Abstract
Culture—the totality of traditions acquired in a community by social learning from other individuals—has increasingly been found to be pervasive not only in humans’ but in many other animals’ lives. Compared with learning on one’s own initiative, learning from others can be very much safer and more efficient, as the wisdom already accumulated by other individuals is assimilated. This article offers an overview of often surprising recent discoveries charting the reach of culture across an ever-expanding diversity of species, as well as an extensive variety of behavioral domains, and throughout an animal’s life. The psychological reach of culture is reflected in the knowledge and skills an animal thus acquires, via an array of different social learning processes. Social learning is often further guided by a suite of adaptive psychological biases, such as conformity and learning from optimal models. In humans, cumulative cultural change over generations has generated the complex cultural phenomena observed today. Animal cultures have been thought to lack this cumulative power, but recent findings suggest that elementary versions of cumulative culture may be important in animals’ lives.
Culture pervades human lives, from technologies and languages to social customs and religions. Cumulative cultural advancements have made possible nearly 8 billion people colonizing all nonpolar land masses, while humans’ closest ape cousins number a mere few hundred thousand, restricted to dwindling habitats in Africa.
For a long time, this extraordinary capacity for, and reliance on, culture has been assumed to be unique to humans. Research in recent decades has revealed a very different picture, however. Culture, defined as all group-specific behavioral patterns that are acquired by learning from other individuals (Laland & Janik, 2006), has been found to be surprisingly widespread in the animal world, profoundly shaping many animals’ behavior patterns and psychology. This brief article can provide only a slim overview of the principal discoveries, but I cite a variety of more in-depth reviews for readers keen to further explore.
Preliminaries: Core Terminologies and Methodologies
I first offer definitions of some key terms, but I preface this by suggesting that no definition can be objectively “correct.” Good definitions instead simply express commonly accepted meanings that facilitate communication across a community.
Defining
Accordingly it is too simplistic to ask, “Does species X have culture?” I have suggested that, instead, it is instructive to decompose culture into aspects such as (a) the ways in which traditions are distributed in space and time, (b) the underlying learning processes, and (c) the behavioral or other specific contents of the traditions (e.g., Whiten, 2017b). The array of forms these aspects of culture take can then be more informatively compared across species.
Once a researcher provides an explicit definition of the phenomenon at stake, the “real science” can begin: empirically and objectively testing the phenomenon’s occurrence in a species. A powerful array of methods has been developed to do so. These methods include systematic observations in the wild, controlled experiments in lab and field, and sophisticated modeling and statistical techniques (Hoppitt & Laland, 2013). Much progress has been driven by the ingenuity of researchers in developing these complementary methodological approaches. For example, the basic experimental paradigm for testing social learning by comparing participants who witness a model do act X with participants who lack that opportunity has been extended to
The Discovery of Animal Cultures
The first inklings of cultural phenomena in nonhuman animals (henceforth, “animals”) emerged in three differing discoveries around the middle of the 20th century (Whiten, 2017a). In one, scientists provisioning Japanese macaques with sweet potatoes on an island beach reported that after a juvenile, Imo, started washing sand off the food in a stream, the behavior gradually spread across her affiliated peers and family members to become a tradition in her group. Later, potato washing was extended to the sea: The macaques repeatedly dipped the potatoes, which salted them. A second influential discovery was that songbirds displayed different song dialects in different geographic locations. Coupled with laboratory experiments demonstrating that many birds learned their songs by listening to the songs of others, this discovery revealed the first vocal cultures in animals. Third, small birds in England started tearing the tops off milk bottles to drink the cream beneath, and the diffusion of this novelty was tracked as it spread across the United Kingdom from just a few initial spots.
As the century progressed, studies of great apes (initially, chimpanzees in Africa and later, orangutans in Asia) added a new dimension to a growing body of evidence about animal cultures. Whereas studies like those outlined in the previous paragraph revealed unitary traditions, such as birdsong dialects, studies of apes identified regional cultures differing in multiple local traditions regarding tool use, grooming styles, and courtship gambits. Different human cultures around the world are distinguished by arrays of different traditions, including traditions of language, dress, and cuisine. A similar principle thus emerged on a more modest scale in the multitradition cultures of apes (Whiten, 2017b).
