Abstract
Oral cancer ranks among the most prevalent malignant tumors of the head and neck, comprising 1%–2% of all global malignancies. Despite advancements in surgery, radiotherapy, chemotherapy, and immunotherapy, challenges remain in oral cancer treatment, including high recurrence rates, severe side effects, and limited efficacy. Hydrogels, biocompatible materials with a three-dimensional network structure, have emerged as promising candidates in oral cancer therapy due to their dual roles in drug delivery and immune modulation. This review examines the contribution of hydrogels to oral cancer treatment through their synergistic control of drug release and localized immune activation. By enabling targeted, stimuli-responsive drug release, and enhancing local immune responses, hydrogels reduce systemic toxicity and improve treatment precision. When integrated with advanced approaches such as gene therapy and immune modulation, hydrogels hold potential to further amplify therapeutic efficacy. Although preclinical results are promising, clinical translation requires addressing scalability, quality control, and long-term safety concerns. With ongoing development, hydrogels are positioned to become vital tools in the treatment of oral cancer, periodontitis, and other oral diseases.

Introduction
Oral cancer, encompassing malignancies of the oral cavity and oropharynx, remains a significant global health challenge1,2. Epidemiological trends are shifting, with human papillomavirus (HPV) now contributing to a marked rise in oropharyngeal cancer incidence, especially in middle-aged and older populations, alongside traditional risk factors such as tobacco and alcohol use3,4. In high-risk populations, organized screening through clinical oral examination has proven effective, significantly reducing mortality 5 . However, the often subtle and asymptomatic nature of early oral cancer results in many patients being diagnosed at advanced stages 6 . Late diagnosis diminishes treatment efficacy and leads to poorer survival outcomes, highlighting the urgent need for more sensitive early detection methods.
Standard therapeutic approaches for oral cancer, such as surgical resection, radiotherapy, chemotherapy, targeted therapy, and immunotherapy7–11, have evolved considerably. However, these treatments are frequently accompanied by notable limitations that challenge their efficacy and tolerability 12 . For example, while surgical resection effectively removes tumor tissue, it often results in functional impairments, such as difficulties with speech and eating 13 . Although radiotherapy and chemotherapy can control disease progression, they are associated with severe toxicities that significantly affect patients’ quality of life. Furthermore, even when initial tumor control is achieved, the risk of local recurrence and distant metastasis remains high, necessitating rigorous post-treatment surveillance 14 . A central challenge in oral cancer management is balancing therapeutic efficacy with the mitigation of adverse effects, which drives ongoing research and innovation in the field 12 .
Hydrogels, three-dimensional polymeric networks known for their biocompatibility, flexibility, and stimuli-responsiveness (e.g. to pH, temperature, or light), offer precise control over drug release. These properties make hydrogels versatile platforms for biomedical applications, including tissue engineering, wound healing, and cancer therapy15,16. In oncology, hydrogels not only enable targeted drug delivery with reduced systemic toxicity but also function as protective barriers to support tissue regeneration 17 . When integrated with advanced therapies like immunotherapy and gene therapy, hydrogels are emerging as transformative tools for developing more precise and effective oral cancer treatments18,19 (Fig. 1).
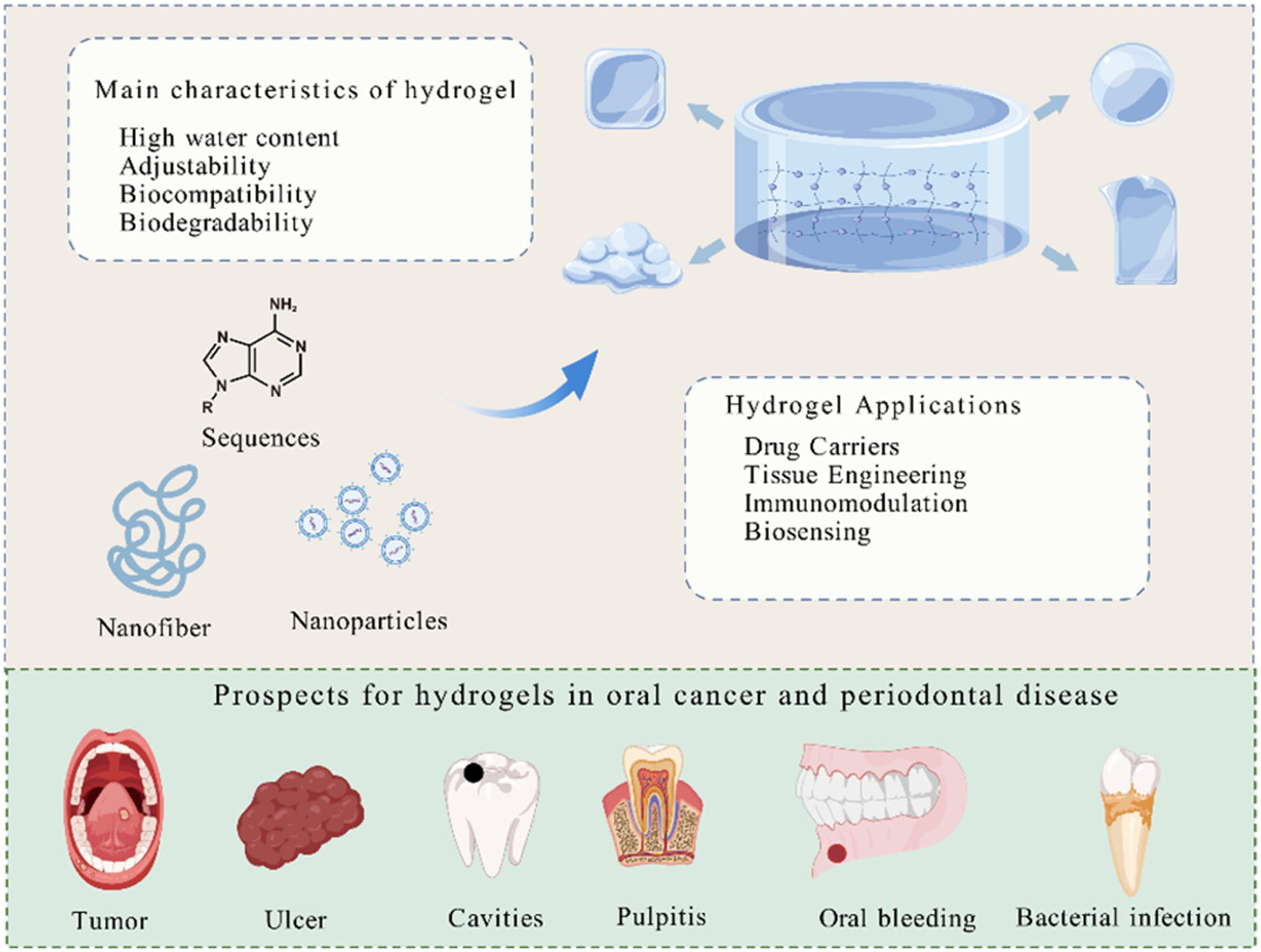
