Abstract
Facial nerve transection frequently results in incomplete functional recovery despite microsurgical repair due to axonal misalignment and limited intrinsic regenerative capacity. Schwann cells (SCs) are essential for peripheral nerve repair, supporting axon elongation, remyelination, and neurotrophic signaling. Human embryonic stem cell–derived Schwann cells (hESC-SCs), including Schwann cell precursors (SCPs) and immature SCs, constitute a scalable source of cells for tissue-engineered nerve grafts. This study investigated whether silicone conduits loaded with hESC-SCs could enhance facial nerve regeneration following complete transection in rats. hESCs were differentiated into SCPs or immature SCs using a stepwise in vitro protocol and purified by fluorescence-activated cell sorting (FACS). Forty-eight Sprague-Dawley rats underwent right facial nerve transection with a 3-mm gap and were randomly assigned to six groups: sham, axotomy only, empty conduit, Matrigel-filled conduit, conduit with SCPs, or conduit with immature SCs. At 12 weeks post-surgery, toluidine blue histochemistry, transmission electron microscopy (TEM), and morphometric evaluation demonstrated significantly larger axonal diameters, thicker myelin sheaths, and improved g-ratios in SC-treated conduits compared with empty conduit and axotomy-only controls (P < 0.05). Behavioral assessments showed partial functional recovery in SC-treated groups, with blink reflex performance significantly superior to controls at 8 and 12 weeks (P < 0.05). No significant differences were found between SCP and immature SC groups. These findings suggest that hESC-SCs incorporated into bioengineered conduits represent a potentially effective cell-based strategy for peripheral nerve regeneration. However, the interpretation of mechanistic effects is limited by the absence of direct engraftment validation and molecular analyses.

Keywords
Introduction
Peripheral facial palsy can result from various etiologies, including congenital anomalies, infectious and inflammatory diseases, trauma, and neoplasms involving the temporal bone and parotid gland1–3. Traumatic causes, such as temporal bone fractures or surgical resection of benign and malignant tumors (e.g. vestibular schwannoma, cholesteatoma), may directly transect the facial nerve, leading to severe functional impairments including facial asymmetry, incomplete eyelid closure, oral incompetence, and psychosocial burden2,3. Although peripheral nerves have intrinsic regenerative potential, recovery after severe facial nerve transection is often incomplete due to misdirected axonal growth, scar formation, and chronic denervation of target musculature4–6.
When addressing gaps in the facial nerve, surgeons typically turn to direct neurorrhaphy or autograft interposition as their preferred methods7–9. These methods can restore anatomical continuity but are limited by donor-site morbidity, limited graft availability, mismatched fascicular structures, and unpredictable functional outcomes7–9. Even after successful coaptation, axonal misdirection and partial target reinnervation often lead to synkinesis and suboptimal recovery of coordinated facial movements. Therefore, more effective regenerative strategies are needed to improve long-term functional outcomes after facial nerve injury.
ESCs have emerged as promising candidates for nerve repair due to their unlimited ability to divide and their capacity to be directed toward specific cell types 10 . Schwann cells (SCs) serve indispensable functions during nerve restoration, including elaboration of growth-promoting molecules, physical guidance of sprouting axons through injury sites, and myelin sheath reconstitution around regenerated fibers11–16. Studies have shown that neural precursors derived from ESCs can enhance axon regeneration and improve motor recovery in animal models of nerve injury 13 . Contemporary refinements in directed differentiation methodologies now permit derivation of anatomically specified neural precursor subpopulations with potential for improved therapeutic outcomes 17 . Exogenously delivered SCs can guide axonal regrowth across nerve gaps and create a microenvironment enriched with neurotrophic and neurotropic factors that further promote nerve regeneration14–16.
