Abstract
This work investigates the effects of core–shell rubber (CSR) nanoparticles on the curing behaviour and thermo-mechanical properties of an epoxy using differential scanning calorimetry and dynamic mechanical thermal analysis approaches. Interaction between CSR nanoparticles and epoxy matrix is detected at a temperature of approximately 97°C in the curing process. This results in an increase in the glass transition temperature (Tg ) of the cured nanocomposites. Given the semi-dynamic curing schedule, the curing process of all the epoxy nanocomposites consists of an abrupt onset stage followed by a slow diffusion-controlled stage. Higher temperature is required to initiate the curing for the epoxy nanocomposites with higher loading of CSR nanoparticles. This is attributed to the physical changes caused by the addition of CSR nanoparticles, such as the increase in the viscosity and the reduction in the density of the reactive groups. The storage modulus of the epoxy decreases in the glassy region but remains constant in the rubbery region due to the incorporation of CSR nanoparticles.
Keywords
Introduction
Epoxies are widely used as matrices of fibre-reinforced plastic composites and structural adhesives due to their prominent properties such as high stiffness, high strength and good thermal stability. However, epoxies exhibit low fracture toughness and poor resistance to crack growth due to the highly cross-linked structure. The addition of second-phase modifiers such as rubber particles, 1 –3 silica particles, 4 –6 carbon nanotubes 7,8 and graphene 9,10 demonstrates a promising method to improve the fracture toughness of epoxies. Among different types of modifiers, the rubber particles show superior performance in epoxy toughening.
Extensive research, as presented in previous studies, 11 –14 has been performed to study the addition of core–shell rubber (CSR) nanoparticles on the mechanical properties, thermal stability and fracture behaviour of epoxies and to establish the structure–property relationship of rubber–epoxy composites. It is generally accepted that blending CSR nanoparticles in epoxies significantly increases the fracture toughness and reduces the stiffness and strength. However, various effects of CSRs on the thermal stability, that is, glass transition temperature (T g), of epoxies were reported. For example, Giannakopoulos et al. 11 and Carolan et al. 12 observed negligible effect of adding CSR nanoparticles on the T g of epoxies, while a slight reduction in the Tg of an epoxy was reported due to the addition of 20% CSRs in the work of Chen et al. 14 In contrary, our previous work 1 demonstrates that blending CSR nanoparticles into an epoxy resulted in a moderate increase of the T g. To fully understand the thermal property–structure relationship of CSR-modified epoxies, it is critical to characterize the network structure and the topology of the nanocomposites. This could be achieved by studying the curing behaviour and the thermo-mechanical properties of the epoxy nanocomposites. To date, limited to no work has been performed to study the effects of CSR nanoparticles on the curing behaviour of the CSR-modified epoxy nanocomposites. Moreover, the interaction between CSR nanoparticles and epoxy matrix in the curing process has never been in situ detected.
Differential scanning calorimetry (DSC) analysis is widely used to study the curing of epoxies 15 –18 due to its ability to give a quantitative measurement of the amount of reaction based on the calorimetric measurement. Nevertheless, the sensitivity of DSC is insufficient to capture the CSR/epoxy interaction due to the negligible calorimetric change generated by the interaction/reaction between the CSR nanoparticles and the epoxy matrix. Hence, no conclusive result is available using solely DSC technique. Dynamic mechanical thermal analysis (DMTA) is very sensitive for detecting the changes in molecular mobility of polymers via in situ measuring the storage modulus and loss modulus. This technique has the potential to monitor the CSR/epoxy interaction. Many studies 19,20 used DMTA approach to study the curing process of resins by employing a sandwich specimen. In that setup, a substrate was fixed to the top (drive) clamp, and a very thin layer of liquid resin was sandwiched between the substrate and the studs mounted on the bottom clamp. The major drawback of this setup is that very small amount of resin is sandwiched and tested between the substrate and the stud. This leads to severe challenge upon the sensitivity of the DMTA to detect the CSR/epoxy interaction.