Characteristics, applications, and prospects of hydrogels in oral cancer and periodontal disease. Hydrogels, with high water content, tunability, biocompatibility, and biodegradability, show great promise in diverse applications, including drug delivery, tissue engineering, immunomodulation, and biosensing. They hold significant therapeutic potential in treating both oral cancer and periodontal disease, targeting conditions ranging from tumors to infections. Figure created with Adobe Illustrator 2020.
Fundamental research on hydrogels
Fundamental research on hydrogels provides a solid scientific foundation for their widespread application in the biomedical field. As polymeric materials with a three-dimensional network structure, hydrogels are characterized by their high water absorbency, biocompatibility, biodegradability, and mechanical flexibility (Fig. 2), making them highly attractive for various medical applications 20 .

Hydrogel properties and biomedical applications. Hydrogels possess unique properties such as high absorbency, degradability, and elasticity, enabling applications in wound healing, tissue repair, drug delivery, and safe absorption or excretion after treatment, such as their versatility in clinical use. Figure created with Adobe Illustrator 2020.
Physicochemical properties of hydrogels
Hydrogels are highly valued in medicine for their remarkable water absorbency 21 , forming a hydrated 3D network that closely mimics the moist environment of human tissues, particularly in areas like the oral cavity and skin, where natural moisture is crucial 22 . This moisture-rich environment supports tissue repair, cell growth, and wound healing. In post-surgical applications for oral cancer, hydrogels effectively prevent drying of the surgical site and grafts, reducing the risk of shrinkage in both normal and grafted tissues 23 . Their high water content also facilitates sustained drug release, enhancing therapeutic outcomes. For instance, Wang et al. developed a supramolecular hydrogel (2-FA) with excellent biocompatibility and antibacterial activity. In a rat tooth extraction model, the 2-FA hydrogel demonstrated a faster degradation rate, less osteoclast activity, and reduced inflammatory infiltration compared to natural healing and commercial hemostatic agents (e.g. gelatin sponges), promoting bone healing without disrupting normal oral functions such as speech and swallowing 24 . From a mechanical standpoint, the flexibility and elasticity of hydrogels provide superior compatibility with various soft tissues. Many human tissues (e.g. skin, muscle, oral mucosa) are soft and elastic, and biomedical materials must match these mechanical properties to avoid discomfort during treatment 25 . Hydrogels, with their soft and elastic nature, conform to tissue movement, minimizing the discomfort typically associated with rigid materials. In oral cancer care, where bleeding is common, this adaptable adherence reduces irritation, promotes healing, and aids in hemostasis, thereby supporting patient recovery 26 .
The degradability of hydrogels represents another significant advantage in biomedical applications. Unlike traditional implants, hydrogels can be naturally absorbed or eliminated through biodegradation, preventing the long-term accumulation of residual materials in the body 27 . This property is particularly advantageous for short-term treatments, such as localized drug delivery and wound healing following oral cancer surgery, where hydrogels can eliminate the need for a second procedure to remove the material, thus reducing surgical burden and infection risk 28 . Additionally, the degradation rate of hydrogels can be chemically adjusted to align with the treatment timeline, optimizing therapeutic efficacy by ensuring the hydrogel degrades at a rate suitable for its intended clinical application.
