Abstract
Reduced graphene oxide (RGO) or graphite is functionalized with hydroxyl groups for linking to the sides of polyurethane (PU) chains. Blended PU with RGO or graphite is prepared as a control for comparison. The PU composites are compared with respect to their spectroscopic, thermal, mechanical, shape memory, and sheet resistance properties. Scanning electron microscopy images demonstrate the good distribution of functionalized graphene oxide (FGO) or functionalized graphite (FG) particles on the inner surface of the PU. The linking of FGO or FG onto PU does not significantly affect the thermal behavior or shape memory properties but sharply improves the tensile strength of the PU composites without a noticeable decrease in tensile strain. The shape recovery of PU composites remains at approximately 90%, regardless of the FGO or FG content. The FG-linked PU composites exhibit a sharp decrease in sheet resistance as the FG content increases, whereas the sheet resistance of the FGO-linked PU composites does not decrease with increasing FGO content. The control PU composites with blended RGO or graphite show significant reductions in their sheet resistance. Considering the ease of functionalization of the graphite surface and the significant improvement in tensile strength, linking FG onto PU is advantageous for the development of PU composites with low sheet resistance.
Introduction
Fillers such as metals,1,2 carbon nanotubes,3,4 graphene, and graphite have been used to improve the electrical conductivity of polymer composites5,6. Graphite has long been used as a conductive filler due to its high electrical conductivity, scalability for bulk applications, abundance and low cost for applications such as electrically conductive engineering plastic, 7 conductive protective coatings, 8 conductive foams, 9 and radio wave-absorbing materials 10 . Expandable graphite has recently been used as an electrically conductive filler in polymer composites due to the uniform dispersion of graphite 11 . Graphene is prepared from graphite by oxidation and exfoliation and improves the electrical conductivity, mechanical strength, and gas barrier properties of the polymer composites 12 . However, the even distribution of graphene particles in polymer composites requires control of the interface compatibility between the graphene and the polymer matrix. Recently, graphene, which is a conductive functional filler, was chemically linked to a polymer matrix via click chemistry 13 . The linking of functional materials, such as graphene or graphite, onto PU can impart various characteristics to polymer composites and extend their potential applications. Polymer composites are usually prepared by melt mixing or solvent blending, but the chemical linking of graphene or graphite to the polymer may provide secure bonding and a uniform distribution of graphene or graphite in the polymer matrix. However, the chemical linking of graphene or graphite onto polymer is scarcely reported due to the poor reactivity of graphene or graphite particles with polymer surface and the potential problems in large scale processing steps14–16. PU is selected as the polymer matrix in this investigation because of its exceptional mechanical and chemical properties, such as high tensile strain and impact resistance, easy modification, and resistance to oxidation17–19. The modification of PU with functional side groups has been used to develop characteristics such as low-temperature flexibility, 20 biocompatibility,21,22 hydrophilicity, 23 pH sensitivity, 24 and antimicrobial activity. 