Abstract
Focal segmental glomerulosclerosis (FSGS) is one of the major causes of nephrotic syndrome, which can progress to end-stage renal disease, leading to kidney transplantation. Following renal transplantation, recurrence of FSGS (rFSGS) occurs in 30%–40% of patients with a high risk of graft loss. rFSGS typically presents with nephrotic-range proteinuria within days after post-transplantation. This review summarizes pathophysiology, biomarkers, and therapeutic strategies for rFSGS. Monogenic causes of FSGS, such as those caused by APOL1 mutation, show variable recurrence, while NPHS2 and ACTN4 show low recurrence of FSGS. Evidence suggests that idiopathic or primary FSGS is strongly associated with rFSGS, owing to podocyte structural damage caused by circulating permeability factors or immune dysfunction. Recent advances have identified biomarkers such as anti-nephrin antibodies, anti-CD40 antibodies, soluble tumor necrosis factor receptor 2 (sTNFR2), and soluble urokinase-type plasminogen activator receptor (suPAR) that help in early detection of recurrent FSGS. Post-transplant monitoring includes measuring urine protein-to-creatinine ratio (UPCR) and 24-h urine protein excretion, and a kidney biopsy. Preventive strategies, although including plasmapheresis and rituximab, show limited benefit and are not recommended for routine prophylaxis. Treatment options include plasmapheresis, immunoadsorption, and immunosuppressive drugs such as cyclophosphamide, rituximab, or calcineurin inhibitors. Recurrent FSGS is a clinical challenge with its multifactorial pathogenesis. Incorporating strategies such as genetic testing, risk stratification, and early detection with the help of biomarkers and early treatment can induce remission and preserve graft survival. Despite these advances, large prospective studies are still required for standardizing prevention and management strategies for rFSGS.
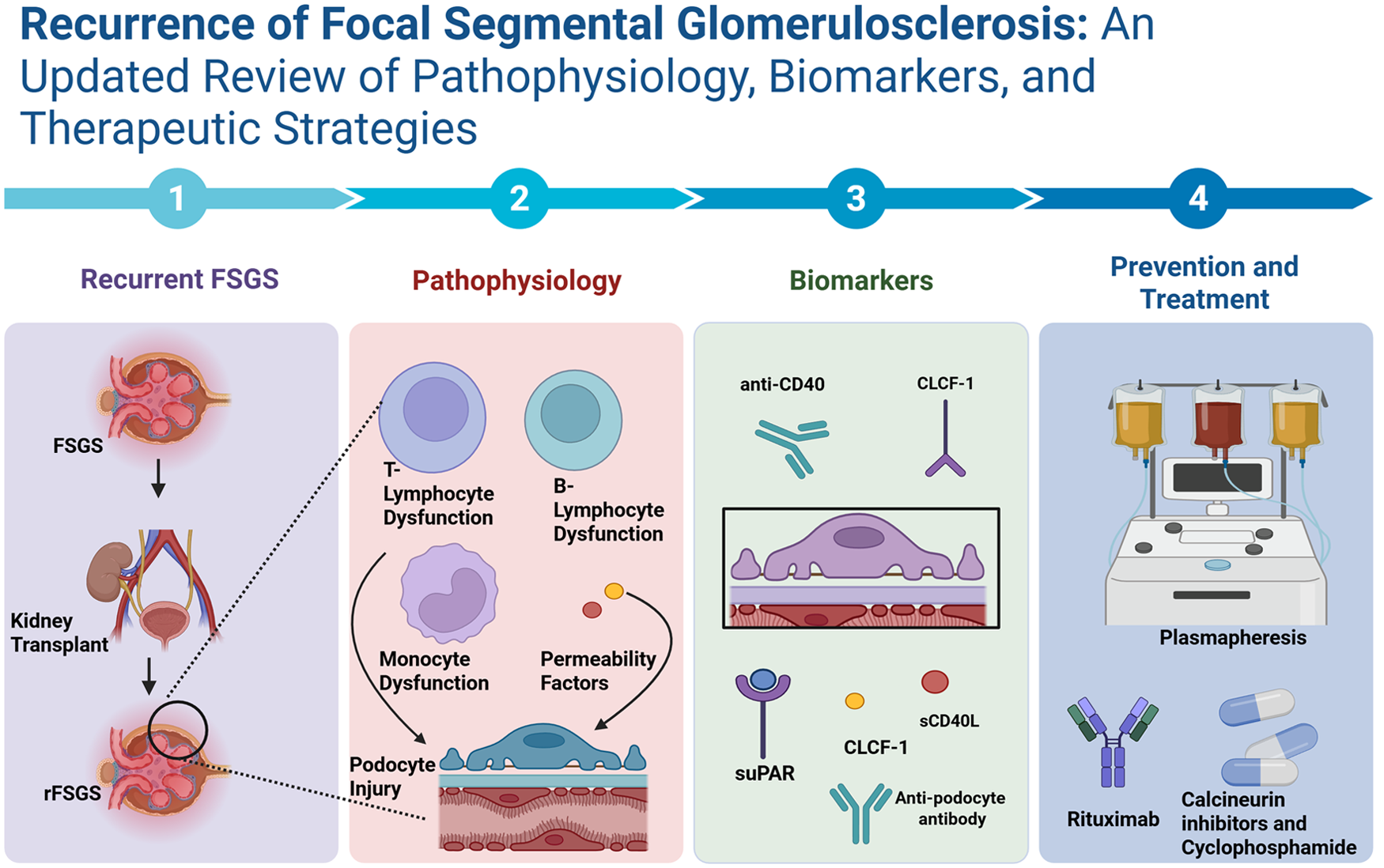
Introduction
Focal segmental glomerulosclerosis (FSGS) is a chronic, nephrotic glomerulopathy that clinically presents with proteinuria, hypoalbuminemia, generalized edema, hyperlipidemia, and lipiduria. FSGS comprises a significant portion of nephrotic syndrome patients, representing 40% of adults and 20% of children 1 . FSGS is often resistant to steroids, the primary treatment for other glomerulopathies such as minimal change disease 2 . As a result, it often progresses to end-stage kidney disease (ESKD) in 40%–60% of patients 3 . FSGS is classified based on etiology into primary, genetic, and secondary FSGS, as shown in Table 14,5.