Hydrogels’ chemical structure can be precisely engineered to impart specific functionalities. Notable progress includes the development of “Smart” hydrogels that respond to microenvironmental stimuli (e.g. temperature, pH, or light) by altering their physical state or drug release properties 29 . This responsiveness enables new opportunities in precision medicine, especially in cancer treatment 30 . In oral cancer, for instance, such hydrogels can be designed to release drugs selectively based on local conditions, such as lower pH or elevated temperature in tumor tissues 31 . This targeted delivery minimizes the exposure of healthy tissue, thereby enhancing treatment specificity and efficacy. For example, Liu et al. 32 developed a PEGPD@SDF-1 hydrogel responsive to gingival proteases, which releases SAMP specifically in response to this stimulus, effectively inhibiting the growth of Porphyromonas gingivalis.
Classification of hydrogels
Hydrogels can be classified into natural and synthetic types based on their origin (Fig. 3). Natural hydrogels are typically derived from biological materials such as collagen, gelatin, alginate, and hyaluronic acid, offering excellent biocompatibility and biodegradability, making them widely used in biomedical applications 33 . These hydrogels integrate well with biological systems, and their degradation products are generally non-toxic and physiologically compatible. However, natural hydrogels often have limitations in physicochemical properties, such as mechanical strength and stability, which can restrict their broader application. In contrast, synthetic hydrogels are generally made from high-performance polymers like polyethylene glycol (PEG), polylactic acid (PLA), and poly(lactic-co-glycolic acid) (PLGA), providing superior control over their properties. Synthetic hydrogels can be precisely engineered during synthesis to achieve desired mechanical properties, degradation rates, and drug release profiles 34 . This level of control offers synthetic hydrogels significant potential for drug delivery and cancer treatment. By tailoring synthesis conditions, the physical and chemical properties of synthetic hydrogels can be finely tuned to meet specific therapeutic needs 35 .

The two primary categories of hydrogels and their key properties: natural hydrogels (e.g. collagen, alginate) and synthetic hydrogels (e.g. polyethylene glycol, polylactic acid). The figure highlights the comparative advantages, such as the biocompatibility of natural hydrogels and the mechanical stability and functionality of synthetic ones, as well as their respective limitations. Figure created with Adobe Illustrator 2020.
Research on the interaction between hydrogels and cells
The interaction between hydrogels and cells plays a critical role in their biomedical application, influencing biocompatibility, therapeutic efficacy, and safety in areas such as drug delivery and tissue engineering, and cancer treatment 36 . For effective in vivo function, hydrogels must support essential cellular processes such as adhesion, proliferation, and differentiation, while avoiding immune rejection or adverse responses 37 .
Hydrogels promote these cellular functions largely due to their excellent biocompatibility. Their soft, hydrated structure mimics the native extracellular matrix (ECM), providing a physiologically relevant microenvironment that supports cell adhesion, proliferation, growth, and function with minimal inflammation or immune rejection. This feature is particularly critical in sensitive regions like the oral cavity 38 . This biocompatibility, combined with the physical support, is essential not only for general tissue repair 39 but also for localized treatments in oral wound healing, post-surgical recovery, and managing conditions such as gingivitis and oral ulcers 40 .
To further enhance hydrogel–cell interactions, researchers often functionalize hydrogel surfaces with bioactive motifs. Incorporating cell-adhesion peptides such as RGD sequences, significantly promotes cellular attachment and functional maintenance 41 . For example, Fraser and Benoit 42 used RGD- and GFOGER-functionalized PEG hydrogels to regulate the activity of periodontal ligament cells (PDLCs), promoting cementum regeneration in rat periodontal defect models. Similarly, Zhang et al. 43 developed GelMA microspheres loaded with human platelet lysate (PL) and nanoclay Laponite, which sustained the release of growth factors, enhanced angiogenesis, and improved the differentiation of human dental pulp stem cells, demonstrating promising potential for pulp tissue regeneration.
Hydrogels as advanced delivery platforms in oral cancer: from drugs to immunomodulators
Hydrogels offer significant potential in oral cancer treatment by enabling controlled drug delivery and localized immune modulation. Their three-dimensional, responsive structure facilitates precise therapeutic release, enhancing efficacy while minimizing systemic side effects44,45. Figure 4 schematically illustrates their preparation process (from raw materials to gel formation) and various delivery systems—such as nanospheres, nanoparticles, nanotubes, and nanofibers—designed to improve drug retention and targeted efficacy in the oral cavity.

Schematic illustration of hydrogel preparation and its applications in oral therapy. The diagram summarizes key steps in hydrogel preparation (weighing, dissolution, sonication, and gel formation) and highlights various hydrogel-based delivery systems (e.g. nanospheres, nanoparticles, nanotubes, and nanofibers) used for targeted treatment in the oral cavity. These systems are designed to enhance drug retention and therapeutic efficacy at the target site. Figure created with Adobe Illustrator 2020.
The porous network of hydrogels functions as a drug reservoir, enabling sustained and localized release that maintains high drug concentrations at the tumor site while reducing off-target toxicity46,47. “Smart” hydrogels can be engineered to respond to tumor-specific signals, such as acidic pH, triggering drug release precisely within the malignant microenvironment 48 . Further functionalization with targeting ligands, such as antibodies, enhances specificity for cancer cells, improving treatment accuracy and sparing healthy oral tissues49,50.