25 Previously, we have linked RGO 26 and graphite 27 onto PU using the PU modification method. However, the electric conductivity of RGO-linked PU was missing and the tensile strength was low compared with the above functionalized PUs. In addition, the graphite content in the graphite-linked PU was excessive as a polymer composite. Therefore, the RGO- or graphite-linked PUs should be investigated again with the redesigned structures. In this investigation, RGO or graphite is functionalized with hydroxyl groups and chemically linked to PU as a filler through allophanate bonding to attain an even distribution and secure binding of RGO or graphite onto PU. The tensile mechanical properties of PU composites are compared with plain PU because the linked RGO or graphite particles may reduce the tensile properties, as has been observed in some polymer composites 28 . Graphene or graphite is known for its electrical conductivity, so the impact of RGO or graphite on the sheet resistance of the PU composites is one of the main focuses of this investigation. PU composites with low sheet resistance have a few advantages over metals, such as easy molding, corrosion and impact resistance, and elasticity, and therefore have potential applications in electromagnetic wave reflection, electrostatic discharge, and high-voltage cables.
Experimental Method
Materials
Poly(tetramethylene ether)glycol (PTMG, Mn∼2,000 g/ mol, Sigma-Aldrich, St. Louis, MO, USA), 4,4'-methylenebis (phenyl isocyanate) (MDI, Junsei Chemical, Tokyo, Japan), and 1,4-butanediol (BD, Junsei) were dried under high vacuum (0.1 Torr) overnight before use. The synthetic graphite powder (soft black hexagonal crystals, particle size <20 μm, density = 2.2 g/cm3, mp = 4,489°C), potassium permanganate, sodium nitrate, and hydrazine hydrate were obtained from Sigma-Aldrich. Isopentylnitrite (TCI, Tokyo, Japan), 2-(4-aminophenyl) ethanol (Alfa Aesar, Ward Hill, MA, USA), and hydrogen peroxide (Junsei Chemical) were used as received. Dimethylformamide (DMF), diethyl ether, and acetonitrile were obtained from Duksan Pure Chemicals (Ansan, Korea), and DMF was distilled over CaH2 before use.
Preparation of RGO
Graphite (10.0 g) was added to a mixture of sulfuric acid (300 mL) and sodium nitrate (5.0 g) at 0°C, and the mixture was magnetically stirred for 10 min. Potassium permanganate (30.0 g) was added slowly to the mixture, and the mixture was stirred for 30 min at 0°C. The mixture was sonicated for 1 h using a sonicator (Powersonic 410, Hwashin Tech, Korea) and magnetically stirred for 3 h at 25°C. The mixture was then diluted with a solution of distilled water (1.2 L) and hydrogen peroxide (300 mL), stirred for 1 h at 25°C, filtered by aspirator suction through filter paper (Whatman 42), and dried at room temperature for one day to obtain graphene oxide (GO) powder. The GO (5.0 g) was dispersed in distilled water (1.67 L), reduced by magnetic stirring with hydrazine hydrate (1.92 mL) for 24 h at 80°C, and filtered to obtain the RGO powder.
Functionalization of the RGO or Graphite Surface
A solution of 2-(4-aminophenyl)ethanol (23.0 g, 167 mmol) dissolved in 300 mL of acetonitrile and 60 mL of a 1.00 M aqueous HCl solution was stirred at 25°C for 30 min. After the addition of 20.0 g of RGO or graphite, the mixture was stirred at 25°C for 1 h. A solution of isopentylnitrite (19.5 g, 167 mmol) in 100 mL of acetonitrile was added dropwise to the above mixture via an addition funnel with vigorous stirring, and the mixture was stirred at 60°C for 12 h. The mixture was filtered through filter paper (Whatman 42) and washed with DMF until the filtrate was clear. The FGO or FG was washed with diethyl ether (100 mL x 3) to remove the remaining DMF and then dried in a vacuum oven at 60°C for 3 days.
PU Composite Synthesis
MDI (MDI-1, 5.00 g, 20.0 mmol) was added to PTMG (40.0 g, ca. 20.0 mmol) in a 500 mL four-neck flask equipped with a mechanical stirrer, condenser, temperature-controlled heating mantle, and nitrogen inlet. The mixture was allowed to react for 3 h at 50°C to prepare the prepolymer. BD (2.70 g, 30.0 mmol) was dissolved in 10 mL of DMF and added to the prepolymer. The mixture was stirred in the presence of BD for 1 h before MDI (MDI-2, 7.50 g, 30.0 mmol) was added to the reaction mixture. After the addition of MDI-2, the reaction was continued for 1 h with the slow addition of 10 mL of DMF via a dropping funnel to prevent a sudden increase in viscosity. MDI (MDI-3), as specified in

