Abstract
Tubal factor infertility remains a major cause of female reproductive dysfunction. Current therapeutic options, such as surgical repair or
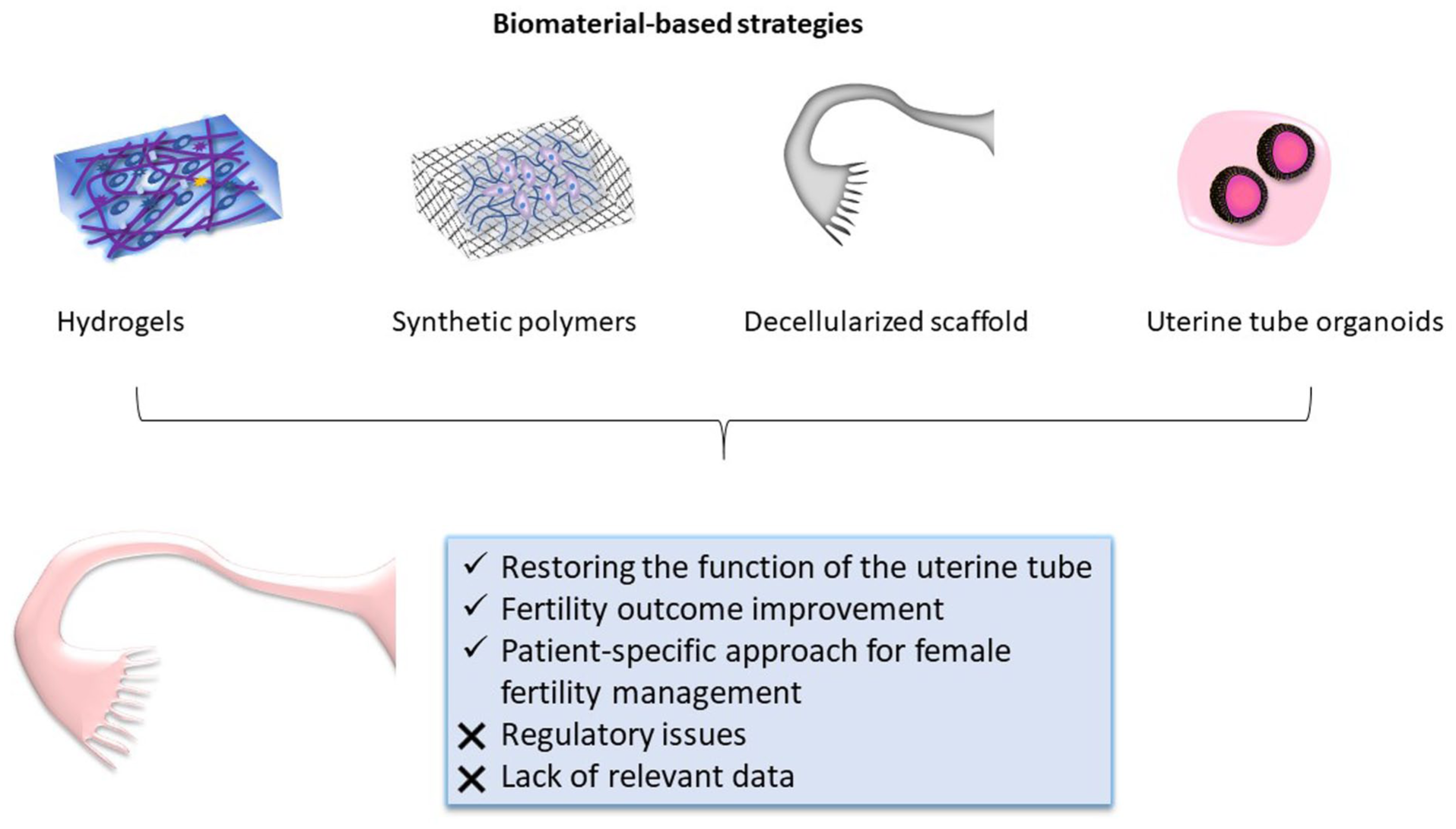
Introduction
The female reproductive system comprises complex anatomical structures responsible for reproduction. The correct interaction and regulation of its major organs, such as the ovaries, uterine tubes, uterus, cervix, and vagina, are essential for maintaining women’s reproductive health 1 . The female reproductive system is responsible for the production of oocytes, the process of fertilization, fetal development, and childbirth. This system functions as a complex network that is directly influenced by endocrine signaling, specifically hormones present in the bloodstream 2 . Endocrine disruptions are directly linked to female fertility dysfunction and can also cause a range of other gynecological conditions, such as polycystic ovary syndrome (PCOS), endometrial hyperplasia, endometriosis, pelvic organ prolapse, reproductive cancers, and so on. In addition, pathologies affecting the women’s reproductive system account for one-third of diseases that have a significant impact on women’s overall health. Unfortunately, their prevalence has been on the rise in recent years 3 . Pharmacological therapy, combined with surgical interventions, represents a conventional treatment approach. However, these strategies often do not yield satisfactory results and can cause adverse side effects 4 . Therefore, there is a pressing need to develop more effective and less harmful therapeutic strategies to preserve normal reproductive physiology in women.
