Abstract
Chitosan-based hydrogels were prepared by grafting with poly(acrylic acid) (PAA) and poly(hydroxyethyl methacrylate) (pHEMA) through ultraviolet polymerization to further improve its hydrophilic nature and enhance its mechanical properties. The epidermal growth factor (EGF) was incorporated inside the hydrogels to stimulate the wound healing. The physical characterization of chitosan-PAA-pHEMA hydrogel indicates that it is highly hydrophilic with adequate properties for wound dressing purpose. The biological characterization shows that chitosan-based hydrogel retains its thrombogenic and antibacterial properties. The in vivo wound healing experiment demonstrates that the hydrogel-incorporated EGF has better wound healing rate than other dressings.
Keywords
Introduction
It is observed that if the wound is exposed to closed and moist conditions, the wound will undergo reepithelialization faster compared to the group maintaining in an open and dry condition. 1 Currently, closed moisture wound dressings have gradually replaced dry wound dressings, becoming a standard way of wound treatment. Nowadays, most commercial dressings are designed to provide the following advantages: reducing wound tissue necrosis, reducing pain around affected areas, accelerating the rate of wound repair and simplifying procedures to protect the wound for the most basic functions. The closed moisture wound dressings accelerate the wound healing process via a variety of mechanisms. The prime concern is to maintain the wound in a moist and isolated environment. According to Bolton’s study, rate of evaporation of water is less than 35 g/m2/h from the surface of closed moisture wound dressing under room temperature due to which faster wound healing. 2 Exposure of wound in air will increase the depth of the wound necrosis. 3 Closed dressings not only accelerate the rate of epithelialization but also promote cell migration, which can accelerate the wound healing effect. In addition, it increases the rate of collagen synthesis. 2
The purpose of the developing the dressings is simply to provide some novel methods and materials, so that the wound can be covered and healed rapidly and permanently. In addition, the appearance and function can recover as same as before the injury. There are three main objectives of developing wound dressings: (1) to accelerate wound healing process; (2) to make the material like skin; and (3) to reduce scars after healing. 4 Nowadays extracellular matrix (ECM) is one of most popular dressing materials. For example, applying proper crosslinking agent, chitosan can form hydrogel on the wound surface. Hydrogel contains a lot of water, which can serve as the ECM. Hydrogel has several advantages on the dressings. First, hydrogels can absorb the exudate released from the wound. Also, after absorbing water molecules, hydrogel can still maintain its three-dimensional structure eliminating contact between the wound and the external environment. 5 It ensures the protection of wound from external factors. Moreover, the characteristics of hydrogel can be improved by changing the functional groups attached to it or adding monomers for copolymerization, thereby making its functionality more diverse.
Recently, chitosan became a promising biomaterial mainly because of its excellent biocompatibility, nontoxic, antimicrobial activity, hemostasis, and biodegradable nature. 6,7 However, a major disadvantage is less hydrophilic nature of the hydrogel. To improve its hydrophilic property and accelerate the healing process, photocrosslinkable chitosan hydrogel was developed. 8,9 To further strengthen its hydrophilic nature and enhance its mechanical properties, this study used two hydrophilic monomers acrylic acid (AAc) and 2-hydroxyethyl methacrylate (2-HEMA) monomer grafted onto chitosan. Ultraviolet (UV) photopolymerization is used in the experiment. An advantage of using UV light for polymerization is that it can use high energy to break bonding which can effectively shorten the reaction time. When the UV light irradiation is used for polymerization, the amount of initiator and the crosslinking agent can be minimized without using promoters, hence a three-dimensional network structure polymeric hydrogel is obtained. As these additives are toxic in nature therefore if they do not react completely and remain in the hydrogel it will do harm to the human body. Using photo-polymerization by UV can reduce the amount of these additives to lower the toxicity of the finished product to a great extent. 10 Since UV radiations are highly energetic, the hydrogel get sterilized effectively making it safe for further applications.
In this study, three types of hydrogels composed of chitosan, AAc, or 2-HEMA were prepared. The physical properties of hydrogels including degree of swelling, mechanical strength, morphology, and water vapor transmission rate (WVTR) were studied. Finally, the optimal composition of hydrogel was subjected for antibacterial assessment, thrombosis test, biological agent release study, and wound healing experiment to assess its potential in quick wound healing as dressing.