(a) Structure of plain PU, (b) hydroxyl functionalization on the RGO or graphite surface, and (c) linking of FGO or FG onto PU
Composition of the PU composites

The CG1 series used RGO.
The CG2 series used graphite.
A specimen with dimensions of 20 × 20 × 1 mm and a known weight (m1) was swollen in 50 mL of toluene in a closed-cap bottle for 24 h. The swollen weight of the specimen (m2) was measured after quickly removing the toluene on the polymer surface with a tissue. The swollen specimen was dried at room temperature for one week and then weighed to obtain the dry mass (m3). The solvent volume (Vs) of the swollen specimen, averaged over five swelling experiments, was calculated using the weight difference between the swollen (m2) and dry (m3) states and the solvent density (0.8699 g/cm3). The volume of the polymer (V) in its dry state was calculated by dividing the polymer dry weight (m3) by the polymer density. The volume fraction of the polymer in the swollen state (v1) was calculated using the equation Vp/(Vs + Vp). The cross-link density was derived using the following method. Specifically, the interaction parameter () between the solvent and PU was determined from equation (1) 29 .

solubility parameters of the solvent and polymer, respectively
molar volume of the solvent (106.3 cm3/mol)
gas constant (8.31 MPa·cm3·K−1·mol−1)
absolute temperature (298 K)
The solubility parameters of toluene (δ1) and PU (δ2) were 18.2 and 20.5 (MPa)1/2, respectively30,31. The cross-link density was calculated from the Flory-Rehner equation (2):

volume fraction of the polymer in the swollen mass
interaction parameter
cross-link density
A Fourier-transform infrared (FT-IR) spectrophotometer (JASCO 300E, Tokyo, Japan) with attenuated total reflectance capability was used to measure the infrared spectra: 25 scans were performed with a resolution of 4 cm−1 and a 2 mm/s scan speed. A differential scanning calorimeter (DSC, TA instruments DSC-Q20, New Castle, DE, USA) was used to obtain the calorimetry data from heating and cooling scans at a rate of 10°C/min between −50 and 250°C. After melting at 250°C for 5 min and cooling quickly to −50°C, the phase transition of 5 mg of specimen was monitored while warming to 250°C at 10°C/min. The dynamic mechanical properties were measured to determine the phase transition at a very low temperature using a dynamic mechanical analyzer (DMA, Triton TTDMA, Lincolnshire, UK), in which the storage modulus and loss modulus were scanned in tension mode between −150 and 100°C at 10 Hz. Scanning electron microscopy (SEM, Hitachi S-4300SE, Tokyo, Japan) images were obtained of fractured samples that had been coated with platinum (thickness of 12 nm) to observe the inner surface; samples were fractured by freezing in liquid nitrogen, followed by fracturing steps. The electrical sheet resistance of the PU composite film was measured using a four-point probe system (CMT Series, Chang Min Tech, Sungnam, Korea) at room temperature.
Tensile and Shape Memory Tests
The tensile mechanical properties were measured on a universal testing machine (UTM, LR10K, Lloyd Materials Testing, West Sussex, UK) according to the ASTM D638 standard using a 20 mm gauge length, a 10 mm/min crosshead speed, and a 500 N load cell. The same UTM equipped with a temperature-controlled chamber was used for the shape memory test. A specimen of length L0 was drawn to 2L0 at 45°C for 2 min and held at 45°C for 5 min. Afterwards, the specimen was cooled to −25°C with liquid nitrogen for 10 min, and the shrunken length (L1) was measured after the upper grip was released. The shape retention (%) was calculated from L0 and L1 using equation (3). The specimen was heated to 45°C for 10 min, and the length (L2) was measured. The shape recovery (%) was calculated from 2L0 and L2 using equation (4).


Synthesis of the PU Composite
The FGO or FG is chemically linked to PU instead of blending to improve the binding and distribution of RGO or graphite in PU and the interface compatibility with PU. The surface of RGO or graphite is functionalized with groups that can react with PU for the chemical linking. The functionalization proceeds by electrophilic substitution between the π bonds of RGO or graphite and the diazonium salt form of 2-(4-aminophenyl) ethanol. The diazotization of 2-(4-aminophenyl) ethanol is conducted with isopentylnitrite instead of sodium nitrite to improve the solubility in acetonitrile. RGO is prepared from graphite through successive oxidation and reduction steps and then functionalized to obtain phenylethyl hydroxyl groups on its surface, whereas graphite is directly functionalized using the same method to produce hydroxyl groups on its surface. The graphite oxidation step may lead to the breakdown of carbon-carbon bonds but, after reduction can provide, hydroxyl groups other than the functionalized hydroxyl groups
32
. The FGO or FG is then used for linking onto PU in the subsequent linking step. PU is composed of hard (MDI and BD) and soft (PTMG) segments, for which the addition of MDI is divided into MDI-1 and MDI-2 to prevent side reactions between MDI molecules. The carbamate bond in the hard segment is the site of linking FGO or FG onto PU using MDI-3 as an activating reagent following the reported methods23,33-35. The steps of linking FGO or FG onto PU are shown in