Uterine tubes are 10- to 12-cm-long paired organs of the female reproductive system. They connect the ovaries with the uterus and play a crucial role in the process of fertilization and early embryo development 5 (Fig. 1). The main function of the uterine tubes is to ensure the transport of the oocyte from the ovary to the uterus as well as to provide a suitable environment for fertilization and early embryonic development 6 . Therefore, their patency and functioning epithelium are crucial for successful gameto-maternal interaction 7 . Medical literature describes that 85% of infertile couples have a known cause, and tubal factor infertility accounts for approximately 35% of these cases 8 .

Anatomy and histology of the female uterine tube. Figure was drawn by authors in PowerPoint.
The gold standard for tubal infertility treatment is

Process of uterine tube organoid formation. This process involves a biopsy of the uterine tube tissue and subsequent isolation (mechanical and enzymatic) of uterine tube epithelial cells. The next step consists of culturing these cells in a specific medium supplemented with various growth factors. To complete differentiation into 3D uterine tube organoids, the cell suspension is embedded in a suitable ECM scaffold (Matrigel), where cells begin to proliferate and self-organize into spheroid organoids. Figure was drawn by authors in PowerPoint.
Despite the implementation of established clinical approaches for tubal infertility, the emergence of TERM offers novel therapeutic possibilities with significant potential not only to address this complex reproductive challenge but also to support the restoration of natural conception. Accordingly, the ability to effectively regenerate oviductal tissue after pathological damage, whether caused by inflammation, hydrosalpinx, malignancies, or ectopic pregnancy, constitutes a key strategy in restoring female reproductive function 14 .
TERM integrates the latest advancements in stem cells, their derivatives, biomaterials, and bioactive molecules to facilitate the restoration of damaged tissues. Among these components, biomaterials have garnered particular interest, as ongoing research and development enable their targeted modification using stem cells, exosomes, or other biologically active agents 15 .
Despite their fundamental role in fertilization and early embryo development, the uterine tubes have long remained underrepresented in reproductive research compared to other gynecological organs, such as the uterus and ovaries. It is challenging to precisely determine this phenomenon. However, pointed-out imbalance may be attributed to several factors, including the anatomical inaccessibility of the oviducts or the historical prioritization of endometrial and ovarian pathologies such as cancer, endometriosis, PCOS, or infertility related to endometrial dysfunction16–19. In addition, advances in assisted reproductive technologies have shifted scientific attention away from restoring tubal function toward bypassing it altogether. Consequently, progress in understanding the biology, regeneration, and pathology of the uterine tubes has lagged, creating a significant knowledge gap that limits the development of targeted therapeutic interventions.
While TERM has been widely explored within the scope of female reproductive health, most research efforts have predominantly targeted organs other than the oviducts. This is why the presented article focuses exclusively on the potential of oviductal tissue engineering, specifically on the application of biomaterials with regenerative capabilities.