In addition to oncology, hydrogels support dental and pulp tissue repair. They can seal the pulp cavity, provide a stable microenvironment for sustained release of anti-inflammatory or antibacterial agents, and promote cell adhesion and regeneration—benefits that extend to oral diseases like pulpitis and periodontitis 51 .
Building on their role as passive carriers, hydrogels are engineered as an ideal localized delivery platform for immunomodulatory agents. Their porous structure protects and gradually releases cytokines, antibodies, antigens, and other small molecules, enabling precise immune activation within the tumor area while minimizing systemic exposure 52 . For example, Han et al. 53 used stiffness-tunable collagen hydrogels to deliver vascular endothelial growth factor (VEGF), promoting dentin-pulp regeneration in immunodeficient mice.
This localized strategy is also effective for inflammatory oral diseases like periodontitis. A chitosan-based thermosensitive hydrogel (TISH) co-loaded with granulocyte-macrophage colony-stimulating factor (GM-CSF) and resveratrol was developed to locally immunomodulate dendritic and T cells, suppressing inflammation and promoting tissue healing in vivo 54 . Similarly, a chitosan/β-glycerophosphate/gelatin hydrogel loaded with aspirin and erythropoietin (EPO) provides early anti-inflammatory action followed by sustained release, significantly reducing inflammation and enhancing periodontium regeneration 55 .
By co-delivering chemotherapeutic agents and immunomodulators, hydrogels can simultaneously target tumor cells and reprogram the local immune landscape, creating a potent synergistic effect against oral cancer.
Mechanistic insights into hydrogel-based therapy
The efficacy of hydrogels in oral cancer treatment relies on two key mechanisms: physicochemical control for drug delivery (including physical adsorption, stimuli-responsive release, and microenvironment targeting) and biological interfacing for immune modulation (such as local immune activation and cell-adhesive retention) (Fig. 5). These integrated mechanisms facilitate synergistic, localized therapy, which is critical for the development of next-generation treatment platforms (Table 1).

Mechanisms of hydrogel-mediated immune modulation in cancer therapy. This schematic illustrates how hydrogels can function as smart platforms for cancer immunotherapy. It emphasizes their capacity for controlled drug release in response to microenvironmental triggers (e.g. pH, temperature), blocking immune checkpoints like PD-1/PD-L1 and CTLA-4, activating T cells, and promoting immune cell adhesion through specific signaling pathways that enhance immune cell interactions with tumor cells. Figure created with Adobe Illustrator 2020.
