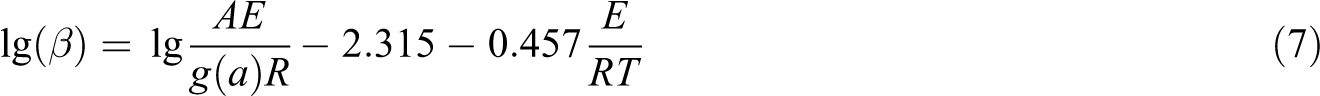Abstract
The thermal stabilities of epoxy resin/diethyl bis(2-hydroxyethyl)aminomethylphosphonate (EP/DBAMP) systems were investigated by thermogravimetric analysis (TGA) under non-isothermal conditions in nitrogen atmosphere. Kissinger and Flynn–Wall–Ozawa methods were used to study the thermal degradation process. The results showed a remarkable increase of activation energy (E) in the presence of DBAMP, which indicated that the addition of DBAMP retarded the thermal degradation of EP. The Flynn–Wall–Ozawa analysis further revealed that DBAMP significantly increased the activation energy in the early stage of EP’s thermal degradation, demonstrating that DBAMP had improved the initial thermal stability and modified the flame retardancy of EP in the thermal degradation process.
Keywords
Introduction
Epoxy resins (EPs) are widely used in chemical products such as surface coatings, adhesives, laminating and insulating materials for electric devices, due to their excellent fluidity, chemical resistance, low shrinkage on cure, superior electrical and mechanical properties and good adhesion to many substrates. 1 -4 However, like other polymeric materials, the flammability of EPs is an obvious shortcoming in certain applications. 5-6 Several approaches have been reported to enhance the thermal properties of EPs through modification of the epoxy backbone. 7 -9
Traditionally, brominated compounds and antinomy oxide are used to reduce the flammability of EPs. Considering the generation of toxic, corrosive and halogenated gases and the release of toxic dibenzo-p-dioxin and dibenzofuran chemicals in combustion should be avoided, there is a trend towards using halogen-free flame retardants. 10 -12 Organic phosphates do not cause any of these problems and are frequently used for replacing brominated compounds in flame-retarding EPs. By forming a carbonaceous char, which acts as a physical barrier to heat transfer from the flame to the polymer matrix and diffusion of combustible gas and smoke, organophosphorus compounds present better ability than halogen-containing compounds as flame retardants for polymeric materials. The products, kinetics and mechanism of the thermal degradation of some EPs have been investigated. However, the thermal degradation behaviour of phosphorus-containing epoxy has not been well investigated so far. The lack of knowledge about the degradation mechanism and kinetics does not allow us to predict the behaviour of such polymers upon heating and correlate flame retardancy with their structure. 13 -16 Moreover, to improve the efficiency of flame retardancy, various halogen-free formulations are often studied and developed, based on high-performance epoxy with added organophosphorus compounds. 17 -20 Thus, it is of great importance to study the thermal degradation process and further establish the flame-retardant mechanism of EP systems with organophosphorus compounds, because the flame-retardant property and processing of polymer materials strongly depend on their thermal stability.
In this article, the influence of diethyl bis(2-hydroxyethyl)aminomethylphosphonate (DBAMP) on the thermal degradation behaviour of EP was studied, and the flame-retardant mechanism of EP/DBAMP systems was revealed through thermogravimetric analysis (TGA) measurements under non-isothermal conditions in nitrogen atmosphere.
Experiment
Materials
The EP used was the diglycidyl ether of bisphenol A (DGEBA) from Shanghai Resin Co. Ltd, China, with a mass per epoxy equivalent of 0.51 eq. 100 g−1. Epoxy curing agent was 4,4′-diamino-diphenyl methane (DDM) from Shanghai Resin Co. Ltd, China. The flame-retardant DBAMP (Figure 1) was supplied (98 wt%) by Zhejiang Wansheng Chemical Co., Ltd, China. All components were used as received without further purification.
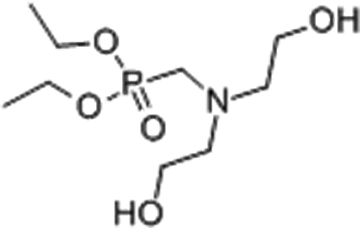
The chemical structure of DBAMP.
Preparation
Briefly, the EP/DBAMP composites were prepared as follows: The DBAMP (10 g) was added to DGEBA (72 g) under mechanical stirring for 5 min. Then, the mixture was heated in a vacuum oven at 50°C for 10 min. Subsequently, DDM (18 g) was added to the mixture and stirred for 30 min. After degassing in vacuum for 10 min to remove any trapped air, the samples were cured at 100°C for 2 h and postcured at 150°C for 2 h. For comparison, pure EP was also prepared with the same processing conditions.
