Abstract
Objective:
The objective of this study was to compare trigger point (TrP) dry needling, TrP electroacupuncture and motor point electroacupuncture of the trapezius muscle for the treatment of myofascial pain syndrome (MPS).
Methods:
This randomised clinical trial included 90 patients divided into three groups. Group 1 was treated with dry needling of TrPs, group 2 with intramuscular electrical stimulation of TrPs, and group 3 with electroacupuncture of motor points and/or the spinal accessory nerve. Each group received seven treatment sessions. The outcomes were the pain score measured by visual analogue scale (VAS) and quality of life evaluated by the 12-item short form (SF-12) health questionnaire. We compared the pain outcome over serial time points using growth curve analysis methods.
Results:
Participants in the three groups experienced significant improvements in pain scores over time. The average pain level of participants in group 3 across the repeated assessments was 0.98 units lower than in group 1 (mean difference (95% confidence interval (CI) = 1.74–0.23)), p = 0.012). There were no significant differences in pain scores between participants in groups 1 and 2, and there were no significant differences in quality of life across the three groups at the end of the treatment period.
Conclusion:
Our results provide evidence that electrical stimulation of motor points and/or of the spinal accessory nerve may be superior in terms of pain relief (but not quality of life) to dry needling and possibly electrical stimulation of trigger points for the management of MPS involving the trapezius.
Trial registration number:
TRIAL-RBR-43R7RF (Brazilian Clinical Trials Registry)
Introduction
Myofascial pain syndrome (MPS) is one of the most common causes of musculoskeletal pain; it is often underdiagnosed with a prevalence ranging from 21% to 93%.1,2 It is a localised muscle disorder associated with the presence of myofascial trigger points (TrPs) within taut bands of skeletal muscles, referred pain and brief contractile responses during mechanical stimulation of TrPs. 3 Evidence supports the understanding that TrPs are associated with dysfunctional motor endplates due to the presence of spontaneous electrical activity, endplate noise or disturbance in muscle spindles. Nociception in MPS involves both local and referred pain; however, its complete pathophysiological mechanism is not exactly known. 4
The cervical and lumbar regions, as well as the hip and shoulder girdles, are the areas of the body most commonly affected in MPS. Importantly, the upper trapezius is the most frequently affected muscle, typically with TrPs that refer to the posterior region of the neck and temporal region.3,5,6
MPS is diagnosed by searching for palpable tense muscle bands in which the TrPs are found. These are extremely painful points that produce local or referred pain at a distance when stimulated by digital palpation or localised needle puncture. MPS often presents with localised visible or palpable muscle contraction – that is, a localised twitch response. 7
Previous studies have attempted to explain TrP formation in MPS. One study suggested that myofascial pain is a form of neuropathic pain of the musculoskeletal system, based on the idea that every denervated structure is hypersensitive. Thus, dysfunction occurs mainly in segmental nerve branches, which may explain the absence of muscle lesions and the presence of sensory, motor and autonomic changes in MPS. 8
Non-invasive treatments including manual therapies (such as myofascial induction, compression, transcutaneous electrical nerve stimulation, heat, cryotherapy and hydrotherapy) are commonly used for the treatment of MPS.9,10 These techniques not only improve physical function but also relieve pain. 9 They may increase mobility and decrease functional disability and pain pressure thresholds. In addition, they have also been shown to improve deep fascial motion and reduce muscle stiffness. These effects are attributed to mechanical strain and changes in cellular physiology. 10
Pharmacological treatments include anti-inflammatory agents, myorelaxants, antidepressants, anticonvulsants, benzodiazepines and opioids. Minimally invasive techniques include TrP dry needling, TrP infiltration with local anaesthetics and electrical stimulation of motor points. 11 Importantly, few studies have compared dry needling of TrPs with electrostimulation of TrPs or motor points for the treatment of MPS.12,13
Needling and electrical stimulation techniques have evolved from acupuncture. Evidence suggests that traditional acupuncture point locations become sensitive to touch in case of muscle dysfunction, and overlap considerably with TrP locations. 14 Interestingly, classical acupuncture point locations also correlate with neural structures such as muscle motor points.15,16
A motor point is defined, from an anatomical perspective, as the region where the terminal area of the motor nerve fibre (motor endplate) is dense - that is, where the nerve from the motor root enters the muscle fibre. From an electrophysiological point of view, a motor point is defined as the point with the highest excitability of the muscle, where muscle contraction can be elicited by the least amount of electrical stimulation.17,18
Needle insertion may be the most effective treatment for MPS when guided by careful inspection for signs of sensory, motor and autonomic dysfunction in the affected skin and muscles. This needling method takes a neurofunctional approach and involves the performance of intramuscular stimulation;8,16 it is referred to as neurofunctional acupuncture.13,16 Intramuscular stimulation can be performed with stimulation of TrPs or motor points present in the palpable tense bands, either by manual or by electrical stimulation.16,19,20
The effectiveness of intramuscular stimulation at relieving chronic neck pain has previously been demonstrated. 21 This technique improves the analgesic efficiency of acupuncture, 20 where low frequencies (2 Hz) increase the release of enkephalins and high frequencies (100 Hz) stimulate the release of dynorphins. 19 The descending inhibitory system is also activated by electroacupuncture. 22
Dry needling consists of deep needling, typically using acupuncture needles in the region of TrPs in the muscle, causing brief contractile responses with consequent changes in muscle fibre length and tension. It also activates mechanoreceptors that generate sensory stimuli in the pain gate system, blocking the nociceptive TrP input and relieving muscle tension and pain. 23 Dry needling does not use electrical stimulation, similar to traditional Chinese acupuncture, and must not be confused with needling of so-called meridians in traditional Chinese acupuncture. 24
Given the lack of previous studies comparing dry needling with different forms of electrical stimulation in MPS involving the trapezius muscle, the aim of this study was to compare motor point electroacupuncture, TrP electroacupuncture and dry needling for the treatment of patients with MPS involving the trapezius by evaluating pain scores and health-related quality of life. Our hypothesis was that electrostimulation used for the treatment of trapezius myofascial pain would be superior to dry needling and electroacupuncture of TrPs.