Human embryonic stem cells (hESCs) represent a renewable source that can be consistently differentiated into large numbers of SCs with stable phenotypic and functional properties10,18,19. While ESC-derived SCs have shown beneficial effects in animal models of peripheral nerve injury, little is known about their efficacy in cranial peripheral nerve injuries, such as facial nerve transection. Combining hESC-derived SCs (hESC-SCs) with bioengineered silicone conduits may provide an “off-the-shelf” translationally viable option compared to autologous nerve grafts for treating complex facial nerve defects. We investigated whether hESC-SCs delivered through silicone conduit implants could enhance nerve repair following facial nerve transection in rats. Our hypothesis was that hESC-SC supplementation would lead to improved axonal regrowth, myelination, and motor recovery compared to control groups, offering essential groundwork for cell-based facial nerve repair strategies.
Materials and methods
Ethics statements
The WA09 (H9) hESC line was obtained from WiCell (Madison, WI, USA) and registered with the Korean National Stem Cell Registry (registration number: hESC22020002). Institutional review board approval was secured for all hESC-related procedures (protocol: KHSIRB-22-501), and appropriate notifications were submitted to the Korean Ministry of Health and Welfare. Animal studies followed the guidelines of the Institute of Medical Sciences at our affiliated hospital (protocol: KHMC-IACUC 2022-044).
Experimental design and animal groups
Forty-eight adult male Sprague-Dawley rats weighing 220–250 g were used in this study. After 1 week of acclimatization, rats were randomly assigned to six groups (n = 8 each): group A (sham surgery with skin incision only); group B (nerve transection with 3-mm gap, no conduit); group C (empty silicone conduit); group D (silicone conduit with Matrigel); group E (conduit with 5 × 104 Schwann cell precursor [SCPs]); and group F (conduit with 5 × 104 immature SCs). Surgery was performed at week 0; functional assessments were conducted at weeks 1, 4, 8, and 12; and tissues were harvested for histopathological analysis at week 12.
Differentiation of hESCs into SCs
Directed differentiation of hESCs toward the SC lineage followed an adapted multi-step protocol (Fig. 1)20,21. Stem cell colonies were enzymatically dissociated to single-cell suspensions and seeded onto Matrigel-coated culture surfaces. Neural induction commenced on day 0 using knockout serum replacement (KSR) basal medium containing the small molecules SB-431542 (10 µM) and LDN-193189 (500 nM). On day 2, the culture formulation was modified to include CHIR99021 (3 µM) and N-[N-(3,5 difluorophenacetyl)-L-alanyl]-S-phenylglycine t-butyl ester (DAPT) (10 µM) alongside the initial inhibitors. Progressive medium transitions occurred on days 5, 6, and 8, shifting the KSR-to-Neurobasal (NB) ratio from 3:1 to 1:1 to 1:3 while maintaining identical small-molecule supplementation. From day 10 onward, pure NB medium enriched with dibutyryl cyclic adenosine monophosphate (AMP) (200 µM) and sodium L-ascorbate (200 µM) supported terminal SC maturation.

Schematic illustration of the experimental design, differentiation timeline, and study groups. hESCs were differentiated into Schwann cells (SCs) through a stepwise protocol (D0–D40) involving neural crest induction, SCP formation, and maturation into immature SCs. Rats were assigned to six groups (n = 8 per group): sham control, axotomy only, empty conduit, Matrigel conduit, conduit with SCPs, and conduit with immature SCs. hESCs: human embryonic stem cells; FACS: fluorescence-activated cell sorting; SCPs: Schwann cell precursors; SCs: Schwann cells. This figure was created by the authors using Adobe Illustrator.
Following 20 days of differentiation, SCPs were isolated through fluorescence-activated cell sorting (FACS) employing a phycoerythrin (PE)-labeled anti-α4-integrin (CD49d) antibody (R&D Systems, Minneapolis, MN, USA; catalog: FAB1354P). Sorted cells remained in NB maintenance medium until day 40, at which point immature and mature SC populations were separated via additional FACS sorting. For transplantation purposes, either SCPs or immature SCs (5 × 104 cells suspended in 5 µL of Matrigel) were deposited into sterile silicone tubes measuring 5 mm in length with 1 mm internal and 2 mm external diameters (Fig. 1).