Characterisation
TGA was carried out using a Mettler Toledo TGA/DSC thermal analyzer (Switzerland). TGA/differential scanning calorimetric thermal analyser and around 4 mg samples were placed on an aluminium pan. The sample was heated from 50°C to 700°C at a set heating rate of 5°C min−1, 10°C min−1, 20°C min−1 or 40°C min−1. All the tests were performed in a nitrogen flow with a flow rate of 80 ml min−1.
Thermal degradation theory
The kinetics of thermal transformation of a solid state chemical reaction is based on the assumption that the reaction rate is 21 -23
where a is the degree of conversion,
where A is the pre-exponential factor, E is the activation energy of the kinetic process, T is the temperature and R is the universal gas constant.
Then, the rate of degradation, which is dependent on the temperature and the change of the sample weight, can be expressed as
Equation (3) is also used in its integral form, which for isothermal conditions becomes
For non-isothermal degradation, equation (3) becomes
where
The Kissinger method 24
The equation of the Kissinger method can be expressed as follows
where
The peak rate temperatures determined at different heating rates allow the activation energy to be calculated by the Kissinger method. Plotting the natural logarithm of
Flynn–Wall–Ozawa method 25,26
The Flynn–Wall–Ozawa expression is as follows
The above equation shows that
Results and discussion
In dynamic thermogravimetry (DTG), the heating rate is a crucial factor affecting the shape of a thermogram and the characteristic temperature of decomposition. The TGA and DTG curves of pure EP and 10 wt% EP/DBAMP (FREP) systems at different heating rates (5°C min−1, 10°C min−1, 20°C min−1 and 40°C min−1) are shown in Figures 2 to 5. All the curves indicate that the systems exhibit only a single weight loss stage in the temperature range of 200–600°C. The char residues of FREP at 700°C with a heating rate of 10°C min−1 are 19.0 wt%, which is about 4.4 wt% more than that of pure EP.

TGA curves of EP at different heating rates in N2.

TGA curves of FREP at different heating rates in N2.

DTG curves of EP at different heating rates in N2.