Methods
This was a prospective, randomised, open-label clinical trial. The study was approved by the Research Ethics Committee of the Botucatu Medical School (Unesp) on 7 May 2016 (CAAE no. 56654416.6.0000.5411; opinion no. 1,621,990) and registered in the Brazilian Registry of Clinical Trials (REBEC) at http://ensaiosclinicos.gov.br (TRIAL-RBR-43R7RF). It is reported in accordance with the CONSORT guideline. 25
Participants were recruited between October 2016 and September 2019 using the webpage of the Botucatu Medical School, newspapers, social networks and local radio stations. Study participants were clinically examined and selected according to the diagnostic criteria for MPS - that is, the presence of the four major criteria and at least one minor criterion that define the syndrome. The major criteria are the presence of tense bands in muscle, presence of intense pain in the TrPs in a tense band, reproduction of pain by pressure at the TrPs, and limitation of range of motion due to pain. The minor criteria are a contractile response evoked visually or upon palpation, a contractile reaction to TrP needling, electromyographic demonstration of characteristic electrical activity in a painful nodule within a taut band, elicitation of pain and sensory abnormality in the distribution of the TrPs corresponding to their compression. 26
In order to meet the inclusion criteria, subjects needed to be >18 years of age, have a history of musculoskeletal pain in the neck and shoulders with involvement of the upper trapezius, and demonstrate the presence of active TrPs with a pain score > 3 on a visual analogue scale (VAS). The exclusion criteria included cervicobrachial pain due to radiculopathy confirmed by clinical evaluation, neurological signs and/or abnormal imaging findings, diabetes mellitus and/or peripheral neuropathies, use of analgesic drugs in the previous 30 days, fibromyalgia, contraindications to the use of electrical stimulation (i.e. use of a pacemaker or other electrical stimulators), use of anticoagulant drugs, and pregnancy.
Groups
The selected patients read and signed the informed consent form and answered general questions regarding their pain using the 12-term short form (SF-12) health questionnaire 27 and the VAS. Subsequently, they were randomised (through the website www.random.com with allocation concealment using opaque envelopes) to one of the following groups (n = 30 participants each): group 1, who received dry needling of TrPs; group 2, who received TrP needling with electrical stimulation; and group 3, who received needling of the motor point and/or the nerve innervating it (accessory nerve) with electrical stimulation. In all groups, the upper trapezius muscle (or the accessory nerve) was the target of the intervention (Figure 1).
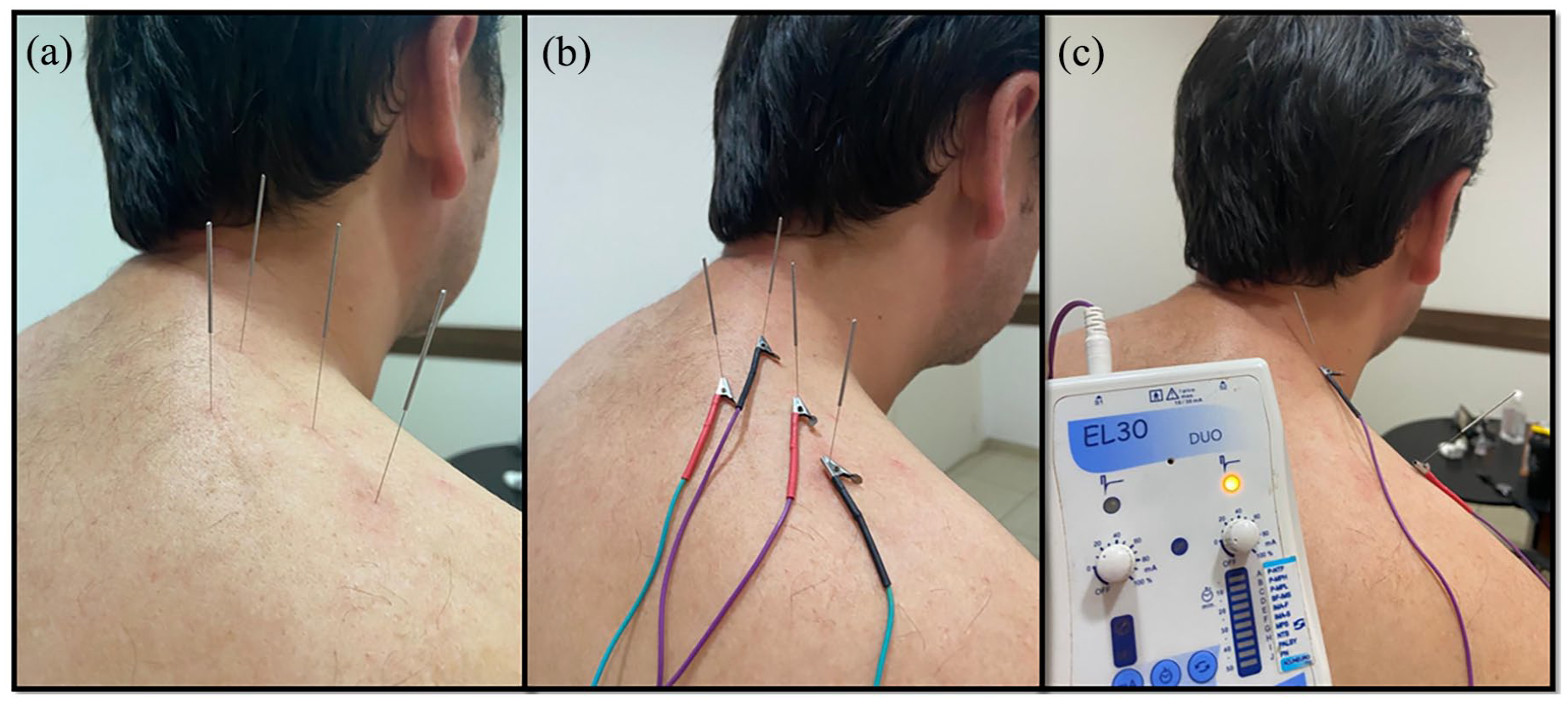
Photographs of the techniques used as interventions. (a) Group 1: dry needling of trigger points. (b) Group 2: needling and electrical stimulation of trigger points. (c) Group 3: needling and electrical stimulation of motor points and/or the spinal accessory nerve. The electrical stimulator used in groups 2 and 3 can be seen in picture C.
