Abstract
Psychiatric research is undergoing significant advances in an emerging subspeciality of computational psychiatry, building on cognitive neuroscience research by expanding to neurocomputational modeling. Here, we illustrate some research trends in this domain using work on proactive cognitive control deficits in schizophrenia as an example. We provide a selective review of formal modeling approaches to understanding cognitive control deficits in psychopathology, focusing primarily on biologically plausible connectionist-level models as well as mathematical models that generate parameter estimates of putatively dissociable psychological or neural processes. We illustrate some of the advantages of these models in terms of understanding both cognitive control deficits in schizophrenia and the potential roles of effort and motivation. Further, we highlight critical future directions for this work, including a focus on establishing psychometric properties, additional work modeling psychotic symptoms and their interaction with cognitive control, and the need to expand both behavioral and neural modeling to samples that include individuals with different mental health conditions, allowing for the examination of dissociable neural or psychological substrates for seemingly similar cognitive impairments across disorders.
Abnormalities in cognitive function are recognized as a critical aspect of schizophrenia. Cognitive impairments in schizophrenia are present before illness onset (Karcher et al., 2022), persist throughout the life span (Karcher et al., 2022; Sheffield et al., 2018), are associated with poor outcomes (Cowman et al., 2021), and are present across the spectrum of psychotic disorders (e.g., Frattaroli et al., 2022; Karcher et al., 2020; Millman et al., 2022). Research has identified many psychological and molecular targets for enhancing cognitive processing in schizophrenia (e.g., McCutcheon et al., 2023). However, we are still far from having adequate treatments for this crucial component of schizophrenia, highlighting the importance of better understanding the nature and causes of cognitive impairment.
Psychiatric research is undergoing significant advances in a new subspeciality of computational psychiatry, complementing cognitive neuroscience constructs with an expanding paradigm of theory-driven neurocomputational modeling as well as data-driven analyses using various forms of machine learning. Here we focus on neurocomputational modeling, which is based on the premise that we can estimate parameters indexing the computations (i.e., algorithm-based transformations of information) performed by neural circuits and that these parameter estimates bridge the gap between neural circuit function, cognition, and behavior, potentially allowing us to better identify core cognitive deficits (Redish & Gordon, 2016) in disorders such as schizophrenia (e.g., Sheffield et al., 2014).
Here, we will illustrate the potential utility of such computational psychiatry approaches using one hypothesis about a core cognitive deficit in schizophrenia—a deficit in actively representing and maintaining goal information needed to guide behavior (e.g., Barch et al., 2009). We refer to this as a deficit in
Computational Frameworks for Modeling Cognitive Control
We can formulate computational models of control functions at different levels of analysis (e.g., Maia & Frank, 2017). At the biophysical level, these models identify synaptic and microcircuit mechanisms that generate complex cortical oscillations. This work has been extended to include information about how different receptor contributions, at different timescales, can generate persistent and recurrent neuronal firing that supports computations for higher-level cognitive processes, such as working memory or cognitive control (Murray & Anticevic, 2017). In such models, disruptions are introduced to the system at various levels (e.g., changes in parameters thought to reflect the function of specific neurotransmitters). Then, putative behavior or neural function patterns are generated and compared with behavioral data to link the computational framework to human behavior and brain activation. Biophysical models are not a single level of analysis but can capture disruptions at multiple levels of analysis, such as at the level of a specific component of a circuit (e.g., a specific type of inhibitory interneuron, inputs to a circuit, computations in a circuit, or readout mechanisms of a circuit).
At a more abstract level are system-level computational modeling approaches, often in the parallel distributed processing framework. These models have generated and tested hypotheses about the role of particular neurotransmitters and brain systems in cognitive control, including dopamine (DA), norepinephrine, dorsal frontal-parietal systems, and dorsal anterior cingulate (dACC; e.g., Cohen & Servan-Schreiber, 1992). Like the biophysical type of models, hypotheses about impairments driving cognitive dysfunction are tested by introducing perturbations to the model and comparing outcomes with human behavioral and/or neural patterns. At another level of analysis, often bridging between more biophysical and systems levels, are mathematical formalisms of cognition and behavior. These models often focus on reinforcement learning or reward processing but have also been expanded to include mechanisms of behavioral control, motivation, and prediction (e.g., Otto et al., 2015). Such models can generate parameters hypothesized to reflect various processes (e.g., learning rates) to identify the source of behavioral or neural deficits.