DTG curves of FREP at different heating rates in N2.
Upon increasing the heating rate from 5°C min−1 to 40°C min−1, the thermal degradation curves of EP shift to higher temperature and the onset degradation temperature is enhanced. These phenomena are attributed to the temperature retardancy. A similar trend is obtained for FREP. Furthermore, it can be obtained from the figures that the maximum weight loss rate of FREP is lower than that of pure EP, and that the FREP has more stability than the pure EP, which is in accord with the change of onset degradation temperature between the EP and FREP systems.
Kinetic parameters of thermal degradation can be used to characterise the thermal stability of polymer and the activation energy generally can be considered as a semiquantitative factor. 27 Using the TGA and DTG curves in Figures 2 to 5, the thermal degradation processes of EP and FREP can be further studied by the Kissinger and Flynn–Wall–Ozawa methods.
Firstly, according to equation (6), the plots of
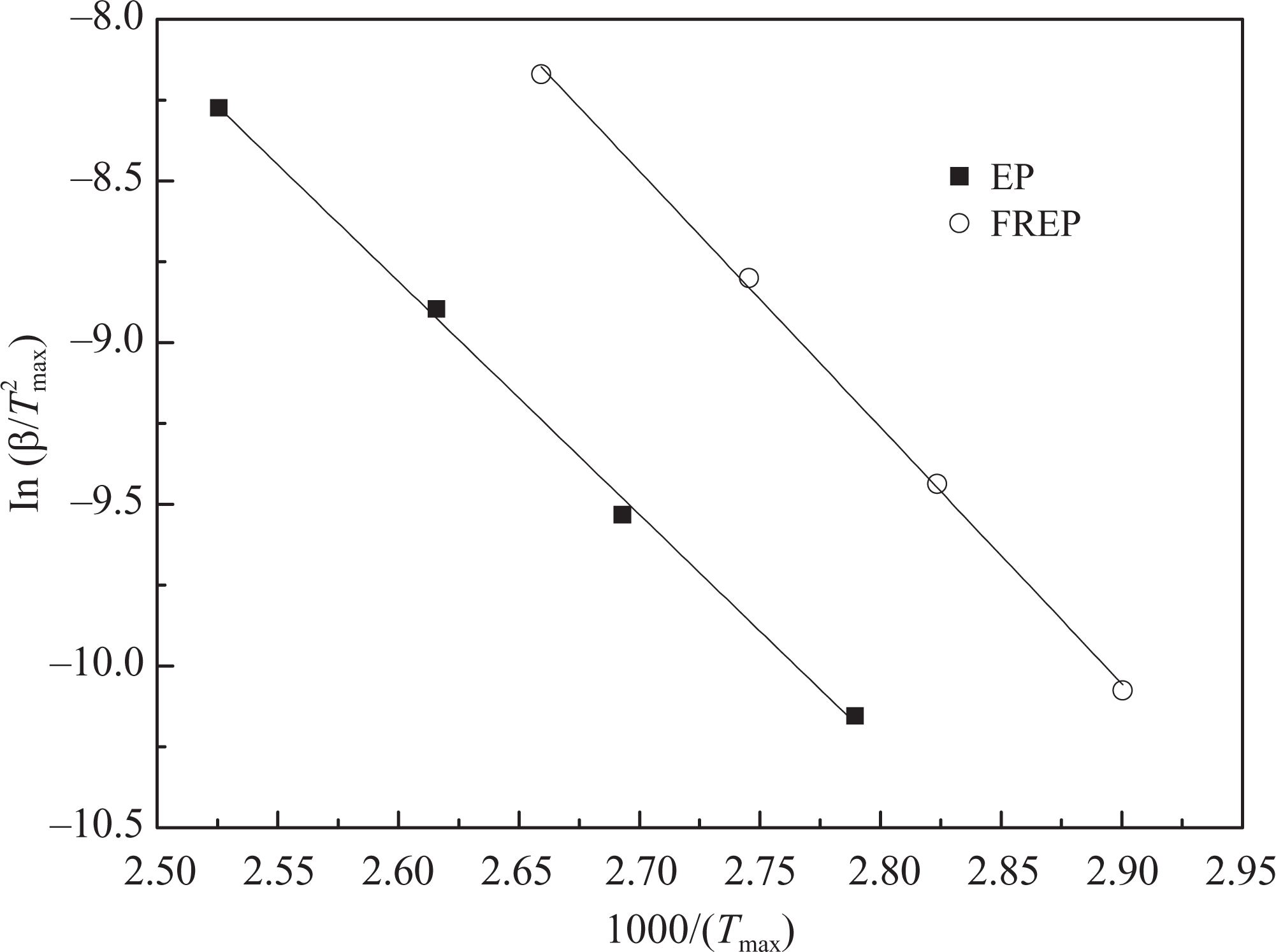
The plots of
Kinetic data for thermal degradation of EP and FREP by the Kissinger method.

FREP: 10 wt% EP/DBAMP; EP: epoxy resin; DBAMP: diethyl bis(2-hydroxyethyl)aminomethylphosphonate.
The activation energy obtained for the thermal degradation process of FREP (65.90 kJ mol−1) is higher than that of pure EP (60.00 kJ mol−1). This means that the flame-retardant additive DBAMP is effective to increase the thermal degradation activation energy and delay the thermal degradation of the EP system. It may be concluded that the addition of DBAMP accelerates the formation of an insulating carbon layer and improves the flame-retardant behaviour, because the char layer plays an important role in the flame retardancy of plastic systems.
Because of the limitations of the Kissinger method, its data can only provide information at the peak temperatures. However, the TGA data were further studied by Flynn–Wall–Ozawa analysis, which is an integral method that can determine the activation energy without information about the reaction order. By this means, more informative kinetic parameters can be obtained.