Procedures
Patients were treated using the different techniques according to group allocation. In group 1, patients were treated with unilateral or bilateral upper trapezius TrP dry needling. No mechanical (in-out movement of the needle) or electrical stimulation was performed. In group 2, TrPs of the upper trapezius were unilaterally or bilaterally needled and electrically stimulated using an electroacupuncture device (NKL 608, Brazil) that delivered direct current (pulse duration 200 ms) at a frequency of 2 Hz and intensity of 3–4 mA (depending on patient’s response). In groups 1 and 2, needles were inserted deep into the trapezius TrPs until the moment the patient experienced brief contractile responses with consequent muscle fibre shortening and tension changes. In group 3, the motor point of upper trapezius and/or the accessory nerve was/were needled and electrically stimulated. The motor point of the upper trapezius, or innervation zone, was determined in keeping with previous studies at the midpoint of the longitudinal axis of the muscle, or halfway between the C7 spinous process and the prominence of the acromioclavicular joint (Figure 2). 28 The same device (NKL 608, Brazil) was used to deliver electrical stimulation using the same parameters as above. Muscle contraction was visually and tactilely analysed and used to confirm the correct needling location.
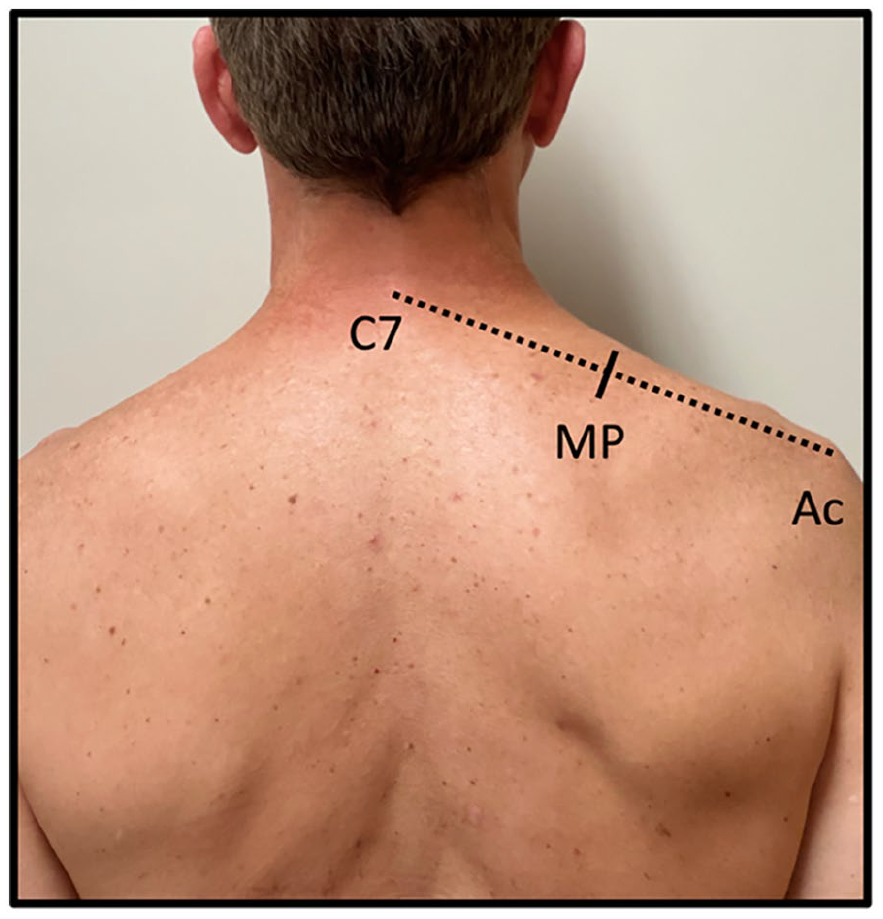
Upper trapezius motor point (MP) located at the midpoint between the C7 spinous process and the lateral margin of the acromion (Ac).
Treatment sessions were repeated weekly for 7 weeks, and participants were evaluated at eight timepoints: before the beginning of the treatment; immediately after each session; and after the end of the seven sessions. At all these timepoints, their pain scores were evaluated. The SF-12 health questionnaire was administered only before the first and after the last session.
All groups were treated for a period of 20 min using 0.25 × 40 mm stainless steel acupuncture stainless steel needles (Dongbang, South Korea), which were inserted to a depth of 20–30 mm, depending on the subcutaneous tissue thickness of the patient. The same acupuncture certified physician (with 20 years’ experience) treated all patients to prevent biases in the execution of the techniques. This physician was not blinded; however, the patients were treated in individual rooms, without being informed about the technique being performed.
Outcomes
The pain score on the VAS was the primary outcome of the study. 29 Health-related quality of life at baseline and at the end of the last treatment session was assessed as a secondary outcome using the SF-12 physical and mental health summary scales. 27
Sample size calculation and statistical analysis
GLIMMPSE 3.0.0 software was used to calculate the required sample size (http://glimmpse.samplesizeshop.org) using the following parameters: power of 90%; two-tailed alpha value for type I error of 0.05; allocation ratio of 1:1:1; eight measurements of the primary outcome variable (VAS); use of the Hotelling–Lawley trace test, equivalent to the proposed multilevel analysis, to test the null hypothesis of similar pain scores between the three groups; assumption that the average pain level at the beginning of the study would be the same across the three groups (VAS = 8); estimated mean VAS decreases of 0.2, 0.4 and 0.6 points at each assessment for groups 1, 2 and 3, respectively; VAS standard deviation of 1.66; 13 baseline correlation of 0.6 between repeated measures; and decay rate of 0.3 over time. Thus, the estimated sample size was 81 participants; this was increased by 10% to consider the possibility of losses, resulting in a final target sample size of 90 participants.