Representative studies of functional hydrogels in oral disease management.
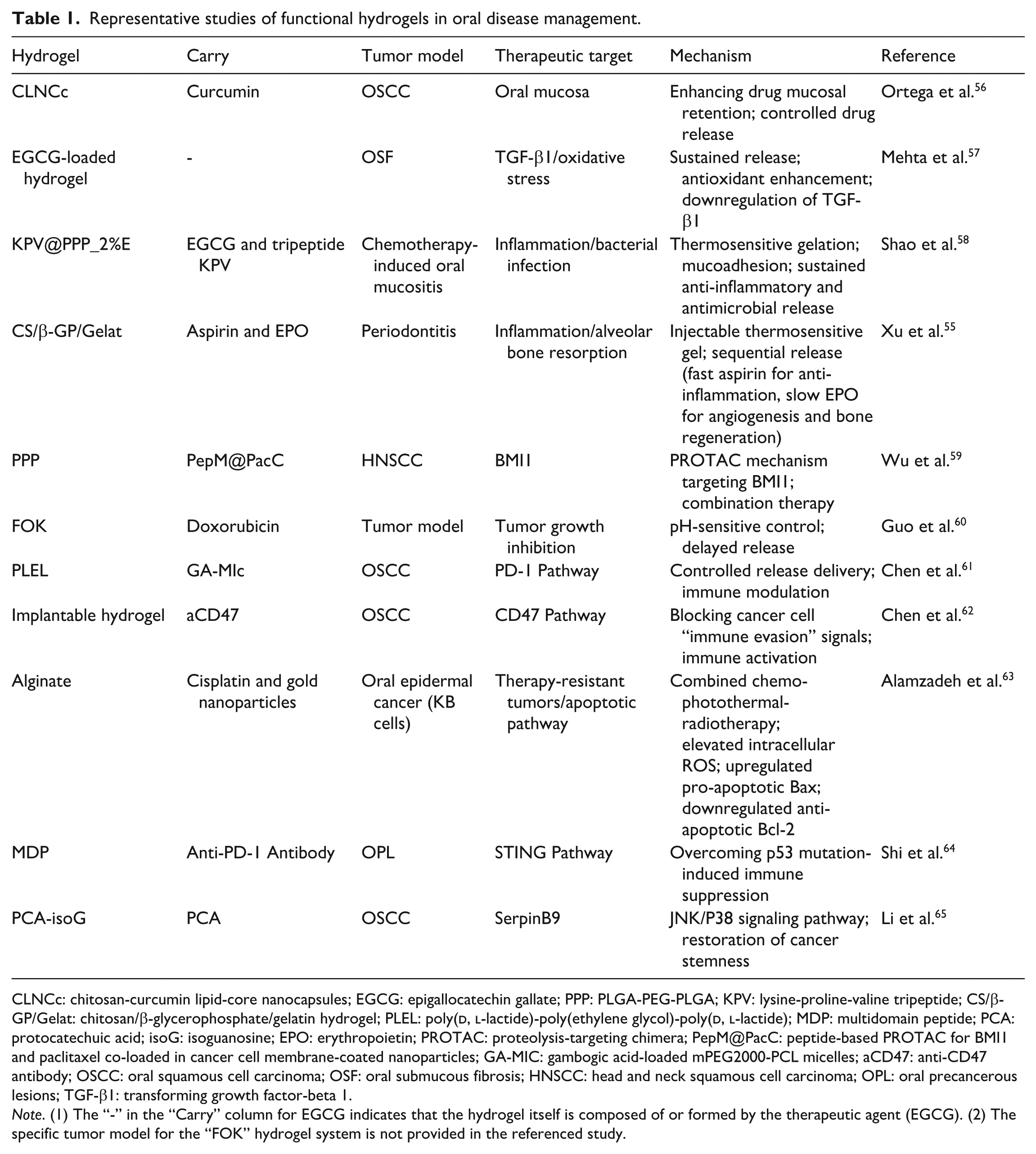
CLNCc: chitosan-curcumin lipid-core nanocapsules; EGCG: epigallocatechin gallate; PPP: PLGA-PEG-PLGA; KPV: lysine-proline-valine tripeptide; CS/β-GP/Gelat: chitosan/β-glycerophosphate/gelatin hydrogel; PLEL: poly(
Note. (1) The “-” in the “Carry” column for EGCG indicates that the hydrogel itself is composed of or formed by the therapeutic agent (EGCG). (2) The specific tumor model for the “FOK” hydrogel system is not provided in the referenced study.
Physical adsorption and controlled release
Hydrogels possess a unique three-dimensional network structure, which allows for the encapsulation of drug molecules through physical adsorption 66 . This structure stabilizes the drug molecules and enables stimuli-responsive release; changes in temperature, pH, or other environmental cues induce swelling or contraction of the polymer chains within the hydrogel, thereby modulating pore size and controlling drug diffusion rates 67 . For example, Ortega et al. developed a thermosensitive hydrogel (chitosan-curcumin lipid-core nanocapsules, CLNCc) that incorporated curcumin-loaded nanocapsules and adhered to the mucosa for up to 8 h. Under low pH or high-temperature conditions, the hydrogel undergoes significant volume changes, which promote faster drug release 56 . Mehta et al. formulated an epigallocatechin gallate (EGCG)-based hydrogel for the treatment of oral submucous fibrosis (OSF), focusing on oral premalignant diseases. In a rat OSF model, this hydrogel enhanced antioxidant capacity, downregulated transforming growth factor-beta 1 (TGF-β1) expression, and significantly facilitated mouth opening 57 (Fig. 6a). Similarly, Shao et al. designed a thermosensitive PLGA-PEG-PLGA (PPP) hydrogel system, infused with EGCG (at 2% to enhance antibacterial effectiveness) and the anti-inflammatory tripeptide LysProVal (KPV) (referred to as KPV@PPP_2%E). This hydrogel rapidly forms at 37°C, exhibits strong adhesion to tissue, and provides prolonged anti-inflammatory and antibacterial effects, significantly improving eating, weight recovery, and reducing inflammation in animal models of oral mucositis 58 (Fig. 6b). Xu et al. expanded this paradigm to periodontal regeneration by creating an injectable, thermosensitive hydrogel composed of chitosan/β-glycerophosphate/gelatin, co-loaded with aspirin and EPO for periodontitis treatment (Fig. 6c). Upon injection, the formulation swiftly gels at body temperature, facilitating localized and sustained dual-drug delivery: an early release of aspirin to mitigate inflammation is followed by a prolonged release of EPO to promote angiogenesis and periodontium regeneration 55 .