SEM images (x 3,000) of the fractured inner surfaces of L, G1-25, CG1-50, G2-50, and CG2-50
The cross-link density of PU composites was determined using the polymer swelling method, in which the swelling of PU is inversely related to the degree of cross-linking (
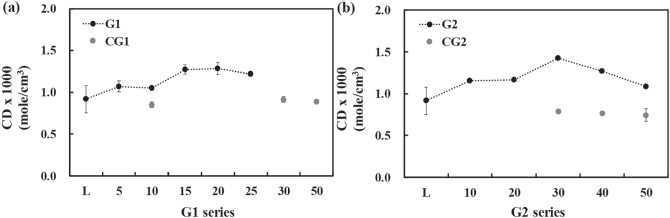
Cross-link density profiles of the (a) G1 series and (b) G2 series
The IR spectra of the FGO and FG in

IR spectra of (a) GO, FGO, (b) graphite, FG, (c) the G1 series and (d) the G2 series, and the resolution of the overlapped carbonyl peaks of (e) L and (f) G2-50
The melting temperature (Tm) of the soft segment and the glass transition temperature (Tg) of the PU composite were investigated by DSC and DMA, respectively. The melting peak of the soft segment is observed between 18 and 21°C in the second heating scan in

DSC heating scans of the (a) G1 series and (b) G2 series, and cooling scans of the (c) G1 series and (d) G2 series
Comparison of the thermal properties of the soft segment of the PU composites

Tg was determined from the loss modulus plot.
The glass transition temperature (Tg) of the soft segment was investigated by DMA, in which the storage modulus and loss modulus were monitored between −150 and 100°C (

Storage modulus plots of the (a) G1 series and (b) G2 series, and loss modulus plots of the (c) G1 series and (d) G2 series
The maximum stresses of the G1 and G2 series sharply increase compared to that of the L form: 38.9 MPa for G1-5, 41.2 MPa for G1-15, 51.7 MPa for G2-10, and 44.8 MPa for G2-30 compared to 15.2 MPa for linear PU (L) (

Maximum stress profiles of the (a) G1 series and (b) G2 series, and strain-at-break profiles of the (c) G1 series and (d) G2 series
The shape recovery and retention results of the G1 and G2 series are compared with those of linear PU (L) in

Shape recovery profiles of the (a) G1 series and (b) G2 series, and shape retention profiles of the (c) G1 series and (d) G2 series
The sheet resistance results of the FGO- or FG-linked PU composites are shown in

Sheet resistance of the (a) G1 series and (b) G2 series
The PU composites linked with FGO or FG are compared with respect to their spectroscopic, thermal, mechanical, shape memory, and sheet resistance properties. Graphene/graphite particle surfaces are functionalized with hydroxyl groups for linking to the sides of PU chains. Blended PU composites with plain RGO or graphite are also prepared as control groups for comparison. The SEM images demonstrate the even distribution of FGO or FG on the inner surface of the PU, except in the G1 series. The linking of FGO or FG onto PU does not significantly affect the thermal behavior or shape memory properties but sharply improves the tensile strength of PU (287% for the G1 series and 344% for the G2 series) without a noticeable decrease in tensile strain. The shape recovery characteristics of G1 and G2 series remain constant at approximately 90% regardless of the FGO or FG content. G2, CG1, and CG2 series exhibit a sharp decrease in sheet resistance as the FGO or FG content increases, whereas the sheet resistance of G1 series does not decrease with an increasing FGO content. The control PU composites prepared with blended RGO or graphite (CG1 and CG2 series, respectively) unexpectedly show reduced sheet resistance. Considering the ease of functionalization of the graphite surface and the significant improvement in tensile strength of G2 series, the linking of FG onto PU is beneficial for the development of PU composites with low sheet resistance.
Footnotes
Acknowledgment
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2016R1D1A1B01014308).