Mechanisms of Control
One model of proactive control, termed the dual-mechanisms-of-control theory (DMC; Braver et al., 2021), distinguishes between proactive and reactive modes of cognitive control (see Fig. 1). With proactive control, goal-relevant information is actively maintained in a sustained or anticipatory manner to drive before or to prepare before the occurrence of cognitively demanding events. Goals refer to the information one needs to accomplish a particular task. In contrast, in the reactive mode, control is recruited as a mechanism mobilized only when required, such as when a cue in the environment triggers the activation of a goal or when an event that creates conflict is detected. Thus, proactive control relies on anticipating and planning for future outcomes, including preventing interference before it occurs. In contrast, reactive control relies on detecting cues about the need for specific behaviors and resolving conflict once it is detected.
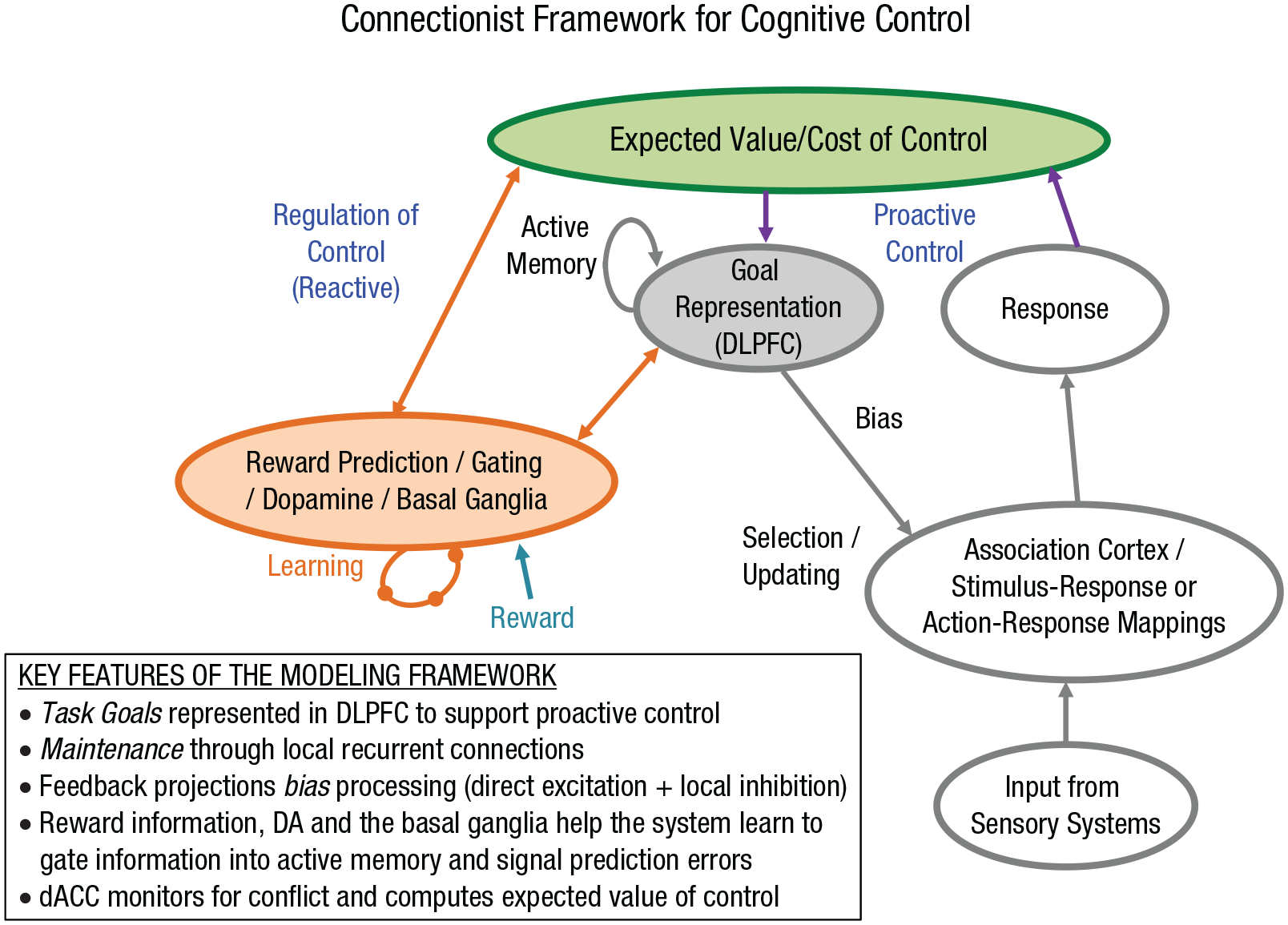
Computational model of cognitive control. Modifications and elaborations to the original model developed by Cohen and Servan-Schreiber (1992; model components in gray) that include mechanisms of both proactive and reactive control. The model includes roles for (a) dopamine (DA) and/or the basal ganglia in gating information into active memory to support proactive control (model components in orange; Braver et al., 2021) and in potentially providing feedback about prediction errors that drive reactive control and (b) dorsal anterior cingulate cortex (dACC) in monitoring for conflict, computing the expected value of control, and dynamically regulating cognitive control (model components in green; Shenhav et al., 2016). DLPFC = dorsolateral prefrontal cortex.
In the social engagement example, proactive control might come into play if you have made a plan to meet a friend for a drink after work and therefore need to take a different route. Proactive control would involve keeping that plan in mind as you leave work so that you make a left turn instead of a right turn at the stop