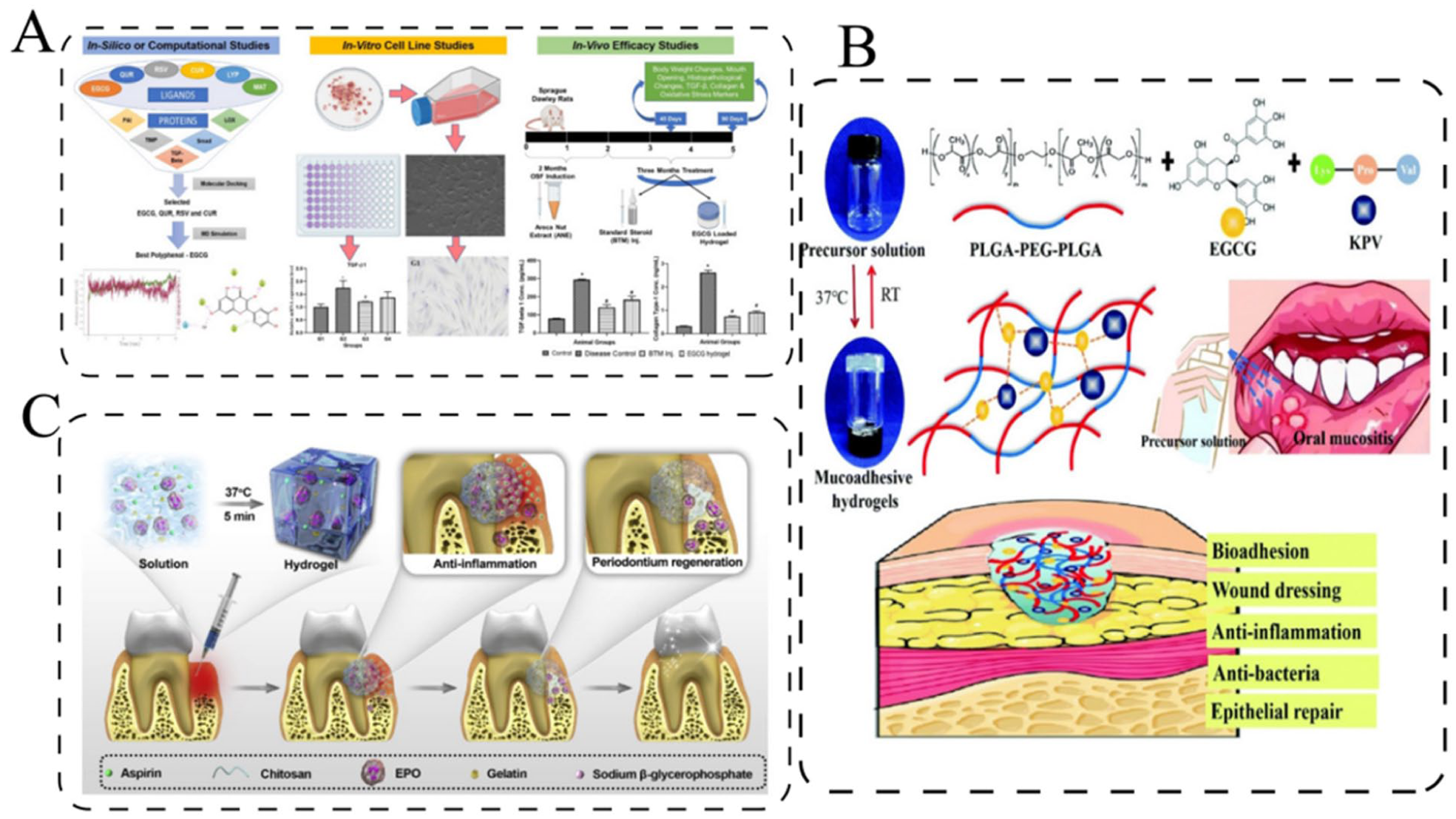
Therapeutic applications of functional hydrogels in oral disease models. (a) Mechanism and therapeutic effects of an epigallocatechin gallate (EGCG)-based hydrogel in a rat model of oral submucous fibrosis (OSF), highlighting enhanced antioxidant defense, reduced TGF-β1 expression, and facilitated mouth opening 57 . (b) Design and function of a mucoadhesive KPV@PPP_2%E hydrogel for the treatment of chemotherapy-induced oral mucositis 58 . (c) Schematic illustration of the preparation and application of the chitosan/β-glycerophosphate/gelatin hydrogel loaded with aspirin and EPO for periodontitis treatment 55 . Figure created with Adobe Illustrator 2020.
Immune activation mechanism
Hydrogels uniquely activate immune cells locally within tumors, providing precise immunomodulation with minimal systemic impact. For instance, a thermosensitive polymer-based hydrogel loaded with CaCO3-imiquimod nanoparticles activates T-cell responses, while cancer cell membrane-coated nanoparticles deliver proteolysis-targeting chimera (PROTAC) peptides for targeted chemotherapy, synergistically enhancing the treatment of head and neck squamous cell carcinoma (HNSCC)59,68. These hydrogels establish a sustained immunoactive tumor microenvironment, where cytokine release amplifies local immune activity and improves cancer cell clearance.
Hydrogels also enable localized and controlled release of immunomodulators, reducing systemic side effects such as fever and autoimmune reactions, in line with the growing trend of localized immunotherapy to overcome the limitations of systemic administration 69 . By concentrating immune activity within the tumor region, this approach is especially beneficial in areas like the oral cavity, where it can help inhibit recurrence after surgery or radiation 70 . Future studies may combine hydrogel-based local immunotherapy with systemic treatments to further enhance both efficacy and safety 71 .
Tumor microenvironment-responsive mechanism
pH-responsive hydrogels exploit the acidic tumor microenvironment to improve targeted drug delivery. By swelling in low pH conditions, they accelerate drug release within tumors, enhancing precision and reducing off‑target effects72,73. For example, an injectable, pH-responsive FOK peptide hydrogel was developed to efficiently load doxorubicin (DOX), enabling precise tumor-targeted drug release and sustained tumor growth inhibition, while minimizing systemic toxicity in a mouse model 60 .
To further enhance precision, hydrogels are also engineered to respond to additional external stimuli, such as light or temperature 74 . Chen et al. developed a thermosensitive hydrogel, gambogic acid-loaded mPEG2000-PCL micelles (GA-MIC)-GEL, which rapidly gels at 37°C to provide sustained release of gambogic acid. In mice with OSCC, this system downregulated PD-1, increased cytotoxic T cells, and improved the tumor immune microenvironment (TIME) 61 . These smart systems allow on-demand control of drug release or immune activation, advancing the potential for localized and personalized cancer therapy.