Characterization of hESC-derived SCs
Sequential differentiation of hESCs (H9 line) toward SCPs and immature SCs employed the stepwise induction methodology detailed earlier (Fig. 2a). Lineage-appropriate differentiation was validated through total RNA extraction at day 20 (SCP stage) and day 40 (immature SC stage), followed by RT-PCR profiling of SC-associated transcripts including SOX10, CHD19, p75, OCT6, MPZ, and Periostin (Figs. 2b–e). Protein-level validation employed immunocytochemical detection of S100β and MPZ at both differentiation timepoints (Fig. 2f, g). Day-20 SCPs demonstrated expression of SOX10, CHD19, and p75, while day-40 immature SCs exhibited OCT6, MPZ, and Periostin positivity, confirming sequential maturation toward an SC phenotype. To further assess relative SC maturation, mean fluorescence intensity of MPZ and S100β was quantified from immunocytochemical images using ImageJ software. In addition, PMP22 isoform expression was analyzed by RT-PCR, and the ratio of variant 1 to variant 2 was used as an indicator of maturation-associated isoform switching (Fig. 2h, i).

Characterization of hESC-derived Schwann cell precursors (SCPs) and immature Schwann cells. (a) Morphology of undifferentiated hESCs and neural crest cells at day 10 of differentiation. Scale bar = 100 μm. (b) Representative FACS plots showing CD49d+ Schwann cell precursors (day 20) and immature Schwann cells (day 40); undifferentiated hESCs served as negative control. (c) Quantitative PCR analysis of CD49d mRNA expression in FACS-sorted CD49d+ and CD49d⁻ populations at days 20 and 40 (n = 6; Student’s t-test; ****P < 0.0001). (d, e) Gene expression of Schwann cell lineage markers in CD49d+ cells. Schwann cell precursor markers (SOX10, CHD19, p75) were enriched at day 20, while differentiation markers (OCT6, MPZ, Periostin) were upregulated at day 40 (n = 9; one-way ANOVA; ****P < 0.0001). (f, g) Immunostaining of CD49d+ cells showing MPZ (red) and S100β (green) expression. Nuclear localization of S100β at day 20 indicated the precursor stage, whereas cytoplasmic localization at day 40 reflected Schwann cell differentiation. (h) Quantification of mean fluorescence intensity for MPZ and S100β in CD49d+ cells at day 20 and day 40, demonstrating significantly higher expression at day 40, consistent with Schwann cell maturation. (i) PMP22 isoform analysis showing an increased variant 1/variant 2 ratio in day 40 cells compared with day 20 cells, indicating maturation-associated isoform switching. Scale bar = 100 μm.
Surgical procedure for nerve axotomy and transplantation model (week 0)
At week 0, all rats underwent right facial nerve transection surgery. Rats were anesthetized using 5% isoflurane (Forane solution; Joongeuk Pharmaceutical, Hwaseong, Korea) in 80% O2 using the VetEquip system, with anesthesia maintained with 3% isoflurane. Under anesthesia, the skin was incised posterior to the right external auditory canal, and dissection proceeded anteromedially to identify the clavotrapezius muscle tendon, thereby exposing the main trunk of the facial nerve. A transection injury was induced in the proximal part of the main trunk of the facial nerve in all rats. A 3-mm defect was created in the trunk of the right facial nerve, and each proximal and distal nerve end was sutured (epineural sutures with 4-point mattress) with a 5-mm silicone nerve conduit using 9-0 nylon (Fig. 3).

Surgical procedure for facial nerve transection and conduit implantation. (a) Exposure of the main trunk of the right facial nerve (yellow arrow) after blunt dissection of overlying tissues. (b) Placement of a 5-mm silicone nerve conduit bridging the transected nerve gap. (c) Final positioning of the conduit secured in place before wound closure.
For control animals, a silicone conduit (5 mm in length) containing only Matrigel was interposed at the facial nerve trunk. Matrigel (5 μL) was loaded into each tube via micropipette prior to implantation. A total of 48 rats were randomly assigned to one of six experimental groups (n = 8 per group). Manipulations included skin incision alone (group A); axotomy with a 3-mm nerve gap (group B); introduction of an empty silicone nerve conduit (group C); introduction of a Matrigel silicone nerve conduit (group D); introduction of a Matrigel silicone nerve conduit with SCPs (group E); and introduction of a Matrigel silicone nerve conduit with immature SCs (group F).