In the current work, a novel DMTA setup, in conjunction with DSC analysis, is proposed to study the curing behaviour of CSR-modified epoxy nanocomposites. The interaction between CSR nanoparticles and epoxy matrix is in situ monitored, for the first time, in the curing process. The effects of the CSR/epoxy interaction on the thermo-mechanical properties of the cured CSR-epoxy nanocomposites were also investigated.
Experimental
The epoxy resin is a standard diglycidylether of bisphenol A (DGEBA) epoxy resin (Epon828 from HEXION, Germany) with an epoxy equivalent molecular weight between 185 g eq−1 and 192 g eq−1. The curing agent is dicyandiamide having fenuron as the accelerator. The CSR nanoparticles, Zeon F351 from Nippon Zeon Chemicals, is supplied as dry powders. They have a structure of an acrylic rubber core surrounded by a poly methyl methacrylate (PMMA) shell. The mean diameter of the CSRs is 203.4 nm, and the mean diameter of the rubber core is 169.6 nm, giving a thickness of the shell of 16.9 nm. 1 The mixing of DGEBA resin, hardener and CSR nanoparticles consists of two steps. Step 1 is the mechanical stirring using an IKA RW20 digital mixer (IKA, Germany) operating at 2000 r min−1 for 1 h. In step 2, an ESCO shear mixer (ESCO-Labor AG, Switzerland) equipped with a vacuum pump was employed for high-shear vacuum mixing for 2 h at 60°C to improve the CSR dispersion and remove the air bubbles. CSR-modified epoxy nanocomposites with following CSR contents were processed: 0, 8, 16, 22, 30 and 38 vol.%. They are referred to as Matrix, C8E, C16E, C22E, C30E and C38E, respectively, in this article.
DMTA on the curing process of the nanocomposites was carried out in a DMA 242E (NETZSCH, Germany) machine using a novel sample holder set. The curing sample holder consists of a container and a pushrod (see Figure 1). In the test, the uncured resin was added into the container to immerse the pushrod ball and then the pushball moved dynamically to detect the storage modulus and loss modulus of the resin. The analysis was carried out under nitrogen atmosphere with a 20-μm amplitude at 1 Hz frequency in compression mode. Then 2 ml uncured nanocomposite was measured and added into the container using a syringe. The temperature schedule for the curing contained a dynamic stage, heating up from 25°C to 180°C at a rate of 2°C min−1, followed by an isothermal stage of 180°C for additional 30 min. It is worth noting that the epoxy nanocomposites studied in this work are widely used as the matrices of carbon fibre composites and structural adhesives in automotive and aerospace industries, and the semi-dynamic curing schedule is chosen to be consistent with the curing schedule for industrial applications.

DMTA sample holder set for curing. DMTA: dynamic mechanical thermal analysis.
DSC measurements of the curing were performed by employing a DSC 204 F1 Phoenix (NETZSCH) machine under nitrogen atmosphere. Approximately 3 g of uncured nanocomposites was secured in an aluminium crucible. The samples were heated from 30°C to 180°C at a heating speed of 2°C min−1 and then remained at 180°C for additional 15 mins.
The thermo-mechanical properties of cured epoxy nanocomposites were probed using DMTA. The samples were machined and polished from a cured plate to a dimension of 10 × 2.5 × 0.6 mm3. The analysis was performed in tension mode at 1 Hz frequency. The scanning temperature range was 20°–150°C with a heating rate of 2°C min−1. T g was determined at peak value of loss factor (tan δ).
Results and discussion
Curing behaviour of the nanocomposites
Typical curves of loss factor (tan δ), storage modulus (E′) and sample temperature (Temp.) versus time from the DMTA curing analysis of C38E are shown in Figure 2. A step increase in E′ is observed at approximately 40 min. This was caused by the interaction between CSR nanoparticles and epoxy matrix and will be discussed later. It is found that the curing initiated abruptly at approximately 65 min (150°C) due to the initial autocatalytic acceleration of the epoxy. This abrupt curing onset resulted in a steep increase in E′ and a corresponding peak on tan δ curve at the same time. Considerable heat was released in short time due to this abrupt exothermic curing, which suddenly increased the sample temperature from 150°C to 165°C in a very short time (see the peak on Temp. curve at approximately 65 min in Figure 2). The DMTA furnace stopped heating to comply with the defined temperature schedule of 2°C increment per minute. After the initial acceleration, the curing retarded as the reaction became diffusion controlled due to the cross-linking of epoxy, 21 which was accompanied with a gradually increase in E′ until the curing completed. The observed curing behaviour is in good agreement with the literature 21 –23 that the curing kinetics of epoxy resins consist of an initial autocatalytic-acceleration stage, which corresponds to the abrupt curing onset at approximately 150°C, followed by a diffusion-controlled stage.