Based on the data of Figures 2 and 3 and the equation of
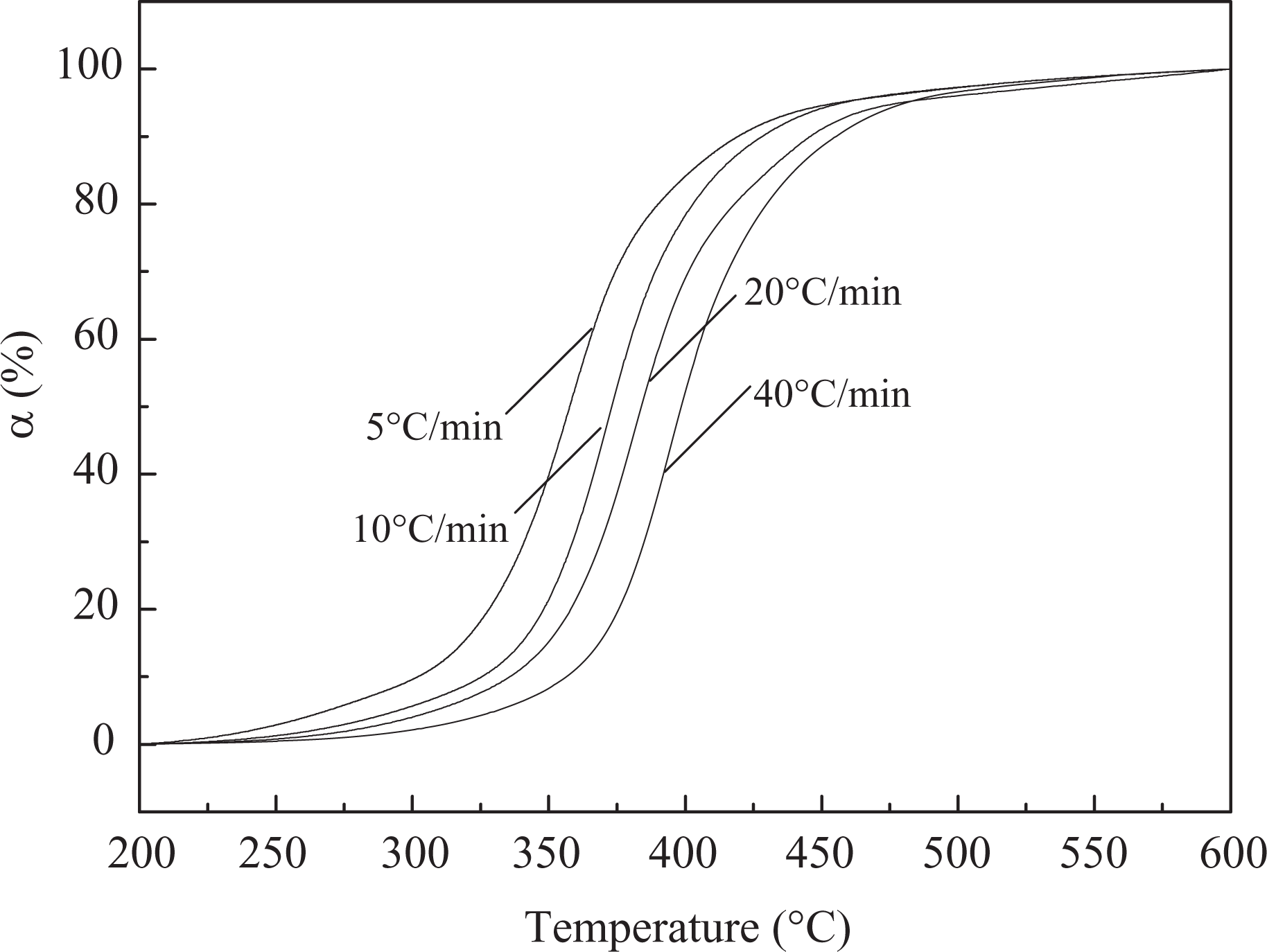
Conversion of EP as a function of temperature.

Conversion of FREP as a function of temperature.

The plots of
According to equation (7), the plots of

The plots of
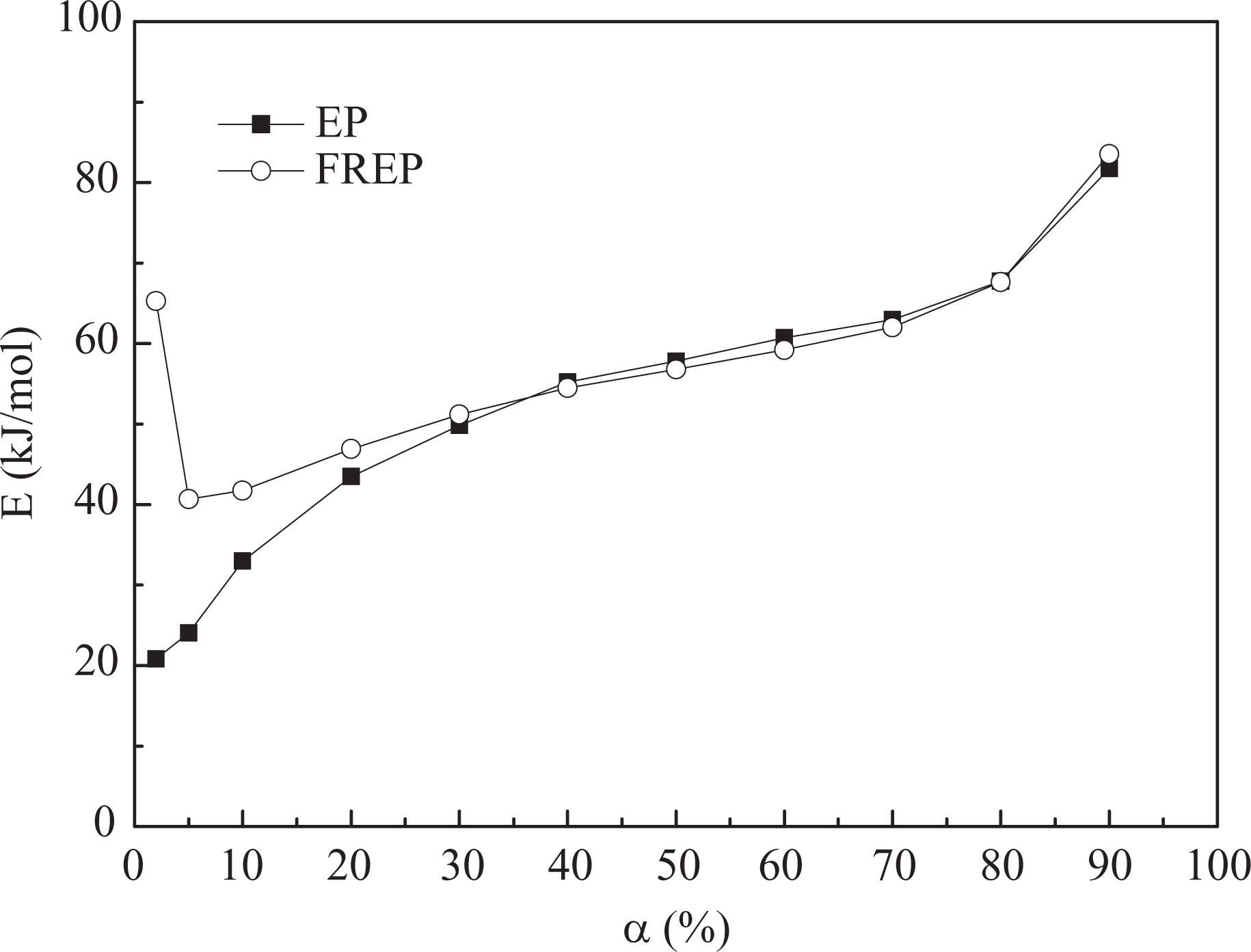
Activation energy curves for EP and FREP systems using the Flynn–Wall–Ozawa method.
It can be observed from Figure 11 that the EP and FREP systems both tend to exhibit three stages of activation energies in degradation processes. In the early stage, the loss of light degradation products, such as carbon dioxide, occurs, and the EP generally completes this stage before 20% weight loss. After this stage, the activation energy of flame-retardant EP rises as the thermal degradation progresses and then remains constant up to 80%, which means the stabilities of the intermediate products are rather similar.
Moreover, it can be observed that the activation energies of FREP are significantly higher than that of pure EP at lower degree of thermal degradation. The resulting data, together with the analysis of the activation energies, indicate that the FREP has better thermal stability than pure EP in the initial thermal degradation stage. In other words, the additive DBAMP causes slower degradation of the EP. In the middle and final stages, the activation energies of EP and FREP systems have a similar trend. Furthermore, the kinetic parameters (