sign where you normally turn right to go home (an environmental trigger indicating conflict between typical behavior and this novel situation). In contrast, reactive control might kick in if you had not been thinking about this when you left work and you turn right at the stop sign, only to have it trigger the need to backtrack to meet your friend. Proactive control might also operate at longer time scales, such as helping to support extended plans to engage in social activities, including calling friends to make plans, organizing transportation, or obtaining money. Reactive control might kick in if you happen to see a friend while you are out and ask them in the moment to go have coffee and now have to figure out transportation or finances. Notably, logistical challenges, such as money or having a way to get places, are types of conflict that can interfere with social engagement among individuals with schizophrenia (Weittenhiller et al., 2021).
Computational Models of Proactive Control
An essential class of models used to investigate cognitive control mechanisms are those coming from the connectionist or parallel distributed processing framework, sometimes referred to as “guided activation theories.” At a simplistic level, in such models, units are summing devices that collect inputs from other units in the model and change their outputs when the inputs change. These models represent information as graded patterns of activity over populations of units where processing occurs as the flow of activity from one set of units to another. Learning occurs by modifying the connection strengths between these units. In some models, this learning is meant to be analogous to the type of Hebbian learning known to drive changes in connection strength between neurons. Connectionist models may help us to understand fundamental principles of neural computation but may only sometimes map onto specific neurobiologically realistic mechanisms. However, learning mechanisms that are putatively more biologically realistic have also been incorporated into such models (e.g., O’Reilly et al., 2019).