Building on these pioneering studies, research in this century has generated an accelerating accumulation of often-surprising discoveries about the range and diversity of culturally inherited behavior in animals (Lachlan & Whiten, 2020; Whiten, 2017a). In the following sections, I summarize a series of discoveries demonstrating the reach of culture in animals’ lives.
The Reach of Culture Across Species and Behavioral Domains
Cultural traditions have been identified across a rapidly growing diversity of species and in many different forms of behavior. Cetaceans (whales and dolphins) have provided many new examples. Their cultural diversity spans such domains as song, migration routes, and foraging specializations (Whitehead & Rendell, 2015). Regionally varying vocal cultures in whales parallel the dialects of songbirds, but in some species, such as humpback whales, greater song complexities are transmitted, and radically new songs are intermittently innovated, subsequently spreading across vast ocean basins (Garland et al., 2011). Many more examples of avian culture have accumulated, too. These include the adoption of particular migration routes, nesting sites, diets, and foraging techniques (Aplin, 2019). Traditions that have been identified in fish include traditions of mate preferences and daily routes over coral reefs (Laland et al., 2011).
The extending reach of cultural transmission is graphically illustrated by recent studies of insects. In one, a bumblebee was trained to scrabble at a string to pull an artificial nectar-laden flower within reach. Fellow bees from the hive then observed this and began to adopt the technique, which subsequently spread to many other hive members. This did not occur in control hives lacking an initial model (Alem et al., 2016). Evidence for cultural transmission has even emerged for the humble fruit fly. Virgin females who watched other females’ preference for mating with one of two differently colored males subsequently showed a mating preference for that color (
In sum, cultural phenomena have now been demonstrated in all major groups of vertebrate animals in the wild, as well as in lab studies such as those on insects, and they span numerous important facets of animals’ behavior (Whiten, 2021).
The Psychological Reach of Culture Across Lifetimes
Much of the transmission of cultural knowledge occurs in the earliest stages of life. For example, some whales migrate annually between breeding and feeding grounds halfway around the world, and the young first travel with their mother, learning from her a route they often follow throughout their lives (Whitehead & Rendell, 2015). Many young mammals and birds learn from existing parental expertise during these intimate early relationships. What is learned socially may involve such varied activities as following particular migratory routes, foraging, avoiding predators, using tools, communicating vocally, and engaging in social customs such as styles of grooming (Aplin, 2019; Whiten, 2017a, 2019, 2021). Tapping this existing expertise offers a high-value shortcut to adaptive behavior, as in the advantages of learning from other individuals about how to evade predators, rather than relying on trial and (likely fatal) error.
Social learning is not limited to infancy. Three phases in lifetime social learning have been distinguished in primates (Whiten & van de Waal, 2008; see Fig. 1). Phase 1 is typically focused on the mother, as described in the previous paragraph. In the second phase, youngsters expand their social network and are increasingly exposed to other, and sometimes improved, models to learn from. For example, young capuchin monkeys begin to attend to the best nut-cracking models in their group, typically not their mother (Ottoni et al., 2005). In strongly dimorphic species, such as gorillas, it may advantage a young male to apprentice himself to an adult male to learn about diets he would not learn about from his mother, a pattern seen in species of both monkeys and apes. Later, sexually maturing males or females of most primate species will disperse to breed further afield, avoiding inbreeding. This gives rise to the third phase: social learning from knowledgeable residents about new local foraging opportunities and predation risks, as well as whom to defer to in social interactions.

Three phases in social learning during primates’ lifetimes (after Whiten & van de Waal, 2018). In Phase 1, social learning is focused on the mother. This phase is illustrated here by a scene from a field experiment by van de Waal et al. (2013). Infant vervet monkeys provisioned with pink and blue corn learned a preference for the color their mothers had been trained to prefer (in this example, the pink corn). In Phase 2, the youngster’s social world and focus of learning begin to expand, as illustrated by a scene from work by Ottoni et al. (2005). In this example, a young monkey watches one of the group’s best nut crackers in action. Phase 3 occurs when sexually maturing males or females disperse to breed further afield. In this phase, the immigrants learn from knowledgeable residents. In van de Waal et al.’s field experiment, immigrant males switched their color preference for corn to the color preferred by their new group.