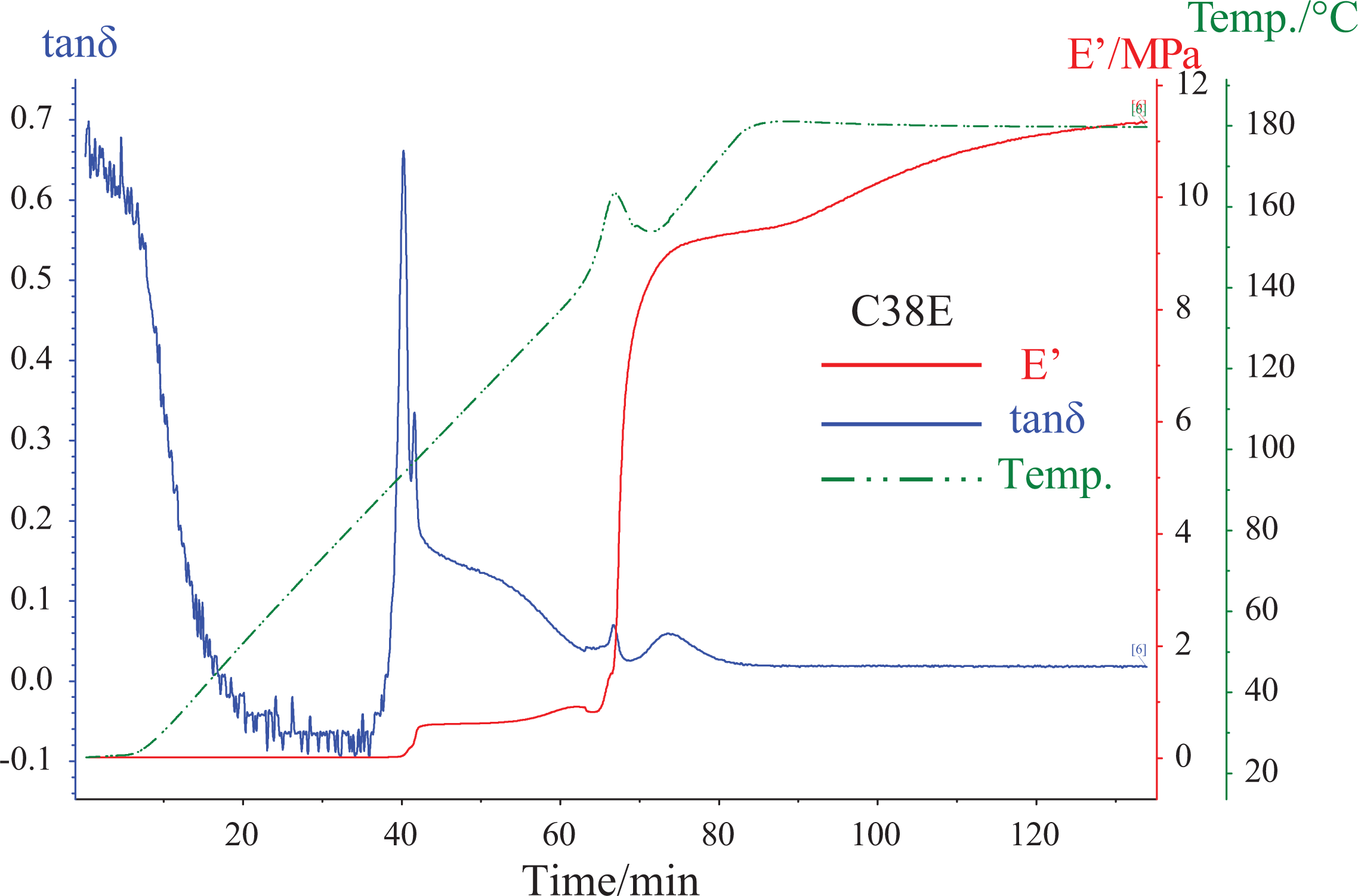
Tan δ, E′ and Temp. versus time from the DMTA curing analysis of C38E. DMTA: dynamic mechanical thermal analysis; Temp.: temperature; E′: storage modulus.
