Abstract
Traumatic brain injury (TBI) remains a major global health challenge with limited effective treatments. This systematic review documents the significant evolution of mesenchymal stem cell (MSC) therapies from traditional cell transplants to advanced cell-free products and engineered delivery systems. A systematic search was conducted across Web of Science, Embase, and PubMed/Medline for studies published from January 2015 to June 2025, resulting in the inclusion of 80 studies for qualitative synthesis. The review identifies four primary therapeutic mechanisms: reducing inflammation, protecting brain cells, maintaining the blood–brain barrier, and supporting tissue repair/regrowth. This PROSPERO-registered systematic review followed Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. Methodological quality and risk-of-bias were rigorously assessed using a multi-tool approach tailored to study design (Cochrane RoB, SYRCLE, NOS/JBI). Recent clinical evidence (e.g. STEMTRA trial) suggests these treatments are safe and can improve function in chronic TBI patients. Despite this, critical research gaps persist in establishing standardized protocols, optimal dosing, and long-term safety data. Emerging trends include (1) shifting toward enhanced exosomes/secretomes, (2) integrating MSCs/derivatives with advanced biomaterials for controlled delivery, (3) exploring alternative MSC sources, and (4) developing combination therapies. With a deepening mechanistic understanding and positive early clinical results, future research must prioritize standardization and personalized treatment plans to accelerate clinical translation.

Keywords
Introduction
Traumatic brain injury (TBI) represents a significant global health challenge, affecting millions worldwide each year. This is reflected in epidemiological studies which indicate that over 50 million people sustain TBIs annually, with substantial percentages of emergency admissions and trauma-related deaths observed in both developed and developing regions 1 . In the United States alone, estimates suggest that more than 1.7 million new cases occur yearly, a figure accompanied by a high prevalence of long-term disabilities that impose a significant economic and societal burden 2 .
This burden is manifested in a range of debilitating long-term sequelae that persist long after the initial injury. Survivors of moderate-to-severe TBI frequently suffer from persistent cognitive dysfunction, including significant deficits in memory, attention, and executive function, which severely impacts their ability to return to work and reintegrate into society 3 . Motor deficits are also a major contributor to long-term disability, with many patients experiencing impaired balance, poor coordination, spasticity, and hemiparesis that limit mobility and independence 4 . Beyond these core deficits, TBI is a leading cause of post-traumatic epilepsy and is strongly associated with an increased lifetime risk of psychiatric disorders, such as major depression, anxiety, and post-traumatic stress disorder (PTSD)5,6. Perhaps most concerning is the established epidemiological link between a history of TBI and an accelerated risk for developing late-life neurodegenerative diseases, including Alzheimer’s disease and chronic traumatic encephalopathy (CTE) 7 .
Current standard-of-care, while essential for acute stabilization, offers limited options for reversing established neurological damage. Treatment primarily consists of supportive care and physical rehabilitation, which often results in incomplete functional recovery and fails to halt the underlying, progressive neurodegenerative processes initiated by the secondary injury cascade 1 . This therapeutic vacuum highlights the urgent clinical need for innovative interventions that can target the multiple, complex injury mechanisms to improve these specific, clinically relevant outcomes.
In China, TBI poses some public health challenge, with long-term consequences extending beyond the initial injury. A large, nationally representative cross-sectional survey by Jiang et al. 8 provides crucial epidemiological insights into this burden. The study, which included nearly 600,000 participants, found a weighted TBI prevalence of 434.9 per 100,000 people9,10. A critical finding from this research is the substantial risk of future cerebrovascular events for those with a history of TBI. While the study identified a weighted prevalence for comorbid stroke and TBI of 29.30 per 100,000 people, the risk was not symmetrical. The data revealed that the prevalence of stroke in patients with a prior TBI was 11 times higher than the prevalence of TBI in patients with a prior stroke. This starkly illustrates that TBI is a major risk factor for subsequent stroke, thus highlighting that the public health impact of TBI includes a heightened, long-term burden of serious secondary complications.
From a pathophysiological perspective, TBI is an alteration in brain function resulting from an external force to the head, leading to a complex constellation of physical, cognitive, and emotional impairments. TBI typically involves both an immediate primary injury caused by mechanical impact and a secondary injury that develops over time due to biochemical cascades, inflammation, and cellular dysfunction 11 . TBI progression is typically understood through two interconnected phases: primary and secondary injuries. The primary injury occurs at the moment of impact, resulting from the direct physical forces on the brain. Following this initial event, a secondary injury phase begins, characterized by a cascade of harmful biological reactions within the brain. These reactions include inflammation, excessive nerve cell stimulation (excitotoxicity), damaging chemical processes (oxidative stress), impaired cellular energy production (mitochondrial dysfunction), and breakdown of the protective blood–brain barrier (BBB)12,13. Because these secondary processes unfold over time after the initial trauma, they represent important targets for potential treatments aimed at limiting ongoing brain damage 14 . Despite detailed knowledge of these secondary injury mechanisms, current medical treatments for TBI are primarily supportive. Conventional approaches have not been effective in halting the complex cascade of secondary events, leaving a significant gap in care for TBI patients 15 . This inability to fully address the secondary injury contributes to poor long-term outcomes for survivors and also leads to substantial economic and societal costs due to prolonged healthcare needs and lost productivity 1 . As a result, researchers are increasingly investigating cell-based therapies. The goal of these therapies is to intervene in multiple damaging pathways simultaneously. Specifically, strategies aim to protect nerve cells, control harmful inflammation, encourage the brain’s natural repair processes, and help maintain the integrity of neural connections14,16.
Among the cell types being explored, mesenchymal stem cells (MSCs) have gained significant interest. MSCs are multipotent stromal cells characterized by their ability to self-renew and differentiate into a variety of cell types, such as osteoblasts, chondrocytes, and adipocytes 17 . MSCs naturally possess therapeutic properties, including the ability to reduce inflammation, modulate the immune system, and release factors that support tissue repair and recovery after brain injury 14 . Studies suggest that MSCs achieve these effects largely through indirect actions, releasing various beneficial molecules (collectively known as the secretome, which includes factors delivered via extracellular vesicles or EVs) that influence the injured brain environment 18 . For this review, we use EVs as the broad term encompassing all secreted lipid-bound particles and reserve the term exosomes only for studies claiming purification of this sub-population. The increasing focus on such vesicles has also necessitated the development of rigorous methodological standards, such as the MISEV2023 guidelines, to ensure reproducibility 19 . In addition, the way MSCs are used therapeutically has also evolved. Early research focused on transplanting whole MSCs, hoping they would replace damaged brain cells. More recent approaches utilize the substances released by MSCs (their secretome), employ genetically modified MSCs to enhance specific functions, or use supportive structures (scaffolds) to improve cell delivery and survival at the injury site14,16. This shift reflects a growing understanding that effectively treating TBI requires tackling multiple injury mechanisms, and MSCs, through various strategies, might act as versatile therapeutic agents.
However, translating promising laboratory findings with MSCs into effective clinical treatments has proven challenging. Early clinical trials have highlighted difficulties, including the need for standardized methods for preparing and administering MSCs, uncertainty about the comparative effectiveness of different MSC-based approaches (e.g. whole cells vs secretome), and an incomplete understanding of precisely how they exert their effects in humans with TBI14,15. Variability in trial results has consequently slowed wider clinical adoption. Thus, TBI remains a significant global health challenge, primarily due to the damaging consequences of the secondary injury cascade. Innovative approaches like MSC-based therapies offer considerable promise by potentially addressing multiple aspects of this complex injury process.
Existing literature and research objective
A critical analysis of recent literature confirms that while valuable reviews on MSC therapy for TBI exist, they form a fragmented landscape, with no single work comprehensively addressing the proposed research objectives in a systematic framework. The current body of review literature can be categorized into several distinct, yet siloed, groups. Foundational overviews, such as those by Hasan et al 11 and Das et al 20 , were instrumental in establishing the scientific rationale for MSC therapy. They summarized early preclinical data; identified the primary mechanisms of action, notably immunomodulation and the paracrine secretion of neurotrophic factors; and discussed the fundamental biology of MSCs. However, as products of their time, these reviews predate the significant and rapid shift toward advanced cell-free therapeutics and bioengineered delivery systems. Consequently, they provide a crucial historical baseline but lack the contemporary data needed to guide modern research, which has moved far beyond conventional cell transplantation.
Building on this foundation, more current works focus on specific facets of this therapeutic evolution with essential depth. For instance, the reviews by Yudha et al 21 on the MSC secretome and Liu et al 22 exclusively on exosomes provide granular analyses of cell-free mechanisms. They delve into specific molecular cargo like miRNAs, detail complex signaling pathways, and discuss the technical challenges of isolation and characterization. Their limitation, however, is that by concentrating on one branch of the evolutionary tree, they do not situate these advanced therapeutics within the broader context of MSC therapy, nor do they systematically compare their progress and potential against the whole-cell or scaffold-based approaches that preceded them.
Other studies adopt a translational or methodological perspective. Carbonara et al. 23 framed MSCs as a promising solution to the historical failures of single-target pharmacological trials in TBI, highlighting their multi-modal action and wider therapeutic window. This provided a compelling clinical rationale but did not systematically analyze or compare the different MSC-based therapeutic modalities themselves. More recently, Yang et al 24 conducted a rigorous network meta-analysis to quantitatively compare the efficacy of EVs from different cellular origins. While methodologically powerful, their analysis was intentionally restricted to preclinical EV studies, excluding whole-cell therapies, clinical trial data, and bioengineered systems from its scope.
What remains missing is a comprehensive, contemporary synthesis that critically documents the entire evolutionary trajectory of MSC-based therapies over the past decade. The field has undergone a clear, stepwise progression, from conventional cell transplantation to advanced cell-free and sophisticated bioengineered systems. No existing review systematically charts this longitudinal progression, analyzes the key innovations at each stage, and places these diverse modalities into a single comparative framework.
Hence, this research seeks to achieve three goals: (1) Analyze the progression of MSC therapies from whole-cell transplantation to advanced cell-free and bioengineered delivery systems. (2) Characterize the mechanisms of MSC action and evaluate their translational progress from preclinical to clinical application. (3) Compare different MSC strategies (sources, delivery, modification) and identify critical research gaps necessary for clinical implementation.
Research significance
This comprehensive analysis addresses the critical need for effective TBI treatments by synthesizing a decade of MSC therapy research into a cohesive framework, clarifying progress and persistent challenges. With TBI affecting millions globally and lacking effective treatments, a systematic evaluation across diverse MSC approaches, from traditional transplantation to sophisticated bioengineered and cell-free strategies, provides timely guidance. This evaluation is expected to document the evolution of these therapies, establishing how the shift enhances therapeutic potential while addressing manufacturing and delivery issues. Furthermore, identifying consistent underlying mechanisms, such as immunomodulation, neuroprotection, BBB preservation, and regenerative support, aims to establish a mechanistic roadmap for developing more targeted interventions.
A substantial contribution involves the critical assessment of translational progress. Analyzing clinical trial outcomes alongside preclinical innovations is expected to pinpoint crucial gaps in standardization protocols, dosing optimization, and patient stratification approaches that must be addressed for widespread clinical implementation. Research trends and comparative analyses of delivery methods and cell sources will offer immediate, practical guidance, directing resources toward the most promising avenues, and potentially reducing research redundancy. Ultimately, by systematically connecting promising preclinical findings with the practical requirements of clinical translation, this work intends to accelerate the development of viable MSC-based therapeutic options for patients and clinicians awaiting effective TBI treatments.
Methodology
Search strategy
A comprehensive literature search was conducted across three major scientific databases (Web of Science, Embase, and PubMed/Medline) for publications between 2015 and 2025, following Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. This systematic review was conducted and reported in accordance with the PRISMA statement 25 . Refer to the PRISMA checklist, a part of the supplementary material. The protocol was registered with PROSPERO (CRD42023449349).
Search parameters
Databases: Web of Science, Embase, PubMed/Medline.
Publication period: Full paper available online by 1 January 2015 to 30 June 2025.
Publication types: Original articles only on Controlled Clinical Trials, Randomized Controlled Trials, Case Reports, Original Articles.
Language: English Only.
Search terms: Five keyword combinations: a. ‘Mesenchymal Stem Cells’ AND ‘Traumatic Brain Injury’. b. ‘Mesenchymal Stromal Cells’ AND ‘Head Injury’. c. ‘Mesenchymal Stem Cells’ AND ‘Brain Trauma’. d. ‘MSCs’ AND ‘Closed Head Injury’. e. ‘Mesenchymal Stromal Cells’ AND ‘TBI’.
Study selection process
The search results from the three major scientific databases (Web of Science, Embase, and PubMed/Medline) were combined, and duplicates were removed using Zotero v.7.0.26 to yield a set of unique records. Subsequently, these unique records were manually screened and managed via a shared spreadsheet. Two independent reviewers then screened the titles and abstracts for relevance against the predefined eligibility criteria. The main reasons for exclusion at this stage were lack of focus on MSC therapy for TBI or the material being non-original research. Following title and abstract screening, two independent reviewers thoroughly examined potentially eligible full-text articles. Reviewers remained blinded to each other’s decisions throughout the process, and any discrepancies were resolved through discussion to reach an agreement. Full quantitative details, including the number of studies at each stage, are presented in the PRISMA flow diagram (Figure 1).