Materials and methods
Materials
Chitosan (low viscous, 550 kDa, DD = 75%), AAc, and N,N′–methylenebisacrylamide (NMBA) were obtained from Fluka (St. Louis, MO, USA). 2-HEMA was purchased from Merck (Germany) and 2-ketoglutaric acid from Acros (Pittsburgh, PA, USA). Pseudomonas aeruginosa (BCRC 51693), Escherichia coli (BCRC 11446), and Staphylococcus aureus (BCRC 10780) were supplied by Bioresource Collection and Research Center, Taiwan. Epidermal growth factor (EGF) was obtained from ProSpec, USA (East Brunswick, NJ, USA). 3 M dressing used in animal studies was purchased from local cosmeceutical store (Taiwan). All other chemicals and reagents used were of an analytical grade.
Preparation of chitosan-PAA-pHEMA hydrogel
Chitosan (2 wt%), 2-ketoglutaric acid, and NMBA were added into AAc or acetic acid solution and stirred by homogenizer (Poly Tron; Kinematica, AG, Switzerland) at 25,000 r/min to dissolve them completely. The compositions for each formulation are listed in Table 1. HEMA was then added and mixed completely. The mixture was then immediately injected into the space between two glass plates using a syringe. The gel membrane thickness was adjusted using a silicone spacer between the two glass plates. The whole fixture was subjected to UV light for polymerization at an intensity of 2200 W for 20 min. After the completion of polymerization, hydrogel was rinsed several times by deionized water to ensure complete removal of unreacted monomers, finally it was dried at room temperature until no water remains.
Compositions for various hydrogel formulations.a

AAc: acrylic acid; HEMA: hydroxyethyl methacrylate; CSPA: chitosan-PAA; CSPH: chitosan-pHEMA; CSPAH: chitosan-PAA-pHEMA; PAA: poly(acrylic acid).
a2-Ketoglutaric acid and N,N′-methylenebisacrylamide are 2 wt%.
Fourier transform infrared spectrum analysis
After UV polymerization of the copolymer, the absorption peaks of functional groups were observed by Fourier transform infrared spectroscopy (FTIR spectroscopy; Spectrum One, Perkin Elmer, Waltham, MA, USA). The hydrogel was dried and grounded into fine powder. The powder was mixed evenly with potassium bromide in the ratio of 1: 99 and then was pressed to form tablets.
Swelling and water content test
In swelling test, the dried hydrogel was placed in 20 ml phosphate buffered saline (PBS) solution at 37°C. At specific time intervals, it was blotted with filter paper and weighed until the weight remained unchanged. The swelling ratio was calculated by the following equation
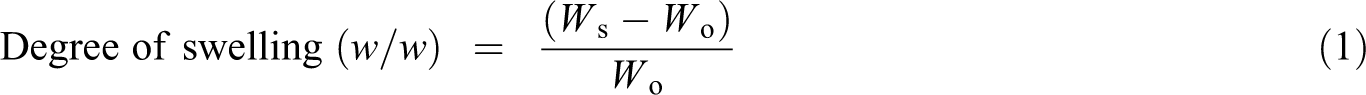
where W o is the weight of dried hydrogel; W s is the weight of the hydrogel after absorbing water.
The procedure of determining water content of hydrogel is the same as mentioned above, except the hydrogel was immersed in PBS solution until it was fully swelled. It was then blotted with filter paper and weighed. The water content was calculated using this equation.

where W o is the weight of the dried hydrogel; W t is the weight of swollen hydrogel after reaching equilibrium.
The data of degree of swelling and water content were reported based on average of three measurements.
Mechanical test
The tensile tests were performed on hydrogel after swelling it completely in PBS (has dimension of 7 × 2 × 2 cm3) using a universal material tester (AG-IS, Shimadzu, Japan) at a rate of 2 mm/min. The data of tensile stress and strain were reported based on average of three measurements.
Interior morphology observation of hydrogel
The hydrogel was immersed in deionized water at 20°C until it fully swelled, then it was frozen at −40°C overnight before being subjected to lyophilization (FD-5 N, EYELA, Japan). Surface area and cross-sectional area of specimen were then observed by scanning electron microscopy (SEM, S-3000 N, Hitachi, Japan). The specimen was fixed on aluminum stubs and coated with gold for 60 s prior to the observation.