In field experiments, van de Waal et al. (2013) trained groups of wild vervet monkeys to prefer either pink or blue corn provisions by making one distasteful, and later, new naive infants’ choices were observed when no distasteful additive was present. All 27 of the 27 infants studied followed their mothers’ preference (Phase 1). When sexually maturing males dispersed to new groups with a different color preference, they readily abandoned the preference of their natal group and switched to the alternative favored in the new group (“When in Rome, do as the Romans do”; Phase 3).
The three phases illustrated in Figure 1 appear to characterize most primate species, but likely not all. It is a task for future research to discover the role social learning plays through the lifetimes of the many different kinds of cultural animals. Having provided evidence that juveniles’ closely focused peering at what other individuals do offers a proxy for social learning (because it commonly predicts what the youngsters try to do shortly afterward), Schuppli and van Schaik (2019) documented two orangutan populations peering in as many as 195 and 125 different contexts, respectively. The authors suggested that prior studies were overly conservative about the scope of animal species’ culture, concluding that immature great apes seem to “learn virtually all of their skills socially” (p. 5).
Although much of this crucial social learning often occurs early in life, researchers now know that it has a role to play at many points in animals’ life histories. If Schuppli and van Schaik (2019) are right, much remains to be learned about this.
Diversity in Social Learning Processes and Their Neural Substrates
Primates’ brains are larger than the average mammals’, and primates get high scores on cognitive tests. The coevolution of these characteristics was originally assumed to be driven by adaptive advantages in foraging and other dealings with the physical world. Later in the 20th century, evidence mounted in support of an alternative social, or
The cultural-intelligence hypothesis posits a spiral evolutionary process. On the one hand, wherever it becomes of greater adaptive advantage to rely heavily on cultural information (e.g., Schuppli & van Schaik, 2019), there will be selection pressure favoring more sophisticated social learning processes, as well as the storage of greater masses of culturally acquired information and hence brain expansion. In complementary fashion, such neural and psychological enhancements may facilitate yet more extended cultural acquisitions, completing a positive-feedback loop. Such spiraling cultural and neural complexity appears to have been a runaway process for much of recent human evolutionary history.
But not all animals that learn socially are as large brained as the apes: Bees and fruit flies are clearly not, so they present an apparent conundrum for the cultural-intelligence hypothesis. The solution to this conundrum may lie in the diversity of social learning processes distinguished in this research field (Fig. 2), another indicator of the reach of culture in animals’ psychological makeup. Some processes in the taxonomy illustrated in Figure 2 appear to be relatively cognitively complex and are perhaps restricted to larger-brained species, such as apes and cetaceans. These processes include imitation and emulation. Copying of what another individual does, by either imitation or reenactment of resultant tool or other object movements, may involve matches in an action’s shape, in its sequential or hierarchical structure, and in inferred causal or intentional links between its components. For example, Boesch et al. (2020) reported that communities of wild chimpanzees in central Africa that use tools to mine subterranean termites do so using regionally distinct sequences of bodily techniques, which apparently require bodily imitation for their transmission, a capacity consistent with results obtained in some lab experiments (Whiten, 2017b).