Prisma flow diagram for MSC in TBI selection.
Data extraction and synthesis
Following the final selection, two independent reviewers conducted data extraction using a pre-tested Microsoft Excel spreadsheet. The extracted data focused on the PICOS elements relevant to the review objectives: intervention details (MSC source, product type, delivery route, modification strategy), comparison details, and outcome measures (behavioral, histological, electrophysiological, molecular markers). Data on study characteristics (e.g. animal species, TBI model, injury severity, follow-up period) were also recorded. Any disparities in the extracted data were resolved through discussion between the reviewers and consultation with a third reviewer if necessary. The review utilizes a narrative synthesis approach, as meta-analysis was not conducted due to the profound heterogeneity across study designs and interventions. The synthesis was performed by grouping findings based on the therapeutic modality (e.g. scaffolds, cell-free derivatives) to analyze patterns and report the main findings, consistencies, and discrepancies across the evidence base.
Quality assessment methods
Due to the inherent heterogeneity of the literature, spanning randomized human trials, non-randomized cohorts, preclinical animal models, and in vitro studies, a multi-tool approach was implemented for methodological quality assessment to ensure internal validity was judged using the most appropriate standards. This approach, which directly informs our strength of evidence judgments, utilized the following type-specific instruments:
Cochrane Risk of Bias Tool (RoB 2.0): Used for assessing bias in the four major randomized controlled trials (RCTs) (e.g. McAllister et al., Kawabori et al.) and their associated protocols.
Modified SYRCLE’s Risk of Bias Tool: Used for assessing bias across all Preclinical In Vivo Animal Studies (most included literature), focusing on domains critical to animal studies (e.g. Random Housing, Blinding of Caregivers, Random Outcome Assessment).
JBI Critical Appraisal Checklist for Case Series and Reports: Used to assess the methodological quality of Case Reports and Case Series (e.g. Kabataş et al., Nabity et al.).
Newcastle–Ottawa Scale (NOS): Used for assessing the quality of Observational Studies/Cohorts where applicable (e.g. non-randomized clinical cohorts like Nguyen et al.).
This ensured that the overall methodological rigor of the body of evidence was accurately and transparently assessed. Tabular and graphical details are in the supplementary material, Tables 18–21 and Figures 2–5.

Cochrane risk of bias assessment (ROB traffic light illustration).

Cochrane risk of bias assessment (ROB summary illustration).

Syrcle risk of bias assessment (summary).

Syrcle risk of bias assessment (traffic light illustration).
Classification and analysis
The foundation for this analysis rests upon the source excerpts detailing the application of MSC therapies in the context of TBI. This body of literature includes preclinical investigations across multiple species (rat, mouse, swine) employing MSCs sourced from bone marrow, umbilical cord, adipose tissue, cranial bone, olfactory mucosa, and placenta. A spectrum of therapeutic approaches is represented, encompassing not only direct MSC transplantation but also the use of MSC-derived products like secretome and EVs (including exosomes), genetically engineered MSCs, and MSCs combined with biomaterial scaffolds or adjunctive therapies. Details are presented in Table 1, tables section, in the supplementary material.
Categories of studies and their defining characteristics.
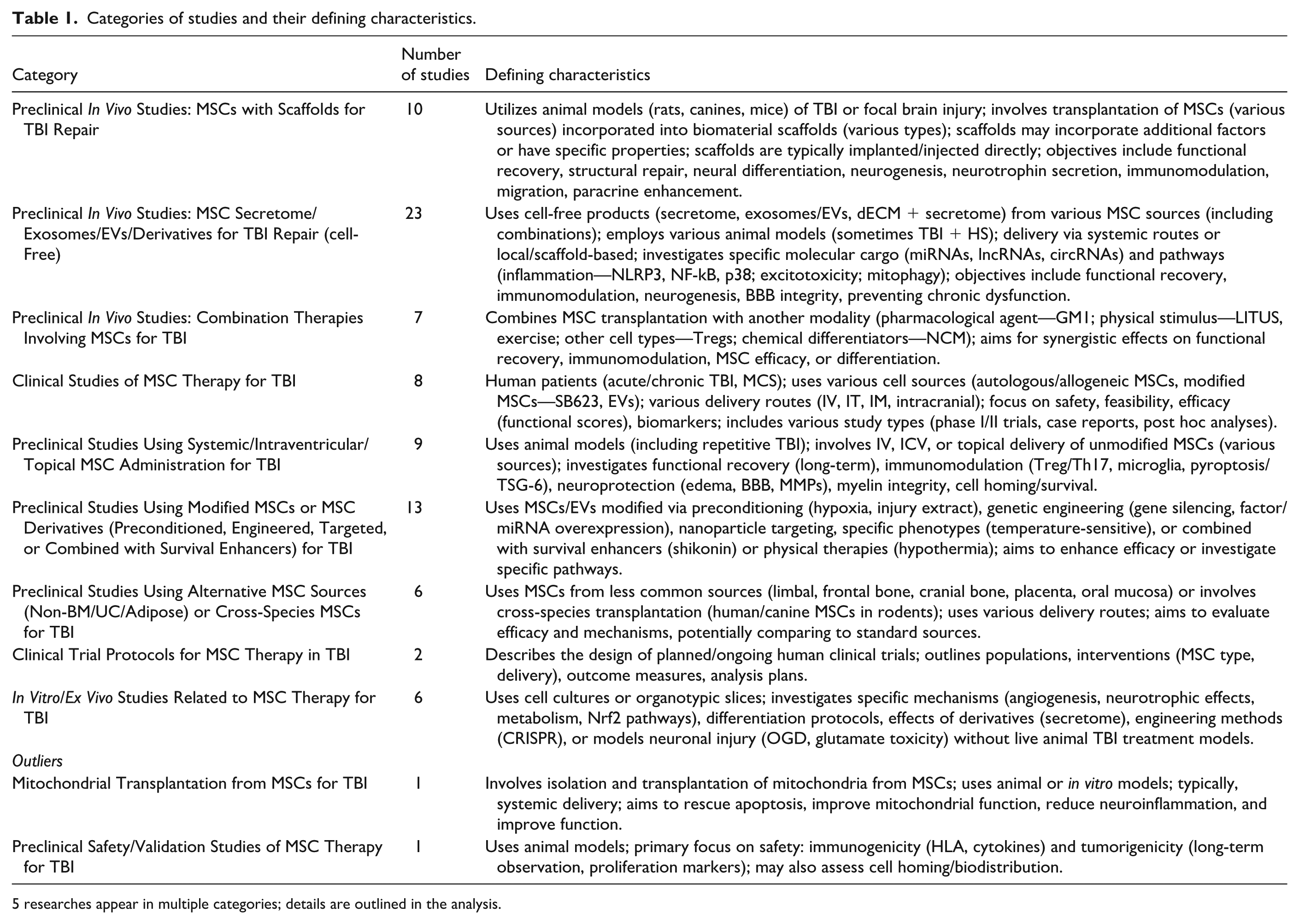
5 researches appear in multiple categories; details are outlined in the analysis.
Intra-category analysis and findings
Preclinical in vivo studies: MSCs with scaffolds for TBI repair
The studies within this category explore innovative therapeutic strategies for TBI using biomaterials and MSCs covering a diverse set of preclinical studies using various animal models of TBI, predominantly rodents and canines. The studies employed different scaffold materials combined with MSCs from various sources and evaluated their efficacy using a range of behavioral, histological, and molecular outcome measures. These include the following:
Scaffold types and properties: Identification and characterization of scaffold materials, their physical and biochemical properties, and functional attributes.
MSC sources and integration: Analysis of MSC sources, isolation methods, characterization, and their interaction with scaffold materials.
Mechanism forms: Examination of proposed mechanisms through which scaffold–MSC combinations exert therapeutic effects.
Outcome measures: Assessment of behavioral, histological, electrophysiological, and molecular methods used to evaluate therapeutic efficacy.
Scaffold types and properties
The reviewed literature showcases a diverse array of scaffold materials, each with unique properties tailored to address the complex challenges of TBI repair. Hydrogels, including gelatin methacrylate (GelMA), hyaluronic acid (HA), sodium alginate (SA)/collagen, and enzyme-crosslinked gelatin hydrogels, are prominent due to their injectability, which allows for minimally invasive delivery to the lesion site, minimizing further trauma26–30. These hydrogels often incorporate modifications to enhance their bioactivity, such as imidazole groups to promote neural differentiation or the capacity for controlled release of therapeutic factors like stromal cell–derived factor-1 (SDF-1α), nerve growth factor (NGF), or Fas ligand (FasL) 30 . Porous scaffolds, exemplified by poly(lactic-co-glycolic acid) (PLGA), collagen/silk fibroin (SF) composites, and peptide-based self-assembling nanoscaffolds like R-GSIK, provide a three-dimensional (3D) architecture that supports cell adhesion, growth, and migration31–33. These scaffolds can be engineered with specific physical properties, such as porosity and mechanical strength, to mimic the native brain tissue environment and facilitate tissue regeneration27,32,34. PLGA, for instance, is highlighted for its biodegradability and processability, making it suitable for drug delivery. Collagen and SF are favored for their biocompatibility and biodegradability, offering a matrix for cell colonization and axonal growth 34 .
In addition, decellularized extracellular matrix (dECM)-derived scaffolds are also gaining attention for their inherent tissue specificity and low immunogenicity 26 . Combining dECM with hydrogels like GelMA aims to recreate the native tissue niche, providing essential cues for neurogenesis and immunoregulation. Furthermore, stimulus-responsive scaffolds, such as the magnetically manipulated DHAC-F hydrogel composed of collagen and modified HA incorporating magnetic nanoparticles, represent a sophisticated approach to dynamically regulate the cellular microenvironment through external cues, influencing neural differentiation and tissue regeneration 27 .
The trend observed is clearly toward more functionalized scaffolds that go beyond simple structural support. The incorporation of bioactive molecules for controlled release and the development of stimulus-responsive materials suggest a move toward precisely engineered microenvironments to guide cellular behavior and enhance therapeutic efficacy28,30,35. While specific materials like PLGA remain common due to their established properties, newer approaches utilizing dECM and stimulus-responsive hydrogels indicate an evolving understanding of the intricate interplay between the scaffold and the host tissue27,32. Summary of these observations across the studies is illustrated in Table 2.
Overview of scaffold types and key properties.