Continuous demographic variables with approximately normal distribution were described by the mean and standard deviation, continuous variables with non-normal distribution were described by the median and interquartile range, and categorical variables were described as absolute numbers and proportions. 30 The chi-square test was used to compare categorical variables at the baseline when the expected values for all cells in the contingency tables were ⩾ 5; otherwise, Fisher’s exact test was used. We used analysis of variance (ANOVA) for the baseline comparisons involving continuous variables with approximately normal distributions, and the Kruskal–Wallis test for variables with non-normal distribution. The same approach was used to compare quality of life scores at the end of the study.
We used growth curve analysis to compare the trajectory of VAS pain scores over time across the three treatment groups. 31 Growth curve analysis is an established multilevel regression method to analyse time course data, which involves multiple repeated observations or measurements over time. Growth curve analysis allows the explicit modelling of change over time and the quantification of both individual-level and group-level differences, considering the correlation between repeated measurements nested within individuals. Accordingly, growth curve analyses are immune to the multiple comparison biases that occur in analyses using other statistical methods where each point in time is treated as a separate outcome. In our growth curve analysis models, we used the variables of treatment group (using group 1 as the reference for comparisons between groups) and time (relative to the moment of each measurement of the pain outcome) as fixed factors, and examined for any interactions. The model also included participants and the time variable using random effects. The time variable was sequentially inserted in the models through linear orthogonal terms and second- and third-order polynomials. The model with the best degree of fit to the data was evaluated by calculating −2 times the log-likelihood change, which follows a chi-square distribution with degrees of freedom equivalent to the number of parameters added to the models. All analyses were performed using R software (version 4.0.5) and the lme4, 32 lmerTest 33 and ggplot2 34 packages at a two-tailed alpha level of 0.05 for statistical significance.
Results
Figure 3 shows the participant inclusion flowchart, as recommended by the CONSORT statement. Table 1 presents the comparison of the three groups at baseline concerning the distribution of clinical and sociodemographic variables. Only the variable related to pain location presented a statistically significant difference between the groups, as group 2 exhibited a higher frequency of pain in the cervical region compared to the other two groups.
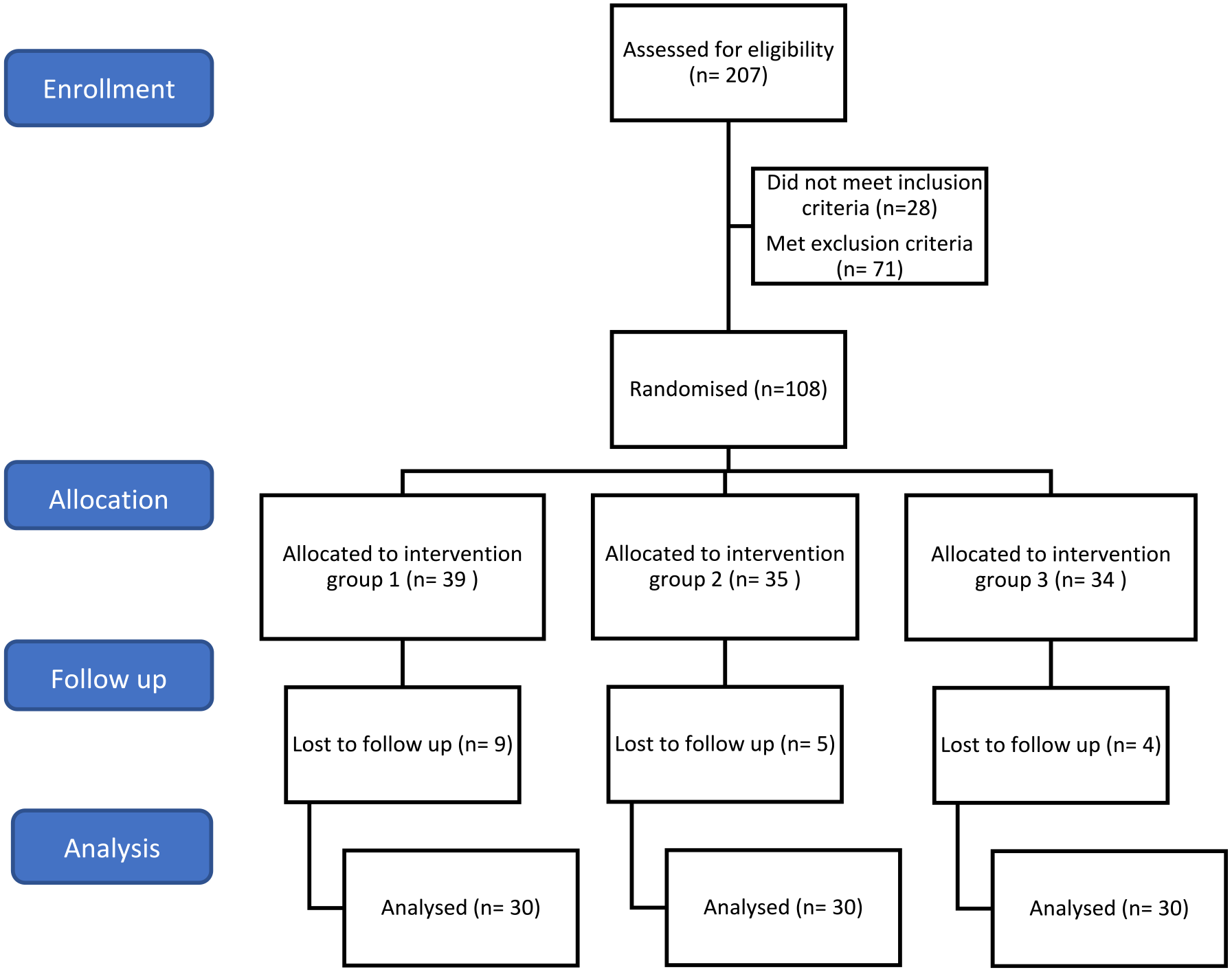
Participant inclusion flowchart.