Smart hydrogels also facilitate combinational therapies that integrate multiple treatment modalities. Chen et al. 62 created an implantable hydrogel that co-delivers photodynamic, photothermal, and immune adjuvant agents, effectively preventing OSCC recurrence and metastasis in a mouse model, providing long-term postoperative immune protection. Similarly, Alamzadeh et al. developed a multifunctional alginate hydrogel co-loaded with cisplatin and gold nanoparticles. This platform combined chemotherapy (cisplatin release), photothermal therapy (via near-infrared irradiation of gold nanoparticles), and radiotherapy. The combined approach significantly increased intracellular reactive oxygen species (ROS) and shifted the pro-apoptotic balance (upregulating Bax, downregulating Bcl-2), inducing tumor cell death and demonstrating potential to overcome therapy resistance in oral cancer models 63 .
Through these stimulus-responsive mechanisms, hydrogels enable targeted drug release tailored to the tumor microenvironment, enhancing treatment precision and safety. Shi et al. utilized a multidomain peptide (MDP) hydrogel loaded with PD-1 immune checkpoint inhibitors to prevent the progression of oral precancerous lesions (OPL) into OSCC. This system controllably releases anti-PD-1 antibodies, activating CD8+ T cells and restoring STING pathway function, which enhances the interferon response and antigen presentation to improve the TIME. Regardless of p53 mutation status, this strategy effectively suppressed the malignant progression of OPL, showing promise in OSCC prevention 64 .
Cell adhesion mechanism
Cell adhesion within the tumor microenvironment plays a pivotal role in supporting anti-tumor immune responses. The adhesive interface provided by hydrogels aids in retaining immune cells and therapeutic agents locally, enhancing immunomodulatory effects. For instance, a Protocatechuic acid (PCA)-based supramolecular hydrogel combined with isoguanosine (isoG) promoted cell adhesion, which in turn improved local immune activity and treatment efficacy 65 . This demonstrates how utilizing the adhesive properties of hydrogels can serve as an effective strategy in cancer immunotherapy 75 .
Clinical prospects of hydrogels
Clinical applications of market-approved hydrogels
Hydrogels have gained significant attention in medical applications, particularly for localized and targeted therapies, owing to their responsive drug release capabilities76,77. While several hydrogel-based products are currently in clinical use, their effectiveness varies across different conditions. Table 2 presents the translational pipeline; for example, in HNSCC, HydroTac® hydrogel dressing did not outperform conventional hyaluronic acid dressing in preventing acute radiodermatitis (ARD), with its use limited by handling challenges and patient adherence 78 . In contrast, an α-mangostin-loaded chitosan-alginate hydrogel film significantly reduced ulcer size and pain in recurrent aphthous stomatitis, demonstrating good mucoadhesion and safety 79 . In transoral robotic surgery (TORS) for oropharyngeal cancer, PuraBond® hydrogel effectively achieved hemostasis, simplified application, and shortened hospital stays without complications 80 . These examples highlight the potential of hydrogels in oncology, from localized drug delivery to postoperative management (Fig. 7). However, clinical translation still faces challenges such as patient compliance and variable outcomes. Future research should focus on optimizing hydrogel design to improve biocompatibility, stability, and therapeutic precision, providing safer and more effective solutions for cancer treatment 81 .
Clinical applications and research progress of hydrogels in oral disease treatment.

ARD: acute radiodermatitis; RAS: Recurrent Aphthous Stomatitis; TORS: transoral robotic surgery; EGCG: epigallocatechin gallate; TGF-β1: transforming growth factor-beta 1; IGF-1: insulin-like growth factor-1; OSF: oral submucous fibrosis; rhEGF: recombinant human epidermal growth factor; COX-2: cyclooxygenase-2; MMP9: matrix metalloproteinase-9; HNSCC: head and neck squamous cell carcinoma; OSCC: oral squamous cell carcinoma; OM: oral mucositis; L-cys_rGO: L-cystine functionalized porous and electroactive reduced graphene oxide (rGO) hydrogel; TNF-α: tumor necrosis factor-α.

Applications of Hydrogel in Oral Health. Hydrogels play diverse roles in oral health, ranging from serving as drug carriers for cavities and providing slow-release medications for pulpitis to offering hemostatic functions for bleeding. They assist in tumor treatments via gene therapy, act as biosensors for ulcer diagnosis, and deliver antimicrobial action to combat infections, contributing to comprehensive oral care. Figure created with Adobe Illustrator 2020.
Postoperative wound healing and tissue regeneration in the oral cavity
Hydrogels are ideal for postoperative oral care due to their unique three-dimensional network structure, biocompatibility, and high adjustability 88 . They create a protective, moist, and breathable barrier that prevents infection, supports healing, and enables comfortable long-term use without interfering with oral function89,90. For example, in the context of treatment-induced injury, Zhao et al. 82 demonstrated that the NAHAO® hydrogel effectively alleviated pain and promoted healing in a rat model of 5-fluorouracil-induced oral mucositis by inhibiting key inflammatory and tissue-degrading enzymes, such as cyclooxygenase-2 (COX-2) and matrix metalloproteinase-9 (MMP9) expression. Beyond wound management, hydrogels can actively promote regeneration by delivering bioactive molecules 91 . Srouji et al. 83 showed that hydrogel scaffolds loaded with TGF-β1 and IGF-1 significantly enhanced bone regeneration in rat mandibular defects, highlighting their potential for repairing surgical sites in patients with oral cancer.