MSC sources and integration
The reviewed studies in this section explore MSCs from diverse sources including bone marrow (BMSCs), human umbilical cord (hUC-MSCs), and adipose tissue (AD-MSCs) for TBI repair. Each source offers unique advantages: BMSCs provide established protocols and neural differentiation potential; hUC-MSCs offer non-invasive collection and enhanced paracrine effects; and AD-MSCs present abundant source tissue with simpler isolation. These MSCs were integrated with scaffolds through various methods including direct seeding with pre-implantation culture periods (as in Jiang et al.’s collagen/SF scaffolds) 31 , encapsulation (Zhang et al.’s dBECM/GelMA hydrogels showing good biocompatibility) 26 , and injection (Sahab Negah et al.’s MSC-scaffold suspension). Assessment of transplanted MSCs revealed promising outcomes: long-term survival with neuronal differentiation markers 31 , enhanced neural marker expression in hydrogel environments, and improved neurological outcomes with reduced neuroinflammation 33 collectively demonstrated the therapeutic potential of MSC–scaffold combinations for TBI treatment.
Mechanisms forms
The primary proposed mechanisms for scaffold + MSC therapy in TBI involve several interconnected functions. Scaffolds are designed to bridge the cavity left by damaged tissue, providing crucial structural support that prevents cavity collapse and offers a physical substrate conducive to axonal regeneration 31 . Beyond mere structure, these scaffolds act as sophisticated delivery vehicles, ensuring the localized and controlled release of MSCs along with vital neurotrophic factors, chemokines, or immunomodulatory agents directly at the injury site30,34. This targeted delivery aims to bolster cell survival, enhance the recruitment of beneficial cells, and promote differentiation28,30. Furthermore, the inherent properties of the scaffold itself, such as its material composition, mechanical stiffness 27 , and the incorporation of specific bioactive cues like imidazole, RGD peptides, or growth factors30,31,35, can actively guide the differentiation of the delivered MSCs toward neural lineages, potentially replacing lost neurons and supporting neurogenesis27,29,31. Concurrently, scaffolds can contribute to local immunomodulation, for instance, FasL-releasing hydrogels are designed to induce apoptosis in cytotoxic T cells, fostering a more favorable environment for MSC survival 36 . This complements the intrinsic immunomodulatory capabilities of MSCs, which can be potentiated by the scaffold microenvironment 33 . Finally, the scaffold provides a protective niche, shielding the transplanted MSCs from the hostile conditions often found in the injured brain, including inflammation, hypoxia, and nutrient deprivation, thereby significantly improving their engraftment, survival, and overall therapeutic efficacy.
Outcome measures
Behaviorally, researchers utilized standardized neurological scoring systems including modified Glasgow Coma Scale (GCS), Purdy scale, Neurological Deficit Score (NDS), and modified Neurological Severity Score (mNSS) to evaluate functional recovery, with subjects receiving scaffold–MSC combinations consistently demonstrating significant improvements compared to controls28,31,33. Jiang et al 31 implemented sophisticated gait analysis using motion capture systems, surface electromyography, and vertical ground reaction force measurements, revealing improved joint motion, more regular gait patterns, increased muscle activity, and enhanced weight-bearing capacity in treated animals. Electrophysiologically, motor evoked potential measurements showed shortened latency and increased amplitude in treatment groups, indicating improved conduction along the corticospinal tract31,34. Besides, advanced imaging techniques provided crucial structural and metabolic insights, with magnetic resonance imaging (MRI) demonstrating better cerebral motor cortex repair in treatment groups 31 . Also, diffusion tensor imaging showing improved corticospinal tract integrity with visible neonatal tracts on injured sides and magnetic resonance spectroscopy revealing higher NAA/Cr, NAA/Cho, and NAA/(Cho + Cr) ratios indicative of enhanced neuronal integrity. Histological and molecular assessments formed the foundation of all studies, with immunohistochemistry and immunofluorescence evaluating expression of markers related to neural regeneration (NeuN, DCX, MAP-2, NEFM), inflammation (GFAP, Iba-1), myelination (MBP), synaptogenesis (Syn), and vascularization (vWF)26,31,32. Advanced molecular, ultrastructural, and inflammatory analyses offered a comprehensive, multi-domain evaluation of therapeutic outcomes26,28,31 (see assessment and measures in Table 3).
Outcomes based on different assessment methods.

Preclinical in vivo studies: MSC secretome/exosomes/EVs/derivatives for TBI repair (cell-free)
The research in this category analyzes exosomes/EVs derived from various MSC sources (bone marrow, umbilical cord, adipose) for TBI therapy. It investigates enhancing therapeutic potential via MSC preconditioning (e.g. hypoxia) and details the characterization (size, morphology, markers) of these secreted products. Mechanisms explored involve specific molecular cargo like miRNAs and proteins acting on key signaling pathways (nuclear factor kappa B (NF-κB), mitogen-activated protein kinase (MAPK), Nrf2, NLRP3). Diverse delivery methods (intravenous (IV), intranasal, intraventricular, scaffolds) are studied. Research also covers biodistribution, cellular uptake by microglia/astrocytes, functional recovery, and brain injury reduction assessments in TBI animal models.
Product type
Research into MSC-based therapies for TBI utilizes different product types, including the total MSC secretome and purified exosomes or EVs. The crude secretome, comprising all soluble factors, EVs, and other molecules secreted by MSCs, has shown therapeutic potential, for instance, secretome derived from hUC-MSCs loaded onto 3D-printed scaffolds aided neural network reconstruction in canines 37 , and injury-preconditioned secretome within scaffolds demonstrated enhanced therapeutic effects 38 . While the secretome offers the full spectrum of MSC bioactive factors with possible synergistic benefits, its complex and variable nature poses challenges for standardization and mechanistic understanding (details are in Table 4). Conversely, purified exosomes/EVs are the focus of much recent research, with studies demonstrating their ability to improve neurological outcomes, reduce lesion size, and decrease inflammation in swine models and attenuate glutamate-mediated excitotoxicity in rats39,40. Purification allows for better characterization and standardization, facilitating clearer mechanistic insights, though the process might exclude some beneficial components found in the broader secretome.
Product types with their merits and limitations.

Beyond crude secretome and purified exosomes, advanced formulations integrating these MSC products with biomaterials or sophisticated delivery systems represent a growing area of TBI research. Examples include electrospun nanofibrous platforms designed for localized delivery of stem cell–derived exosomes, 3D-printed collagen/SF scaffolds carrying hUC-MSC secretome, and 3D-printed implants loaded with hypoxia-pretreated MSC exosomes36,41,42. These strategies aim to optimize the delivery, retention, and sustained release of therapeutic agents directly at the brain injury site, potentially offering greater efficacy than administering exosomes or secretome alone. Although direct comparative studies between these product types are limited, purified exosomes/EVs are currently the most widely investigated approach, likely due to advantages in characterization and standardization. However, advanced formulations that combine exosomes with biomaterial scaffolds show significant promise, particularly for enhancing localized, sustained therapeutic effects in cases of severe TBI involving substantial tissue damage.
Source and preconditioning
The therapeutic effectiveness of MSC-derived exosomes, also known as EVs, in TBI is heavily dependent on the specific cellular source of the MSCs (see Table 5). Studies have examined exosomes from various sources, including bone marrow (BM-MSCs), umbilical cord (UC-MSCs), adipose tissue (AD-MSCs), and clonal lines. BM-MSC exosomes have demonstrated the ability to reduce glutamate excitotoxicity, create neuroprotective changes in the brain transcriptome, and modulate microglial polarization for recovery42,47,48. UC-MSC exosomes promote neurological recovery by inhibiting glial activation and provide neuroprotection through pathways like lncRNA TUBB6/Nrf2 and PINK1/Parkin-mediated mitophagy, showing strong potential in reducing neuroinflammation41,49,50. Also, AD-MSC exosomes mitigate secondary neuroinflammation via the NLRP3 pathway and promote microglial M2 polarization via circ-Scmh1 delivery, exhibiting significant immunomodulatory effects. Clonal MSCs have also shown promise in reducing cell death pathways and may offer greater consistency51–53; while BM-MSCs offer balanced neuroprotective and regenerative properties, UC-MSCs and AD-MSCs seem particularly potent in their anti-inflammatory actions.
Cellular sources and preconditioning overview.