Post-transplant recurrent FSGS (rFSGS) is defined as the development of proteinuria and hypoalbuminemia in a transplant recipient with prior primary FSGS. Prevalence of rFSGS is approximately 30% in post-transplant recipients, and nearly 39% developed graft loss over a median follow-up duration of 5 years 7 . A meta-analysis of 22 studies with 966 patients reported an overall recurrence rate of 38% (95% confidence interval (CI): 31%–44%). Subgroup analyses by country demonstrated recurrence rates of 38% in the United States, 29% in Spain, 32% in Korea, 45% in France, and 62% in Japan. Stratification by study size showed recurrence rates of 39% (95% CI, 31%–48%) in cohorts with >30 patients compared with 35% (95% CI, 26%–44%) in smaller studies. Among 11,742 kidney transplant recipients screened, 176 patients had idiopathic FSGS. Among them, 57 (32%; 95% CI, 25%–39%) experienced recurrence, and 39% progressed to graft loss over a median of 5 years (interquartile range (IQR), 3.0–8.1 years).
In a comparison of kidney health outcomes in children, adolescents, and adults with FSGS, Gipson D et al. examined whether age at diagnosis influenced kidney health outcomes among these patients with FSGS. The study included 127 (26%) children, 102 adolescents, and 253 adults. Overall, the median time to ESKD was 11.9 years (IQR, 5.2–19.1 years). The risk of ESKD among children versus adults did not differ significantly (hazard ratio (HR), 0.67; 95% CI, 0.43–1.03) or between adolescents and adults (HR, 0.85; 95% CI, 0.52–1.36). The median time to the composite endpoint was 5.7 years (IQR 1.6–15.2 years), with HR estimates of 1.12 (95% CI, 0.83–1.52) for children versus adults and 1.06 (95% CI, 0.75-1.50) for adolescents versus adults. These findings suggest that the impact of FSGS on kidney survival is substantial and consistent across age groups. Approximately 30% of pediatric FSGS cases are associated with monogenic disorders, whereas the majority are idiopathic and thought to result from unidentified circulating factors 8 . Idiopathic FSGS, presumed to have an underlying immunologic mechanism, presents a substantial risk of recurrence following kidney transplantation and is a leading cause of allograft failure, yet comprehensive data remain limited due to the rarity of this condition 9 . Estimates of the prevalence of this phenomenon vary widely between 6% and 57% 10 . This rate depends largely on demographic and clinical characteristics of patients being studied.
Despite substantial investigative efforts, the underlying mechanisms of FSGS remain incompletely understood, and effective preventative strategies are still limited. Fortunately, several recent breakthroughs offer new avenues to prevent this phenomenon. A recent focus has been placed on risk stratification through predictive biomarkers, circulating permeability factors (CPFs), and the integration of immunologic and genetic factors for risk stratification that informs preventative treatment strategies. Other research has identified emerging therapeutic targets. This review aims to review rFSGS risk factors based on genetics, pathophysiology, emerging biomarkers, treatment, and preventive strategies, with particular emphasis on risk stratification.
Risk factors for rFSGS
In adults, a large, multicenter, retrospective cohort study containing 253 patients with primary FSGS, Uffing et al. found 57 patients developed recurrence of FSGS after transplantation, with a median time of 1.5 months to recurrence. Twenty-two of the 57 patients with recurrent FSGS went on to develop graft failure. Specifically, patients with recurrence of FSGS were identified to have an HR of 4.80 (P < 0.001) for graft loss. Although this article attributes a higher HR to older-aged patients developing recurrent FSGS, other sources have suggested the contrary 7 . Higher recurrence incidence in white patients may be confounded by differences in allelic frequencies of causal risk-associated variants in APOL1 or causal variants in NPHS2. Another meta-analysis of 22 studies with 966 patients showed no association between rFSGS and factors such as human leukocyte antigen (HLA) mismatches, duration of dialysis before kidney transplantation, sex, living donor, tacrolimus use, and previous transplantation. Risk factors for rFSGS are summarized in Table 27,11.
Risk factors for recurrent FSGS.

HR: hazard ratio; SMD: standardized mean difference; OR: odds ratio.
Genetic risk factors
Genetics plays a complex role in the development of FSGS (Table 3)12–37. Estimates of the prevalence of causal variants in FSGS patients vary widely based on the age range of patients being studied, case definition, and how variants are classified as causal.
