C/min, the nitrogen flow rate is 100 mL/min.
Results and discussion
Orthogonal experiment results of continuous heat treatment
The effect of process factors on fiber mechanical properties is studied by continuous heat treatment (Figure 1③). An L25 (53) orthogonal table is used to design the orthogonal experiment, in which the temperature, speed, and tension are selected as factors, and five levels are selected for each factor. A total of 25 experiments are conducted. The experiment results are shown in Table 2.
Orthogonal experiment results of continuous heat treatment for Aramid III.

Statistical analysis is performed on the orthogonal experiment data. The experiment results calculate the sum and average values of tensile strength and elastic modulus of three factors at five different levels. Then the range of tensile strength and elastic modulus of each factor at different levels is calculated (Tables 3 and 4), and the influence of temperature, tension, and speed on Aramid III fiber is obtained (Figure 2).
Statistical analysis of the influence of different factor levels on tensile strength.

Statistical analysis of the influence of different factor levels on elastic modulus.


The effect of temperature, speed, and tension on the mechanical property of the fiber.
Temperature, tension, and speed are the three main influencing factors in the continuous heat treatment process of Aramid III fiber. The order of influence on fiber tensile strength is temperature > speed > tension, and the influence on fiber elastic modulus is temperature > tension > speed. Therefore, in the continuous heat treatment process of Aramid III, the temperature has the most significant influence on the fiber’s tensile strength and elastic modulus and is the most critical factor.
The following study focus on the influence of temperature on the chemical structure, surface morphology, crystallinity, orientation, mechanical properties, and thermal properties of Aramid III fiber during continuous heat treatment.
Fiber structure
Chemical structure
The infrared spectra of Russian Armos fiber and Aramid III fiber prepared at different heat treatment temperatures are shown in Figure 3.

IR spectrum of Armos and fiber prepared by different heat-setting temperature.
Figure 3 shows that the N-H stretching vibration absorption band in the amide group of Aramid III molecular structure is near 3300 cm−1. The absorption band at near 1650 cm−1 is the stretching vibration of the C = O in the amide group (amide I band), the absorption band at near 1475 cm−1 is the bending vibration of the N-H in the amide group (amide II bands), and the C-N stretching and N-H bending coupling vibrational absorption band of the amide group (amide III bands) are found near 1260 cm−1. The absorption band at near 1510 cm−1 is the skeleton vibration of the benzene ring, and the absorption band at near 830 cm−1 is the bending vibration of the C-H in the substituted benzene. The absorption band near 1640 cm−1 is the stretching vibration of the C = N group in the imidazole unit. The weak absorption peak near 1590 cm−1 is the characteristic ring vibration of conjugation between benzene and benzimidazole rings. 19
The IR spectra of the Aramid III fibers prepared at different heat treatment temperatures are analyzed. The absorption bands of each fiber are the same, showing that the chemical structure of Aramid III is changed little under different heat treatment temperature conditions, and no functional group is produced or disappears in the different heat treatment temperatures.
The infrared absorption bands of Russian Armos fiber and Aramid III fiber prepared at different heat treatment temperatures are compared. The positions of the absorption bands of the two fibers are the same, but the intensity of the absorption bands is slightly different. The two kinds of fibers have the same monomer but different monomer ratios.
Surface morphology
Surface morphology is critical to the surface properties of the fiber, especially when the fiber is used in preparing high-performance composites. The SEM images of Russian Armos fiber and Aramid III fiber are shown in Figure 4.