Modeling Proactive Control in Schizophrenia
An important set of guided activation models developed by Cohen and Servan-Schreiber (Botvinick & Cohen, 2014) instantiated the hypothesis that proactive control arises from interactions between the DA neurotransmitter system and the prefrontal cortex, inspiring the basis for the DMC model shown in the figure (Braver et al., 2021). These models suggested that goal-related information is actively maintained in the prefrontal cortex and used to bias stimulus-response mappings represented in posterior cortex, serving as a source of top-down support for controlling behavior. The task goal module was explicitly associated with the functions of the dorsolateral prefrontal cortex (DLPFC). Active maintenance in the absence of external inputs was assumed to occur via recurrent excitation. This model assumed that projections from the goal representation module biased processing in other systems via direct excitation. However, competition between representations within a processing stream occurred via lateral inhibition. The effects of DA were assumed to be modulatory, such that changes in DA activity could either increase or decrease activity depending on the nature of the inputs that DA modulated. Increases in DA were assumed to increase the signal-to-noise ratio of a unit’s activation value in relation to its input. The effects of perturbations in DA are simulated as a change in the model’s gain parameter, which relates a unit’s activity to its input. With excitatory input, higher gain means that the same level of input leads to higher activation. With inhibitory input, higher gain leads to more negative values and lower activation.
The same basic model captures the patterns of behavioral data in many different tasks thought to depend on the ability to represent and maintain goal information in DLPFC. One such task is called the AX-CPT (Henderson et al., 2012). In this task, people are shown a series of letters and are asked to hit one button to indicate “target” when the letter
These models have also been used to test the hypothesis that reductions in DA input into DLPFC lead to impairments in proactive control, which in turn leads to deficits in a range of cognitive tasks among individuals with schizophrenia (Cohen & Servan-Schreiber, 1993). In the AX-CPT, a reduction in proactive control leads to failures to respond “target” on
Modeling Value and Effort Allocation in Relation to Cognitive Control
Recent computational models of cognitive control have emphasized mechanisms for computing the expected value of control. The use of cognitive control mechanisms comes at a price of computational effort. Therefore cost-benefit analyses need to be generated to guide when it may be most valuable to increase control exertion (e.g., Shenhav et al., 2016). Disturbances in the mechanisms that compute the expected value of control might lead individuals to either overestimate or underestimate the cost of control and fail to adjust control accordingly. Shenhav and colleagues hypothesized that the dACC may help compute the expected value of control, potentially accounting for why the dACC is central to cognitive control and decision-making. Individuals with schizophrenia show greater “discounting” of rewards as a function of mental effort, meaning they seem to place less value on rewards that require more effort. This discounting is strongly related to experiential negative symptoms (Culbreth et al., 2023).
Further work is needed to determine whether such discounting reflects an objectively greater cost of cognitive control (i.e., cognitive impairment), dysfunction in the systems that compute the expected value of control, or less value placed on rewards in schizophrenia. In the social engagement example, reductions in allocating effort toward social interactions could arise because individuals anticipate experiencing less pleasure in social interactions, because the activities needed to obtain social interactions are objectively more effortful for them (needing to take a bus instead of driving) or because they overestimate how much effort it will take. Recent mathematical modeling frameworks allow for independent estimation of parameters that index the contribution of sensitivity to effort and reward sensitivity to decision-making (Le Heron et al., 2018), providing a means to disentangle the source of such impairments. This is done by independently manipulating how much effort is needed to achieve an outcome across trials (e.g., press a button 50, 100, 150, or 200 times, measuring sensitivity to cost) as well as how much reward can be obtained (e.g., $0.50, $1.00, $1.50, or $2.00, measuring sensitivity to reward). Independently manipulating reward and effort when people make choices between doing a harder task for various amounts of reward versus an easier task for less reward (e.g., 25 button presses for $0.25; Umbricht et al., 2021) allows for separate estimates of reward and cost sensitivity as well as their interaction. Relatedly, we know that schizophrenia, like most forms of psychopathology, is heterogeneous in presentation and potentially in causes. Incorporating modeling of such variability (e.g., severity of specific kinds of symptoms, such as motivational symptoms, or varying levels of genetic risk) into models focused on cognitive function may broaden the utility of these models for understanding key symptom domains of psychopathology.