Clinical characteristics, mechanism, and recurrence of focal segmental glomerulosclerosis risk by gene.

MS: Meacham syndrome; FS: Frasier syndrome; DDS: Denys-Drash syndrome.
Genetic causes of FSGS include pathogenic variants in NPHS1, NPHS2, WT1, COL4A, ACTN4, TRPC6, INF2, PAX2, ANLN, CRB2, LMX1B, CD2AP, MYO1E and others (Figure 1). Although sometimes thought of as monogenic causes of FSGS, these rare variants often have a relatively high prevalence in healthy patients, suggesting multifactorial inheritance or an environmental role 38 . Pathogenic variants of these genes are linked to syndromes of which kidney disease is a feature; however, FSGS patients who screen positive for these variants often present with no other syndromic features16,39,40. Similarly, patients with FSGS often have common variants in APOL1 (G1 and G2), which are also associated with other kidney diseases 41 . By contrast, NPHS2 is the most common monogenic cause of early-onset steroid-resistant nephrotic syndrome (SRNS) 42 and presents in a non-syndromic fashion 41 . Generally, recurrence is considered less common in patients with an identifiable genetic cause, though gene-level analysis is limited to more common variants, including NPHS2 and APOL143–45. Table 4 summarizes studies of recurrence rates of patients with primary FSGS (Table 4)6,44–49. Smaller older studies of recurrence in NPHS2 demonstrate significant recurrence though larger, more recent studies demonstrate recurrence rates of near zero. One small study of INF2 demonstrates one FSGS recurrence in a small number of transplant recipients 49 . In addition, a case study of one patient with an ACTN4 mutation presented FSGS recurrence 5 years after kidney transplantation 50 . Notably, we found no studies reporting recurrence in patients with COL4A mutations despite its high prevalence in adult-onset FSGS patients 44 .

FSGS recurrence by gene. Heatmap demonstrating the estimated recurrence risk of focal segmental glomerulosclerosis (FSGS) after kidney transplantation based on underlying genetic etiology. Genes are arranged from lowest to highest recurrence risk. Color intensity corresponds to the assigned risk category: “0” = none, “1” = very low, “2” = low, and “3” = variable. APOL1-associated disease shows the highest recurrence variability. (Created Using R Statistical Software.)
Genetic risk factors of rFSGS.

Superscripts “t” and “m” refer to heterozygous (“t”) and compound heterozygous or homozygous (“m”).
In genetic FSGS, recurrence rate is generally rare as mutations cause structural defects rather than circulating factors. In the most recent large cohort of 117 patients with homozygous or compound heterozygous NPHS2 variants and SRNS (predominantly FSGS), FSGS recurred in only one patient 45 . Another study conducted in 2011, 0/11 patients experienced recurrence 6 . In contrast, APOL1 risk alleles (G1 or G2) have a greatly elevated risk of developing FSGS 51 and can incur podocyte injury via mitochondrial dysfunction, activation of inflammasomes, disruption of endolysosomal trafficking, ionic imbalances, and ultimately lead to cell death 51 . However, Sanna-Cherchi et al. reported, APOL1 missense variant p.N264K (c.792C>A), rs73885316 confers strong protection against G2-mediated FSGS and kidney disease. The study found that the toxicity of G1 and G2 alleles was markedly reduced when expressed on the haplotype containing the p.N264K variant. Population studies show a high prevalence of APOL1 alleles associated with Afro-descendant patients with chronic kidney disease (CKD) or high blood pressure52,53. Among other monogenic forms such as ACTN4, ANLN, CD2AP, CRB2, MYO1E, LMX1B, and PAX2, rFSGS has not been reported.
Pathophysiology of rFSGS
Recurrent FSGS is proposed to be caused by Tlymphocyte dysfunction and permeability factors, causing podocyte damage (Table 5) 5 . Evidence suggests that disease progression is influenced by clonally expanded cytotoxic T cells. Findings highlight that T-cell interactions suggest that these can be used as biomarkers for rituximab treatment. Treg memory cells deficiency in patients leads to corticosteroid dependence for treatment. Increasing evidence also supports the role of immune-mediated mechanisms involving CPFs and autoantibodies that compromise podocyte integrity53,54.
Pathophysiology of recurrent FSGS 4 .

Not all patients experience recurrence, and among those who do, the timing, severity, and response to therapy vary widely, suggesting the existence of distinct pathophysiological pathways 55 . In certain cases, recurrence of nephrotic-range proteinuria occurs within hours to days post-transplant, supporting a systemic, non-anatomically confined disease process 54 . Experimental models and clinical observations have implicated these factors in triggering cytoskeletal injury of podocytes, leading to effacement, foot process detachment, and glomerular scarring 56 .
Biomarkers for rFSGS
Biomarkers are produced when the immune system damages podocytes, making them indicators for recurrence. One of the most widely studied biomarkers is anti-nephrin antibodies, which target nephrin, an essential component of the slit diaphragm that maintains the glomerular filtration barrier. Under physiological conditions, podocyte foot processes form interlocking slits bridged by nephrin-based complexes that selectively retain plasma proteins. Autoantibodies binding to nephrin impair podocyte structure, leading to proteinuria and foot process effacement56,57. The evolution of biomarkers for FSGS diagnosis is represented in Figure 2.