SEM diagram of Armos and fiber prepared by different heat-setting temperature.
It shows no micropores and cracks on the surface of Aramid III fibers from 340°C to 420°C, but there are a small number of strip grooves along the fiber axis caused by the process characteristics of wet spinning fibers. There is no noticeable difference in the surface morphology of the above five fiber samples, indicating that the heat treatment temperature has no pronounced effect on the surface morphology of Aramid III.
In contrast, the surface of Armos is rougher. There are more and deeper strip grooves on the surface of Armos fiber in Figure 4. Both fibers are manufactured by wet spinning technology, only different heat treatment processes (Armos is produced by intermittent while Aramid III is produced by continuous heat treatment). Therefore, the special surface morphology of Armos should be the result of its intermittent heat-treatment process.
Crystallinity
Fiber is a kind of two-phase material. The crystalline diffraction peaks in the X-ray diffraction curve tend to be overlapped partly due to dispersion. The crystalline peaks and amorphous peaks are generally completely overlapped or mostly overlapped. Therefore, the key to calculating the crystallinity is accurately decomposing the X-ray diffraction intensity curve of the material into crystalline and amorphous parts.
The WAXD diffraction spectrum and crystallinity value of Aramid III fiber prepared at five different test temperatures are shown in Figure 5.
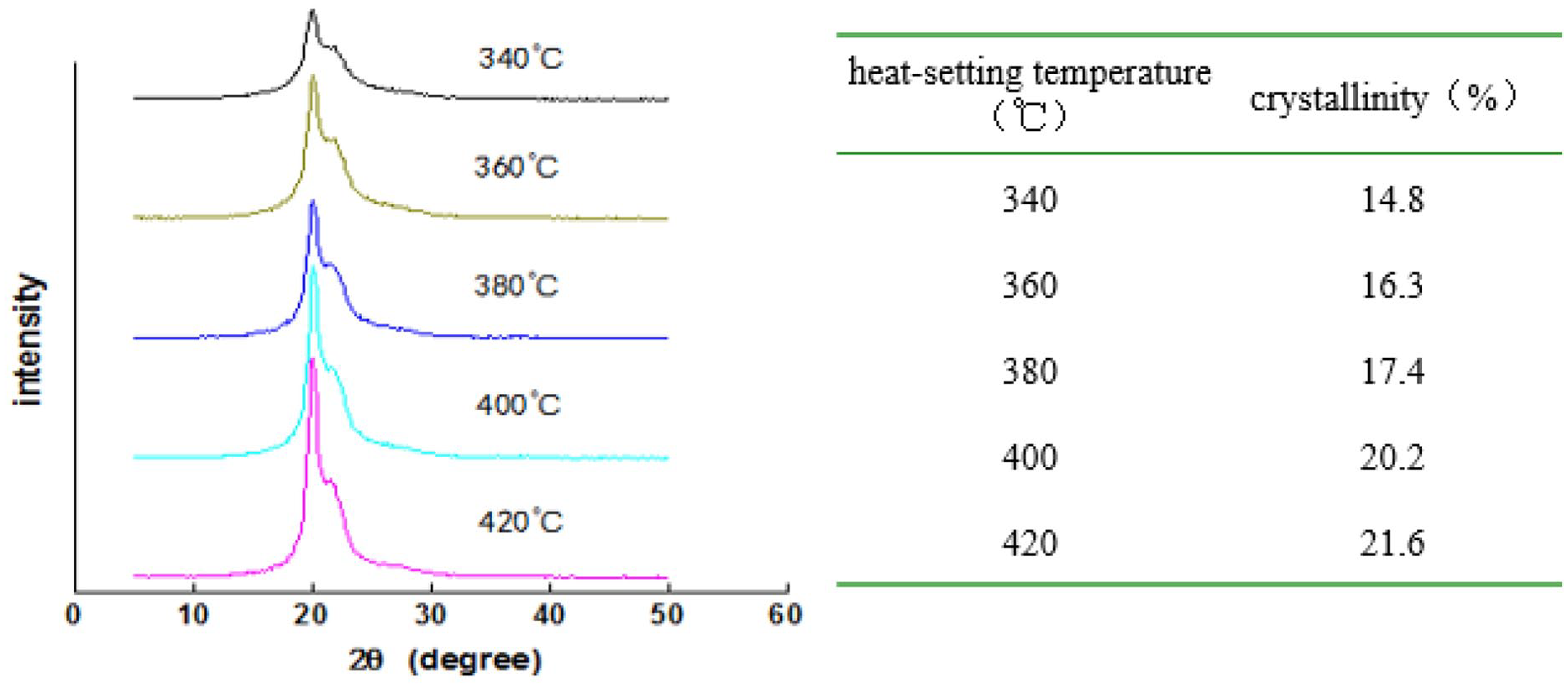
XRD spectrum and crystallinity of fiber prepared by different heat-setting temperature.
The X-ray intensity in the WAXD spectrum of Aramid III fiber includes two parts: coherent scattering and incoherent scattering. Before peak splitting, the incoherent scattering part should be deducted from the test curve. Johnson’s deduction method is adopted, that is, two points of 2θ = 5° and 2θ = 50° are connected into a straight line as the bottom line.
After deducting the background, the Gauss curve is used to express the peak shape functions of the crystal and amorphous regions of the fibers in this experiment. The least-square method is used to fit the peak in the peak separation program of Origin software, 20 the crystallinity value of the fiber is calculated.
It can be seen from Figure 5 that with the increase of heat treatment temperature, the diffraction peak intensity of the fiber becomes more significant, the peak shape is getting sharper, which qualitatively indicates that the crystallinity of the fiber increases with the increase of temperature. The crystallinity quantitatively calculated by the computer peak separation fitting program is entirely consistent with the results reflected by the diffraction peak spectrum.
Aramid III fiber is a block copolymer with a rigid molecular chain. The above XRD results show that the flexibility of the rigid molecular chain of the fiber increases with the increase of temperature. In the same heat treatment time, the diffusion and directional arrangement of the chain segments to the crystalline surface are faster, and the crystallization ability of the polymer is enhanced, so the crystallinity of the fiber is higher.
Orientation degree
The orientation degree of fiber is an important parameter to characterize the supramolecular structure of fiber materials. The orientation of polymer chains along the tensile direction increases the axial mechanical strength and decreases the radial mechanical strength, resulting in the anisotropy of the axial and radial mechanical properties in the aggregated structure.
The influence of heat treatment experiments at different temperatures on fiber orientation is shown in Table 5.
Sound velocity orientation of the fibers.