Model-Based Decision-Making as a Form of Cognitive Control
An additional modeling framework popular in neuroscience and relevant to cognitive control, but applied to somewhat different domains, posits that decision-making arises from two competing systems (e.g., O’Doherty et al., 2017). A “model-free” learning system is thought to learn the value of actions retrospectively by repeating previously rewarded actions. Model-free learning is putatively driven by reward prediction error signaling driven in part by DA signals, which refers to mismatches between an action’s expected and observed outcomes (O’Doherty et al., 2017). Learning in this system is computationally straightforward but relatively inflexible to rapid environmental or outcome value changes. Using our social engagement example, this system could help you learn that a friend you see in the lunchroom at work every day frequently smiles back at you when you smile at them, leading you to smile at them each time you see them. However, this system may lead you to ignore the fact that today, that person is engaged in an intense conversation with another person, creating disappointment when they do not respond to your welcoming smile. A second, “model-based” learning system is thought to implement more goal-directed behavior. It learns the value of actions prospectively by leveraging a cognitive “map” of actions and their future consequences, implementing a form of proactive control over decision-making. This system may allow you to integrate factors such as the current state of the other person (talking to someone else, upset expression on their face, etc.) when deciding whether to try to smile at the person or whether their failure to smile back should “update” your general expectation that they are a person who typically smiles back at you. Because such mental maps are constructed “on the fly,” model-based learning allows for rapid updating of action values. However, simulating future states requires high levels of representational space and maintenance, making model-based learning computationally expensive (O’Doherty et al., 2017).
Work has shown reduced reliance on model-based learning in schizophrenia (Culbreth et al., 2016), consistent with other evidence of impairments in cognitive control, though other work has suggested impairments in model-free learning in psychosis (Brandl et al., 2023). Further, several other disorders, including binge eating, drug use, and obsessive-compulsive disorder, also show a bias toward model-free learning (e.g., Voon et al., 2017). This may reflect the ubiquitous role of cognitive control deficits in psychopathology. However, future work needs to rule out a differential difficulty explanation (i.e., do individuals tend to avoid using model-based learning less because it is more difficult), as has been done for other paradigms assessing proactive control. One way to do this may be to examine the degree to which people can increase cognitive control when offered greater incentives to do so (Patzelt et al., 2019). If a failure to use model-based learning reflects the greater difficulty of doing so, enhancing its benefits may reveal an intact ability to deploy such learning when the benefits overcome the costs. Such an approach might also reveal dissociations across forms of psychopathology that all seem to involve a bias toward model-free learning, with some showing an increase in the use of model-based learning when incentivized to do so and others not able to do so.
Frank and colleagues (Gold et al., 2013) have been using a variant of this framework to understand value-based decision-making and its impairments in schizophrenia. This framework postulates that there are two systems involved in value representation and reinforcement learning. One is a system involving the orbitofrontal cortex (OFC) and, some argue, also the DLPFC, thought to map expected values and update representations in response to feedback. This system aligns with model-based learning. The other is a system involving the basal ganglia, thought to support prediction-error (PE) computations that are more analogous to model-free learning. The argument is that most people use a combination of these two systems to learn to achieve rewarding outcomes, with greater reliance on “explicit” value representations supported by OFC-DLPFC early in learning and more involvement of “PE-based” representations later in learning. In multiple studies, individuals with schizophrenia show reduced reliance on explicit valuation representations via the OFC-DLPFC system compared with healthy controls (e.g., Geana et al., 2022).
Summary and Future Directions
Here, we have highlighted computational formalisms that help us to understand how proactive cognitive control deficits in schizophrenia may arise out of biologically plausible mechanisms that support a range of neural and psychological processes. There are several key next steps in this line of work. First, we need to understand more about the psychometric properties of computationally derived parameters (e.g. reliability over time, floor/ceiling effects) if they are to be used to understand the source of cognitive dysfunction at the individual level or in treatment studies. It is likely that the psychometrics of computationally derived parameters are constrained by the psychometrics of the basic behavior on which the models are based. For example, in recent work, we used mathematical models to generate computational estimates of capacity on a working memory task that asked people to detect changes in the colors of varying numbers of dots. The test-retest reliability (interclass correlation coefficient [ICC]) of accuracy across conditions that varied one, two, or five dot colors ranged from a high of .8 to a low of .4, with a value of .75 for the capacity parameter estimate and a much lower value (.22) for a parameter estimating attentional lapses. It would be interesting to determine whether using only the task conditions with higher ICCs for accuracy would improve the reliability of the parameter estimates.