Behavioral tests (Weeks 1, 4, 8, and 12)
Functional recovery was monitored through behavioral examination at postoperative weeks 1, 4, 8, and 12 22 . Injury severity and recovery extent were quantified via whisker movement observation using an established grading scale, complemented by corneal reflex testing using a modified scoring system for eyelid closure and blink response. Animals were positioned in a restraining apparatus for standardized whisker and blink reflex evaluation; blink testing involved air puff stimulation delivered from a syringe at 5 cm distance (Supplementary Figure S1). Both whisker mobility and blink response were graded using ordinal five-point scales, with scores representing consensus determinations by two independent observers reviewing video recordings. Maximum whisker recovery (score 5) corresponded to symmetric anterior whisker positioning with movement matching the uninjured contralateral side. Lower grades were assigned as follows: score 4, normal whisker mobility with posterior resting position; score 3, prominent whisker tremor with posterior positioning; score 2, minimal whisker tremor at posterior position; score 1, absent whisker movement with fixed posterior orientation22,23. Blink reflex grading employed a modified five-point ordinal scale wherein corneal air puff stimulation elicited graded responses 24 : score 5, complete palpebral closure with brisk reflex; score 4, greater than two-thirds eyelid apposition; score 3, less than one-half eyelid closure; score 2, orbicularis oculi contraction without lid movement; score 1, no observable response 22 .
Animal sacrifice, tissue harvesting, and histological analysis (week 12)
Tissues were harvested at 12 weeks after surgery. After the final behavioral tests, animals were deeply anesthetized, and conduits containing regenerated nerve tissue were removed and fixed in 4% paraformaldehyde. Specimens were then dehydrated through a graded ethanol series, infiltrated with paraffin, and embedded for cross-sectional cutting at 1 µm thickness. Sections were stained with toluidine blue (Fig. 4) and examined under bright-field microscopy to assess axonal and myelin content.