Measurement of WVTR
A cup equipped with a cap having 6.20 mm internal diameter and 8.45 mm external diameter was used to measure the WVTR. The specimens were dried out and cut into pieces larger than internal diameter but smaller than external diameter of the cap. The original top of the cap was removed and the specimen was fastened to the top of the cap. The measuring cup filled with water was placed inside the desiccator at 25°C and 0% relative humidity for 30 min, then the initial weight of the measuring cup was weighted immediately. After that, the measuring cup was moved to another desiccator set at 37°C and 35% relative humidity. Cups were weighed and readings were recorded after the equilibrium state was reached.
The WVTRs of the specimen were calculated at a given time interval using the following equation

where m = mass of water loss over the specified time interval (g); △t = time interval (h); and A = the effective transfer area (m2). WVTR values were averaged from three measurements.
Antibacterial assessment
The standard strains Pseudomonas aeruginosa, Escherichia coli, and Staphylococcus aureus were firstly dispersed in the liquid medium, separately at 37°C, pH 7.4, and rotational speed of 150 r/min to culture and activate for 24 h. Then, medium (1 ml) containing bacteria with a bacterial concentration of 1×106 colony forming unit (cfu)/ml was taken and added to new liquid medium (10 ml). Subsequently, a fixed amount of hydrogel with 1 cm diameter was added and tested on preset time interval. After a certain time interval, the UV absorbance (Lambda 25, Perkin Elmer) of sample was measured at the wavelength 620 nm to observe the antimicrobial property of hydrogels.
Blood clotting test
The dried sample was immersed in PBS at 37°C for 48 h until it swelled completely. After that 1 ml of anticoagulant acid citrate dextrose solution was mixed with 9 ml of fresh rabbit blood and a solution was prepared. The swollen hydrogel was then placed into the solution, and 0.02 ml 10 M calcium chloride solution was added as an initiator. The reaction was carried out for 45 min and then 5 ml of distilled water was added to terminate the reaction. Finally, 5 ml of 36% formaldehyde was added to solidify the blood onto the sample.

EGF-2 release studies
Firstly, the standard releasing curve of EGF was plotted by measuring the absorbance of formulations containing various concentrations of EGF at 450 nm using enzyme-linked immunosorbent assay (ELISA reader, Anthos 2001, Anthos Labtec Instruments, GmbH, Eugendorf, Austria). The dried hydrogel (5 mg) was placed in 1 ml PBS containing EGF (with a concentration of 1 μg/ml) at 4°Cfor 48 h until it swells completely to incorporate EGF into hydrogel. The amount of EGF loaded in each specimen was calculated as the difference between the initial and final concentrations in the surrounding solution, determined by ELISA at 450 nm. The changes in the concentration in the loading medium over time were recorded, and the loading was considered to be finished when equilibrium was reached (generally after 48 h).
The swollen hydrogel was then placed in 1 ml of PBS solution. After a desired period of time, certain amount of aliquot was withdrawn and the releasing amount of EGF was analyzed by ELISA. The sample was placed in another bottle containing 1 ml of PBS, and then the absorbance was determined until EGF was no longer released. EGF release study was carried out at 37°C.
Animal studies
Experimental animals were divided into four groups, group 1 was non-treatment control, group 2 was covered with CSPAH hydrogel only, group 3 covered with CSPAH hydrogel with EGF, and group 4 covered with commercial 3 M dressings. The four groups of rats were firstly anesthetized and their dorsal hair was clipped and the wound site was sterilized with alcohol. Full-thickness round wounds (about 1 cm2, a Sprague Dawley® rat for three wounds) were created on each rat using a sharp pair of scissors and a scalpel. The appropriate dressing material was placed on the wounds according to the groups. The bandage was fixed to the SD rat’s head and neck. Each rat was fed separately with an adequate supply of food and water. Photographs were taken immediately after excision, and the wound area from day 0 to 25 was measured using a slide caliper. All rats were euthanized after the measurements.

where A o is the initial wound area and Ad is the wound area of d day.
All animal experiments were performed in compliance with guidelines approved by the Animal Use and Care Administrative Advisory Committee at the Southern Taiwan University of Science and Technology, Tainan, Taiwan.