In addition to the cellular source, preconditioning methods applied to MSCs before exosome collection significantly enhance their therapeutic profile for TBI treatment. Hypoxic preconditioning, in particular, has shown consistent benefits, with studies demonstrating that exosomes from hypoxia-treated MSCs loaded into implants promote neural regeneration, angiogenesis, and reduce apoptosis more effectively than normoxic exosomes 55 . This method alters the exosomal cargo to be richer in pro-angiogenic, anti-inflammatory, and neuroprotective factors, potentially better mimicking the brain’s injury environment. Other methods include injury-induced preconditioning, where exposing MSCs to injury-related stimuli yielded secretomes with enhanced efficacy in scaffolds and growth factor preconditioning, such as using bFGF, which also improved the therapeutic ability of hUC-MSC-derived secretomes in scaffolds 41 . Comparative analysis suggests hypoxic preconditioning may be the most broadly effective strategy for boosting exosome efficacy across different MSC sources for TBI, significantly enhancing their ability to promote angiogenesis, neurogenesis, and reduce inflammation (details are in Table 5).
Cargo and mechanism
Understanding the molecular contents of MSC-derived exosomes and the signaling pathways they influence is key to harnessing their therapeutic potential for TBI. These exosomes carry diverse bioactive molecules, with microRNAs (miRNAs) being particularly significant. Specific examples include miR-21 from umbilical cord MSCs (UC-MSCs), which inhibits microglial overactivation by targeting the PTEN/AKT/NF-κB pathway, and miR-124-3p from bone marrow MSCs, which reduces glutamate excitotoxicity via the p38 MAPK/GLT-1 axis42,56. In addition, adipose-derived stem cell exosomes deliver circ-Scmh1, promoting anti-inflammatory M2 microglial polarization 52 . Other important miRNAs identified include miR-146a-5p, miR-126, and the miR-17-92 cluster. Beyond miRNAs, exosomes contain crucial proteins and growth factors like brain-derived neurotrophic factor (BDNF), noted for upregulation post-EV treatment, as well as NGF, vascular endothelial growth factor (VEGF), and anti-inflammatory proteins (interleukin-10 (IL-10), transforming growth factor-beta (TGF-β)) that promote neurogenesis, angiogenesis, and immunomodulation 48 .
Lipids and metabolic molecules also contribute to exosome structure and function, potentially aiding cellular survival. MSC-derived exosomes exert their therapeutic effects by modulating multiple intracellular signaling pathways within target brain cells. Key pathways inhibited include the NF-κB pathway, leading to reduced neuroinflammation; the p38 MAPK pathway, which lessens excitotoxicity and inflammasome activity; and the NLRP3 inflammasome, resulting in decreased inflammatory damage51,57,58. Conversely, exosomes can activate protective pathways like the Nrf2 pathway for enhanced antioxidant defense and the PINK1/Parkin-mediated mitophagy pathway to clear damaged mitochondria and reduce cell death50,54. These effects are mediated through interactions with various cell types: exosomes primarily target microglia, shifting them toward an anti-inflammatory M2 state and inhibiting overactivation; they also act on astrocytes to inhibit activation and regulate glutamate transport, provide direct neuroprotection to neurons, and interact with endothelial cells to promote angiogenesis and maintain BBB integrity47–49,54,57.
Delivery and biodistribution
The delivery route and subsequent biodistribution critically influence the therapeutic success of MSC-derived exosomes in treating TBI. So far, IV administration is commonly used due to its clinical ease and systemic reach, enabling neurological recovery, although challenges like rapid clearance by the liver and spleen and crossing the BBB exist39,43. The reason is that intranasal delivery offers a promising alternative, potentially bypassing the BBB for more direct brain access, as demonstrated by Kodali et al. who showed inhibition of detrimental signaling pathways and improved cognitive recovery. Local administration, using methods like electrospun nanofibers or 3D-printed implants, maximizes exosome concentration at the injury site but typically requires invasive procedures50,55,58. Biodistribution studies confirm that exosomes can cross the BBB, particularly when compromised post-TBI and are taken up by various brain cells, notably microglia and astrocytes, though systemic IV administration leads to significant accumulation and clearance by organs like the liver and spleen within hours39,42,49. To overcome delivery challenges and enhance therapeutic outcomes, various strategies are being explored. Scaffold-based delivery systems, such as 3D-printed implants loaded with exosomes or secretome-carrying scaffolds, offer localized, sustained release; protect exosomes from degradation; and create a supportive microenvironment, proving particularly useful for significant tissue damage55,59. While less explored specifically for TBI, surface modification of exosomes with targeting ligands could potentially improve specific cell uptake and reduce off-target distribution.
Furthermore, optimizing the dose and timing of administration is crucial; research indicates dose-dependent functional recovery and suggests an optimal therapeutic window exists, potentially extending days post-injury with early administration showing significant benefits in some models39,60. Tailoring these delivery parameters and strategies to specific TBI scenarios is essential for maximizing the therapeutic efficacy of MSC-derived exosomes.
Preclinical in vivo studies: combination therapies involving MSCs for TBI
This category highlights the evolving landscape of MSC combination therapies for TBI, driven by the potential to overcome monotherapy limitations. Investigations explore novel combinations, like integrated biomaterial-drug systems, and refine existing ones by addressing complexities such as optimal administration timing and potential cell–cell interactions. These sources present preclinical studies, primarily using rat and mouse models, to evaluate the efficacy and mechanisms of these combination approaches in improving outcomes after TBI.
Combination types
Research into MSC therapies for TBI is actively exploring diverse combination strategies to enhance therapeutic outcomes. One major avenue involves combining MSCs with physical modalities. For instance, MSCs have been paired with low-intensity transcranial ultrasound (LITUS), where they were injected directly into the injury site followed by continuous LITUS application61,62. Similarly, human cranial bone–derived MSCs (hcMSCs) were administered intravenously and combined with subsequent treadmill exercise rehabilitation, testing both high and low frequencies63,64. Another approach focuses on combining MSCs with biochemical agents or other cell types. The hUMSCs were studied in conjunction with monosialotetrahexosylganglioside (GM1), a neuro-active molecule, administered systemically alongside local UC-MSC transplantation 63 .
Co-administration of MSCs with other therapeutic cell types, specifically human cord blood-derived regulatory T cells (Tregs), was also investigated via IV infusion, exploring both concurrent and staggered delivery timings64,65. Furthermore, innovative delivery systems and differentiation aids are being integrated. One study utilized an injectable, self-assembling peptide hydrogel (SLNAP) as a vehicle to deliver both human MSCs (hMSCs) and a potent neuro-regenerative chemical modulator (NCM) directly to the TBI site, leveraging the hydrogel for localized scaffolding and sustained release 66 . These varied examples show a trend toward multi-modal approaches, aiming to harness the regenerative and immunomodulatory capacities of MSCs while potentially amplifying their effects through synergistic interactions with physical therapies, biochemicals, other cells, or advanced biomaterials.
Synergy evidence
Studies in this category also indicate synergistic or enhanced effects when combining MSCs with other therapies for TBI. For instance, combinations like MSCs + LITUS, UMSCs + GM1, and hcMSCs + high-frequency exercise demonstrated significantly better functional recovery (cognitive or motor) and more robust increases in key neuroregenerative markers (like GAP-43, BDNF, NGF, neuron-specific markers) compared to either treatment alone61–63,67. Similarly, in vitro, Treg + MSC combinations showed augmented immunosuppressive potency against activated immune cells, surpassing monotherapy results, and the NCM-loaded hydrogel greatly enhanced hMSC differentiation into functional neurons65–67. Specific timings, such as staggered Treg and MSC delivery, also appeared crucial for achieving synergistic reduction of neuroinflammation in vivo. The proposed mechanisms driving this synergy vary with the combination. Physical modalities like LITUS and exercise may enhance MSC proliferation, differentiation, and the release of neurotrophic factors62,67. Biochemical agents like GM1 or NCM likely directly promote MSC differentiation into desired neural lineages, with hydrogels potentially enhancing this through sustained, localized delivery 67 . For cell combinations like Treg + MSCs, synergy might arise from complementary immunomodulatory pathways (potentially involving mediators like PGE2 from MSCs and AREG from Tregs) and Tregs possibly improving the MSC survival or function within the inflammatory TBI environment.
MSC role
In these combination strategies, MSCs themselves are positioned as the core therapeutic agent due to their inherent potential. They serve as a source of regenerative cells, capable of differentiating into neural cell types or promoting neurogenesis through paracrine signaling, potentially enhancing nerve growth and functional recovery. Specific types like hcMSCs are noted for high neurogenic and neuroprotective effects, while human umbilical cord–derived MSCs (hMSCs) demonstrate significant transdifferentiation capabilities63,67. Beyond regeneration, MSCs exert crucial immunomodulatory effects, inhibiting local inflammation post-transplantation and modulating both innate and adaptive immune responses, often attributed to their paracrine release of trophic and repair factors 67 . The combination therapies investigated aim largely to amplify or direct these intrinsic MSC functions. Physical modalities like LITUS are suggested to boost MSC proliferation and differentiation, while exercise may enhance MSC neuroprotective effects62,63. Also, partnering cells, such as Tregs, might improve MSC survival in the host environment and potentially augment their anti-inflammatory mediator production (such as PGE2). Biomaterial approaches, like the SLNAP peptide hydrogel, provide a supportive microenvironment mimicking the native extracellular matrix, facilitating MSC adhesion, differentiation, and serving as a localized reservoir for sustained release of differentiation cues like NCM. Conversely, MSCs also play a role in augmenting the partner therapy, for example, in the Treg+MSC combination, MSCs contribute significantly to overall immunomodulation, potentially working alongside Tregs via distinct pathways to suppress inflammation65,67.
Timing and regimen
The timing of intervention and the administration regimen are critical variables explored across these MSC combination therapy studies for TBI. Several protocols initiated treatments relatively early after injury. For instance, MSCs were injected in situ within 24 h post-TBI before commencing continuous LITUS treatment for 28 days. Similarly, hcMSCs were delivered intravenously 24 h post-injury, followed by exercise rehabilitation starting 24 h later 61 . The concurrent Treg + MSC combination also involved IV infusion at 24 h post-TBI. In contrast, other studies employed later or varied intervention time points. Umbilical Cord Mesenchymal Stem Cells (UMSCs) were transplanted locally 7 days after TBI initiation, coinciding with the start of daily systemic GM1 administration62,67. The potential importance of timing was highlighted in the Treg + MSC studies, where staggering the IV infusions, Treg at 24 h followed by MSC at 72 h, yielded more promising results in reducing neuroinflammation compared to administering both at 24 h63,67. Furthermore, the duration and frequency of the secondary treatment varied significantly, from continuous daily LITUS application to daily or intermittent exercise regimens. Administration routes also differed, including direct in situ injection into the injury site, IV infusion, and localized topical delivery via an injectable, thixotropic peptide hydrogel designed for sustained release at the injury focus65,66. This hydrogel approach aimed specifically to overcome delivery barriers and maintain therapeutic concentrations locally. The varied timings, frequencies, durations, and routes underscore the complexity of optimizing these combination therapies and suggest that the specific ‘therapeutic window’ and delivery method likely influence treatment efficacy63,66,67.
Clinical studies of MSC therapy for TBI
The included studies encompassed a range of study designs, from case reports to RCTs. The analysis focused on the following key parameters:
Study Phase and Design: Study phase (I, II), randomization status, control groups, blinding procedures, and patient population characteristics (acute vs chronic TBI, specific deficits).
Intervention Details: Cell type and source (autologous vs allogeneic), processing methods, delivery route, dose, and administration frequency.
Safety Outcomes: Adverse events, serious adverse events, and safety concerns related to specific cell types or delivery routes.
Efficacy Outcomes: Functional scales used, evidence of efficacy, correlation with specific patient groups, cell types, or delivery parameters, and biomarker changes.
Data extraction and analysis were conducted to identify patterns, consistencies, and discrepancies across studies. The quality of evidence was assessed based on study design, sample size, outcome measures, and follow-up duration.
Study phase and design
The reviewed studies represented a range of clinical trial phases and designs, from early-phase safety studies to more advanced efficacy trials as shown in Table 6. Most studies here focused on patients with chronic TBI (>12 months post-injury), with only one study addressing acute TBI. The most robust evidence came from phase II RCTs, particularly the STEMTRA trial reported by both McAllister et al 68 and Kawabori et al 69 , which included 61–63 patients with chronic motor deficits secondary to TBI. Most studies included patients with moderate to severe TBI, as classified by standard measures such as the Glasgow Outcome Scale-Extended (GOS-E) or GCS. The sample sizes varied considerably, from single case reports to larger trials with over 60 participants, reflecting the progressive development of MSC therapy from proof of concept to more advanced clinical testing.
Overview of observed study phases and design options.

Intervention details
The studies utilized various sources of MSCs and different delivery methods (summarized in Table 7). The most common cell sources were bone marrow–derived MSCs (BM-MSCs; five studies) and Wharton’s jelly–derived MSCs (two studies) as shown in Table 7. One innovative study utilized MSC-derived extracellular vesicles (MSC-EVs) rather than whole cells. The majority of studies (six out of eight) used allogeneic cells, which offers logistical advantages over autologous approaches by eliminating the need for bone marrow harvesting from injured patients (see Table 7). Three delivery routes were prominently featured: intracerebral implantation, intrathecal administration, and IV infusion. Two studies employed a combined approach using multiple routes simultaneously. The dosing regimens varied widely, from single administrations in intracerebral implantation studies to multiple doses over several months in studies using less invasive delivery methods. The SB623 cells used in the STEMTRA trial represent a unique approach, as they are bone marrow MSCs modified through transient transfection with a Notch-1 intracellular domain plasmid to enhance therapeutic potential. It is important to point out Cell Processing Innovation (SB623 Cells): SB623 cells represent an innovative approach to MSC therapy. These cells are produced by the transient transfection of bone marrow MSCs with a plasmid containing the human Notch-1 intracellular domain. This modification lowers the potential for cells to differentiate into bone, cartilage, or adipose cells while increasing their ability to secrete trophic factors, chemotactic factors, and deposit extracellular matrix proteins which may support damaged neural cells. The transfection is considered transient as the plasmid is lost rapidly during cell expansion and passaging.
Comparative MSC cell sources and delivery attributes.