Evolution of biomarkers in FSGS diagnosis. Timeline summarizing key biomarker discoveries in primary FSGS from 1999 to 2024, including circulating factors, gene-expression findings, proteomic markers, podocyte specific biomarkers, and emerging antibody and receptor-based biomarkers. (Created in BioRender. Rayarakula, N. (2025), https://BioRender.com/auu4rh6.)
Torra et al. screened approximately 9000 candidate serum antibodies in retrospectively collected samples from patients with recurrent FSGS, identified the seven most predictive, and validated them in a prospective cohort. Among these, anti-CD40 demonstrated the highest predictive utility for recurrence. CD40 is a co-stimulatory receptor found on antigen-presenting cells and podocytes; its activation may contribute to T cell–driven podocyte injury and promote sustained immune-mediated damage (Figure 3). Notably absent from the 9000 antibody panel were anti-slit and anti-nephrin 58 .

Overview of key circulating, immune, and podocyte-specific biomarkers implicated in recurrent FSGS, highlighting pathways involving permeability factors, inflammatory cytokines, mitochondrial stress, cytoskeletal remodeling, and associated therapeutic targets. Created in BioRender. Rayarakula, N. (2025), https://BioRender.com/cjwdp7w.
An additional non-antibody biomarker that has been extensively investigated is soluble urokinase-type plasminogen activator receptor (suPAR). This has been implicated in promoting podocyte dysfunction through the activation of podocyte β3 integrin signaling, leading to cytoskeletal rearrangement and foot process effacement 59 . The previously known fact that serum from patients with FSGS can activate podocyte ß3 integrins, causing massive pedicellar fusion, suggested the possibility that suPAR plays a role in the pathogenesis of FSGS. However, several other factors, such as tumor necrosis factor, can play similar actions on podocyte integrins and indeed exert profound modifications on cellular suPAR expression.
Increased levels of suPAR have been observed in both primary and recurrent FSGS cases, though its diagnostic utility is limited due to similar elevations observed in non-glomerular inflammatory and infectious diseases 60 . Nevertheless, evidence shows an inverse relationship between suPAR levels and estimated glomerular filtration rate (eGFR), indicating its potential role as a dynamic biomarker for disease activity and renal impairment in FSGS 36 . Various biomarkers used in rFSGS are summarized in Table 658–64.
Biomarkers for recurrent FSGS.

The heterogeneity of recurrence risk across individuals with otherwise similar clinical and histological profiles has led to increasing interest in classifying FSGS patients into specific biological subgroups, a process known as endotype stratification. Several studies suggest that patients with idiopathic FSGS may represent an immune-driven endotype characterized by autoantibody-mediated podocyte injury, such as through the presence of anti-nephrin or anti-CD40 antibodies. These patients may be identifiable through serologic profiles and may respond preferentially to immunomodulatory therapies57,67. In contrast, individuals with monogenic forms of FSGS, such as those involving NPHS1, NPHS2, or COL4A mutations, typically exhibit a significantly reduced risk of recurrence. These cases are often resistant to immunosuppressive therapy, further reinforcing the distinction between systemic and genetically intrinsic disease mechanisms 68 .
Although no singular permeability factor has been conclusively identified, proteomic and immunologic profiling of FSGS patients has facilitated the development of multi-marker panels. These panels aim to facilitate individualized transplant risk assessments and allow for targeted management strategies57,69. Growing knowledge of the pathogenesis of FSGS and its recurrence post-transplant has also led to therapeutic insights that are now being explored.
Risk stratification
Through examining patients’ genetics, circulating factors, and demographic risks, it is possible to predict patients’ risk of recurrence with a relatively high accuracy. Currently, no studies examine risk factors across modalities. Circulating factors are generally most predictive. The best single predictor of FSGS is anti-nephrin antibodies. One patient with diffuse podocytopathy found that pretransplant anti-nephrin antibodies had a 38% sensitivity and 100% specificity for predicting future diffuse recurrence; however, this study included a significant number of non-FSGS patients and patients with prior disease recurrence. Analysis of supplemental data from this study revealed up to a 50% sensitivity and 100% specificity in first-time transplant recipients with an FSGS history and a detectable anti-nephrin level (n = 22) 68 . A study in Japan showed similar findings where anti-nephrin was seen in all patients with non-genetic FSGS experiencing recurrence (n = 11), but not in patients without recurrence (n = 3). With multiple predictive circulating factors, one might suggest a multivariate risk score to assess recurrence risk.
A significant breakthrough in risk prediction for recurrence was seen in a study that examined a panel of 9000 antibodies for those that were most predictive of FSGS recurrence. This screening yielded seven antibodies: FAS, CD40, CGB5, PTPRO, P2RY11, APOL2, and SNRPB2, among which anti-CD40 was the most predictive marker 58 . Soluble tumor necrosis factor receptor 2 has shown promise as a predictive biomarker for progression of FSGS to ESRD (area under the receiver operating characteristic curve (AUROC) = 0.848); however, it has not been investigated as a predictor of FSGS recurrence 64 .