The curves of E′ and tan δ versus time from the DMTA curing analysis of all the epoxy nanocomposites are shown in Figures 3 and 4, respectively. Figure 3(a) shows the full time scale of the curing process, while Figure 3(b) and (c) are zoomed in around 40 min and 65 min, respectively. From Figure 3(a), it is found that a step increase in E′ took place at approximately 40 min for some formulations, while E′ of all the nanocomposites increased steeply at approximately 65 min. Figure 3(b) shows the E′ versus time curves in the time scale of 35–50 min. One can see that the step increase in E′ occurred at approximately 40 min (97°C) for all the epoxies with CSR nanoparticles. This step increase corresponds to two relaxation peaks on the tan δ curves in the same time range (Figure 4). Moreover, both the step increases in E′ and the height of the corresponding peaks on tan δ curves increased with the increasing content of CSR nanoparticles. It is worth noting that the step increase of E′ and the corresponding peaks of tan δ were not observed for the neat epoxy (Figures 3 and 4). This demonstrates the step increase was caused by the addition of CSR nanoparticles. The question arises here is why there are two relaxation peaks on the tan δ curves of the CSR-modified epoxies (Figure 4). The first peak of tan δ corresponds to the glass transition of the PMMA shell (given to be around 95°C by the supplier) of the CSR nanoparticles. The transition of the PMMA shell from the glassy state to the rubbery state resulted in higher flexibility and mobility of the PMMA molecule, which benefitted the interaction between the CSR nanoparticles and the epoxy matrix. Hence, the second peak on the tan δ curves corresponds to the interaction between the CSR nanoparticles and the epoxy matrix. This resulted in the step increase of E′. The steep increase of E′ at approximately 65 min is observed for all the epoxies (Figure 3(b)). This is due to the initial curing acceleration of the epoxy. It is clear that longer time and higher temperature was required to initiate the curing for nanocomposites with higher CSR content. This was mainly attributed to the physical changes caused by the addition of CSR nanoparticles, such as the increase in viscosity and the reduction in density of the reactive groups. 24
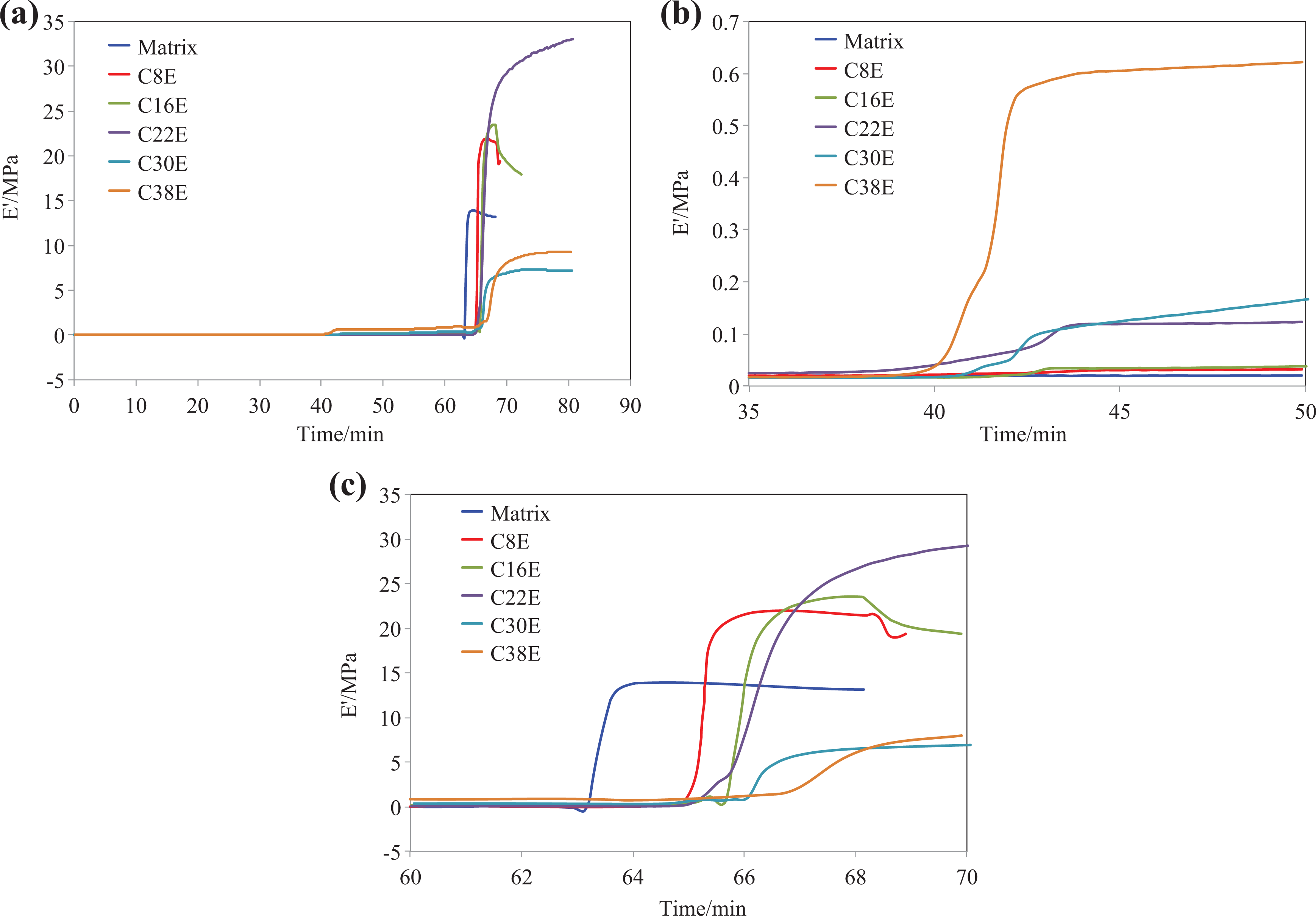
E′ versus time of the curing process for all formulations: (a) full time scale and (b) and (c) zoomed in around 40 min and 65 min, respectively. E′: storage modulus.