Once rats were anesthetized, after a certain time interval, wound tissues from the surrounding and subcutaneous skin were retrieved. Wound tissues were placed in a bottle containing 10% formalin for paraffin embedding and section. The slice with 5 μm thickness was fixed on a slide coated with poly-L-lysine. After drying at 45°C, the samples were analyzed with hematoxylin and eosin (H & E) staining.
Statistical analysis
Results are given as means ± SD. Statistical analysis was performed using Student’s t test. Statistical significance was set at p < 0.05.
Results and discussion
Physical characterization of chitosan-PAA-pHEMA hydrogel
Chitosan due to its excellent biocompatibility, nontoxic, biodegradable nature, hemostasis, and antimicrobial activity has received much attention on research for wound dressing. 6,7,9,11,12 However, the main drawback is it is hydrophobic in nature. In this study, chitosan-based hydrogels were prepared by grafting with PAA and pHEMA through UV irradiation to further improve its hydrophilic nature and enhance its mechanical properties. Three different compositions of hydrogels, that is, chitosan-PAA (CSPA), chitosan-pHEMA (CSPH), and chitosan-PAA-pHEMA (CSPAH) were prepared. FTIR analysis was conducted to investigate whether chitosan was successfully copolymerized with AAc and HEMA. From Figure 1(a), the ν as-NH at 3400 cm−1 and ν s-NH at 3300 cm−1 are assigned to chitosan. These two absorption peaks overlap with ν −OH (3300 cm−1) and ν s-NH 3+ (3100–2000 cm−1) from chitosan; thus, it is not distinguishable. The absorption peak at 2850–3000 cm−1 is ascribed to ν sat-CH. After graft polymerization with AAc and HEMA using UV light, the ν c=o absorption peaks from AAc and HEMA started appearing at 1730 cm−1, as shown in Figure 1(b) to (d). The original δ N–H peak at 1660 cm−1 shifts to 1640 cm−1 due to graft polymerization of AAc or HEMA. However, the ν s–NH 3+ absorption peak (3100–2000 cm-1) is still shown in Figure 1(b) and δ N–H absorption peak remains unchanged. Therefore, it proves that chitosan crosslinks with AAc through the interactions between NH3 + from chitosan and COO− from Aac. 13 Furthermore, the δ N–H absorption peak (1640 cm−1) shown in Figure 1(c) is weakened and the C–N absorption peak at 1450 cm−1is found to be stronger. Thus, it demonstrates that the bonding between chitosan and HEMA occurs at −NH2 group. 13,14 As shown in Figure 1(c) and (d), the intensity of absorption peaks at 1275 cm−1 and 1075 cm−1 that belong to C–O–C increase, which further suggests that HEMA polymerizes with chitosan. In summary, based on the FTIR analysis, AAc and HEMA successfully polymerize with chitosan to form CSPAH hydrogel. The schematic reactions among chitosan, AAc, and HEMA via UV irradiation are shown in Figure 2. The interactions between NH3+ from chitosan and COO− from AAc lead to formation of CSPA hydrogel. In CSPH hydrogel, covalent bonds are formed between chitosan and HEMA. For CSPAH hydrogel, the bondings are dominated by ionic interactions between chitosan and AAc with HEMA as a chain extender in the structure.

FTIR analysis of (a) chitosan, (b) CSPA, (c) CSPH, and (d) CSPAH hydrogels.