Advancements in ex vivo modeling demonstrate a strong opportunity for prediction, prevention, and treatment of rFSGS, A novel cell-based assay utilizes luciferase-based reporters to monitor pro-apoptotic genes such as IL1β, BMF, and IGFBP3 that are upregulated in podocytes exposed to rFSGS patient’s plasma. The test demonstrated good diagnostic performance, with sensitivities and specificities above 80% (area under the curve (AUC): 0.81) for the IL1β and BMF reporter genes 70 . Building on this concept, Gupta et al. used stem cell–derived organoids to predict FSGS recurrence. Plasma of patients with FSGS on organoids from non-FSGS donors induced multiple histological and biomarker signatures consistent with FSGS. Interestingly, however, fewer apoptotic cells were seen in treated organoids as patients received sequential treatments with plasmapheresis (PP). 71 Ex vivo tissue modeling represents a promising technology that may have future implications for risk stratification, preclinical investigations, and clinical decision-making in pretransplant induction therapy.
Post-transplant monitoring of rFSGS
High-risk patients undergo intensified surveillance in addition to KDIGO recommendations, with urine protein-to-creatinine ratio measured on post-operative day 1, at discharge, weekly for 4 weeks, followed by monthly for 1 year [2C] (Figure 4).
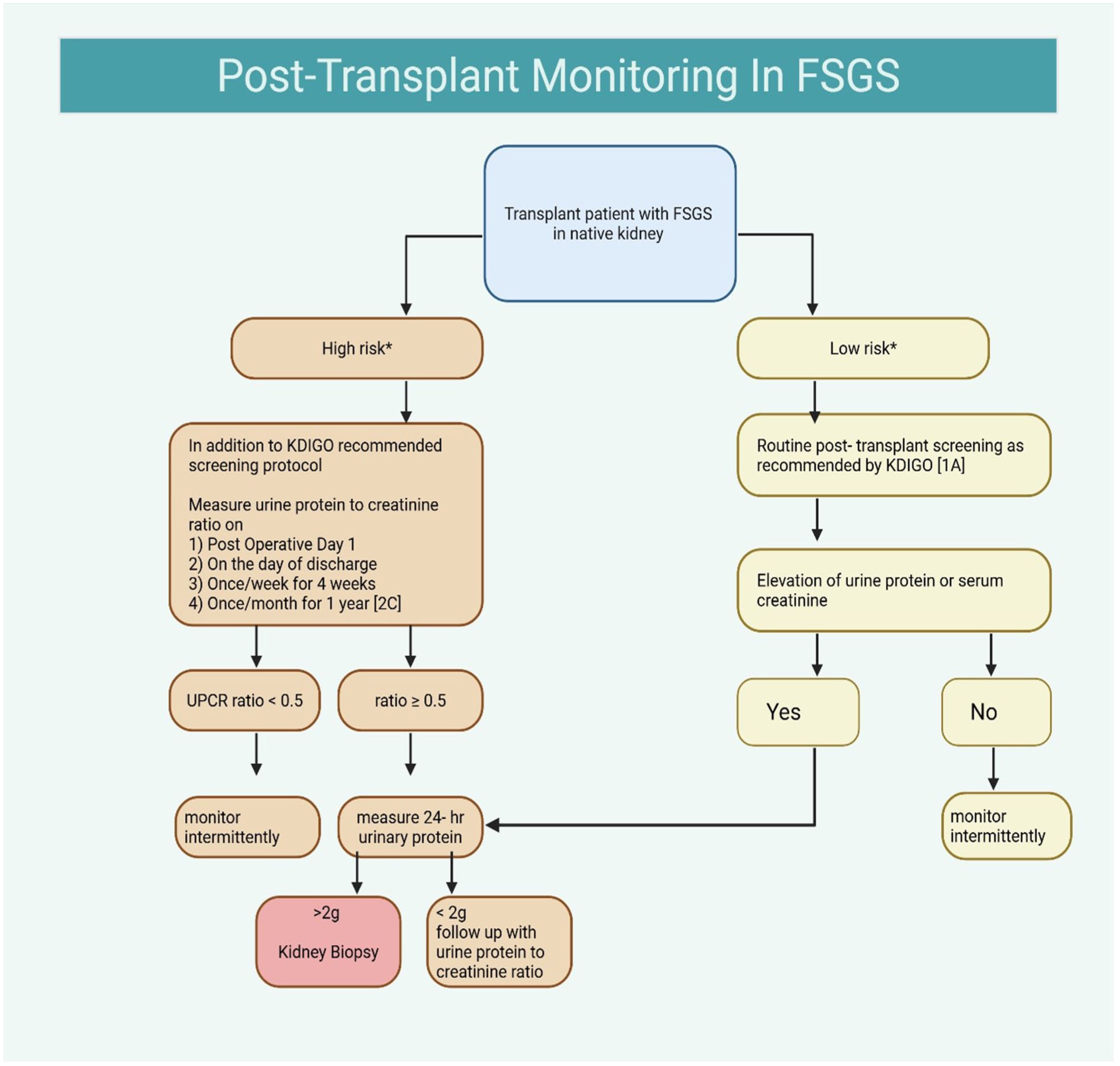
Algorithm for post-transplant monitoring in patients with FSGS, outlining KDIGO-based surveillance, high- versus low-risk pathways. Created in BioRender. Rayarakula, N. (2025), https://BioRender.com/az67ytw.