Controlled drug release
Controlled drug release is critical for enhancing efficacy and minimizing side effects in oral therapy. The tunable network structure of hydrogels makes them ideal carriers, enabling sustained, localized drug delivery through adjustments in polymer structure, crosslinking density, and degradation rates 92 . This approach maintains therapeutic drug levels locally, reduces dosing frequency, and improves patient compliance92,93. For instance, Bhardwaj et al. developed a dual nano-ion carrier hydrogel based on gellan gum and lipids for the localized co-delivery of cisplatin and paclitaxel in HNSCC. This hydrogel prolonged drug retention at the tumor site, minimized systemic exposure, and synergistically enhanced radiosensitivity with a favorable safety profile 84 . Furthermore, controlled release facilitates the long-term management of chronic oral conditions. For example, in treating OSF, an EGCG hydrogel outperformed traditional corticosteroids by inhibiting collagen expression and improving mouth opening in preclinical models, offering a safer and more effective local therapy 57 .
The responsive design of hydrogels allows drug release to be modulated by specific biological features of the tumor microenvironment. Zheng et al. studied a silver nanoparticle-loaded hydrogel that responds to the dysbiotic state of the intratumoral microbiome in oral squamous cell carcinoma (OSCC). By selectively suppressing competing bacteria and enriching beneficial Streptococcus salivarius, this system actively corrects microbial imbalances, thereby enhancing anti-tumor immunity. When combined with PD-1 inhibitors, it illustrates how hydrogels can be engineered to sense and correct pathological biological signals, thereby amplifying the outcomes of immunotherapy 85 .
Advanced and emerging therapeutic platforms
Beyond established applications, hydrogels are being engineered as advanced platforms for next-generation therapies and diagnostics. One emerging frontier is gene therapy, which offers promise for cancer treatment by directly modifying cellular genetic material. Hydrogels serve as ideal carriers for this purpose, providing a protected, localized, and controllable delivery platform that minimizes systemic side effects 94 , a critical advantage in oral cancer. Their three-dimensional network protects genetic payloads from degradation and enables triggered release at the target site95,96. Although clinical translation in oral cancer is still developing, the foundational design principles of hydrogels lay a strong foundation for future gene-based interventions.
Hydrogels are transforming diagnostics and therapeutic monitoring through advanced biosensing. Their biocompatibility and adaptable structure allow seamless integration with biological tissues for real-time monitoring. For instance, Dai et al. developed a wearable, multifunctional dual-network hydrogel incorporating PANI@BTO nanoparticles. This hydrogel visually senses oral pH via polyaniline and generates antibacterial ROS via barium titanate under ultrasound, combining early detection with non-invasive therapy for cavity prevention and whitening
86
. Pushing sensitivity further, Kapil et al. created an innovative electrochemical immunosensor based on
Future trends and challenges of hydrogels in oral disease treatment
The future of oral disease therapy lies in precision and personalization. Hydrogels, due to their highly tunable properties, are positioned at the forefront of this evolution. By customizing their composition, drug-release profiles, and responsiveness (e.g. to pH or temperature), patient-specific regimens can be developed to optimize efficacy and minimize side effects 97 .
However, translating this potential from the laboratory to clinical settings presents significant challenges. Key obstacles include achieving scalable and reproducible manufacturing, understanding in vivo degradation and clearance to ensure long-term safety, and rigorously validating biocompatibility through clinical trials. Overcoming these hurdles will require advancements in material design, novel production technologies, and close collaboration across scientific and clinical disciplines to standardize and regulate hydrogel-based therapies and products 98 .
Conclusion
As an emerging biomaterial, hydrogels offer considerable potential for treating oral diseases due to their unique physicochemical properties and excellent biocompatibility. Recent developments have expanded their application in drug delivery, immunomodulation, gene therapy, and tissue regeneration—particularly for oral cancer, periodontitis, dental pulp repair, and mucosal healing. By enabling localized and targeted delivery, hydrogels enhance treatment precision while minimizing systemic side effects, providing patients with safer and more effective options. In pulp and periodontal regeneration, hydrogels offer structural support and actively promote tissue repair, while in mucosal repair, their adhesive and biocompatible nature accelerates healing and alleviates discomfort.
Looking ahead, research will increasingly focus on the development of multifunctional and personalized hydrogel systems, integrating technologies such as immunotherapy, gene editing, and biosensing to achieve comprehensive treatment and real-time monitoring. Despite their promise, clinical translation still faces challenges, including production quality control, understanding in vivo degradation pathways, and ensuring long-term safety. Nevertheless, hydrogels are poised to become transformative tools in oral healthcare, significantly improving both patient quality of life and therapeutic outcomes.
Footnotes
Acknowledgements
Not applicable.
Ethical approval
Not applicable.
Consent statement
Not applicable.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (82360198).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Not applicable.
Statement of human and animal rights
This article does not contain any studies with human or animal subjects.
Statement of informed consent
There are no human subjects in this article and informed consent is not applicable.