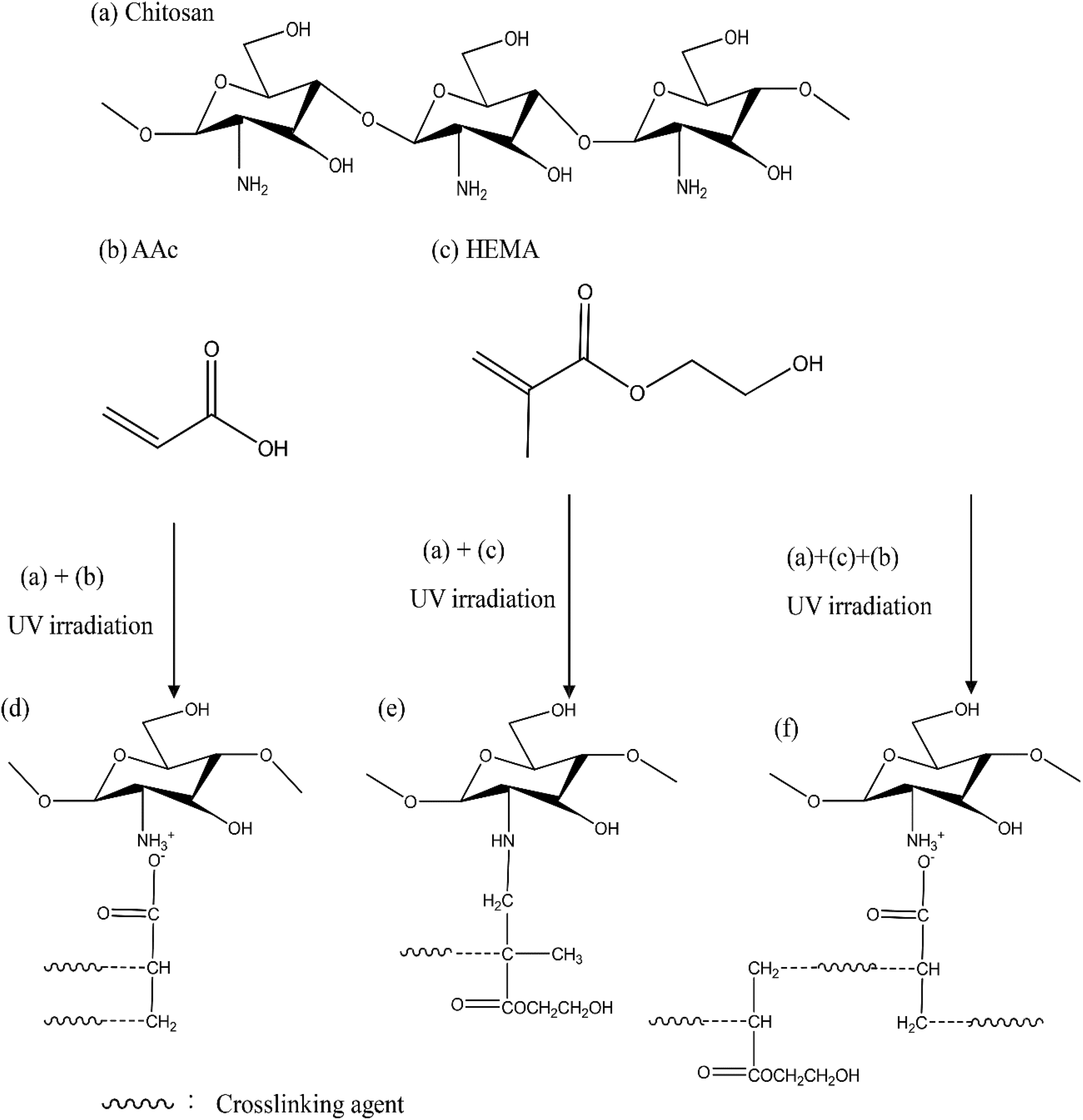
Schematic reactions among (a) chitosan, (b) AAc, and (c) HEMA via UV irradiation to form (d) CSPA, (e) CSPH, and (f) CSPAH hydrogels.
After drying, CSPA, CSPH, and CSPAH hydrogels were placed at 37°C in PBS to observe their swelling behavior. Chitosan polymerizes with different monomers showing different degrees of swelling. As listed in Table 2, CSPA (degree of swelling 4.8 and water content 83% after 50 h immersion) has better swelling and water content than CSPH (degree of swelling 1.0 and water content 49% after 50 h immersion). It is because COOH group from AAc is more hydrophilic than OH group from HEMA. While, comparing CSPAH with CSPA, since HEMA is added to CSPAH hydrogel, the hydrophilic performance of CSPAH hydrogel is not inferior to CSPA, with up to 4.3 swelling degree and 80% water content at 50 h.
Physical characterization of CSPA, CSPH, and CSPAH hydrogels.

WVTR: water vapor transmission rate; CSPA: chitosan-PAA, CSPH: chitosan-pHEMA, and CSPAH: chitosan-PAA-pHEMA; PAA: poly(acrylic acid); PBS: phosphate buffered saline.
a The data are obtained after 50 h immersion in PBS.
For wound dressing, a higher degree of swelling can enhance the absorption of fluids. It is better to have an excellent degree of swelling on the first day, which can effectively recover a huge loss of moisture of the first day. In this study, CSPA and CSPAH hydrogels, both have a 4.0 degree of swelling after 24 h immersion (data not shown), hence, are suitable for wound dressings.