Patients with a ratio <0.5 are monitored intermittently, while those with a ratio ≥0.5 undergo 24-h urinary protein measurement; values <2 g/day prompt continued follow-up with spot urine testing, whereas >2 g/day warrant a kidney biopsy. Low-risk patients follow standard KDIGO post-transplant screening [1A], with further evaluation triggered by any rise in serum creatinine or urine protein. This approach emphasizes early identification of recurrent FSGS through structured proteinuria monitoring, enabling timely intervention to preserve graft function.
Prevention and treatment of rFSGS
The therapeutic interventions act on the same mechanisms that drive rFSGS by removing autoantibodies, suppressing T cell–mediated injury, and preserving podocyte skeletal structure. Among the most widely used interventions is PP, aimed at removing CPFs that contribute to podocyte damage (Figure 5). In a retrospective pediatric study, Ohta et al. 72 reported a substantial reduction in recurrence rates—33% in patients who received perioperative PP versus 67% in those who did not, indicating a nearly 50% relative risk reduction. Similarly, Gohh et al. 73 evaluated ten high-risk adult transplant recipients who underwent eight sessions of perioperative PP, finding that 70% remained recurrence-free over extended follow-up, compared with an expected graft survival of just 40% without intervention.
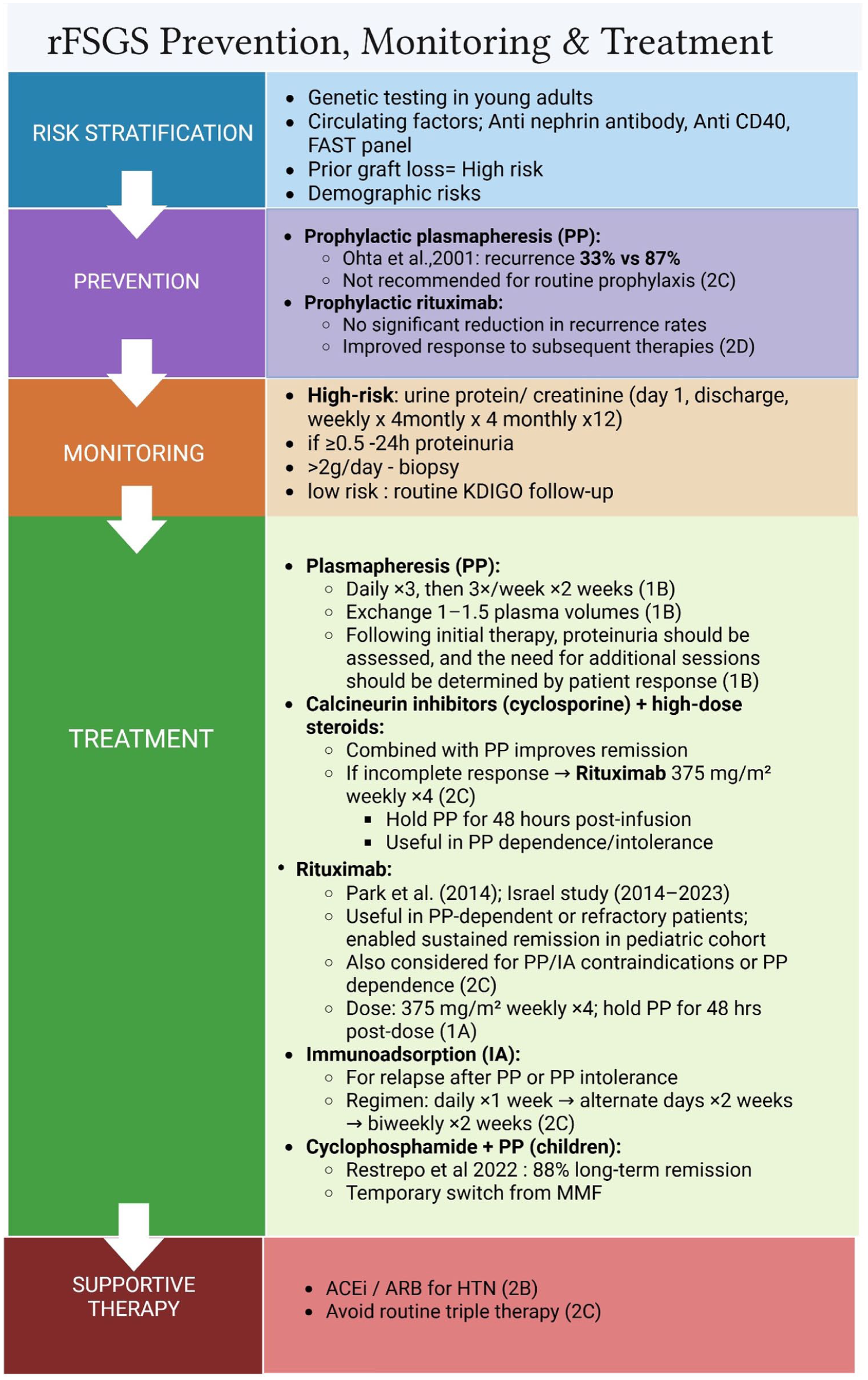
Summary of risk stratification, prevention, monitoring, and treatment strategies for recurrent FSGS, including genetic and biomarker-based risk assessment, KDIGO-aligned surveillance, and evidence-based therapeutic options such as plasmapheresis, calcineurin inhibitors, rituximab, immunoadsorption, and supportive therapy. Created in BioRender. Rayarakula, N. (2025), https://BioRender.com/auu4rh6.