Safety outcomes
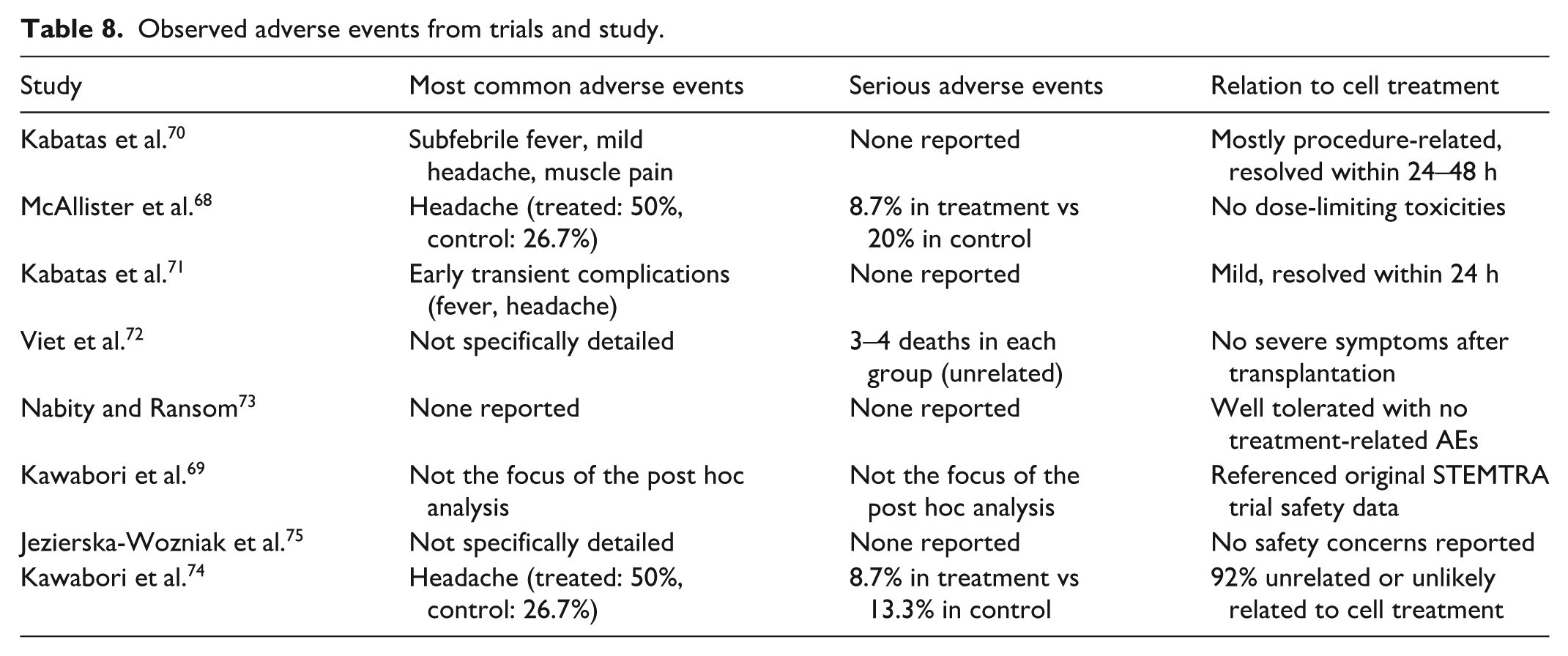
Safety profiles were generally favorable across all studies, with most adverse events being mild to moderate in severity and often related to the delivery procedure rather than the cell product itself (see Table 8): From the comparison, headache was the most reported adverse event, particularly in studies using intracerebral implantation. However, the STEMTRA trial demonstrated that the incidence of serious adverse events was lower in the treatment group (8.7%) compared to the control group (13.3%–20%), suggesting that the procedure was well-tolerated. In the STEMTRA trial, MRI examination revealed parenchymal and subdural hematomas in approximately 20% of treated patients, but most resolved without further treatment. Only one case required intervention for a hematoma, which was classified as probably related to the surgical procedure rather than the cell treatment. Notably, despite the use of allogeneic cells in most studies, immune-related adverse events were rare. In the STEMTRA trial, only 1 out of 46 treated patients (2.2%) developed antibodies to donor human leukocyte antigen (HLA) antigens, with no clinical consequences reported. This suggests that immunosuppression may not be necessary for allogeneic MSC transplantation, at least for the cell types and delivery methods used in these studies.
Observed adverse events from trials and study.
Efficacy outcomes
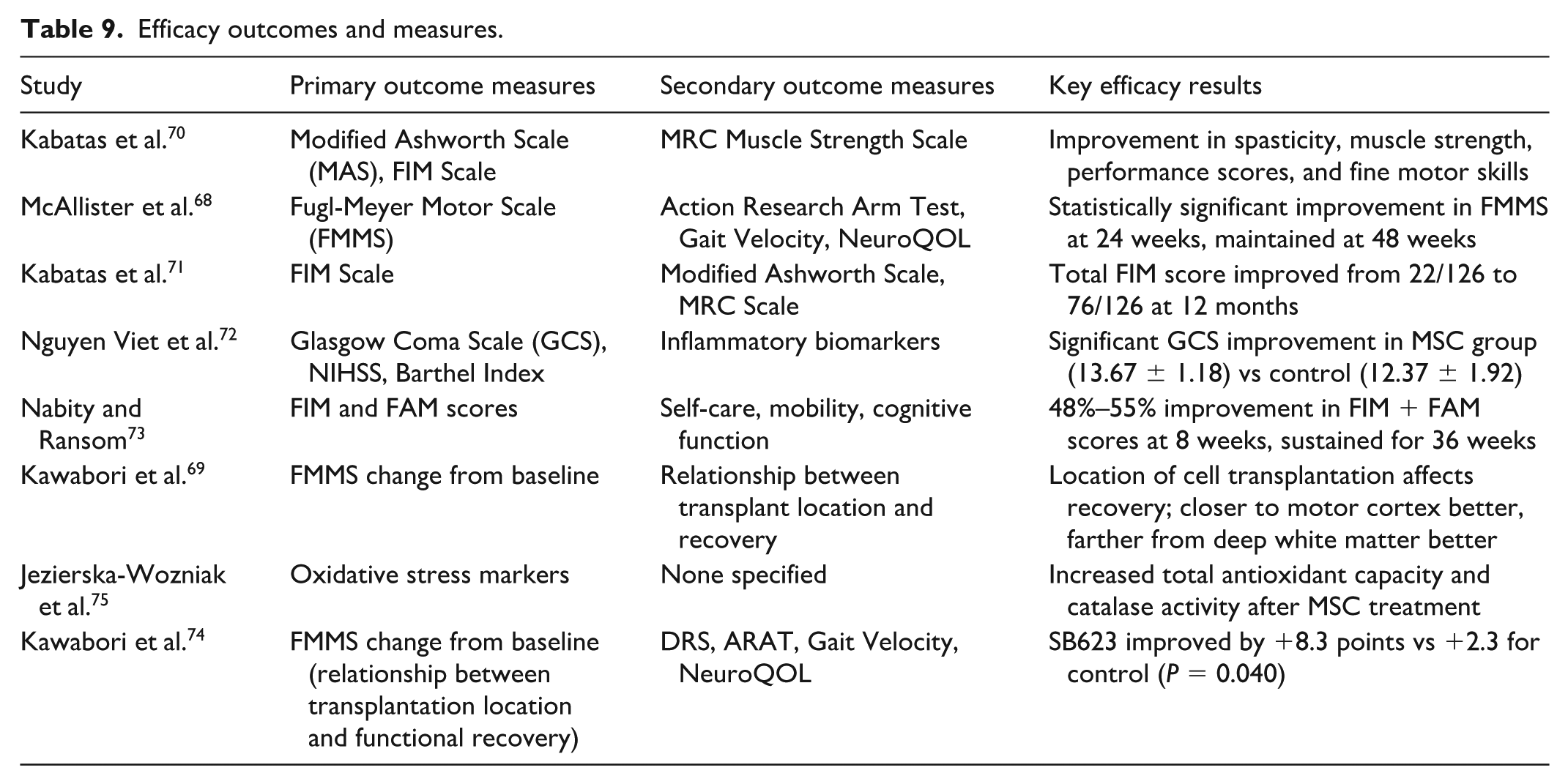
Efficacy outcomes varied across studies, with different functional scales used to assess motor, cognitive, and overall functional improvements (see Table 9). Studies assessing therapeutic outcomes frequently evaluated motor function using scales like the Fugl-Meyer Motor Scale (FMMS), Modified Ashworth Scale (MAS), and MRC Muscle Strength Scale, with the STEMTRA trial showing a significant FMMS improvement of 8.3 points in the treatment group versus 2.3 in controls (P = 0.040), and 39.1% of treated patients achieving clinically meaningful gains (≥10 points) compared to 6.7% of controls69,74. Altogether, functional status, commonly measured by the Functional Independence Measure (FIM), also demonstrated substantial improvements in various studies, including a rise from 22/126 to 76/126 over 12 months in one case and sustained 48%–55% improvements in FIM/FAM scores in another study using MSC-EVs70,71. Cognitive function improvements were noted where assessed, and one study linked MSC treatment to increased antioxidant capacity, suggesting a potential mechanism. Importantly, a post hoc analysis of STEMTRA revealed that optimal cell transplantation location varied by injury type, favoring proximity for motor cortex damage but distance for deep white matter damage, indicating the need for tailored implantation strategies. Dose–response relationship in STEMTRA trial: an interesting finding from the STEMTRA trial was the lack of a classical dose–response relationship. Patients receiving the middle dose (5.0 × 106 cells) showed the greatest improvement in motor function (+10.9 points vs +2.4 points in control, P = 0.002), with 53.3% achieving clinically meaningful improvement. The higher dose (10.0 × 106 cells) did not provide additional benefit and was associated with increased variability in response68,72,73,75. This suggests that beyond a certain threshold, the beneficial effects of additional cells may be counterbalanced by locally increased inflammation due to increased cell death or that a biological asymptote had been reached.
Efficacy outcomes and measures.
Preclinical studies using systemic/intraventricular/topical MSC administration for TBI
Data were extracted from the selected studies according to predefined categories:
Study Phase and Design: Type of study (phase I, II), study design (randomized, controlled, open-label), and patient/animal population characteristics.
Intervention Details: Cell type and source, autologous versus allogeneic, route of delivery, dosage, and frequency.
Safety Outcomes: Adverse events, complications, and safety concerns related to specific cell types or delivery routes.
Efficacy Outcomes: Functional scales used, demonstrated efficacy, correlation with specific patient groups or delivery parameters, and biomarker changes.
Mechanism of Action: Mechanisms by which MSCs exert their therapeutic effects in TBI.
The quality of the included studies was assessed based on methodological rigor, sample size adequacy, control of confounding variables, appropriate outcome measures, and comprehensive reporting of results.
Study characteristics (study phase and design)
The reviewed studies employed various TBI models to evaluate MSC therapy efficacy (see Table 10). Most of these studies utilized controlled cortical impact (CCI) or fluid percussion injury (FPI) models, which are well-established and reproducible TBI models. The studies typically evaluated moderate TBI, with Wang et al 76 being unique in investigating repetitive mild TBI. 67 All studies were conducted in rodents (rats or mice), with no studies in larger animal models59,76–83. Study durations ranged from 6 days to 9 months, with Wang et al. being the only study to evaluate long-term outcomes beyond 1 month.
TBI models and trial observations.

MSC intervention details
The studies reviewed utilized MSCs from various tissue sources. The different delivery routes for these MSCs are categorized and summarized in Table 11. MSC sources included bone marrow (BMSC), adipose tissue (AD-MSC), umbilical cord (UC-MSC/HUCPVC), and placenta (PDMSC). IV administration was the most common delivery route (7/9 studies), with one study using topical application and one using intracerebral injection81,83. Cell doses ranged from 1 to 4 million cells, with most studies administering cells within 24 h post-injury. Wang et al. 76 were notable for using a later administration time (4 days post-injury), whereas Ma et al. 83 uniquely compared normoxic and hypoxic-preconditioned MSCs, finding that hypoxic preconditioning enhanced the therapeutic effects. This suggests that MSC preparation methods may influence efficacy.
MSC utilized sources and delivery routes.