From the above results, it can be found that the activation energies of EP thermal degradation calculated by the Kissinger and Flynn–Wall–Ozawa methods are very similar. Therefore, the above two methods can be used to study the thermal stability behaviour of EP and the flame-retardant mechanism of DBAMP in EP systems.
Conclusions
The thermal degradation behaviour of EP flame-retarded by DBAMP has been investigated by TGA under isothermal conditions in a nitrogen atmosphere. During non-isothermal degradation, the Kissinger and Flynn–Wall–Ozawa methods were successfully employed to analyse the thermal degradation process. The results showed that a remarkable increase of activation energy was observed for thermal degradation in the presence of DBAMP, which indicated that the addition of DBAMP retarded the thermal degradation of EP. The Flynn–Wall–Ozawa method further revealed that DBAMP increased the activation energy of EP thermal degradation in the initial stage, which indicated that the DBAMP stabilised the char residues and improved the flame retardancy of EP in the initial period of the thermal degradation process.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Ningbo Natural Science Foundation (2017A610067), Zhejiang Provincial Innovation Training Plan for University Students (2017R424009), National Undergraduate Training Program for Innovation and Entrepreneurship (201811058002), Zhejiang Provincial Natural Science Foundation of China (LQ13E030002) and Ningbo Municipal Program for Leading and Top-Notch Talents for financial support.