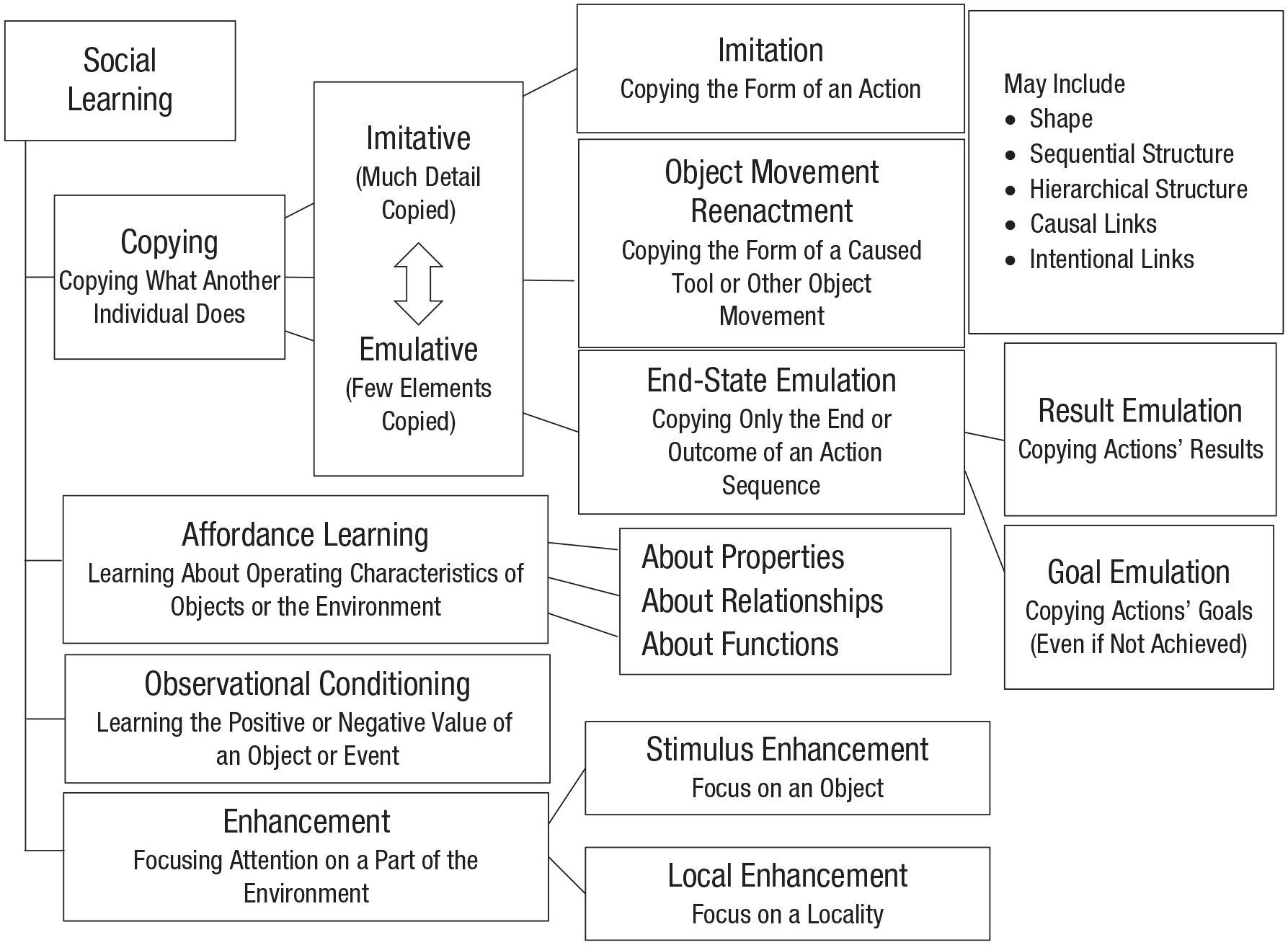
A taxonomy of forms of social learning (after Whiten et al., 2004), from the relatively complex (at the top) to the relatively simple (at the bottom).
In the alternative and sometimes “cleverer” (in the sense of more creative) process of emulation, only the endpoint of a witnessed act is copied, and it is achieved by novel means. Emulation may involve copying the observed result of another individual’s actions or copying the assumed goal, if the individual is perceived to have pursued a goal but not achieved it. For example, Tennie et al. (2010) showed captive chimpanzees how pouring a bottle of water into a flask made a peanut rise up and become graspable. When tested with no bottle available, some individuals took water from their drinker and spat it into the flask instead, thus creating the observed result but by different means. Whether imitation is significant in apes’ lives or they rely instead on emulation continues as a fierce controversy in the field (Henrich & Tennie, 2017; Tennie et al., 2009; Whiten, 2017b).
At the simpler end of the processes distinguished in Figure 2 is enhancement, in which the activity of an individual merely draws a learner’s attention to a particular item, location, or route. In this case, only basic associative learning seems to be required, as when a youngster learns from other individuals what to eat and where to find it, or follows a parent along a traditional migration route. Enhancement is thus probably the most widespread of all these kinds of social learning in the animal kingdom, yet functionally highly important.