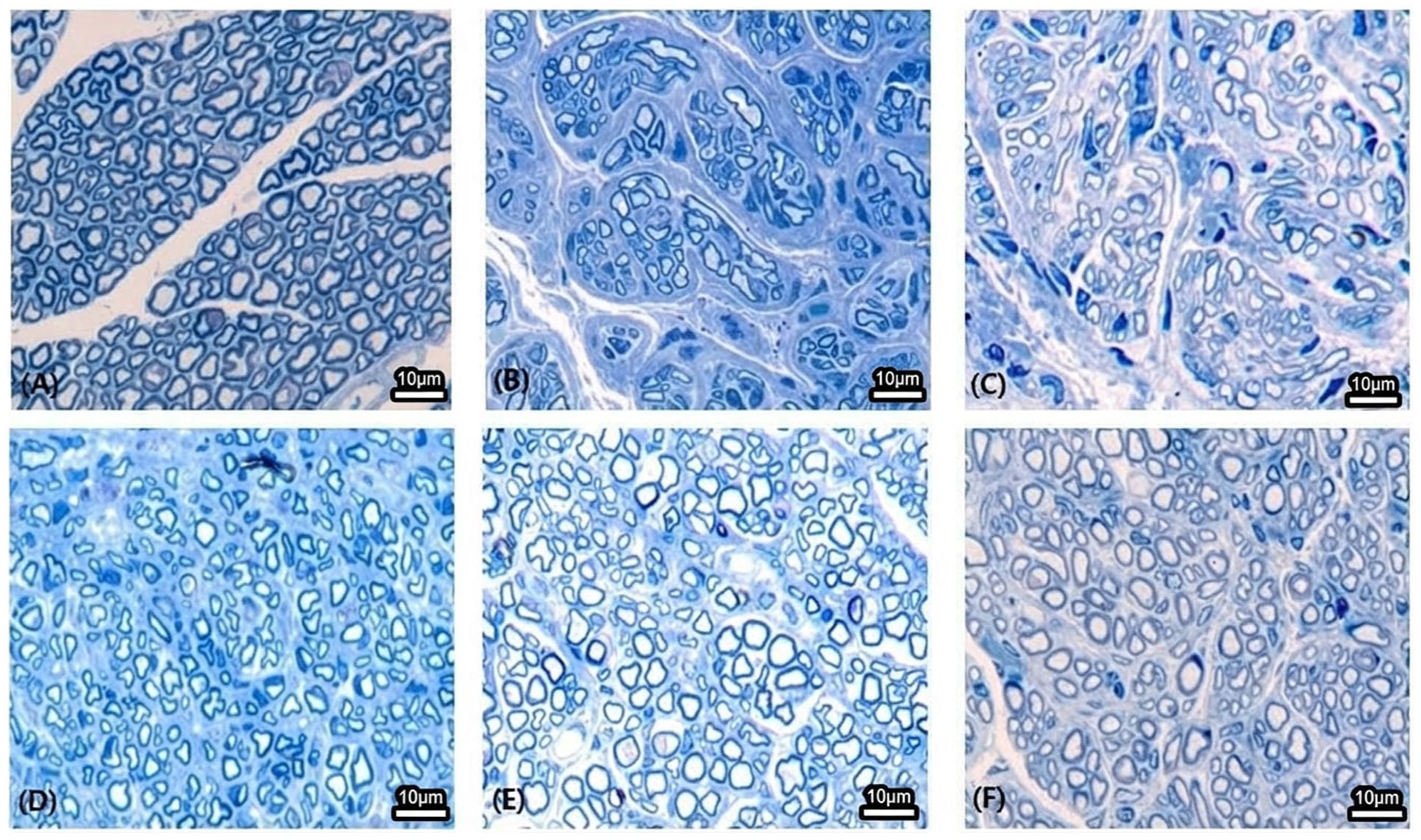
Toluidine blue staining of regenerated facial nerve fibers. Representative light micrographs of semi-thin transverse sections (10 μm) stained with toluidine blue at 12 weeks post-surgery. (a) Sham control group, (b) axotomy-only group, (c) empty conduit group, (d) Matrigel conduit group, (e) conduit with Schwann cell precursors (SCPs), (f) conduit with immature Schwann cells. Scale bar = 10 μm.
Transmission electron microscopy and morphometric analysis
Ultrathin sections (70 nm) were placed onto copper grids (F-200; Nisshin EM Co., Ltd., Tokyo, Japan) and double-stained with 2% uranyl acetate followed by lead citrate (Sigma-Aldrich, USA). Ultrastructural observation was performed using a Hitachi H-7600 TEM operated at 80 kV (Fig. 5). From each specimen, five representative micrographs at ×1500 magnification were captured, and morphometric parameters—including axon diameter, fiber diameter, myelin thickness, and g-ratio (axon-to-fiber diameter ratio)—were quantified for approximately 100 myelinated axons per sample using Photoshop CS2 (Adobe Systems, USA). Myelin thickness was determined as the average of three randomly selected points around each axon, and the g-ratio was determined as the ratio of axonal diameter (d) to myelinated fiber diameter (D) (Supplementary Figure S2).

Transmission electron microscopy (TEM) of regenerated myelinated fibers. Representative TEM micrographs of transverse sections of regenerated facial nerve fibers harvested at 12 weeks post-surgery. (Group A) Sham control, (group B) axotomy only, (group C) empty conduit, (group D) Matrigel conduit, (group E) conduit with Schwann cell precursors (SCPs), (group F) conduit with immature Schwann cells. Groups B and C were combined (B+C) because no significant differences were observed between axotomy-only and empty conduit groups in Bonferroni post-hoc comparisons (P > 0.05). Similarly, groups E and F were combined (E+F) because no significant differences were observed between SCP and immature SC groups (P > 0.05). See Supplementary Table 2 for full pairwise statistical results. Labeled structures: A = axon, M = myelin sheath (white annotation), scale bar = 2 μm.