A good dressing needs adequate mechanical strength, which may avoid breakage and make it easier to replace. As listed in Table 2, the maximum tensile stress of CSPH (0.47 MPa) is the largest among the three hydrogels. This is because strong covalent bonds are formed between chitosan and HEMA by breaking the double bonds of HEMA. The bonding between chitosan and AAc is basically the weak ionic interactions between NH3 + and COO−, which renders the lowest stress (0.14 MPa) among others. For CSPAH hydrogel, the bonding is mainly the ionic interactions between chitosan and AAc, while HEMA plays a role as a chain extender in the structure due to which stress (0.34 MPa) and elongation (53%) are upgraded effectively. Catanzano et al. 15 prepared alginate–hyaluronan composite hydrogels to accelerate wound healing process. The compressive tests of these hydrogels performed in quasi-static conditions had stresses ranging between 200 and 300 Pa at 10% deformation. They claimed that the investigated hydrogels showed sufficient strength to be handled, cut or packed without experiencing any difficulty. In this study, tensile tests were conducted instead of compressive tests and the maximum tensile stresses observed were promising; therefore, it is assumed that the prepared hydrogels have adequate strength for wound dressing application.
To investigate the actual hydrophilic nature of the prepared hydrogels, their surface and interior morphologies were observed by SEM. From Figure 3(b) and (c), we can observe that the surface of the hydrogel is very dense without any pores, mainly due to the highly hydrophilic nature of CSPA and CSPAH hydrogels, which contracts to the extreme and form skin layer after freeze-drying. On the other hand, the surface of CSPH hydrogel (Figure 3(a)) is not as dense as CSPA and CSPAH, since it is more hydrophobic than these two hydrogels. As shown in Figure 3(d) to (f), neat and uniform pore structures within the hydrogels can be seen. However, all three of them have great diversity in the pore size and shape. In Figure 3(d), CSPH hydrogel has grid-like cross-section pores (pore size 25 μm) with curved cell walls, while CSPA has circular pores (pore size 9 μm) and CSPAH is equipped with the previous two hydrogels morphology with well-rounded pores (pore size 19 μm). In this study, all the hydrogels have open pore structure and they are sufficient to absorb body fluid. On the other hand, they are suitable to be used as scaffolds for cells to attach and migrate.

Surface and interior morphologies observed from SEM: (a) surface CSPH, (b) surface CSPA, (c) surface CSPAH, (d) interior CSPH, (e) interior CSPA, and (f) interior CSPAH. (×1 k)
The prepared hydrogels were subjected to homemade water vapor transmission device under proper temperature and humidity conditions to measure their WVTRs. The principle is, when water vaporizes, it will adsorb on the surface of the sample and the adsorbed water molecules will release from the other side by diffusion. The result (Table 2) indicates CSPA (WVTR = 1666 g/m2/day), CSPH (WVTR = 1887 g/m2/day), and CSPAH (WVTR = 1732 g/m2/day) hydrogels compared to blank group (not covering with anything, natural moisture loss, WVTR = 1223 g/m2/day) have higher values of WVTR because these three hydrogels have high hydrophilic property and can absorb much water vapor. The water vapor then diffuses through the hydrogel membrane. Among these three, CSPH has the highest WVTR, because CSPH does not form a skin layer on its surface as observed from SEM making water molecules to pass through it faster than the other two.