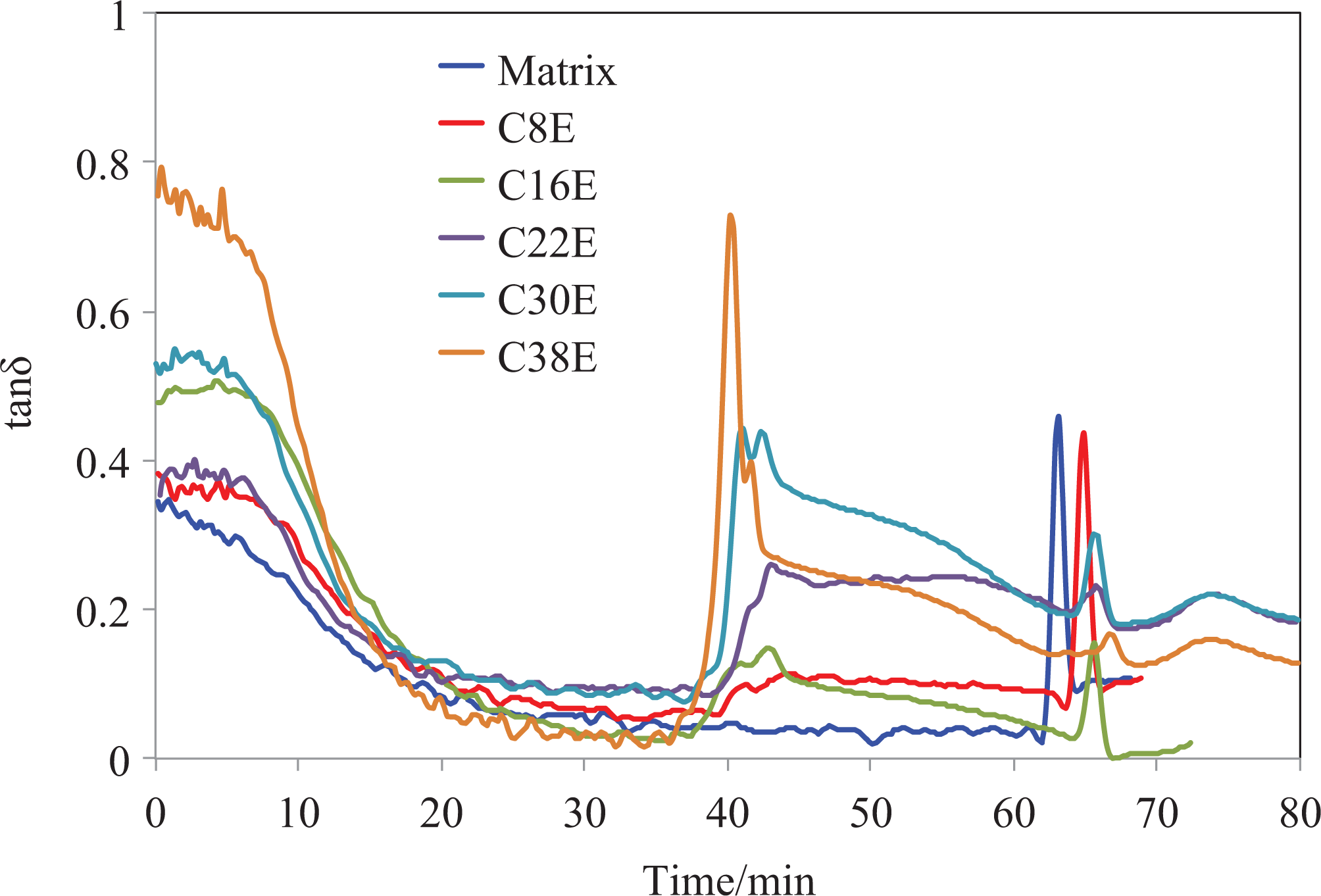
Tan δ versus time of the curing process for all formulations.
The curing curves of all the epoxy nanocomposites from DSC measurements are shown in Figure 5. The peaks of the DSC curves at around 150°C correspond to the exothermic curing reaction, and the onset curing temperature, peak curing temperature, and heat of reaction (area of peak) are summarized in Table 1. It was found that the onset curing temperature and peak curing temperature increased as the CSR content increased. This is consistent with the results of the DMTA curing analysis. The heat of reaction decreased as the CSR content increased, which implies decreased released heat from the curing of per unit nanocomposite as the CSR content increased. This was attributed to the increase in the viscosity of nanocomposites and the reduction in the density of reactive groups due to the addition of the CSR nanoparticles.

The curing curves of all nanocomposite formulations measured by DSC. DSC: differential scanning calorimetry.
Results obtained from DSC curing measurements.
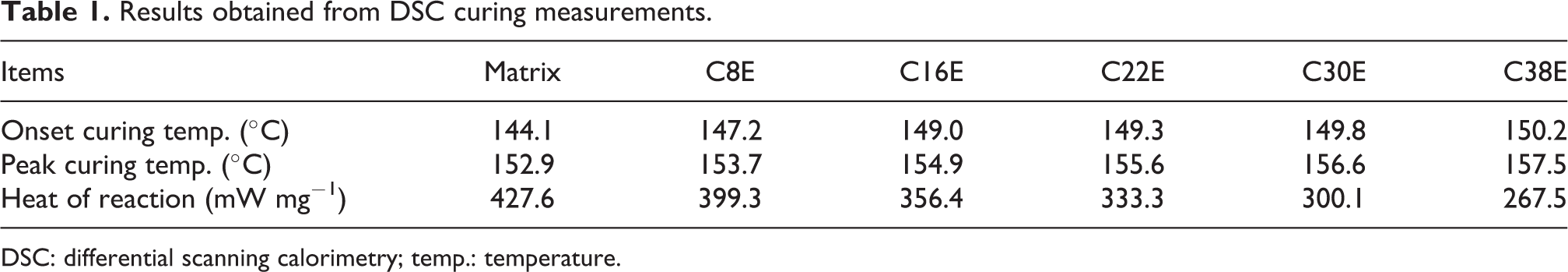
DSC: differential scanning calorimetry; temp.: temperature.
