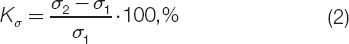Abstract
The aim of this study was to investigate the influence that different fatty acid zinc salts had on the rheological, curing and mechanical properties of natural rubber based composites filled with silica and containing bi-functional organosilanes in the presence or absence of zinc oxide. The results demonstrated that the combination of zinc oxide and zinc soaps had a strongly pronounced anti-reversion effect. The absence of reversion in the cure curves of the rubber compounds comprising a combination of zinc oxide, stearic acid and zinc soaps, results in retention of their mechanical properties, even after overcure.
Keywords
Introduction
The accelerated sulfur vulcanisation of rubber is a complicated chemical process. The detailed reaction scheme of the vulcanisation process is still not well studied, although the practical application of rubber vulcanisates has been known for more than 150 years 1 .
The mechanical properties of rubber vulcanisates are dependent to a great extent on the type of the vulcanisation process and the chemical nature of the crosslinks. Those also determine the application of rubber composites. For instance, vulcanisates crosslinked by peroxides find application in components subjected to static loads – usually under pressure when a low compression set and good heat aging resistance are required. Sulfur-containing vulcanisates find application in articles subjected to dynamic deformation hence need to be more elastic. Having lower dissociation energy polysulfide crosslinks are destroyed easier under any deformation, but their chemical nature affords their ability to transform, hence to lower the stress2,3.
It is known that efficient vulcanisation of rubber by elemental sulfur or by sulfur donors requires the presence of an activator. There are many substances known, mainly metal oxides (e.g. CdO, PbO, MgO etc.), activating to a different degree the accelerators during the sulfur vulcanisation 4 . Yet, zinc oxide is the most efficient and widely used in the practice inorganic activator4–8. The activating effect of the zinc oxide is stronger expressed in the presence of fatty acids like stearic, palmitic, oleic ones 9 , etc.
The high activity of zinc oxide can be explained on the basis of the chemistry of complex formation. A preceding reaction with stearic acid forms the rubber hydrocarbon-soluble zinc stearate and releases water before the onset of cross-linking. Furthermore, ZnO is in many vulcanisation systems a precursor to zinc-derived accelerators 4 . There are other investigations supposing that Zn2+-ions form such active complexes with accelerators, which are more reactive than the accelerator itself 10 .
It seems that the complex, formed of accelerator, sulfur, divalent zinc from the zinc oxide and a fatty acid is the actual accelerating agent, whose activity controls the formation of sulfur cross-links during the vulcanisation. Action, similar to that of the fatty acids, is also performed by their zinc salts - so called zinc soaps 11 . The zinc soaps of a short aliphatic chain (up to C8) display an activating effect in the absence of zinc oxide 12 , too. The substitution of zinc oxide by zinc soaps in the rubber compound, results in a decrease of zinc ions content, defined as eco-toxic 13 . Zinc salts of the organic acids with a longer aliphatic chain (C16-C18) have a less pronounced activating effect, but adding them to rubber compounds in which zinc oxide and thiazole accelerator are present, leads to improved plasticity of the compounds as well as to higher tensile strength and elasticity of their vulcanisates 12 .
Depending on the fatty acids, structure, their zinc salts exhibit different effectiveness in the rubber compounds. Their properties are determined by the ratio of the organic portion to their metal content, as well as by their structure 12 .
The influence of various soaps of organic acids has been investigated in natural rubber compounds in order to demonstrate their properties vs. structure dependency. The investigated rubber compound is intended for tyre production. It has been found that in some cases, the reversion resistance during vulcanisation is considerably improved 13 .
The reversion is a thermo-initiated process related mainly to the overcure. The reversion includes reactions leading to desulfurisation of the polysulfide cross-links and changes in the vulcanisation structure 14 . That results in worsened mechanical properties of rubber vulcanisates 15 . The reversion is most characteristic for natural rubber compounds vulcanised at over 155°C 10 .
The tendency in the production of tyres is the replacement of carbon black, used as a filler, by silica. When using silica as a filler, the presence of bi-functional organosilanes improves the filler dispersion during mixing like the zinc soaps do, but also sets up “polymer-filler” interactions during the vulcanisation. That results in an improved mechanical and dynamic properties of the vulcanisates what is obligatory for rubber compounds16–19.
As mentioned above, a shortcoming of natural rubber is the reversion observed upon vulcanisation at temperatures over 155°C. That shortcoming can be overcome by the usage of various polyfunctional additives, e.g. different zinc salts of the fatty acids.
There are no literature data on the effect that zinc soaps have upon the reversion processes occurring during the vulcanisation of a natural rubber composite filled only with silica. A greater part of the studies dealing with the matter are on compounds filled mainly with carbon black or a combination of carbon black and a small amount of silica. Therefore, the aim of the present work is to investigate the influence of zinc soaps of different aliphatic units (fatty – zinc stearate; cyclic –zinc resinate and a mixture of fatty acids zinc salts (mainly unsaturated) – Plastikol) on the curing (including reversion resistance), rheological and mechanical properties of silica filled composites, based on natural rubber, in the presence and absence of zinc oxide.
Experimental
Materials
The investigations were performed using the following materials:
Natural rubber SVR 10 supplied by Hong Thanh Rubber Pty. Ltd. was used as a polymer matrix. Silica, Ultrasil 7000 GR from Evonik Industries was used as reinforcing filler. It had the following characteristics: Specific surface area (N2) 175 m2/g; specific surface area (CTAB) 160 m2/g; loss on drying 5.2%; pore density 270 g/l; electrical conductivity ≤ 1300 μS/cm. The zinc resinate was purchased from Chemos GmbH, Germany. Plastikol was a trade product, comprising a mixture of different zinc salts of fatty acids. It was manufactured by the “Industrial Chemistry” Ltd company in Bulgaria. The acids in Plastikol were mainly unsaturated – about 50% oleic and 15% linoleic one. Plastikol incorporated also saturated fatty acids – about 30% of stearic and 5% of palmitic acid. The zinc content is about 8%. The other ingredients such as zinc oxide (Struktol WB 700A), stearic acid, zinc stearate, bis(triethoxysilylpropyl)tetrasulfide (TESPT, Si 69 – by Evonik Industries), N-tert-butyl-2-benzothiazolesulfenamide (TBBS – Vulkacit NZ/EGC, produces by Lanxess) and sulfur (Struktol SU 95AF) were also of commercial grades.
Measurements
Mooney viscosity of the investigated rubber compounds was determined according to ISO 289-1:2014. The vulcanisation characteristics of the rubber compounds were determined at 160°C on a Moving Die Rheometer (MDR 2000 Alpha Technologies) according to ISO 3417:2008.
The molecular weight of a rubber segment between two cross-links (Mc)
of the investigated composites was calculated by the Flory-Rhener (1) equation
after determining their equilibrium swelling degree in an organic solvent.
The mechanical properties of the composites studied were determined according to
ISO 37:2011. The Shore A hardness of the composites studied were determined
according to ISO 7619–1:2010, while the abrasion -according to ISO 4649:2010.
Accelerated heat aging of the vulcanisates studied was tested according to ISO
188:2011. The coefficients of aging in terms of tensile strength (Kσ)
and elongation at break (Kε) were calculated by equations (2) and
(3).
The formulations of the compounds based on natural rubber – SVR 10 are presented
in
Compositions of the investigated rubber compounds (phr)