Statistical analysis
Statistical analyses were performed using R (version 4.0.1; R Foundation for Statistical Computing, Vienna, Austria) and SAS (version 9.4; SAS Institute, Cary, NC, USA). Behavioral data were analyzed using mixed-effects models to account for repeated measurements, and morphometric data were compared using one-way ANOVA with Bonferroni correction for multiple comparisons. Group differences in surgical outcomes were assessed by univariate ANOVA with Bonferroni post-hoc tests (P < 0.05). Repeated-measures ANOVA was used to evaluate changes over time, with week 1 as the baseline. Baseline comparisons between groups were made using one-way ANOVA, and treatment-by-time interactions were analyzed with mixed-model ANOVA across all groups (A–F).
Results
Based on these in vitro characterization data indicating relatively distinct maturation states of the transplanted populations, both SCPs and immature SCs were subsequently used for in vivo transplantation experiments. All silicone nerve conduits were successfully retrieved at 12 weeks post-transplantation without evidence of infection, swelling, or displacement. Regenerated nerve tissue in all groups was encapsulated by granulation tissue, and no neuroma formation was observed.
Histological and ultrastructural findings
Toluidine blue staining and transmission electron microscopy demonstrated clear group-dependent differences in nerve regeneration (Figs. 4 and 5). Groups B (axotomy only) and C (empty conduit) showed sparse, thinly myelinated fibers with smaller axon diameters and thinner myelin sheaths and did not differ significantly from each other. In contrast, group D (Matrigel) and both SC-treated groups (E: SCPs; F: immature SCs) exhibited thicker regenerated fibers with more robust myelination and improved ultrastructural organization, consistent with qualitative histological impressions, although systematic axon counting was not performed. No discernible differences were observed between groups E and F. Detailed morphometric values are provided in Table 1.
Morphometric analysis of the regenerated nerves on transmission electron microscopy images.

Data are presented as mean ± SD. The different alphabetical letters mean that each group is significantly different by the Bonferroni post-hoc analysis.
P < 0.001.
Quantitative morphometric analysis
Morphometric measurements confirmed significant differences among the six groups (one-way ANOVA, all P < 0.001; Table 1; Supplementary Table 2). Post-hoc comparisons showed that groups B and C did not differ from each other, and groups E and F also showed no significant differences, indicating comparable regenerative effects of SCPs and immature SCs. Both SC-treated groups exhibited significantly larger axon diameters, larger myelinated fiber diameters, and thicker myelin sheaths than groups B, C, and D (all P < 0.001). Based on these similarities, groups B+C and groups E+F were combined for visualization in Fig. 6, while complete pairwise comparisons are provided in Supplementary Table 2.

Quantitative morphometric analysis of regenerated myelinated fibers. Bar graphs showing (A) myelin thickness, (B) myelinated fiber diameter, (C) axonal diameter, and (D) g-ratio (axon/fiber) in each experimental group at 12 weeks post-surgery. Group A: Sham, B: axotomy only, C: empty conduit, D: Matrigel conduit, E: conduit with Schwann cell precursors (SCPs), F: conduit with immature Schwann cells. Groups B and C were combined (B+C) because Bonferroni post-hoc comparisons revealed no significant differences between axotomy-only and empty conduit groups (P > 0.05). Likewise, Groups E and F were combined (E+F) because no significant differences were found between SCP and immature SC groups (P > 0.05). See Supplementary Table 2 for the complete pairwise comparison matrix. Data are expressed as mean ± standard error of the mean. Groups showing no significant differences in Bonferroni post-hoc analysis (B vs C, E vs F; all P > 0.05) were combined for visual clarity. *P < 0.05, **P < 0.01, ***P < 0.001 compared with indicated groups; #P < 0.05 vs. axotomy-only group.
Behavioral assessment
Blink reflex scores improved progressively over 12 weeks in all groups, with modest but significant improvement in the SC-treated groups (E and F) compared with groups B and C at weeks 8 and 12 (P < 0.05) (Fig. 7; Table 2). No significant differences were observed between groups E and F at any time point. Vibrissae movement exhibited substantial interindividual variability and did not differ significantly among groups; group-level trajectories are shown in Supplementary Figure S3.