The orientation degree of Aramid III fiber precursor has reached 87.9%. The reason is that the whole rigid molecular chain is easy to stretch under the action of external force and will be arranged along the direction of the force, forming a higher overall orientation. After passing through the heat treatment duct, the orientation degree of Aramid III fiber is further improved, but the increase is slight. The temperature is higher in the heat treatment process than the Tg of Aramid III (≈275°C). 17 Therefore, the fiber molecular chain segment will further adjust its configuration and orient under the action of external force. The process eliminates the structural defects of the internal stress of the fiber and forms a more uniform and compact structure.
Fiber property
Effect of heat treatment temperature on mechanical properties of fiber
The effects of different heat treatment temperatures on the tensile strength, elastic modulus, and elongation at break of Aramid III fiber are investigated with other process parameters unchanged, as shown in Figure 6.
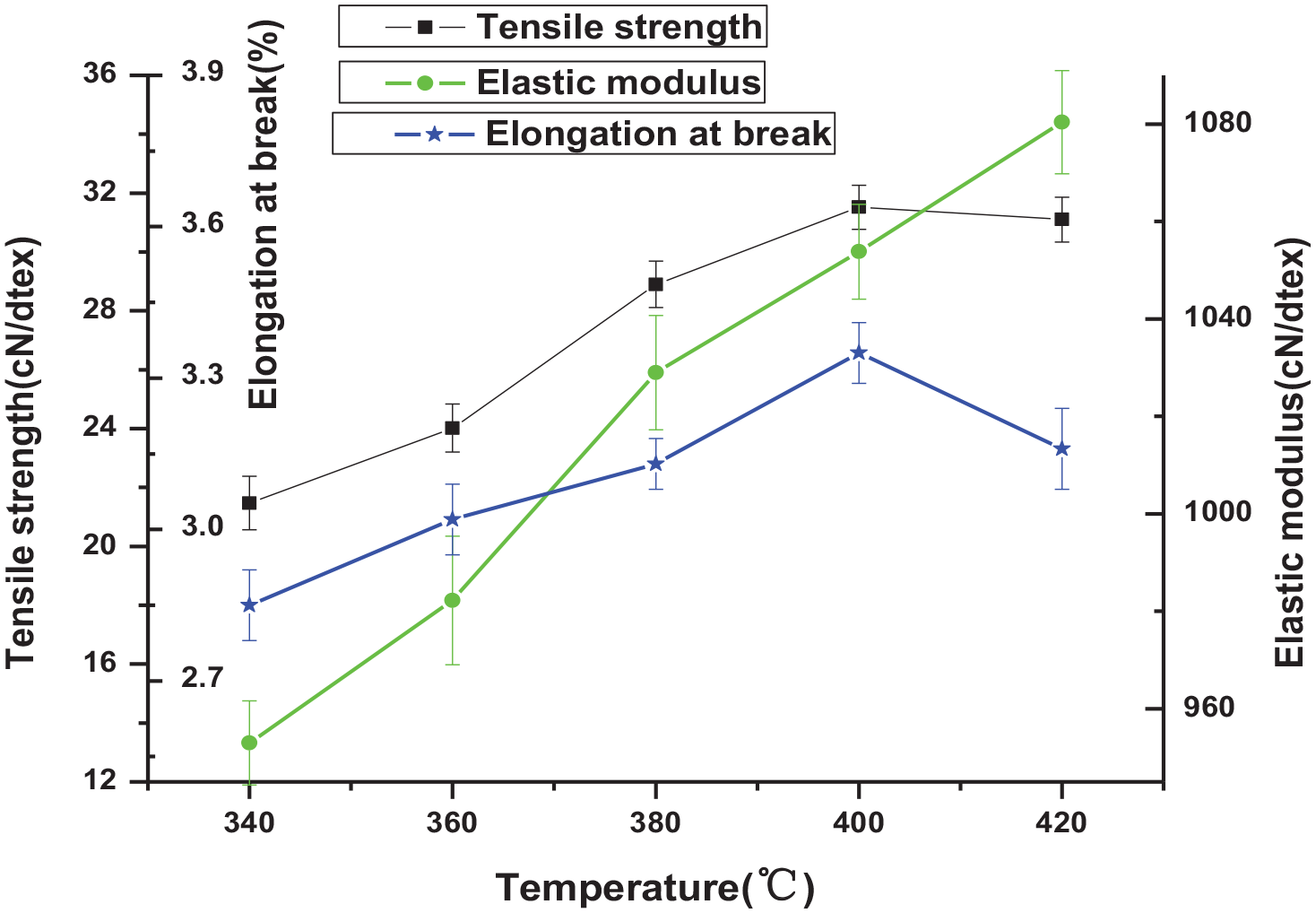
Influence of heat-setting temperature on the mechanical properties of fiber.
It can be seen from Figure 6 that Aramid III fiber is a brittle material with high strength, high modulus, and low elongation. With the increase of temperature, the tensile strength of fiber increases gradually, and when reaching a specific temperature, the tensile strength decreases gradually with the increase of temperature. Similarly, at the initial stage, the elongation at break of the fiber increases with the increase of temperature, reaches the maximum value, and decreases with the increase of temperature. In the scope of test temperature, the elastic modulus of the fiber increases gradually with the increase of temperature.
The increase of temperature is conducive to higher crystallinity of the fiber, leading to the decrease of the elongation at break and increasing the fiber’s elastic modulus.
Therefore, according to the different application fields of the fiber, the corresponding process parameters can be changed, and the process can be controlled to obtain the Aramid III fiber applied to different purposes to meet the requirements of different customers.
Effect of heat treatment temperature on thermal properties of fiber
Thermogravimetry (TG) is used to measure the relationship between mass and temperature under program-controlled temperature. The decomposition temperature of the materials is determined by the tangent method, which is the intersection of the baseline and the tangent line in the descending section of the TG curve, the epitaxial starting temperature.
The derivative thermogravimetric (DTG) curve reflects the relationship between the mass change rate and temperature. Compared with TG, DTG can more accurately reflect the initial reaction temperature, the temperature to reach the maximum reaction rate, and the temperature to terminate the reaction. 21
The TG and DTG spectrum and thermal property data of Aramid III fibers are shown in Figure 7.