Baseline characteristics of the study groups.
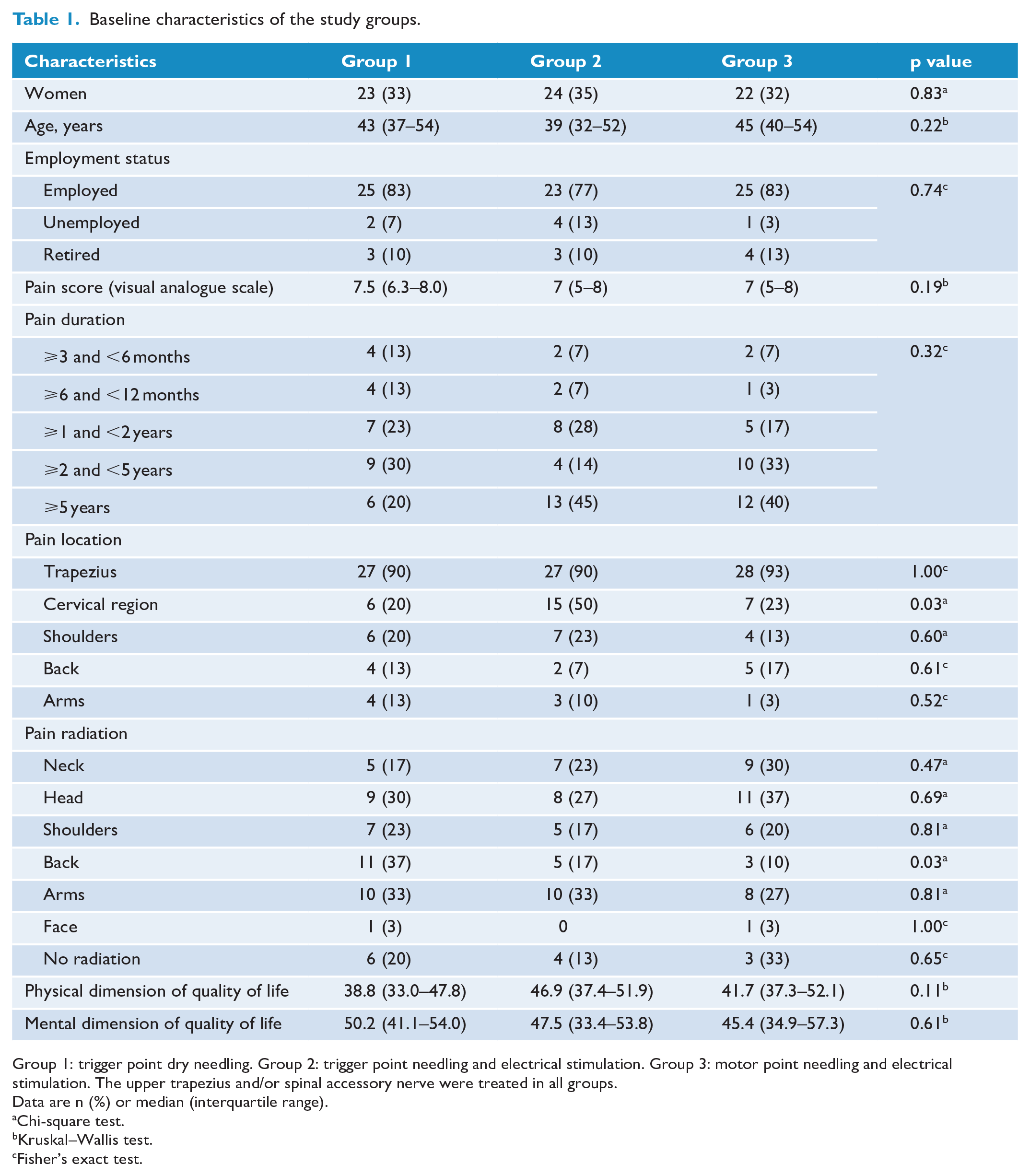
Group 1: trigger point dry needling. Group 2: trigger point needling and electrical stimulation. Group 3: motor point needling and electrical stimulation. The upper trapezius and/or spinal accessory nerve were treated in all groups.
Data are n (%) or median (interquartile range).
Chi-square test.
Kruskal–Wallis test.
Fisher’s exact test.
Of the three models analysed, the one where the time variable was inserted through a second-order orthogonal polynomial term achieved the best fit to the observed data (p < 0.001). Figure 4 shows the progression of the participants’ VAS pain scores in the three groups over the eight evaluations, including the baseline assessment.