Figure 6 shows the zoomed in 50–120°C of the curing curves from DSC measurements. The curves are shifted either up or down for easier observing. An endothermic transition, starting at around 89°C and finishing at around 95°C, was observed for all the epoxy nanocomposites except the matrix. This is the glass transition of the PMMA shell of the CSR nanoparticles, which was also observed in the DMTA curing studies. However, the CSR/epoxy interaction, which was clearly observed from the DMTA curing analysis (Figures 3 and 4), is not observed from the DSC measurements. This might be attributed to two reasons. The first is the sensitivity of the DSC approach is insufficient to detect the CSR/epoxy interaction. The second is no calorimetric change occurred from the CSR/epoxy interaction.
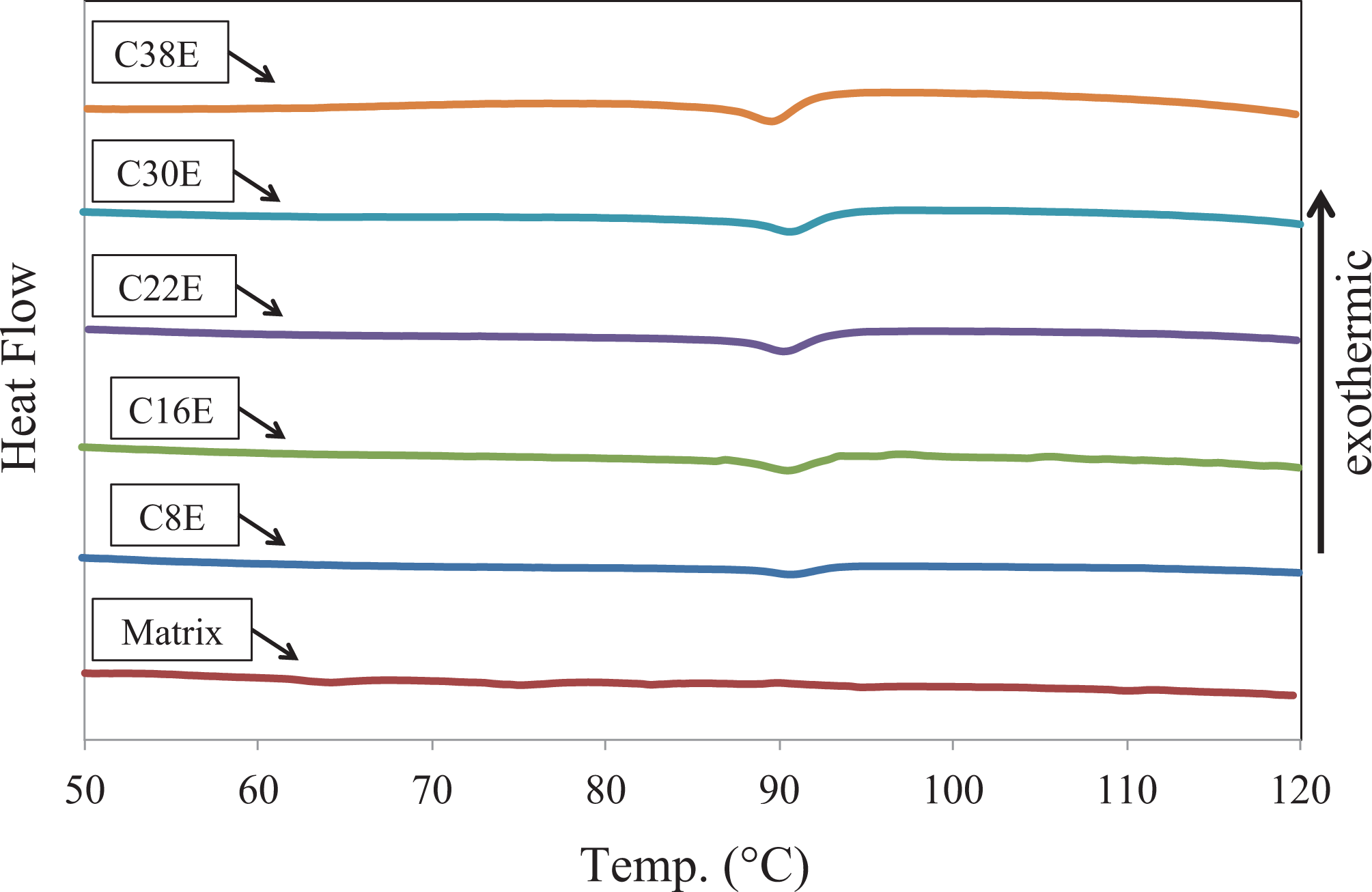
Zoomed in 50–120°C of the curing curves measured by DSC. DSC: differential scanning calorimetry.