Safety outcomes
The reviewed studies reported minimal adverse effects associated with MSC administration. No studies reported tumor formation, immunological reactions, or other serious adverse events. This is consistent with the generally favorable safety profile of MSCs observed in other preclinical and clinical studies. Wang et al. 76 reported improved survival rates in MSC-treated rats compared to untreated TBI controls over a 9-month period (71% vs 62%), suggesting potential long-term safety benefits. Most studies did not systematically report adverse events, focusing primarily on efficacy outcomes. This represents a gap in the current literature, as comprehensive safety data will be essential for translation to clinical trials.
Efficacy outcomes—structural effects
Studies investigating the therapeutic effects of various MSC types following TBI reveal multifaceted benefits across several pathological domains. Regarding cerebral edema and BBB integrity, topical hypoxic-preconditioned adipose-derived MSCs (AD-MSCs) were found to decrease brain water content by downregulating matrix metalloproteinase 9 (MMP9) and normalizing aquaporin-4 (AQP4) distribution, suggesting effects on both vasogenic and cytotoxic edema 83 . IV human umbilical cord perivascular cells (HUCPVCs) reduced vascular permeability, increased the tight junction protein occludin, and preserved pericyte-endothelial interactions, while placenta-derived MSCs (PDMSCs) attenuated MMP expression, correlating with reduced BBB disruption 78 .
MSCs also impact white matter integrity; BM-MSCs attenuated white matter thinning, especially in the corpus callosum, for up to 9 months after repetitive TBI and HUCPVCs helped preserve capillary density and pericyte–endothelial interactions, protecting the neurovascular unit78,82. Furthermore, MSCs exert significant immunomodulatory effects, reducing neuroinflammation. Canine AD-MSCs inhibited microglia and astrocyte activation, promoting an anti-inflammatory M2 microglial phenotype, while BM-MSCs decreased tumor necrosis factor-alpha (TNF-α)-containing microglia long-term across multiple brain regions76,81. UC-MSCs modulated the Treg/Th17 balance toward an anti-inflammatory state, and MSCs generally protected against TBI-induced pyroptosis59,77. Finally, evidence supports roles in neuronal survival and regeneration, as intravenously administered BMSCs homed to the injury site, proliferated, and differentiated into neural-like cells expressing NeuN, MAP2, and GFAP 80 . AD-MSCs increased NeuN expression and improved the anti-apoptotic Bcl-2/Bax ratio 81 , and HUCPVCs reduced β-amyloid precursor protein accumulation, a marker of axonal injury 82 .
Efficacy outcomes—functional effects
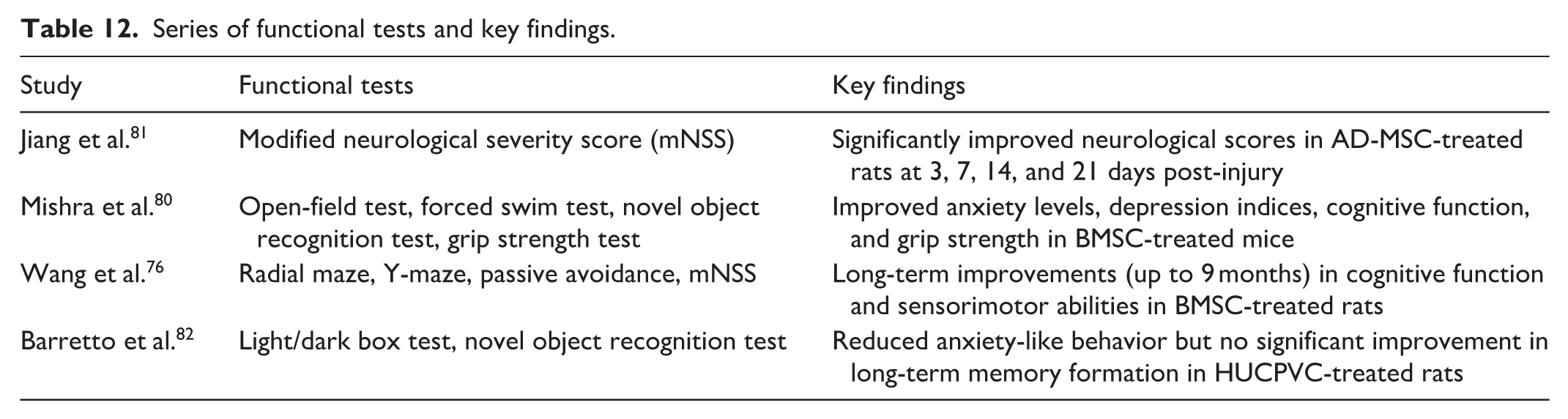
Significant improvements with MSC therapy in functional outcomes were consistently reported across all studies analyzed (see Table 12). Regarding functional outcomes, MSC therapies have shown promise in promoting recovery after TBI, though the extent and nature of improvements can vary. Wang et al 76 conducted the most extensive long-term evaluation, finding that BM-MSCs provided lasting benefits for up to 9 months post-injury. These benefits included enhancements in cognitive abilities, as measured by radial maze, Y-maze, and passive avoidance tasks, as well as improved sensorimotor function assessed via the mNSS. In contrast, Barretto et al 82 reported more specific effects with HUCPVCs, observing a reduction in anxiety-like behavior but no significant improvement in long-term memory formation, indicating potential differential impacts across functional domains. Shorter-term assessments also showed positive results; both Jiang et al. 81 , using AD-MSCs, and Kumar Mishra et al. 80 , using BM-MSCs, documented significant improvements in neurological function within a 21-day post-treatment period.
Series of functional tests and key findings.
Mechanisms of action
Based on the reviewed studies, MSCs appear to exert their therapeutic effects in TBI through multiple distinct mechanisms. A key mechanism is immunomodulation, whereby MSCs alter the inflammatory environment by encouraging microglial polarization toward the anti-inflammatory M2 phenotype 81 , decreasing the quantity of TNF-α-containing microglia 76 , adjusting the Treg/Th17 balance 59 and suppressing pyroptosis, a pro-inflammatory cell death pathway 81 . The variety of identified mechanisms strongly suggests that the therapeutic impact of MSCs results from a synergistic combination of immunomodulatory, neurotrophic, and structural protective actions, rather than relying on a singular mechanism.
Preclinical studies using modified MSCs or MSC derivatives (preconditioned, engineered, targeted, or combined with survival enhancers) for TBI
Modification strategies
To enhance the therapeutic potential of MSCs and their EVs for treating TBI, researchers employ several modification strategies aimed at improving their survival, efficacy, and delivery. Genetic modification is a frequently used approach, involving engineering MSCs or their exosomes to overexpress specific beneficial factors. Examples include enriching exosomes with miRNA clusters like miR-17-92 to promote neurogenesis and angiogenesis 84 , boosting levels of neuroprotective growth factors such as BDNF to support neuronal survival 85 , or increasing the production of anti-inflammatory cytokines like IL-10 to modulate the immune response and reduce neuroinflammation 86 . Genetic engineering can also involve silencing detrimental genes, for instance, using RNA interference to silence Rac1, which improves MSC survival and therapeutic effect 87 or implementing controlled expression systems for transient cytokine production, like IL-4, allowing for precisely timed immunomodulation post-TBI 88 . Beyond direct genetic changes, preconditioning strategies prepare MSCs for the challenging TBI environment by exposing them to specific stimuli before administration. Hypoxic preconditioning, for example, has been shown to enhance MSC differentiation into mature oligodendrocytes and improve cell survival via the mTOR/HIF-1α/VEGF pathway 89 . Similarly, exposing MSCs to media conditioned by injured cells can enhance their secreted factors, leading to improved neuroprotective effects 90 .
Temperature preconditioning, such as combining temperature-sensitive MSCs with mild hypothermia, has also demonstrated enhanced therapeutic outcomes through better cell survival and function 91 . Pharmacological pretreatment offers another avenue; treating MSCs with compounds like shikonin can regulate autophagy via the AMP-activated protein kinase (AMPK)/mammalian target of rapamycin (mTOR) pathway and decrease apoptosis, thereby improving MSC survival in the tissue surrounding the injury 92 . Modifications are also applied to the delivery systems themselves. Recent advances include engineering EVs with enhanced targeting capabilities, such as incorporating paramagnetic nanoparticles to improve guidance and retention following intranasal delivery 93 . Furthermore, incorporating MSCs or EVs into biomaterial scaffolds is being explored to provide sustained therapeutic release and improved localization directly at the injury site.
Targeted enhancements
To enhance the therapeutic potential of MSCs and their EVs for treating TBI, researchers employ several modification strategies targeting key functional improvements including survival, specific factor secretion, immunomodulation, and differentiation potential. Genetic modification is common, for example, silencing Rac1 enhances MSC survival post-transplantation by modulating apoptosis and oxidative stress susceptibility 87 . Engineering MSCs or exosomes to overexpress factors like the miR-17-92 cluster promotes neurogenesis and angiogenesis 84 , BDNF enhances neuroprotection 85 , and IL-10 targets the inflammatory cascade, promoting a regenerative immune phenotype 86 . Controlled systems for transient IL-4 release allow timed immunomodulation, while MSC treatments can also suppress neuroinflammatory ferroptosis88,94.
Preconditioning strategies also prepare MSCs, such as hypoxic preconditioning which improves survival and differentiation into oligodendrocytes, or exposure to injury-conditioned media which enhances the neuroprotective secretome89,90. Pharmacological pretreatment, using agents like shikonin, further bolsters MSC survival by regulating autophagy and reducing apoptosis 92 . Improving MSC migration, homing, and delivery is another critical focus area for enhancing therapeutic efficacy after TBI. Strategies like combining temperature-sensitive MSCs with mild hypothermia have shown promise in enhancing migration, and understanding mechanisms like the HIF-1α/SDF-1/CXCR4 axis offers potential targets for boosting recruitment to the injury site91,95. Delivery system modifications are also advancing, including engineering EVs with paramagnetic nanoparticles for better guidance and retention or using biomaterial scaffolds for sustained local release 93 . Emerging trends point toward combining these different modification approaches, an increasing focus on modified EVs as cell-free therapies, and the development of sophisticated, temporally controlled systems to better align therapeutic delivery with the dynamic pathophysiology of TBI, all aimed at maximizing the targeted enhancements for improved clinical outcomes.
Mechanisms of enhanced effect
MSCs and their derived EVs leverage enhanced mechanisms for therapeutic benefit, notably through potent anti-apoptotic actions. Studies show shikonin-treated MSCs regulate autophagy via AMPK/mTOR, while others reduce neuronal apoptosis using the HIF-1α/SDF-1/CXCR4 axis92,95. Furthermore, miR-17-92 cluster-enriched exosomes inhibit apoptosis by targeting Pten and activating PI3K/Akt signaling 84 . Immunomodulation is also enhanced; IL-10-overexpressing MSCs promote anti-inflammatory macrophage activation as does transient IL-4 expression, while MSC treatment can suppress neuroinflammation by inhibiting ferroptosis86,88,94. These modified cells boost neurogenesis and angiogenesis, with miR-17-92 exosomes enhancing neural progenitor activity and vessel formation, BDNF-overexpressing MSCs promoting plasticity via TrkB receptors, and hypoxia-preconditioned MSCs fostering oligodendrogenesis through the mTOR/HIF-1α/VEGF pathway84,85,89. Beyond these functions, modified MSCs and EVs exhibit stronger anti-oxidative effects, attenuating oxidative stress and ferroptosis by upregulating antioxidant defenses with injury-preconditioned MSCs secreting higher levels of antioxidant factors90,94. A key element unifying these improvements is enhanced paracrine signaling. Injury preconditioning boosts the MSC secretome profile, increasing the release of neurotrophic and anti-inflammatory factors 90 . Engineered EVs provide concentrated delivery of therapeutic payloads directly to injury sites, enhancing effects without needing cell survival 93 . Specifically, miR-17-92–enriched exosomes exemplify this by transferring therapeutic miRNAs to modulate target cell gene expression via improved paracrine mechanisms 84 .
Delivery methods and efficacy
MSCs and EVs have been delivered through various routes for TBI treatment, including minimally invasive IV administration,84,87 targeted but more invasive intracerebral transplantation 86 , intranasal delivery to bypass the BBB, and intraperitoneal administration 94 . Studies consistently show that these modified MSCs and EVs, such as those enriched with miR-17-92 84 , silenced for Rac1 87 , overexpressing IL-10 74 or BDNF 85 , or subjected to hypoxic preconditioning 89 , offer enhanced therapeutic efficacy compared with their unmodified counterparts, leading to improved neurological recovery, reduced inflammation, and enhanced neuroprotection. The timing of delivery is critical, with administration typically occurring within 24–48 h post-injury to target the acute phase, as early intervention can effectively suppress detrimental processes like ferroptosis88,94. Investigations into dose–response relationships suggest that higher doses of modified MSCs or EVs, such as miR-17-92 cluster-enriched exosomes or engineered EVs, generally lead to greater therapeutic benefits up to a certain threshold84,93. Furthermore, studies assessing long-term efficacy indicate that the benefits of these modified therapies can be sustained. For example, BDNF-overexpressing MSCs provided protection against cognitive impairments for at least 35 days 85 , MSC treatment attenuated persistent cognitive deficits from repetitive mild TBI 94 , and IL-10-overexpressing MSCs induced lasting changes in the inflammatory microenvironment for up to 28 days 86 , demonstrating the potential for long-lasting neuroprotective effects.
Preclinical studies using alternative MSC sources (non-BM/UC/adipose) or cross-species MSCs for TBI
The analysis focused particularly on direct comparisons between alternative MSC sources and traditional sources (BM-MSCs, UC-MSCs, AD-MSCs), unique properties of each source, insights from cross-species applications, and potential barriers to clinical translation.
Each study was systematically evaluated for the following key aspects:
Source and characteristics of MSCs used.
Animal model specifications and TBI induction method.
Therapeutic efficacy based on functional recovery assessments.
Proposed mechanisms of action.
Comparative advantages over traditional MSC sources and cross-species applications (when applicable).
Overview of MSC sources and their applications in TBI
The reviewed studies investigated a diverse range of MSC sources for TBI treatment. Table 13 provides a comparative overview of these sources and their key characteristics63,78,79,81,96,97.
MSC sources, TBI models, and findings.