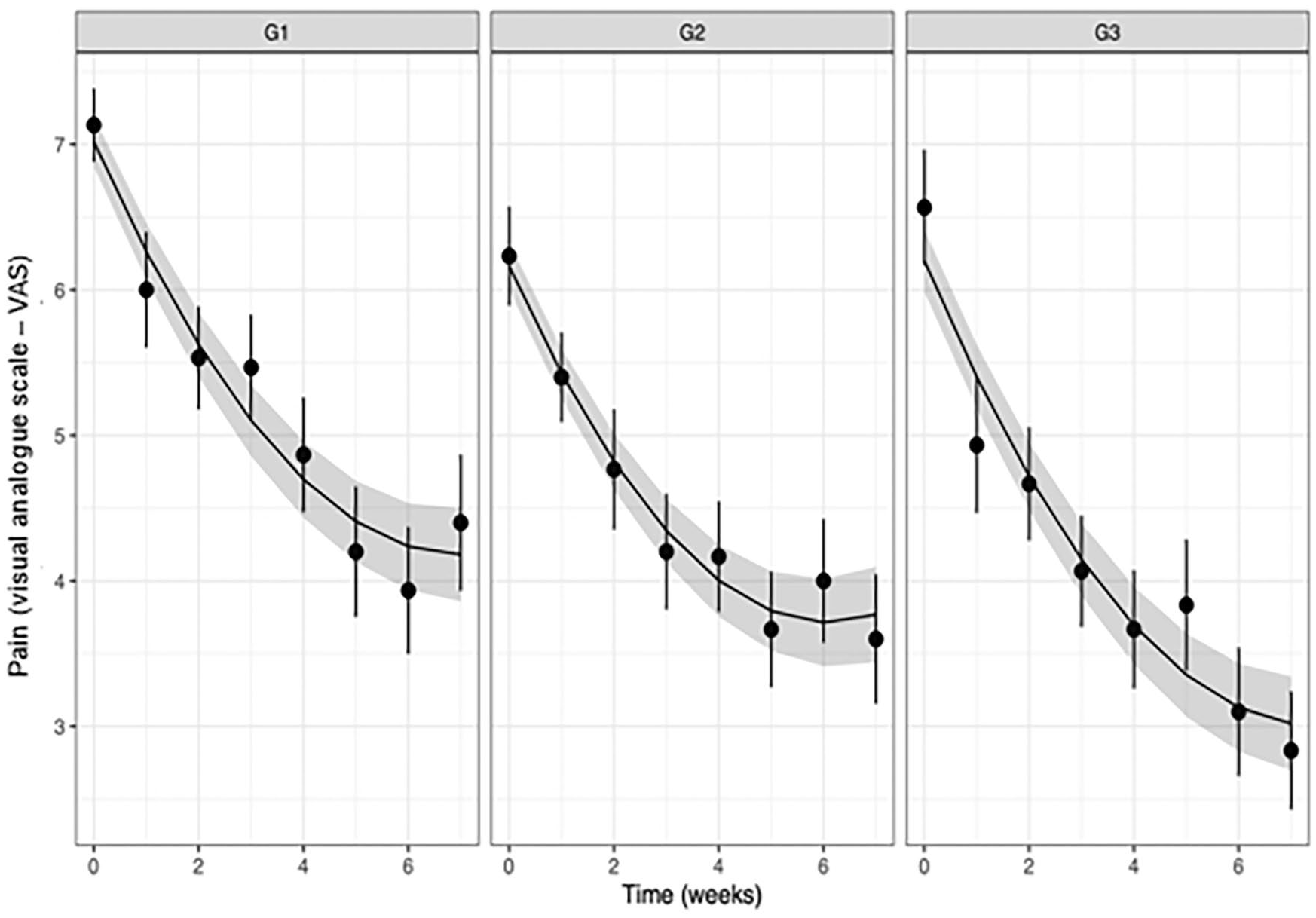
Pain progression of participants in the three groups over time. Group 1: trigger point dry needling. Group 2: trigger point needling and electrical stimulation. Group 3: motor point needling and electrical stimulation. The upper trapezius was treated in all groups. The points represent mean pain values, the bars represent a unit of standard error, and the curve and the band around it represent the predictions of the statistical model used in the analyses and the 95% confidence interval associated with such predictions.
Table 2 describes the estimated β coefficients for the variables examined in the growth curve regression models. The β coefficient value of the intercept corresponds to the mean pain VAS score in group 1 over the eight evaluations. The β coefficients of groups 2 and 3 refer to the comparisons of the mean pain of these groups over the eight evaluations in relation to the group 1 mean. Only the average pain in group 3 was statistically significantly lower than that of group 1, by approximately one point on the pain VAS (mean difference (MD) = 0.98, 95% confidence interval (CI) = 1.74–0.23, p = 0.012). The time variable linear and quadratic term values refer to the slope of the pain decline curve in group 1. The time variable linear and quadratic interaction terms with groups 2 and 3 quantify the comparison of the slope of the pain decline curves of these two groups in relation to group 1 and indicate that the slopes from groups 2 and 3 were not significantly different from that of group 1.
Parameter estimates of the analysed variables in the growth curve regression models of the experimental groups.
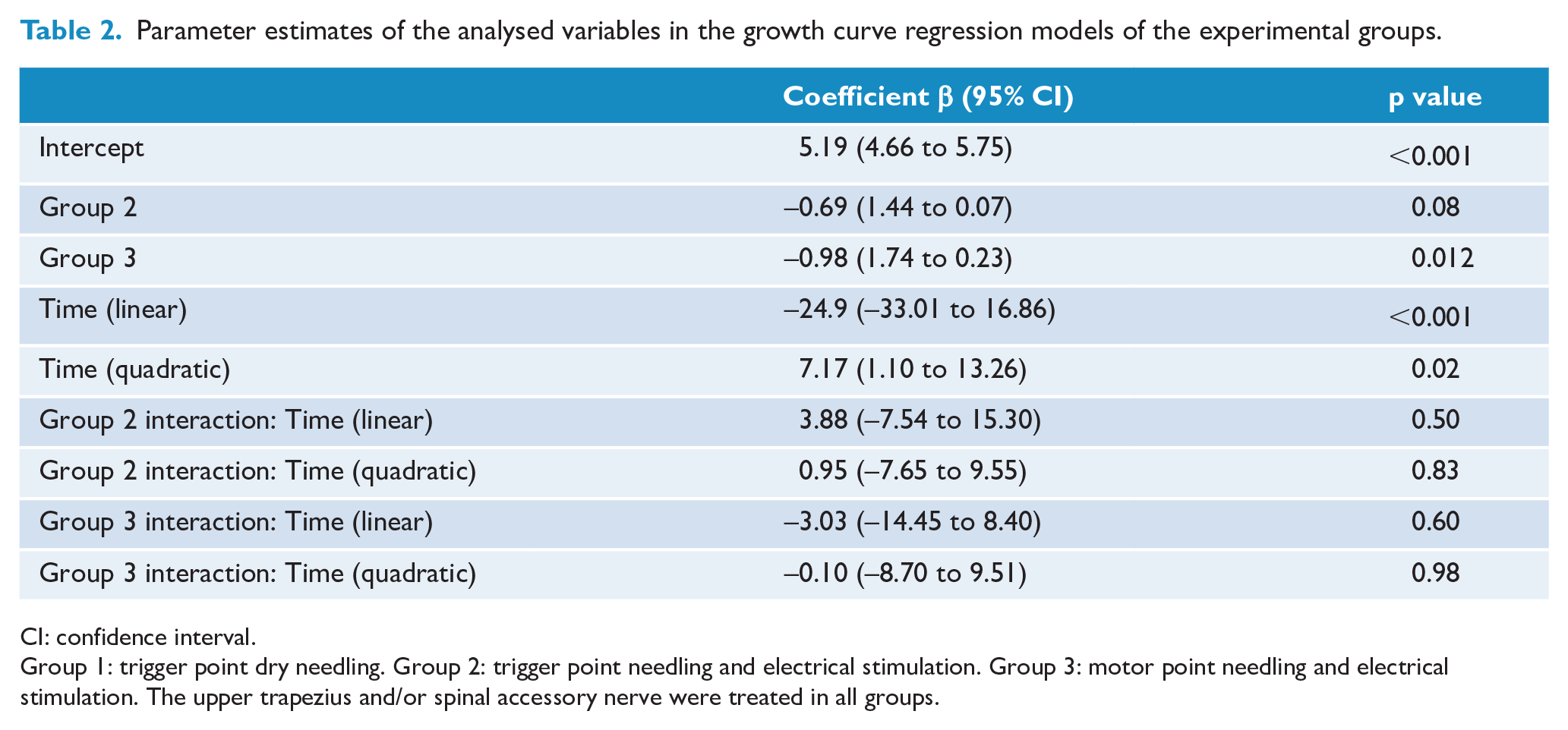
CI: confidence interval.