Second, this selective review focused more on computational analyses of behavioral performance informed by neurally inspired models; however, computational analysis of a variety of neural measures themselves is also essential. As such, we need more behavioral and neuroimaging studies with computationally informed paradigms that include many forms of psychopathology so that we can begin to understand whether cognitive control deficits across disorders (e.g., McTeague et al., 2017) are the results of a shared set of computational, psychological, and neural mechanisms that cut across mental health conditions or are the equifinal outcome of diverging distal pathways to impairment. For example, both individuals with schizophrenia and individuals with depression can experience difficulties with engaging in social activities. However, people with schizophrenia may show increased “cost” of effort to engage in social activities, whereas people with depression show reduced sensitivity to rewards associated with social interactions. If so, the seemingly similar behaviors might reflect different psychological mechanisms that could be revealed by computational analyses. However, it is also possible that both might show a reduction in reward sensitivity, but neuroimaging analyses could reveal that this is associated with disruptions in the striatum in schizophrenia and the ventral medial prefrontal cortex in depression, suggesting that different brain regions or systems might be contributing to seemingly similar behavioral challenges.
Third, we need to move toward incorporating modeling approaches at differing levels of analysis to benefit from their combined strengths (e.g., merging guided activation and reinforcement learning) as well as integrating more biophysical components to these models to help delineate more detailed aspects of neurobiology and their role in cognitive control. Fourth, recent work is demonstrating the power of computational approaches in addressing long-standing questions about the nature of psychosis itself (namely, delusions and hallucinations; e.g., Sterzer et al., 2018). These primarily Bayesian-inspired models reveal the role of placing too much weight on previous information (i.e., “overweighting of priors”), belief updating, and sensory integration in psychotic symptoms, which may interact with cognitive control alterations in the manifestation of schizophrenia.
It is never wise to overhype a new approach as a panacea for addressing challenging scientific questions, such as determining the core cognitive deficits in schizophrenia and their causes. Nonetheless, the computational psychiatry approach outlined here holds promise for contributing novel and vital insights into this challenging puzzle, hopefully moving us toward effective treatment and prevention of cognitive deficits in schizophrenia. Using the social engagement example again, a computational analysis might help determine whether a particular individual was not engaging in social activities because of the effort associated with the process or because of reduced sensitivity to future rewards. The treatment plan could then be tailored to the particular mechanism driving the reduction in social activities (e.g., ways to overcome costs, how to more realistically estimate costs vs. recall of previous positive social interactions, savoring interventions).
Recommended Reading
Corlett, P. R., & Fletcher, P. (2021). Modeling delusions as temporally-evolving beliefs.
Geana, A., Barch, D. M., Gold, J. M., Carter, C. S., MacDonald, A. W., 3rd, Ragland, J. D., Silverstein, S. M., & Frank, M. J. (2022). (See References). Example of computational parameters revealing dissociations among forms of psychopathology that were not seen in a more standard analysis approach.
Gillan, C. M. (2021). Recent developments in the habit hypothesis of OCD and compulsive disorders.
Huys, Q. J. M., & Browning, M. (2022). A computational view on the nature of reward and value in anhedonia.
Yip, S. W., Barch, D. M., Chase, H. W., Flagel, S., Huys, Q. J. M., Konova, A. B., Montague, R., & Paulus, M. (2023). From computation to clinic.
Footnotes
Acknowledgements
Parts of this manuscript were revised and updated with permission from a previous article: D. M. Barch, A. Culbreth, and J. M. Sheffield, “Systems-Level Modeling of Cognitive Control in Psychiatric Disorders: A Focus on Schizophrenia,” pages 145–173 in