Temporal course of behavioral recovery following facial nerve injury and treatment. Blink reflex scores over 12 weeks post-surgery. Rats in groups E (SCPs) and F (immature SCs) showed significantly improved blink reflex recovery compared with groups B (axotomy only) and C (empty conduit) at weeks 8 and 12 (*P < 0.05). Group B: axotomy only, C: empty conduit, D: Matrigel conduit, E: conduit with Schwann cell precursors (SCPs), F: conduit with immature Schwann cells (SCs).
Behavioral recovery scores over time (Blink reflex).

Data are presented as mean ± SD. The asterisk (*) denotes significant differences with group B by the Bonferroni post-hoc analysis.
Indicate statistically significant differences between groups within the same time point, whereas values sharing at least one common letter are not significantly different.
p < 0.05.
Discussion
This study demonstrates that transplantation of hESC-derived SCPs and immature SCs is associated with enhanced structural regeneration of the transected facial nerve in a rat model. Although the degree of functional improvement was modest, the histological and ultrastructural findings consistently indicate that SC transplantation can promote axonal maturation and myelin formation following severe peripheral nerve injury.
The regenerative effects observed after SC transplantation align with the established roles of SCs in peripheral nerve repair. SCs provide trophic support, direct axon outgrowth, and promote myelin formation through dynamic interactions with regenerating axons11,12,14. hESC-derived Schwann lineage cells can also adopt a pro-regenerative state marked by upregulation of repair-associated gene programs, thereby enhancing the pro-regenerative microenvironment 25 . The increased myelin thickness and improved ultrastructural organization observed in this study are consistent with these mechanisms.
A notable finding is that SCPs and immature SCs produced comparable regenerative outcomes. Several factors may explain this similarity. First, the two populations share overlapping phenotypic features and may converge toward similar functional states after transplantation due to cues from the host microenvironment 26 . Second, because lineage tracking was not performed, the survival, differentiation, and direct contribution of transplanted cells cannot be determined. Third, the absence of quantitative axon counting limits detection of subtle differences in axonal density. Finally, the purity of FACS-sorted SCP and immature SC populations was not quantified, making direct comparison between the two groups more challenging. Future studies incorporating fate mapping, phenotype analyses, and rigorous cell characterization will be essential for determining whether SCPs and immature SCs exert distinct biological effects in vivo.
Although blink reflex recovery improved in SC-treated groups at later time points, the magnitude of functional benefit remained modest. Moreover, vibrissae movement did not show significant differences among groups, indicating that functional improvements were limited to specific motor functions rather than representing comprehensive facial nerve recovery. These findings likely reflect inherent limitations of behavioral testing for rodent facial nerve function. The blink reflex score is an ordinal measure with limited sensitivity, assessments were not blinded, and vibrissae movement demonstrated high inter-individual variability. These factors reduce the ability to detect subtle treatment-related differences. Given these methodological constraints, claims regarding functional recovery must be interpreted cautiously. Furthermore, functional recovery dynamics may not be fully captured at the 12-week endpoint. Incorporating quantitative kinematic or electromyographic analyses in future studies may provide a more sensitive evaluation of facial motor function 24 .
Because this study involved xenotransplantation of human Schwann lineage cells into rats, potential immune responses must be considered. Peripheral nerve tissue exhibits relative immune privilege owing to limited vascularity and reduced antigen-presenting capacity, which may mitigate acute xenogeneic rejection27,28. SCs additionally shape the immune environment by promoting M2-like macrophage polarization and producing anti-inflammatory cytokines such as IL-10 and TGF-β29,30. These factors may partly explain the absence of overt inflammatory or rejection-related changes in the present study. However, systematic immunophenotypic analyses were not performed, and subtle immune reactions cannot be excluded. Immune profiling should be incorporated in future experiments to better define host–graft interactions and the durability of xenogeneic SC survival.
The silicone conduit model used in this study provides a controlled platform for assessing nerve regeneration, but it has limited clinical applicability because silicone is non-biodegradable and associated with chronic foreign-body reactions 31 . Thus, the present findings should be interpreted as proof of concept rather than a direct translational strategy. Biodegradable and clinically relevant conduits should be used in future studies to more accurately model potential therapeutic applications.