Source comparison: alternative versus traditional MSC sources
The studies have directly compared alternative sources of MSCs against more established sources like bone marrow (BM-MSCs), adipose tissue (AD-MSCs), or umbilical cord (UC-MSCs), often revealing unique advantages for specific applications like TBI. For instance, Qin et al 96 investigated ectoderm-derived frontal bone MSCs (FB-MSCs), noting their enhanced neurogenic potential linked to their neural crest origin compared with traditional mesoderm-derived MSCs. Their research demonstrated that FB-MSCs were particularly effective at mitigating glutamate excitotoxicity via the FGF1 pathway, a significant mechanism in TBI pathology. Similarly highlighting the importance of developmental origin, Karim et al. examined human cranial bone–derived MSCs (CB-MSCs) and found they possessed greater neurogenic differentiation potential than AD-MSCs. The authors attributed this difference to the neural crest origin of CB-MSCs, stating, ‘CB-MSCs, derived from neural crest origin, appear to possess intrinsic neural differentiation capabilities that exceed those of mesoderm-derived MSC populations’ 63 .
Other comparisons focused on different advantages and sources. Jiang et al. (2024) reported that canine AD-MSCs offered comparable therapeutic benefits to human BM-MSCs in a rat TBI model, effectively reducing neuroinflammation and aiding functional recovery. This study emphasized the practical benefits of canine AD-MSCs, such as easier access and larger harvest volumes, and noted their efficacy even in a xenogeneic context without significant immune rejection 81 . Meanwhile, Yang et al 78 compared PDMSCs with BM-MSCs, finding that PDMSCs exhibited superior capabilities in diminishing matrix metalloproteinase (MMP) activity and oxidative stress following TBI. They observed, ‘PDMSCs demonstrated enhanced anti-inflammatory and antioxidant properties compared to BM-MSCs in our model, potentially due to their unique secretome profile’, suggesting another promising alternative MSC source with distinct therapeutic mechanisms.
Unique properties of alternative MSC sources
The reviewed studies revealed several unique properties and mechanisms specific to alternative MSC sources that may confer advantages in treating TBI. MSCs originating from the neural crest, such as FB-MSCs and CB-MSCs, appear to possess enhanced neurogenic potential compared with traditional mesoderm-derived cells. Specifically, FB-MSCs were shown by Qin et al 96 to express higher levels of FGF1, which helped alleviate glutamate excitotoxicity by regulating glutamate transporters EAAT1 and EAAT2, thereby reducing neuronal damage. Similarly, Karim et al 63 noted that CB-MSCs expressed higher levels of neural markers even prior to differentiation, suggesting an intrinsic neural predisposition. Another distinct source, limbal MSCs (LMSCs), demonstrated superior secretion of neurotrophic factors like BDNF and GDNF compared to BM-MSCs, which Derakhshani et al. 97 suggested could explain their enhanced efficacy in cortical lesions; these cells also showed a notable ability to integrate into injured tissue and aid neural circuit reorganization. Furthermore, PDMSCs exhibited unique capabilities, particularly in inhibiting MMPs (MMP-2 and MMP-9) crucial for BBB integrity post-TBI, primarily through TIMP-1 upregulation, a mechanism less pronounced in BM-MSCs—and also showed enhanced antioxidant effects by activating Nrf2/HO-1 pathways 78 .
Cross-species findings
Several studies utilizing xenogeneic transplantation, despite inherent immunological differences between species, have provided important insights into MSC therapy for TBI. Jiang et al. 81 demonstrated that canine AD-MSCs transplanted into a rat TBI model yielded significant therapeutic benefits even without immunosuppression, experiencing minimal immune rejection, which led the authors to propose that ‘the immunomodulatory properties of MSCs transcend species barriers,’ highlighting potential feasibility for cross-species veterinary applications. Echoing the finding of cross-species efficacy, Ruppert et al. 79 observed that human AD-MSCs were effective in treating both acute and sub-acute TBIs in rats; they concluded that the human cells survived and exerted their effects primarily through ‘paracrine mechanisms rather than direct cellular integration or replacement,’ indicating that secreted factors likely possess cross-species activity. Furthermore, Karim et al. 63 found that hcMSCs were not rejected in a mouse TBI model and demonstrated that combining this xenogeneic cell therapy with high-frequency exercise significantly improved motor recovery, suggesting that ‘mechanical stimulation may enhance the integration and therapeutic potential of xenogeneic MSCs’. These cross-species findings suggest the following:
MSCs possess intrinsic immunomodulatory properties that enable them to survive in xenogeneic hosts without extensive immunosuppression.
The therapeutic mechanisms of MSCs (particularly paracrine effects) appear to function across species barriers.
The hMSCs can be effectively studied in animal models, potentially accelerating translation to clinical applications.
Mechanisms of action
The reviewed studies highlighted several key mechanisms driving the therapeutic effects of alternative MSC sources in TBI models. A primary and consistently reported mechanism across different MSC types was their anti-inflammatory action. For instance, Jiang et al. 81 showed that canine AD-MSCs reduced pro-inflammatory cytokines like IL-1β and TNF-α while boosting anti-inflammatory IL-10 levels in the injured brain. Similarly, Yang et al. 78 found that PDMSCs suppressed neuroinflammation partly by shifting microglial polarization toward an anti-inflammatory M2 phenotype.
Complementing inflammation control, neurotrophic support was highlighted by Derakhshani et al. 97 , who emphasized that LMSCs promoted neural repair through enhanced secretion of factors like BDNF, GDNF, and NGF, which correlated positively with improved behavioral outcomes post-transplantation. Beyond these broader effects, specific MSC types demonstrated unique mechanisms targeting distinct TBI pathologies. Qin et al. 96 identified that FB-MSCs, owing to their neural crest origin, specifically countered glutamate excitotoxicity; they stated, ‘FB-MSCs significantly increased the expression of EAAT1 and EAAT2 in astrocytes surrounding the injury site, facilitating glutamate clearance and reducing excitotoxic damage,’ an effect mediated by FGF1. Addressing vascular damage, Yang et al. 78 uncovered a novel role for PDMSCs in preserving BBB integrity by inhibiting MMPs, noting, ‘PDMSCs treatment significantly reduced MMP-9 expression and activity in the perilesional cortex, which correlated with reduced BBB permeability and edema formation’. Last, Karim et al. 63 presented a unique finding where the efficacy of human CB-MSCs was significantly boosted by concurrent high-frequency exercise, proposing that exercise may create a more supportive microenvironment for MSC survival and function through mechanisms like increased blood flow and growth factor upregulation.
Clinical trial protocols for MSC therapy in TBI
The MATRIx trial, an adaptive phase II study, treats severe TBI patients (GCS ≤8) within 48 h, randomizing them initially to 80 or 160 million bone marrow MSCs or placebo to determine an optimal dose for further testing, using MRI and biomarkers over 12 months 98 . In contrast, the phase III TRAUMACELL trial enrolls unresponsive severe TBI patients (Glasgow <12) approximately 15 days post-injury 99 . Participants receive three weekly IV injections of either 2 million/kg Wharton’s jelly umbilical cord MSCs or placebo. Assessments include positron emission tomography–magnetic resonance imaging (PET-MRI) (with TSPO genotyping), comprehensive biomarkers, and clinical evaluations up to 12–14 months post-treatment. These trials highlight key differences in administration timing (acute vs sub-acute), cell source, dosing strategies, and assessment tools. Both MSC protocols employ scientifically distinct primary outcome measures to assess efficacy. Table 14 provides a comparative observation of the MATRIx and TRAUMACELL trials alongside their respective outcome categories. The MATRIx trial utilizes plasma neurofilament light (NfL) at 14 days as its primary endpoint to measure axonal damage, supported by evidence of NfL’s progressive increase over the first 2 weeks and correlation with clinical outcomes, analyzing results via Student’s t-test on the logarithmic scale while monitoring serious adverse drug reactions for safety 98 . The TRAUMACELL trial measures [18F]-DPA-714 signal intensity 99 , in corpus callosum via dynamic PET-MRI at 6 months as its primary endpoint, reflecting post-traumatic neuroinflammation in a region showing homogeneous patterns of injury in 75%–90% of TBI patients, with statistical comparison of standard uptake value ratios between treatment arms and comprehensive secondary outcomes including neuroinflammation in multiple brain regions, white matter integrity markers, neurological assessments, treatment feasibility, and biomarker predictors of therapeutic success.
Comparative overview of the MATRIx and the TRAUMACELL trials.

In vitro/ex vivo studies related to MSC therapy for TBI
The analyzed studies span a range of in vitro and ex vivo models, including cell culture systems, organotypic brain slices, and biomaterial-based approaches, providing a comprehensive overview of current research in this field.
Model systems used in MSC-TBI research
The reviewed studies employed diverse model systems to investigate the therapeutic potential of MSCs in TBI. These models ranged from simple cell culture systems to more complex organotypic slice cultures, each with varying degrees of physiological relevance to human TBI pathology. Table 15 provides a comparative overview of the model systems used and their relevance to TBI.
Model system explored and their relevance to TBI pathology.

The model systems employed in these studies represent a spectrum of complexity and physiological relevance13,66,100,101,103,104. At the simpler end, Hu et al. 100 used an oxidative stress model in MSCs themselves to evaluate how genetic modification might enhance their therapeutic potentials. More complex models included co-culture system that incorporated interactions between modified MSCs and neural or glial cell lines. The most sophisticated model was the organotypic cortical slice culture with CCI described by Pischiutta et al. 104 , which preserved the 3D architecture and cellular diversity of the brain tissue while allowing for a biomechanical injury closely resembling clinical TBI.
Mechanism focus of MSC-TBI studies
The studies investigated various cellular and molecular mechanisms underlying the therapeutic effects of MSCs in TBI. These mechanisms can be broadly categorized into antioxidative, anti-inflammatory, angiogenic, neuroprotective, and neuroregenerative pathways. Research has explored enhancing MSCs’ intrinsic antioxidative properties via the Keap1-Nrf2 pathway and the effects of Nrf2-overexpressing MSCs on neural cells and inflammation through pathways like PI3K/AKT and STAT6/AMPKα/SMAD3100,103. Others have focused on the angiogenic potential influenced by culture conditions for MSC-EVs, examining effects on endothelial cells and factors like VEGF-A, or the neurotrophic and metabolic benefits of the secretome from induced pluripotent stem cell (iPSC)-derived MSCs on neuronal survival, function, and energy metabolism101,102. These studies increasingly suggest that the paracrine effects mediated by MSC secretomes and EVs are primary drivers of therapeutic benefits, rather than direct cell replacement. However, the potential for cell replacement was explored by Jana et al. 66 , who demonstrated the transdifferentiation of MSCs into electrophysiologically functional neurons using peptide hydrogels and neural differentiators. In addition, studies like Pischiutta et al. 104 assessed neuroprotection by examining neuronal damage markers and the modulation of microglial and astrocyte responses. Collectively, these investigations highlight MSCs’ multifaceted ability to counteract various TBI pathologies, including oxidative stress, inflammation, neuronal death, and vascular dysfunction, through a range of cellular and molecular interactions.
MSC products and delivery methods
The reviewed studies utilized various MSC products and delivery methods to target TBI pathology. These ranged from whole cells (with or without genetic modifications) to cell-free approaches using secretome components or EVs, as detailed in Table 16: MSC products, sources, and delivery methods.
MSC products, sources, and delivery methods.