Lamke et al. 16 reported that WVTR of normal skin is 204 g/m2/day; WVTR of a burn skin is 5138 g/m2/day. An ideal wound dressing must be able to control the rate of moisture loss from the wound to prevent excessive exudate and dehydration. Typically, WVTR of wound is around 2000–3000 g/m2/day, for no dehydration and excessive exudate residue. The WVTR of the hydrogel prepared in this study is close to the standard value allowing the wound to maintain certain moisture and absorb exudates effectively. This makes it easy to enhance cell migration and form new epidermal cells. 14
Antimicrobial assay of chitosan-PAA-pHEMA hydrogel
Infection occurs in wounds when one or more microorganisms (mainly bacteria and sometimes fungi) compete with the host’s natural immune system. Staphylococcus aureus, Escherichia coli, and Pseudomonas aeruginosa species are the most common causes of infection and delay of wound healing. 4 In this study, antibacterial activities in terms of optical density of the prepared hydrogels (CSPA, CSPH, and CSPAH hydrogels) were evaluated against these three bacteria (Figure 4). With the increase in incubation time, the absorbance of CSPA and CSPAH hydrogels against these three bacteria decreases as compared to the control group (without the addition of hydrogel). When growth level has reached an off state, CSPA and CSPAH can inhibit bacterial growth very effectively. This is due to the interactions between the protonated NH3+groups of chitosan molecules and the negatively charged constituents (e.g. phosphate, carboxyl, teichoic acid, nucleic acid) on the surface and inside of microbial cells. 17 These electrostatic forces then hinder the process of formation of bacteria, so as to achieve the inhibition of bacterial growth. However, CSPH does not have positive charges so the inhibiting effect is not evident. Moreover, the hydrophilic OH groups from CSPH can dissolve more oxygen than the control group; therefore, its bacterial growth rate is higher than the control group. CSPAH hydrogel can inhibit bacteria up to almost 99% which indicates that chitosan maintains its antibacterial ability even after bonding with AAc and 2-HEMA. The result implies that a series of local and systemic host responses such as purulent discharge, painful spreading erythema, or symptomatic cellulitis around a wound that leads to soft tissue destruction can be prevented. 18,19 This hydrogel can be safely used as a wound dressing.

The impact of antimicrobial property of CSPA, CSPH, and CSPAH hydrogels against (a) Staphylococcus aureus, (b) Escherichia coli, and (c) Pseudomonas aeruginosa.
Thrombogenicity
Typically, polymers do not have blood clotting properties, 20 whereas chitosan biomaterials show superior thrombogenic effects by causing platelets to aggregate and form pseudopodal shapes by facilitating the platelet activities to expedite the in vitro coagulation processes in normal subjects, therefore they can accelerate wound healing. 21
Figure 5 shows after modification with chitosan, CSPA and CSPAH hydrogels have a good ability of thrombosis because the red blood cells can effectively bond with surface electrostatic charge of cationic amino (−NH3+) groups to form a cross-linked substance. The blood forms clot to stop bleeding and achieve hemostasis purposes which can accelerate wound healing. 22,23 The CSPH hydrogel does not have amino groups; therefore, the effect of blood clotting is not significant. This result shows that it is possible to obtain a chitosan-based hydrogel with some hydrophilic character by mixing HEMA and AAc on graft copolymerization of chitosan without losing its thrombogenic properties.

Test of blood clotting (a) CSPA, (b) CSPH, and (c) CSPAH hydrogels.
EGF release from hydrogel
The use of growth factors to promote wound healing has always been considered one of the possible therapeutic approaches to overcome the problem of chronic wounds. Growth factors are a class of biomacromolecules locally secreted by the ECM capable of regulating biological processes by transferring signals between cells and their local environment, regulating proliferation, migration, and differentiation of cells. 4,24,25 Polymeric wound dressings were successfully developed for the incorporation of free growth factors using biocompatible biomaterials such as gelatin, 26,27 dextran, 11 collagen, 28 fibrin gel, 29 or chitosan. 12 In this study, the prepared dried hydrogels were simply immersed in PBS containing EGF at 4°C until it swells completely to incorporate the growth factor. This process can protect growth factors during the formulation and production phases to achieve a long-term exposure which is a characteristic required for the delivery of growth factors to chronic wounds.
The EGF release from various hydrogels is shown in Figure 6, which indicates that EGF release from CSPAH hydrogel on the day 1 is about 50%, from CSPA hydrogel is about 61%, and from CSPH hydrogel is about 37%. On the first day, the release is mainly from the EGF adsorbed on the surface of the hydrogel. When the swollen hydrogel comes in contact with the releasing medium (PBS solution), the EGF release becomes very fast due to concentration gradient. From the 2nd day to the 7th day, EGF releases mainly from the internal pores of hydrogel which is a diffusion-controlled process. 30 CSPA and CSPAH have more important role in EGF release than CSPH, which is mainly because these two have COO− group attached to the main chain, which can attract and fix positive EGF. Therefore, the EGF content of CSPA and CSPAH hydrogels is larger than CSPH hydrogel.

EGF release profiles from CSPA, CSPH, and CSPAH hydrogels.