This study has several limitations. First, quantitative axon counts were not performed, restricting interpretation of axonal density or total regenerative capacity. Second, the fate and functional contribution of transplanted hESC-derived Schwann lineage cells were not evaluated; without lineage tracking, we cannot determine whether these cells survived, directly contributed to remyelination, or exerted primarily paracrine effects. Third, although the relative maturation states of SCPs and immature SCs were characterized using molecular and protein-level markers, absolute cellular purity of each differentiation stage was not quantified, which may limit direct comparison between the two populations. Fourth, behavioral assessments were unblinded and relied on coarse ordinal scales. Finally, immune responses to xenotransplanted human cells were not systematically analyzed. These factors should be addressed in future studies to strengthen mechanistic interpretation and translational relevance.
Conclusions
In summary, transplantation of hESC-derived SC lineage cells was associated with improved structural parameters of facial nerve regeneration in a rat transection model. However, because cell survival, lineage fate, and direct myelinating roles were not assessed, the present findings should be interpreted as evidence that SC transplantation modifies the regenerative microenvironment rather than confirming a direct myelinating contribution of the transplanted cells. These results provide proof-of-concept support for SC transplantation as an adjunctive strategy, but further mechanistic validation is required before clinical translation.
Future studies should incorporate lineage tracing, immune profiling, and quantitative axon counting to clarify the specific mechanisms by which transplanted Schwann lineage cells influence nerve regeneration. Earlier post-injury time points and higher-resolution functional assessments may provide additional insight into the dynamics of facial nerve recovery. In addition, the use of biodegradable conduits and optimization of SC purity will be essential for advancing SC transplantation toward clinical translation.
Supplemental Material
sj-docx-3-cll-10.1177_09636897261422301 – Supplemental material for Effects of human embryonic stem cell–derived Schwann cells for nerve regeneration after facial nerve injury
Supplemental material, sj-docx-3-cll-10.1177_09636897261422301 for Effects of human embryonic stem cell–derived Schwann cells for nerve regeneration after facial nerve injury by Myung Chul Yoo, Jong Ha Lee, Jae Min Lee, Yong Jun Kim and Seung Geun Yeo in Cell Transplantation
Supplemental Material
sj-jpg-1-cll-10.1177_09636897261422301 – Supplemental material for Effects of human embryonic stem cell–derived Schwann cells for nerve regeneration after facial nerve injury
Supplemental material, sj-jpg-1-cll-10.1177_09636897261422301 for Effects of human embryonic stem cell–derived Schwann cells for nerve regeneration after facial nerve injury by Myung Chul Yoo, Jong Ha Lee, Jae Min Lee, Yong Jun Kim and Seung Geun Yeo in Cell Transplantation
Supplemental Material
sj-jpg-2-cll-10.1177_09636897261422301 – Supplemental material for Effects of human embryonic stem cell–derived Schwann cells for nerve regeneration after facial nerve injury
Supplemental material, sj-jpg-2-cll-10.1177_09636897261422301 for Effects of human embryonic stem cell–derived Schwann cells for nerve regeneration after facial nerve injury by Myung Chul Yoo, Jong Ha Lee, Jae Min Lee, Yong Jun Kim and Seung Geun Yeo in Cell Transplantation
Footnotes
Ethical approval
All animal procedures were approved by the Institutional Animal Care and Use Committee (IACUC) of Kyung Hee University Hospital (approval number: KHMC-IACUC-2022-044; date of approval: November 25, 2022).
Author Contributions
Myung Chul Yoo: Writing—original draft, Visualization, Methodology, Formal analysis, Data curation, Conceptualization, Funding acquisition. Jae Min Lee: Writing—Supervision, Conceptualization. Yong Jun Kim: Writing—review & editing, Supervision. Jong Ha Lee: Writing—review & editing, Supervision. Seung Geun Yeo: Writing—review & editing, Supervision, Funding acquisition.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (NRF 2022R1A2C1091779). This research was supported by a grant from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: RS-2024-00509118). This work was also supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (RS-2024-00346622).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Not applicable.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Statement of Human and Animal Rights
All animal procedures were approved by the Institutional Animal Care and Use Committee (IACUC) of Kyung Hee University Hospital and were conducted in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