Group 1: trigger point dry needling. Group 2: trigger point needling and electrical stimulation. Group 3: motor point needling and electrical stimulation. The upper trapezius and/or spinal accessory nerve were treated in all groups.
A reparameterization of the regression model assigning group 2 as the reference for treatment comparisons did not reveal any significant differences between groups 2 and 3 regarding average pain levels over time or the slope of the pain decline curves.
Finally, we conducted a post hoc sensitivity analysis where the final statistical model was adjusted for pain location as a fixed effect, as this was the only variable with a statistically significant difference in the comparisons between groups at baseline (neck pain, p = 0.03, χ2). In this new analysis, the difference between the mean pain in group 2 versus group 1 was statistically significant (β = −0.89, 95% confidence interval (CI) = −1.64 to −0.13, p = 0.02), as was the difference between the mean pain score in group 3 versus group 1 (β = −1.04, 95% CI = −1.77 to −0.32, p = 0.005). There were no other relevant differences regarding the remaining parameters in the model.
In addition, no differences were observed between the quality-of-life dimensions between the three treatment groups, as shown in Table 3.
Comparison of the physical and mental dimensions of quality of life between the three groups.
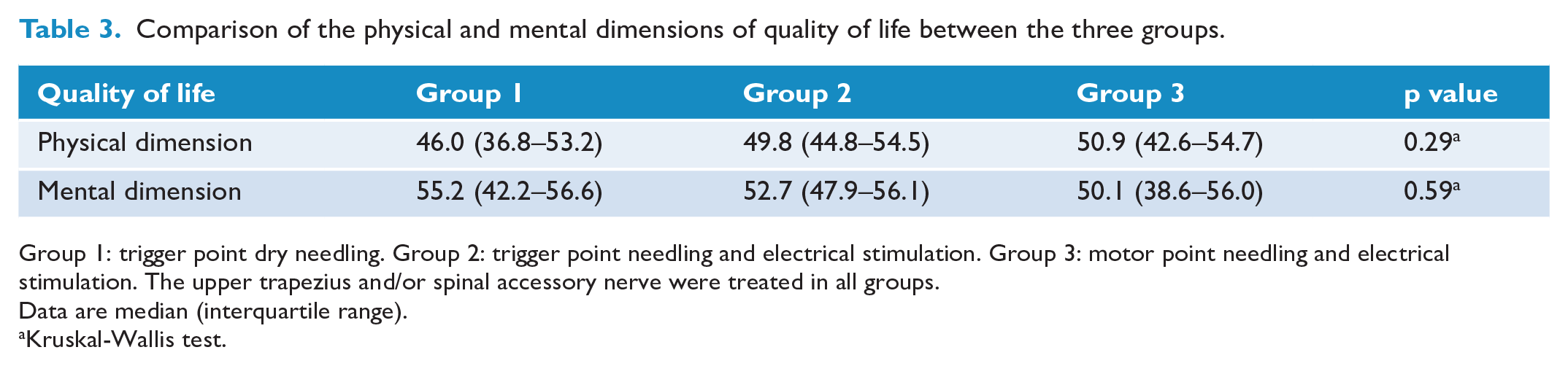
Group 1: trigger point dry needling. Group 2: trigger point needling and electrical stimulation. Group 3: motor point needling and electrical stimulation. The upper trapezius and/or spinal accessory nerve were treated in all groups.
Data are median (interquartile range).
Kruskal-Wallis test.
Discussion
To the best of our knowledge, this is the first study to compare three different techniques for the treatment of MPS involving the trapezius. We found that all three interventions improved pain and that electrical stimulation of motor points was superior to dry needling of TrPs in that regard. As for the quality-of-life outcome, we did not find any significant differences between the three treatment groups.
In our study, the female participants were most affected by MPS, corroborating the results of other studies.35,36 According to other authors such as Skootsky et al., 1 Kaergaard and Andersen 2 , Gerwin 36 and Wolfe et al., 37 MPS is more prevalent in participants of working age, who were represented by 87% of the patients included in our study, with 81% being employed.