TG and DTG spectrum and thermal property data of fiber prepared by different heat treatment temperature.
It can be seen that the decomposition temperature of Aramid III fiber obtained under five temperature conditions in the test is the same, all of which are 533°C, the decomposition rate is slightly different. The residual carbon content at 800°C shows that with the increase of heat treatment temperature, the decomposition rate of finished fiber decreases slightly, and the thermal stability of the fiber is getting better. The DTG curves show that the initial reaction temperature, the maximum reaction rate temperature, and the reaction termination temperature of the five different fibers are the same.
The mechanical properties, crystallinity, and orientation of Aramid III fibers prepared at different heat treatment temperatures are different, but the thermal decomposition temperature is the same, and the decomposition rate is slightly different. It shows that the heat resistance of the fiber mainly depends on the macromolecular chain structure of the polymer, while the aggregation structure of the fiber has little effect on it.
Conclusions
In the continuous heat treatment process of Aramid III, the temperature is the most critical factor, dramatically influencing the fiber’s tensile strength and elastic modulus. The chemical structure of Aramid III is the same under different heat treatment temperature conditions, and no functional group is produced or disappears in the different temperatures. The infrared absorption peak of Aramid III and Armos fiber is the same, but the peak intensity is slightly different. It indicates that the two fibers have identical monomers but different monomer ratios. There is no noticeable difference in the surface morphology of the five fiber samples, and the heat treatment temperature has no obvious effect on the surface morphology of Aramid III. The surface of Aramid III with continuous heat treatment process is relatively smooth, while the surface of Armos with intermittent heat treatment process is relatively rough. Within the experimental temperature range, the crystallinity of the fiber increases with the increase of heat treatment temperature. Compared with the Aramid III precursor, the finished fiber’s orientation is higher after heat treatment. In the heat treatment temperature range of 340°C–420°C, with the increase of temperature, the tensile strength and elongation at break of the fiber increase and gradually decrease after reaching the maximum value, while the elastic modulus of the fiber increases with the increase of temperature. Under different heat treatment temperatures, the thermal decomposition temperature of Aramid III keeps unchanged at 533°C. However, the fiber’s decomposition rates are different. Compared with the lower temperature, the decomposition rate of fibers obtained by heat treatment at 420°C is slower.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is financially supported by the Sichuan Science and Technology Planning Project (Project No. 2019ZDZX0016). Project name: High efficient preparation of Aramid III and its composite products research and application.