Social learning may also be facilitated by a form of
Adaptive Canalization of Social Learning
The social learning processes outlined in Figure 2 are further modulated by what are sometimes called biases, or social learning strategies, which are generally thought to exist because they canalize, or funnel, learning in adaptive ways (Kendal et al., 2018). One example of such a bias is conformity, preferring to adopt whichever of the available options is displayed by a majority of the perceivable population. Evidence for this bias has emerged in species from birds (Aplin et al., 2015) to fruit flies (Danchin et al., 2018). A primate example (van de Waal et al., 2013) is shown as an illustration of Phase 3 in Figure 1. Phase 2 in that figure illustrates another useful bias, learning from the most successful or skilled individuals (Ottoni et al., 2005). Such biases may be combined in complex ways. Bono et al. (2018) found that to predict a vervet monkey’s social learning of one foraging action over an alternative, one needs to know three different things: the sex of the learner, the sex of the individual watched, and the relative profitability of the action option witnessed.
The reach of culture has thus been shown by these recent studies to include not only the evolution of diverse social learning processes distinguished in Figure 2, but also their refinement through a further range of modulating biases. Kendal et al. (2018) described no fewer than 25 different varieties of these typically adaptive biases.
Does the Reach of Animal Culture Extend to Cumulative Buildup Across Generations?
It is now widely accepted that many animals display socially learned traditions, but one core aspect of human culture has been argued to remain unique: cumulative buildup across generations. Think of the evolution of architecture over the centuries, or of digital technology in more recent times. Animal cultures may certainly be sustained across generations, as in the case of chimpanzees’ nut cracking using natural rock hammers, traces of which were found at depths corresponding to 4,300 years ago, in archaeological digs in Africa (Mercader et al., 2007). However, there has been no indication of change, progressive or otherwise, since then.
Intriguingly, recent years have seen the first empirical signs that some measure of cumulative culture can occur in animals, and may even be quite common, although, of course, in not nearly as elaborate a form as in humans (Whiten, 2019). An unexpected experimental demonstration of cumulative cultural progress came in a study of homing pigeons. Sasaki and Biro (2017) allowed two pigeons to home together repeatedly and recorded their tracks, then replaced one with a naive bird and repeated the whole process. Doing this several times revealed faster and more direct homing tracks as the experiment progressed, so there must have been social transmission of the stepwise improvement in homing skill across these “cultural generations.” Further evidence, this time from the wild, came from another unexpected source: bighorn sheep that had been translocated to areas where populations of their species had earlier been cleared. Over two centuries and multiple generations, the sheep began to migrate seasonally to better pastures, and became progressively skilled at finding good grazing areas (Jesmer et al., 2018). This echoed the cross-generation progress seen in the pigeon experiment. Such cycles of social transmission and individual exploration may be more common in nature than researchers have realized, creating cumulative evolutionary effects—one more indicator of the extending reach of culture in animals’ lives. More testing for such patterns in other species and other domains of behavior is now clearly warranted.
Recommended Reading
Allen, J. A. (2019). (See References). A review spanning the animal kingdom, written by a cetacean researcher and with a particular focus on bidirectional linkages between community structures and culture.
Kendal, R. L., Boogert, N. J., Rendell, L., Laland, K. N., Webster, M., & Jones, P. L. (2018). (See References). The most recent comprehensive review of (mostly adaptive) biases in social learning in humans and other animals.
Lachlan, R. F., & Whiten, A. (2020). (See References). An annotated bibliography of more than 90 core publications on animal social learning, culture, and cultural evolution, as well as foundational publications focused on human cultural evolution.
Schuppli, K., & van Schaik, C. P. (2019). (See References). Supported by extensive field studies, a radical argument that animal culture is much underestimated, together with proposals for how this might be remedied by future objective empirical research.
Whiten, A. (2017b). (See References). The most recent comprehensive survey of the observational and experimental evidence for social learning and culture in humans’ closest living primate relatives, along with comparisons with the cultural worlds of human children.
Footnotes
Acknowledgements
I thank Erica van de Waal for the photographs illustrating Phases 1 and 3 in Figure 1 and Tiago Falotico for the photograph illustrating Phase 2 in Figure 1.
Transparency
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