Rituximab, a CD20+ B-cell monoclonal antibody, has also been explored as an adjunct to PP. In a retrospective cohort of adult kidney transplant recipients, Park et al. compared outcomes in patients receiving no prophylaxis, PP alone, and PP with rituximab. While recurrence rates did not significantly differ among groups (22.2% for combination therapy vs 27.7% without prophylaxis), those who did receive rituximab in addition to PP were more likely to respond to subsequent therapies, suggesting potential benefits in recurrence management 74 . Preventative strategies primarily focus on identifying patients at elevated risk—particularly those with early-onset disease or prior post-transplant recurrence. For such individuals, prophylactic protocols involving perioperative PP, with or without rituximab, are often employed despite limited consensus on standardized regimens 72 . The addition to the treatment of another immunosuppressor, as cyclophosphamide, in combination with calcineurin inhibitor and steroid therapy regimen has been highly successful in inducing high remission rates with recurrent FSGS. In a single-center retrospective review by Restrepo et al. of 17 children transplanted due to FSGS, 88% had recurrent disease and were treated for their recurrence with a protocol using PP and augmentation of immunosuppression, including substituting cyclophosphamide for Mycophenolate mofetil (MMF) for 2 months before returning to MMF. Among children with recurrence, 88% achieved a long-lived complete remission. In all but one case, the remissions occurred rapidly and required the addition of rituximab after incomplete remission with PP and cyclophosphamide. All patients who achieved remission stayed in remission remained relapse-free throughout follow-up (median 4 years; IQR 2.2–7.8 years) 75 .
In children, where the data are scarcer, some reports show the FSGS experience, one of them is the 25-year experience by Zilleruelo et al., which examined outcomes in pediatric renal transplantation of patients with FSGS across two treatment eras: Era 1, before the use of daclizumab (anti-IL-2R), and Era 2, after daclizumab. Over the 25 years, 179 pediatric patients younger than 21 years underwent kidney transplantation and FSGS was confirmed in 27. Recurrence occurred in 16 of 28. In Era 1, only 6 of 16 (38%) recurred in the allograft, while 10 of 12 (83%) recurred during Era 2. After a follow-up of 27 ± 15 months, proteinuria decreased by 94 ± 8% from baseline. Based on their findings, the authors suggested a limited course of PP followed by maintenance therapy with AB and MMF improves symptoms and may preserve allograft function. Additional insights from Francis et al. described current therapies and treatment response across four centers in Australia and New Zealand. Their cohort included children younger than 18 years with recurrent FSGS. Median time to first recurrence was 4 days (IQR 2–5 days). The most common therapies were plasma exchange (20/24 patients, 83%), cyclosporin (15/24, 63%), and methylprednisolone (9/24, 38%). Complete remission occurred in 15 patients (63%), partial remission in 2 (8%), and no remission in 7 (29%) patients 76 .
For patients with no response to treatment with PP and high dose of cyclosporine, recent data provide insight into second-line strategies. A retrospective study evaluated the efficacy and safety of early PP followed by rituximab for inducing and maintaining remission in rFSGS. Between 2014 and 2023, 8 of 65 pediatric kidney transplant recipients with idiopathic FSGS underwent kidney transplantation. All patients achieved partial or complete remission with PP, with response times ranging from 8 to 379 days (median 13 days). Rituximab was administered to five PP-dependent patients; three patients sustained remission, while the remaining two showed improved proteinuria and reduced PP frequency. Reported adverse effects included one case of rituximab-induced serum sickness and one mild allergic reaction. One patient lost the graft secondary to humoral rejection, but no grafts were lost to rFSGS. Early PP followed by rituximab therapy effectively induces remission in most post-transplantation rFSGS cases, is well tolerated, and prevents graft loss 9 . The ongoing PRI-VENT FSGS randomized controlled trial (NCT03763643), which includes both pediatric and adult patients, is currently assessing the efficacy of PP plus rituximab compared with standard care, with results expected in late 2026 65 .
Together, current evidence supports early and aggressive intervention with PP in high-risk patients, with rituximab offering additional benefit in selected cases 72 . Further data from prospective trials will be critical in shaping more definitive treatment and prevention guidelines for rFSGS.
According to KDIGO, candidates with primary FSGS should not be excluded from kidney transplantation; however, the risk of recurrence should be considered and discussed with the candidate (1B). Loss of a prior graft due to recurrent FSGS indicates a high risk of recurrence upon subsequent transplants and should be a key factor in determining candidacy (Not Graded). KDIGO also recommends that genetic testing be performed for children and young adults with primary FSGS to inform the recurrence risk (2C). Routine pre-transplant plasma exchange or rituximab reduces the risk (2D).
PP: Early initiation of PP in patients with nephrotic-range proteinuria following transplantation is recommended. The suggested regimen is daily PP for 3 days, followed by three sessions per week for 2 weeks (total nine treatments) [1B]. We suggest exchanging 1–1.5 plasma volumes, using citrate or heparin anticoagulation, with replacement by human albumin or hemofiltration solution; fresh frozen plasma is used as replacement fluid if plasma fibrinogen is low [1B]. Following initial therapy, proteinuria should be assessed, and the need for additional sessions should be determined by patient response [1B]. Routine use of PP for prophylaxis in high-risk patients is not recommended [2C].