Compositions of the investigated rubber compounds (phr)

The rubber compounds were prepared in two stages according to the mixing protocol
presented in
Mixing protocol of the investigated rubber compounds

The vulcanisation process of the natural rubber based compounds was carried out in an electrically heated hydraulic press using a special homemade mould at temperature 160°C and under pressure 10 MPa.
Curing properties

Mooney viscosity of the investigated rubber compounds
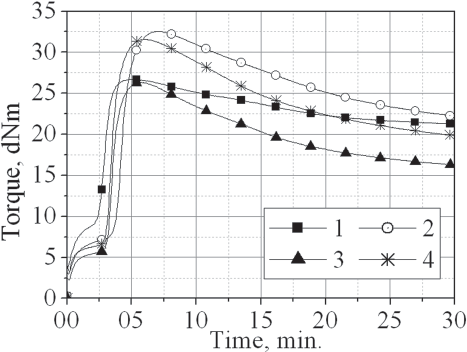
Cure curves of the investigated rubber compounds in the absence of zinc oxide taken at 160°C
The figure shows that the vulcanisation of all the compounds studied starts very rapidly. The scorch time (Ts2) is in the order of 30 s, which is a very short time for compounds produced with a sulfenamide accelerator. Obviously, after reaching the cure time (T90), a strongly pronounced reversion occurs in the course of the cure curves for all investigated compounds. The least reversion is for the control compound (1) containing no zinc soap. According to the figure, the compounds containing zinc stearate (2) or Plastikol (4) have values of maximum torque (MH) higher than those of the compounds that were free from soap (1) or containing zinc resinate (3).
The cure curves of the compounds with various zinc soaps in the presence of zinc
oxide and stearic acid, are presented in
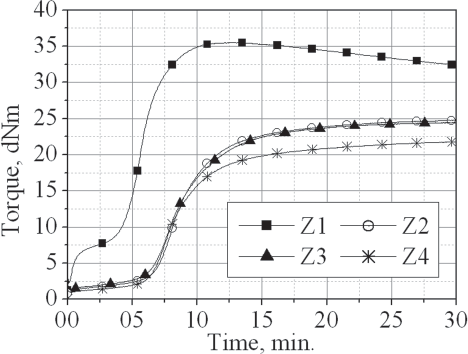
Cure curves of the investigated rubber compounds comprising zinc oxide taken at 160°C
Curing properties of the investigated rubber compounds