Some studies employed genetically modified MSCs, such as Hu et al. 100 using Keap1 knockout MSCs to boost antioxidative capacity and Pischiutta et al. 104 using Nrf2-overexpressing MSCs, representing a strategy to enhance specific intrinsic therapeutic properties. A significant portion of research focused on cell-free approaches; Phelps et al. 101 , Kawatani et al. 102 , and Pischiutta et al. 104 utilized MSC-EVs or the complete secretome, which offers benefits like reduced immunogenicity, improved storage and handling, and potentially better quality control and dosing precision. Notably, Phelps et al. 101 highlighted manufacturing considerations, showing how bioreactor conditions affect the angiogenic potential of MSC-EVs. An innovative delivery strategy was demonstrated by Jana et al. 66 , who combined MSCs with an SLNAP and a neural differentiator, aiming to create a supportive microenvironment for enhanced cell survival and integration via minimally invasive injection. Furthermore, the studies sourced MSCs diversely from adipose tissue, umbilical cord, and olfactory mucosa, reflecting the multiple available tissue origins; the use of iPSC-derived MSCs by Kawatani et al. 102 is particularly noteworthy as it offers a pathway toward a potentially limitless and standardized source for therapeutic applications.
Mitochondrial transplantation from MSCs for TBI
The methodology used in the research by Bamshad et al. 105 involved isolating mitochondria from hUC-MSCs through centrifugation, followed by their delivery into the right lateral ventricle of TBI rat models via stereotaxic injection. Successful internalization of these transplanted mitochondria into brain cells was confirmed using MitoTracker Red CMXRos labeling and fluorescence microscopy. The proposed mechanism addresses a core pathology of TBI: the provision of healthy mitochondria counteracts ATP deficiency and reactive oxygen species (ROS) accumulation characteristic of brain trauma. This directly rescues neuronal cells, reduces apoptosis, and alleviates astrogliosis and microglial activation. Efficacy was demonstrated through multiple outcome measures: significant improvements in motor function (assessed via beam walk, grid walk, horizontal bars, and cylinder tests), reduction in apoptotic brain cells, decreased levels of inflammatory markers Iba1 and GFAP, preservation of brain tissue architecture, and restoration of serum creatine phosphokinase (CPK) levels 105 .
Preclinical safety/validation studies of MSC therapy for TBI
The study by Wang et al. 106 evaluated immunogenicity by checking for human leukocyte antigen II (HLAII) expression and measuring serum pro- and anti-inflammatory cytokine levels. Tumorigenicity was assessed through histological examination of major organs and the brain, checking for abnormal cell proliferation, and using specific markers like EGFRvIII and PCNA. Biodistribution was tracked using in vivo imaging and immunofluorescence staining for markers like CD29. The study used allogeneic human umbilical cord mesenchymal stem cells (huMSCs) obtained from human amniotic membrane and umbilical cord vascular tissue. Safety was evaluated using two delivery methods: direct stereotactic injection into the brain injury site (in situ) and IV injection via the tail vein. Safety monitoring, including short-term assessments up to 28 days and long-term observation for tumorigenicity up to 12 months, assessed immunogenicity (HLAII expression, cytokine levels), tumorigenicity (histology, EGFRvIII/PCNA markers), and biodistribution (imaging, CD29 staining). Key findings indicated a good safety profile for TBI treatment, with no significant immunogenicity issues; huMSCs showed negligible HLAII expression and positively modulated cytokine levels 106 .
Discussion
Research into MSC therapies for TBI reveals a dynamic field exploring diverse approaches, from traditional cell transplantation to cell-free and bioengineered methods. Across these varied strategies, a consistent set of therapeutic mechanisms emerges, directly addressing the second objective of this review. This systematic analysis confirms that MSCs and their derivatives effectively modulate the immune response, shifting the post-injury environment toward neuroprotection by promoting anti-inflammatory M2 microglia and regulating T-cell activity59,81. They also provide neuroprotection by reducing neuronal death, glutamate excitotoxicity, and oxidative stress96,104. Furthermore, MSC therapies help maintain BBB integrity by regulating factors like MMPs and support the brain’s limited regenerative capacity by enhancing neurogenesis and axonal growth31,78,80,83.
A central finding of this review, fulfilling our first research objective, is the documentation of a distinct paradigm shift in therapeutic strategy over the last decade. Early approaches focused on conventional cell transplantation, based on the hope of cell replacement. However, this analysis charts a clear progression toward more advanced and targeted modalities. This includes the integration of MSCs with biomaterial scaffolds to create supportive microenvironments for localized delivery, alongside the genetic modification and preconditioning of cells to enhance the potency of their secretomes. Concurrently, the field is undergoing a significant evolution, moving from a focus on cell replacement to a deeper understanding of paracrine mechanisms. This has fueled a major trend toward cell-free products like exosomes, which offer potential advantages in safety, scalability, and manufacturing standardization. This shift, from using the cell as a ‘bioreactor’ to isolating the therapeutic ‘product’ itself, represents a maturation in the field’s mechanistic understanding.
Despite promising preclinical results, translating MSC therapies into clinical practice faces significant hurdles, even as early trials like STEMTRA show acceptable safety and potential efficacy68,74. Key challenges include determining the optimal timing for intervention, as different studies suggest varying therapeutic windows98,99,106. The findings also highlight the difficulty in establishing effective dose–response relationships, which can be non-linear 68 , as demonstrated by the STEMTRA trial where a medium dose proved more effective than a high dose. This complexity, along with the need for tailoring treatments to individual patient injury characteristics and confirming long-term safety, underscores the substantial work still required for successful clinical translation 69 .
A critical barrier to this translation, which this review brings into sharp focus, is the pronounced heterogeneity in the neurological recovery parameters used to evaluate therapeutic efficacy. Across the dozens of preclinical studies analyzed, outcome measures ranged from broad behavioral scoring systems (e.g. mNSS, NDS) to highly specific tests of motor function (e.g. beam walk, grip strength) and cognition (e.g. Morris water maze). The clinical landscape is similarly fragmented, utilizing scales like the FMMS for motor deficits, the FIM for daily function, and various cognitive assessments. This variability severely impedes cross-study comparison and meta-analysis, making it difficult to establish a consensus on the true magnitude of therapeutic effects. There is no clear agreement on which parameters are the most reliable or clinically meaningful, representing a critical gap between promising preclinical data and definitive clinical validation.
While the evidence strongly documents a profound technological shift in TBI therapeutics, the strength of the evidence supporting definitive clinical benefits remains constrained. Consistent with our comprehensive methodological quality assessment (which revealed a high proportion of ‘Some Concerns’ or ‘High Risk’ in preclinical studies), the overall strength of evidence for key outcomes is consistently low. The strength of evidence for Preclinical Mechanisms (Cell-Free Secretome/EVs, etc.) is rated Very Low, primarily due to the high methodological risk-of-bias in animal studies, pervasive heterogeneity in models, and the high degree of translational indirectness61,79. The strength of evidence for Clinical Functional Recovery is rated Low, limited by small patient sample sizes in efficacy trials, profound heterogeneity in outcome measures, and the complex, non-linear nature of dose–response relationships 68 . Finally, the strength of evidence for Long-Term Safety is rated Moderate at best, constrained by a clear deficiency in long-term follow-up data69,96. To bridge this gap, future research must move beyond isolated feasibility studies. We propose the urgent need for trials focused on (1) defining optimal timing windows and comparative routes (e.g. intranasal EVs vs intracerebral whole cell) in large animal models; (2) performing rigorous dose-finding studies for cell-free products to establish the therapeutic index; and (3) designing clinical trials that incorporate functional biomarkers to stratify patients by injury phenotype and measure true responder populations.
This comprehensive, decade-long review serves to synthesize and contextualize the findings of previous, more narrowly focused research. While foundational reviews by Hasan et al. 11 and Das et al. 20 established the primary paracrine roles of MSCs, this work provides a crucial update by systematically documenting the subsequent technological evolution that has leveraged these very mechanisms. More specialized reviews on the secretome by Yudha et al. 21 and on the exosomes by Liu et al. 22 offer essential deep dives into cell-free mechanisms; our review complements theirs by placing these innovations within a broader comparative framework that includes whole-cell and bioengineered therapies. Similarly, while the network meta-analysis by Yang et al. 24 provides a powerful quantitative comparison of different EV sources in preclinical models, our systematic approach offers a qualitative synthesis across the full spectrum of therapeutic modalities, including clinical trial progress, thereby addressing a wider translational scope.
Ultimately, by systematically charting a decade of research, this review addresses our third objective by identifying the critical, overarching gaps that must be bridged. First, the lack of standardization in MSC manufacturing, characterization, dosing, and particularly outcome measures remain the single greatest obstacle. Second, there is a clear deficiency in long-term safety and efficacy data, with most preclinical studies limited to short follow-up periods. Finally, significant hurdles in translational considerations persist, including an over-reliance on rodent models that may not accurately predict human responses. Addressing these challenges through multicenter collaborations to establish consensus on standards, prioritizing long-term and large animal studies, and designing stratified clinical trials will be essential to finally translate the immense promise of MSC-based therapies into a viable treatment for patients with TBI. With respect to the Risk of Bias and Quality Appraisal Results, methodological quality was assessed using a multi-tool approach tailored to study design (Cochrane, SYRCLE, JBI/NOS). For the Clinical Trials (Cochrane RoB, Fig. A1/A2), the risk was generally Low. In contrast, the extensive body of Preclinical In Vivo Studies (SYRCLE, Fig. A3/A4) showed an overall judgment dominated by ‘Some Concerns’ and ‘High Risk’. This finding was primarily driven by major reporting deficiencies in Blinding of Caregivers/Investigators and Random Housing, which were overwhelmingly rated as Unclear or High Risk. The non-randomized human studies were rated as Good/Fair quality (JBI/NOS), confirming transparent reporting of intervention and outcomes but not mitigating the overall low strength of evidence from the preclinical tier.
Trends, gaps, and future studies
The therapeutic landscape for MSC therapy in TBI is rapidly evolving toward more refined, targeted, and integrated strategies. A major trend involves a shift from whole MSCs to cell-free therapeutics, such as EVs, exosomes, and secretomes, which demonstrate comparable or superior efficacy in reducing inflammation and offer manufacturing scalability and improved safety profiles by avoiding risks like tumorigenicity43,90. Simultaneously, research is focusing on precision enhancement of MSC products through strategies like genetic engineering to enrich exosomes with specific miRNAs for neurogenesis, MSC preconditioning (e.g. via hypoxia) for more potent secretomes, and the selection of cells from optimal developmental origins like neural crest sources46,84,96. Furthermore, there is growing emphasis on the integration of MSC therapies with advanced delivery systems, utilizing biomaterial scaffolds (hydrogels, PLGA) for supportive, localized, and sustained release, alongside minimally invasive approaches like injectable hydrogels and intranasal delivery66,93. Finally, combination therapeutic approaches are emerging, involving the pairing of MSCs with physical therapies (like exercise), pharmacological agents (e.g. GM1 ganglioside), or complementary cell types (like Tregs) to achieve synergistic effects and enhance therapeutic outcomes.
Despite these technological advances, critical research gaps significantly hinder the clinical translation of MSC therapies for TBI. A major hurdle is the pervasive lack of standardization and comparability, evidenced by inconsistencies in MSC characterization, manufacturing methods for derivatives, and heterogeneous outcome measures across studies, which impede reproducibility and consensus-building. This challenge is compounded by severe deficiencies in long-term safety and efficacy data, as most preclinical studies have short follow-up periods (unlike Wang et al., 2024), necessitating more thorough investigation into long-term immune responses and potential off-target effects. Translational hurdles also persist due to the field’s heavy reliance on rodent models, which lack sufficient validation in large animals, and a limited understanding of how patient-specific factors (e.g. age, injury severity) and common TBI comorbidities impact outcomes. Last, a critical incomplete mechanistic understanding remains, making it unclear which specific molecular components within MSC derivatives mediate therapeutic effects and how their mechanisms evolve over time post-injury, demanding further clarification.
To accelerate clinical translation, future studies must be strategically designed to directly address these identified gaps by leveraging the current emerging trends. Research should prioritize standardization efforts through multicenter collaborations to harmonize protocols for MSC manufacturing and outcome assessment. Methodologically, future work should prioritize long-term safety and efficacy investigations in clinically relevant large animal models to validate findings before advancing to human trials. Mechanistic studies must focus on isolating and defining the specific molecular cargo (e.g. miRNAs, proteins) within the next-generation exosomes and secretomes derived from modified or preconditioned MSCs. Finally, the development of optimized and personalized combination therapies, integrated with advanced, controlled delivery systems, should be a priority to maximize therapeutic effect and bring MSC-based interventions to TBI patients more quickly.
Limitations
Despite its comprehensive scope, this review is constrained by several limitations within the existing literature that, in turn, illuminate a clear path for future research.
A primary constraint concerns the scope of our literature search. Our systematic search was restricted to three major scientific databases (Web of Science, Embase, and PubMed/Medline). We acknowledge that excluding other comprehensive platforms, such as Scopus or the Cochrane Library, means that certain relevant studies may not have been captured, potentially impacting the comprehensiveness of our evidence base. While a broader search could further strengthen the evidence, it was not feasible within the scope of this review. Future systematic reviews should aim to expand their search strategies to include these additional key sources.
Furthermore, we acknowledge a critical flaw in the search strategy itself, as key therapeutic terms, such as ‘exosomes,’ ‘EVs,’ and ‘Extracellular vesicle,’ were omitted from the Boolean search strings. These two limitations mean that certain relevant studies were likely not captured, potentially impacting the comprehensiveness of our evidence base. Future systematic reviews should aim to expand their search strategies to include these additional key sources and ensure the comprehensive inclusion of all relevant product-specific keywords.
Beyond our search scope, significant limitations exist within the primary studies. The prevalence of short-term preclinical studies, often with follow-ups under 1 month, leaves the long-term efficacy and safety of MSC therapies uncertain. This issue is compounded by a major translational gap, as the field’s over-reliance on rodent models may not accurately predict the complex pathophysiology and therapeutic response in human TBI. Therefore, future research must prioritize long-term investigations in clinically relevant large animal models to generate more translatable data before advancing to larger human trials.
Finally, significant mechanistic and clinical questions remain. While paracrine action is established, the specific bioactive molecules driving therapeutic effects are poorly defined. This mechanistic uncertainty directly impedes progress in patient stratification, as most studies have not adequately examined how factors like injury severity, age, or comorbidities impact outcomes. Future work should leverage multi-omics approaches to identify these key therapeutic agents and enable the development of precisely engineered, next-generation cell-free products. This must be paired with biomarker-driven clinical trials designed to stratify patient populations, a crucial step toward personalized medicine for TBI.
Conclusion
The past decade has been marked by a definitive evolution in MSC therapy for TBI, representing a critical shift from cell replacement concepts to sophisticated, paracrine-based interventions. Our systematic analysis reveals that this trajectory from conventional whole-cell strategies to engineered cell-free products signifies a fundamental maturation of the field. The focus has moved beyond a regenerative–replacement paradigm to one centered on the precise, mechanistic modulation of the post-injury microenvironment. This progress, culminating in promising early clinical data, confirms that MSC-based approaches hold tangible potential.
However, our comprehensive analysis reveals a critical disconnect: this rapid technological advancement has outpaced the development of standardized methodologies. The profound heterogeneity in MSC manufacturing, dosing, and particularly in the clinical and preclinical outcome measures used to define ‘success’ remains the most significant barrier to meaningful cross-study comparison and effective clinical translation. Without consensus on what to measure and how to measure it, the true comparative efficacy of these innovative therapies remains obscured, hindering progress.
Therefore, the future of MSC-based interventions for TBI depends not only on refining the promising cell-free and bioengineered platforms identified in this review but, more urgently, on a concerted, community-wide effort to establish and adopt standardized protocols and clinically meaningful, validated endpoints. Overcoming these systemic hurdles is the critical next step. Only by aligning methodological rigor with technological innovation can the field finally translate a decade of scientific evolution into a tangible therapeutic reality for patients suffering from TBI.
Supplemental Material
sj-docx-1-cll-10.1177_09636897261417625 – Supplemental material for Evolution of mesenchymal stem cell therapies for traumatic brain injury: A decade of advances, mechanisms, and translational prospects
Supplemental material, sj-docx-1-cll-10.1177_09636897261417625 for Evolution of mesenchymal stem cell therapies for traumatic brain injury: A decade of advances, mechanisms, and translational prospects by Peining Zhu and Yufei Gao in Cell Transplantation
Footnotes
Acknowledgements
Not applicable.
Ethical Considerations
This manuscript is a review article based on previously published, peer-reviewed studies. No new primary data involving human participants or animals were collected for this review. The authors relied on the ethical approvals and patient consent statements obtained by the original investigators for each study included in this review. All source materials are appropriately cited. Therefore, direct ethical approval for this specific review manuscript was not required.
Consent to Participate
Not applicable.
Author Contributions
Peining Zhu: Conceptualization, Methodology design, Formal Analysis (Quality Assessment), Visualization (Figure/Table Creation), Writing – Original Draft Preparation, Writing – Review & Editing, and Supervision.
Yufei Gao: Data Curation and Acquisition (Literature Search and Screening), Formal Analysis (Data Extraction), Validation of Results, and Writing – Review & Editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data supporting the findings of this systematic review are either included within the article and its supplementary information files, or they are fully available from the cited primary literature.
Supplemental Material
Supplemental material for this article is available online.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Trial registration Number/Date
Not applicable.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