This high prevalence of myofascial TrPs in the functionally active age group has also been reported by others. Repetitive work activity and muscle stress probably predispose to the onset of MPS in the presence of active TrPs. Generally, advancing age and decreasing work activity intensify the stiffness and range of motion restriction caused by latent TrPs compared to the pain caused by active TrPs. 36
Despite being the focus of the study, the trapezius was the only muscle affected in most patients (51.1%), corroborating the existing literature. 36 Fischer also identified this muscle as being the most sensitive to algometer pressure. 5
In a large percentage of patients, the pain lasts for more than 3 months (96.7%), reinforcing the dogma that MPS is a chronic disease that often progresses from an acute event not properly treated, with consequent central sensitisation.3,35 It is well documented that, in MPS, all referred pain is due to sensitization of the central nervous system. 4
At the beginning of treatment, all groups exhibited similar mean pain VAS scores, which decreased over the sessions. However, over the course of the repeated pain assessments performed during the 8 weeks of this study, only the average pain VAS scores of participants from group 3 were significantly lower than those from participants from group 1, who were treated with dry needling.
Ga et al. 12 compared the efficacy of electrical stimulation of TrPs and C3 to C5 nerve root electrical stimulation (intervention group) relative to the injection of lidocaine into TrPs (control group). They observed that participants treated with the electrical stimulation intervention had superior results in the treatment of MPS involving the upper trapezius. However, Couto et al. 13 compared TrP and nerve root intramuscular stimulation with 1% lidocaine injection and a transcutaneous electrical nerve stimulation placebo and demonstrated the superiority of intramuscular stimulation compared to the other treatments. Another study reported improved pain when treatment with intra-muscular stimulation in general was compared to classical acupuncture and dry needling methods. 38 In this study, the nerve roots innervating the trapezius were not stimulated, preventing a direct comparison with previous studies.
The three groups exhibited clinically significant improvements in pain VAS scores (>23% compared to baseline values). Regardless of the technique, dry needling is an effective treatment for upper trapezius MPS, as has been reported by other authors.9,12,13,16,19 –21,38 –40
Based on the technique proposed by Gunn, 8 MPS (represented by its TrPs and muscle shortening) actually responds better to intramuscular stimulation, whether by needle manipulation or electrical stimulation, than dry needling.12,13,38 Therefore, it was expected that groups 2 and 3 would experiences better pain outcome in this study.
Gunn proposed this technique based on the theory that MPS is a neuropathic pain that affects the musculoskeletal system. Whatever the muscle affected by MPS, the mechanisms of pain are believed to be the same, that is, hypersensitivity and the effects of muscle shortening due to increased muscle tone. The prolonged muscle shortening causes pain, strains tendons, and damages bones and joints where they attach. 8
Manifestations of this ‘neuropathy’ are almost always found in both the dorsal and ventral branches of the segmental nerve, thus becoming a kind of ‘radiculopathy’. Signs and symptoms are projected to the dermatomes, myotomes and sclerotomes supplied by the affected nerve root. 8
Also, in MPS there is an interaction with the autonomic and sudomotor system, causing sudomotor and pilomotor reflexes, increasing permeability of blood vessels and leading to oedema and other trophic changes. The aim of acupuncture and electrical muscle stimulation is to release muscle shortening by needling the regions of muscle contraction and the paraspinal muscles of the involved spinal segment. Thus, these needling techniques promote relaxation of contracted muscles, decrease impulses from the C fibres of affected nerves and inhibit the sympathetic-excitatory reflex.13,41,42
As previously mentioned, this study did not detect any significant differences in health-related quality of life, evaluated using the SF-12, in the three studied groups. A prior study conducted with MPS patients, also using SF-12 to evaluate the subjects, similarly found no significant difference between groups, although the needling techniques used were different from our study. 13 Those authors had conducted a single-blind randomised clinical trial to investigate the efficacy of a vacuum myofascial therapy device on pressure pain threshold, among other outcomes, in non-specific neck pain. The SF-12 physical and mental components similarly did not differ among the groups.
Kiralp et al. 43 proposed hyperbaric oxygen as a new treatment modality for MPS. They found that the mental and physical health SF-12 scores improved significantly in the intervention group after 3 months compared with pre-treatment values. Ata et al. 44 compared the effects of kinesiotaping and lidocaine injections to treat MPS and found a significant improvement in the intervention group, with respect to the SF-12 physical component evaluations, 1 month after the last treatment. Masarwa et al., 45 in a preprint manuscript, evaluated the effect of TrP compression therapy as an adjunct to an exercise programme to treat MPS and found, upon evaluation 1 month after the final intervention, that SF-12 life quality scores improved in the intervention group. This study is not directly comparable as we did not evaluate the patients at the same time point, relative to the final session of treatment, as other published studies.
This study revealed pain improvements in patients treated by TrP electrical stimulation and motor point and/or spinal accessory nerve electrical stimulation. However, it is worth mentioning that motor point and spinal accessory nerve stimulation use fewer needles and arguably improve the comfort of the patient by avoiding the needling of hypersensitive TrPs. Thus, when motor points and/or the spinal accessory nerve are stimulated, only one needle is placed in the affected muscle. On the other hand, when TrPs are the target of stimulation, multiple needles may be needed. For this reason, there is a potential clinical role of motor point stimulation for MPS patients.
Future studies may investigate possible mechanisms underlying how motor point electrical stimulation improves pain in MPS involving the trapezius, including the study of this technique in other muscles affected by this syndrome.
It is important to assess the strengths and limitations of this clinical trial. Although we were able to detect statistically significant differences in average pain between groups 1 and 3, our sample size was likely not large enough to confirm a difference between group 2 and the other two groups in that outcome. In addition, we were unable to assess the possibility of the recurrence of pain in the long term, several months after the end of the intervention. Administering several sessions in a large number of patients is technically difficult. Needling was performed entirely by the same professional, with no blinding of the performing physician. To our knowledge, this is the first study to compare the three techniques presented.
In conclusion, our results provide evidence that the treatment of MPS involving the trapezius with dry needling, TrP electroacupuncture or motor point electroacupuncture and/or spinal accessory nerve electrical stimulation is associated with decreased pain scores over time. In terms of the primary pain outcome, motor point electroacupuncture and/or spinal accessory nerve electrical stimulation were superior to dry needling.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
The data sets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