As shown in Figure 6, CSPA, CSPH, and CSPAH can be effectively delay the release of EGF. The EGF release can be up to seven days or more, which enables long-EGF activity at the wound in vivo. Furthermore, it can take the advantage from hydrogel, since it is prone to water absorption, which can release the EGF and absorb wound exudate, keeping the wound moist and accelerating the wound healing process.
In vivo wound healing experiments
To determine changes in the wound surface area, a digital camera was used to capture images of the wound, to record the size of the healing of the wound area over time, and to compare difference among the different dressings; open wound without any artificial dressing (control), CSPAH, CSPAH containing EGF, and commercial (3 M waterproof breathable dressing) dressings.
The photographic findings of wound covered with three types of dressings and control on day 0, 10, and 20 are shown in Figure 7. The photographs represent three rats in each group. As shown in the figure, wound closure without any dressing, that is, an open wound was unable to keep the wound moist, thus reducing the rate of wound healing. For CSPAH and CSPAH containing EGF dressings, the wound retained moisture throughout the wound healing process, which accelerates the healing especially for the group of CSPAH containing EGF. A commercial dressing shows improvement in wound healing. However, we found that using commercial dressings can be dangerous as during changing, it caused secondary damage and was difficult to be peeled. Thus, the wound surface can easily experience new trauma. Therefore, ease of removal of dressing is another very important feature during the wound healing. If the wound dressing is easy to strip off a second injury and additional pain can be prevented. 31

The representative images of wound surface healing over time in CSPAH, CSPAH-incorporating EGF, commercial dressings, and control at day 10 and 20.
Figure 8 shows the percentage of wound closure versus healing time for these four groups. The healing capabilities of the groups of CSPAH hydrogel and commercial dressing were similar, while CSPAH hydrogel containing EGF was shown to be better than the other three groups with about 94% wound closure within 25 days (Figure 8). On day 4, 10, 14, and 25, the wound contraction of EGF-loaded CSPAH hydrogel is significantly higher than commercial dressing, p < 0.001. Both CSPAH hydrogel and CSPAH hydrogel containing EGF have the ability to keep the wound moist. However, the latter one is even better for wound healing because EGF can stimulate proliferation of most epithelial cells, fibroblasts, endothelial cells, and even collagen deposition. 32

The percentage of wound closure versus healing time in control, CSPAH, CSPAH-incorporating EGF, and commercial dressings. Statistical analysis was conducted by comparing each dressing with control and pair comparison between CSPAH-incorporating EGF and commercial dressings at each time point. (*p < 0.05; **p < 0.001)
Histological analysis
To investigate the situation of wound healing, the histopathological analysis was conducted.
For control group (Figure 9), there was no epithelial cell migration phenomenon and epithelialization observed over time, only proliferation of fibrous tissue became apparent. On the other hand, for CSPAH, CSPAH containing EGF and commercial wound dressings, it can be observed that the skin attached to dressings appeared epithelium on the second day. Furthermore, more intense epithelial cells migration phenomenon, macrophages, neutrophils, and bold vessels can be observed in the group containing EGF on day 2 (Figure 9). Comparing the first 2 days of each group, we found that the CSPAH containing EGF can heal the wound more quickly, which indicates that EGF promotes wound healing. 33 Hydrogel contains a lot of water, which can serve as the ECM. Thus, the cells can enter the water network structure, increasing migration of epithelial cells which can further promote wound healing.

H & E stain patterns of control, CSPAH, CSPAH-incorporated EGF hydrogels, and commercial dressing observed at day 2. In the upper panel, the arrow points the epithelial cells. In the lower panel, macrophages, neutrophils, and blood vessels are observed in EGF-loaded CSPAH hydrogel at day 2.
Conclusions
Chitosan-based hydrogels are successfully prepared by UV polymerization exploiting the interactions with PAA and pHEMA for wound dressing application. The physical characterizations including swelling ratio, water content, and mechanical properties indicate chitosan-PAA-pHEMA hydrogel has adequate properties for wound healing. Chitosan-based hydrogels after modification retain its thrombogenic and antibacterial properties. The animal studies demonstrated that EGF-incorporated chitosan-PAA-pHEMA hydrogel has better wound healing rate than other dressings. These results reveal that chitosan-PAA-pHEMA hydrogel is a potential candidate for wound dressing application.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