Immunoadsorption: Immunoadsorption may be considered in patients who respond to initial sessions of PP but later relapse. It may also be used as a second-line treatment in those who develop side effects to therapeutic PP [2C]. The recommended protocol involves exchanging 2.5–3 plasma volumes daily for 1 week, followed by tapering to alternate-day sessions for 2 weeks, and then biweekly sessions for another 2 weeks [2C].
Rituximab: We recommend the use of rituximab in patients who have a contraindication to PP or immunoadsorption. Rituximab should be administered as an intravenous infusion at a dose of 375 mg/m2 weekly for 4 weeks [2C]. Screening for hepatitis B and C and human immunodeficiency virus before administration of rituximab is recommended [2B]. PP should be withheld for 48 h after each rituximab infusion, to prevent drug removal [1A].
Cyclosporine: We recommend considering cyclosporine, in combination with high-dose steroids and apheresis procedures, as a first-line drug for the management of FSGS [2C].
Renin-angiotensin system blocking agents: We recommend the use of angiotensin-converting enzyme (ACE) inhibitors or angiotensin receptor blockers in rFSGS hypertensive patients [2B]. However, evidence of the efficacy of triple therapy for rFSGS is limited and is not recommended for routine use [2C].
Discussion
Reported recurrent FSGS rates, although they range from 30% to 40% overall, may reach up to 86% in pediatric patients with idiopathic resistant nephrotic syndrome (SRNS) 71 . Outcomes are influenced by pretransplant history, ethnicity, and variability in recurrence, underscoring their heterogeneous nature. To improve preventive and treatment strategies, subcategorization of rFSGS is recommended.
Ex vivo tissue modeling and recurrence prediction using CPFs each represent unique opportunities for early prevention and treatment. These approaches demonstrate moderate predictive accuracy that may further be enhanced when integrated in a multimodal framework. Ex vivo tissue models in particular offer opportunities to individualize induction therapy by monitoring recurrence activity generated by recipient plasma samples prior to transplantation. Furthermore, they represent a noninvasive means of testing recurrence-prevention therapies without risks associated with human trials.
Therapeutic strategies, including PP, immunoadsorption, calcineurin inhibitors, and rituximab, remain the cornerstone of management. Early treatment with PP in combination with rituximab or cyclophosphamide has the best chance for inducing remission. Routine nephrectomy before transplantation is not advised, but special attention should be given to patients with prior recurrence, as they are at the highest risk.
Another therapeutic option is Liposorber D, a hemoperfusion device that selectively removes lipoproteins such as low-density lipoprotein (LDL) and triglycerides. Preliminary data demonstrate promise in treating drug-resistant FSGS as well as its ease of setup, shorter treatment times, and selective lipid adsorption, which help mitigate podocyte injury 76 .
Despite these advances, recurrent FSGS still poses a threat to graft survival. Prospective studies are needed to standardize diagnosis, prevention, and treatment strategies. Integration of novel therapies, biomarker assays, and genetic testing holds the greatest promise for improving long-term outcomes in the future.
Conclusion
Recurrent FSGS following kidney transplantation remains a major cause of allograft failure and a persistent clinical challenge. Idiopathic forms recur in nearly one-third of cases, often shortly after engraftment, and carry a substantial risk of graft loss. The underlying mechanisms driving recurrence are multifactorial and complex, involving genetic susceptibility, circulating pathogenic mediators, and immune dysregulation.
Emerging research highlights the value of genetic and molecular profiling in risk stratification. Researchers are examining mutations in monogenes, such as APOL1 variants, and mutations affecting slit diaphragm proteins, such as nephrin and podocin, for their predictive value in FSGS recurrence. Understanding these genetic predispositions enables targeted preventive strategies and improved patient-specific transplant protocols. Recent studies have emphasized the utility of biomarkers in early diagnosis and management of recurrent FSGS. Investigations of these markers, such as anti-nephrin antibodies and CPFs, may aid in early detection and facilitate personalized therapeutic approaches. Future efforts should focus on integrating multi-omic data to improve recurrence prediction; developing standardized multicenter registries; validating emerging biomarkers such as anti-nephrin, anti-CD40, and suPAR; and designing targeted therapeutic trials based on immune–podocyte injury mechanisms.
Although FSGS recurrence continues to pose a significant graft failure burden, continued research into its genetic, immunologic, and molecular foundations holds significant promise. Future studies should focus on large-scale clinical trials, a deeper exploration of the pathogenic mechanisms, and enhanced biomarker development to optimize prevention and treatment strategies to improve outcomes for kidney transplant recipients affected by recurrent FSGS.
Footnotes
Acknowledgements
Not applicable.
Ethical Considerations
Not applicable.
Author Contributions
Z.O., D.J, T.G., L.K., S.B, and N.R. contributed to the literature search, figure development, and drafting of the manuscript. M.A., J.M.R., and R.R contributed to critical revisions and supervision. R.R provided expert review and final manuscript approval. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Not applicable as this review article does not contain original datasets.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.