The Table shows that the compounds containing no zinc oxide (1, 2, 3 and 4) have considerably higher cure rates and very short scorch times (Ts2). The presence of zinc oxide (Z1, Z2, Z3 and Z4) results in a considerable decrease of the cure rate. That is in compliance with Coran's theory 10 for formation of a chelate complex between the zinc oxide, the thiazole ring of accelerator and the sulfur molecule bound to it, which results in a decrease of the cure rate and formation of a curing network in which the cross-links are of lower polysulfidity, yielding a more stable cross-link network.

Molecular weight of rubber segment between two cross-links (Mc) of the investigated composites
Mechanical properties of the investigated composites, vulcanised for
T90

Mechanical properties of the investigated composites, vulcanised for T90

As seen from
The mechanical properties of the investigated composites, obtained after overcure
(4xT90) are shown in
Mechanical properties of the investigated composites, vulcanised for 4xT90

According to the Table, after overcure (4xT90), the values of modulus
100 and modulus 300 of the composites comprising no zinc oxide (1, 2, 3 and 4)
decrease in comparison to those of the relevant composites vulcanised for the
cure time (T90 –
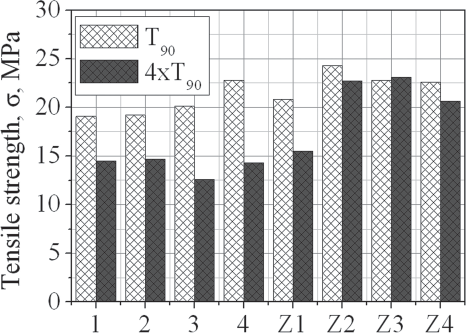
Tensile strength of the investigated composites
The abrasion data of the investigated vulcanisates are presented in
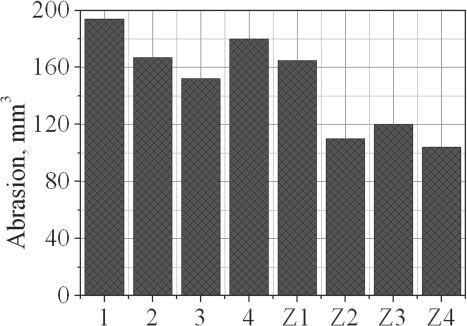
Abrasion of the investigated composites

Aging coefficient of aging of the investigated composites in terms of tensile strength (Kσ)
The vulcanisates containing zinc oxide (Z1, Z2, Z3 and Z5) are considerably more heat aging-resistant, since no change in the coefficient of aging (Kσ) according to the time of vulcanisation is to be noticed. Noteworthy on this point is that the presence of zinc soaps exerts additional positive influence on the aging resistance. Most probably, the cross-link network with lower polysulfidity of the cross-links, formed in the presence of zinc oxide or a combination of zinc oxide and zinc soaps, is the reason for the higher heat aging resistance of these vulcanisates.

Aging coefficient of the investigated composites in terms of relative elongation at break (Kε)
One can see from the results presented that the composites obtained in the presence of a combination of zinc oxide, stearic acid and zinc soaps have better mechanical properties, both before and after accelerated heat aging, even after overcure (4xT90). The reason is in the absence of a reversion of the cure curves of these composites. Probably, a co-ordination complex is formed between zinc oxide, stearic acid, zinc soaps, accelerator and sulfur which yields composites with a more stable vulcanisation structure wherein cross-links of lower sulfidity prevail.
The influence that zinc soaps with different aliphatic parts (fatty – zinc stearate, cyclic – zinc resinate and a mixture of zinc salts of fatty acids (mainly unsaturated) – Plastokol) have on the rheological, curing and mechanical properties of composites based on natural rubber and filled with silica has been investigated. It was found that:
The presence of the investigated zinc soaps results in a considerable decrease of Mooney viscosity of the rubber compounds studied, which is much more pronounced in the presence of zinc oxide and stearic acid.
The combination of zinc oxide and zinc soaps has a strongly pronounced anti-reversion effect. Probably, the reason is the formation of a co-ordination complex between zinc oxide, zinc soaps, accelerator and sulfur, what produces composites of a more stable vulcanisation structure.
The absence of reversion results in retention of the mechanical properties of the investigated composites, regardless of the duration of their vulcanisation.
Footnotes
Acknowledgements
The present research is a result of an international collaboration program between the University of Tabuk, Tabuk, Kingdom of Saudi Arabia and the University of Chemical Technology and Metallurgy, Sofia, Bulgaria. The authors gratefully acknowledge the financial support from the University of Tabuk.