Thermo-mechanical properties
The curves of tan δ as a function of time from the DMTA measurements on cured nanocomposites are shown in Figure 7. T g of the nanocomposites was taken from the peak of tan δ curves and is summarized in Table 2. T g of the neat epoxy was measured to be 122.5°C. The incorporation of CSR nanoparticles steadily increased T g from 122.5°C to 128.6°C of the C38E nanocomposite. This is caused by the detected interaction between the CSR nanoparticles and the epoxy matrix, which took place at approximately 97°C in the curing process. This interaction restricted the mobility and retarded the dynamics of the epoxy molecule.

Tan δ versus time from DMTA measurements on cured specimens. DMTA: dynamic mechanical thermal analysis.
Tg of different formulations.

T g: glass transition temperature.
The curves of the storage modulus (E′) versus time for all the cured epoxy nanocomposites are presented in Figure 8(a). It is found that adding CSR nanoparticles into the epoxy decreased the E′ at the glassy state but had negligible effects on the rubbery plateau modulus. This is because the CSR nanoparticles have a lower E′ than the epoxy matrix. The differences in E′ of the nanocomposites with different CSR contents are more pronounced at a lower temperature, as shown in Figure 8(a). This might be attributed to the difference in the coefficient of thermal expansion (CTE) between the epoxy matrix and the CSR nanoparticles. 25 Since the CSR nanoparticles possess a higher CTE than the epoxy matrix, a residual tensile stress exists at the interface of the CSR and the epoxy matrix. This caused an additional stress transfer between the CSRs and the epoxy when the sample was under loading. The higher stress transfer, which corresponds to the higher residual tensile stress, caused a bigger difference in E′ between different formulations at lower temperature. Figure 8(b) presents the curves of the loss modulus, E″, as a function of time for all the cured nanocomposites. The transition peak becomes less intense due to the addition of the CSR nanoparticles, and the peaks shift to higher temperature and become narrower as the CSR content increased. The peak shifting and narrowing on the E″ curves are caused by the CSR/epoxy interaction, which restricted the mobility of epoxy molecules.

E′ and E″ as a function of temperature for all formulations: (a) E′ versus Temp. and (b) E″ versus Temp. E′: storage modulus; E″: loss modulus; Temp.: temperature.
Conclusions
The effects of CSR nanoparticles on the curing behaviour of an epoxy were investigated using the DMTA and DSC approaches. An interaction between the CSR nanoparticle and the epoxy matrix was in situ detected, for the first time, at approximately 97°C in the curing process. This interaction caused a step increase in the E′ in the curing process and increased the T g of the cured nanocomposites. All the epoxy nanocomposites started to cure rapidly once the temperature reached certain values. After the onset stage, the curing rate dropped as the cure kinetic changed to be diffusion controlled. It required longer time and higher temperature to initiate the curing for the nanocomposites with higher CSR content. This was caused by the increasing viscosity and/or the reducing density of the reactive groups due to the addition of CSR nanoparticles. A decrease in the E′ of the cured nanocomposites at glassy state was observed, as expected.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of following financial support for the research, authorship, and/or publication of this article: This work was financially supported by Irish Centre for Composites Research, China Scholarship Council and Henkel (Ireland).