Materials and methods
This manuscript presents a comprehensive review of recent advancements in uterine tube TERM, with particular emphasis on the application of biomaterials, organoids, and 3D bioprinting. The literature review was conducted using the PubMed database between 27 June and 30 July 2025, employing search terms including natural polymers, synthetic polymers, hydrogels, organoids, oviducts, uterine tubes, regeneration, 3D bioprinting, as well as their combinations. Additional pertinent publications were identified through other databases, such as Google Scholar and Scopus (Elsevier). Only peer-reviewed articles published in English were included. Publications primarily addressing preclinical or clinical studies relevant to uterine tube regeneration were selected for analysis. Initially, the dataset was screened for duplicates (n = 13), after which all retrieved manuscripts were manually reviewed to verify their accuracy. Although articles primarily focused on other reproductive organs were excluded from the dataset, several selected studies addressing tissue engineering strategies for related organs are mentioned throughout the manuscript to provide broader context and enhance the conceptual completeness of the topic. Duplicates, commentaries, editorials, and conference proceedings without full text were also omitted. Through a screening process, we identified 42 publications exclusively focused on uterine tube TERM and analyzed them in detail. Presented review provides a thorough evaluation of natural and synthetic polymers, hydrogels, organoid systems, and 3D bioprinting, emphasizing their translational potential for promoting uterine tube regeneration and improving female reproductive outcomes.
Results
Biomaterials for uterine tubes regeneration
Uterine tubes are highly specialized and anatomically complex structures that play an essential role in female reproduction, facilitating oocyte capture, fertilization, and early embryo transport 20 . Within the framework of TERM, successful restoration or reconstruction of the oviduct must account for the organ’s distinct regional anatomy, specialized cellular composition, and dynamic physiological environment. Anatomically, the uterine tube is divided into four segments: the infundibulum, ampulla, isthmus, and intramural segment, each exhibiting unique histological architecture and region-specific functional roles. These differences include variations in epithelial cell types, mucosal folding, muscular layering, and ciliary activity 21 . Consequently, the design and selection of biomaterials and scaffolds for uterine tube regeneration should reflect these regional characteristics to support proper physiological function and tissue integration.
Natural biomaterials for uterine tubes regeneration
In tissue engineering, natural polymer-based scaffolds have gained significant attention due to their excellent biological properties, mainly reflected in biocompatibility, bioactivity, biodegradability, and ability to mimic the extracellular matrix (ECM) of the native tissues
22
. In addition, these materials facilitate favorable cell–material interactions, which can further enhance cellular proliferation and promote lineage-specific differentiation
23
. On the other hand, natural polymer-based matrices also exhibit unfavorable characteristics, such as poor mechanical strength and stability due to a rapid biodegradation rate. This might limit their application in dynamic
Collagen scaffolds
Collagen is the most abundant protein in mammals and a key structural component of ECM. Its widespread use in biomedical applications stems from its excellent biocompatibility and favorable biological properties
26
. Collagen-based scaffolds have consistently demonstrated enhanced cellular responses, including improved cell attachment, migration, proliferation, and differentiation
27
. Moreover, collagen can be easily modified or combined with bioactive molecules to further promote tissue regeneration. However, its practical application is limited by high production costs and rapid biodegradation
28
. These drawbacks can be mitigated by blending with other polymers, particularly synthetic ones, to improve its mechanical stability and control degradation rates
29
. A pioneering study regarding collagen-based scaffolds was conducted by Venkataraman et al.
30
. A collagen matrix was applied to culture rabbit uterine epithelial cells under
Decellularized scaffolds
Decellularized scaffolds are obtained from native tissues or organs in which all cellular components have been removed while preserving the structure, composition, and mechanical properties of ECM 31 . Their close biological resemblance to native tissue makes them highly attractive for TERM applications. In addition, they are biocompatible and support neovascularization during tissue regeneration 32 . However, incomplete decellularization, mechanical fragility, challenges in sterilization, and difficulties in large-scale production remain major drawbacks that limit their standardized clinical use 33 . Current research is therefore focused on addressing these limitations, particularly by optimizing decellularization protocols and improving the mechanical stability of the resulting scaffolds
Martínez-López et al. evaluated the biological characteristics of decellularized porcine oviduct ECM obtained through the immersion–agitation method 34 . This native-tissue-derived scaffold was assessed in the context of potential repair of reproductive organs. Outcomes revealed that the chosen decellularization protocol achieved satisfactory results, and crucial components of ECM, which in the case of uterine tubes are collagen I and III, proteoglycans, laminin, glycoprotein, and elastic fibers, were preserved. A novel approach was studied by testing the scaffold’s biocompatibility and biotoxicity by co-cultivation with spermatozoa. Results showed that sperm viability and motility were maintained, thus confirming the safety of the decellularization process, as well as the favorable biological response of the oviduct-based scaffold. Decellularization protocols were also assessed by Sanguansook et al. in order to harvest a natural collagen-based scaffold derived from organs of the female upper genital tract (animal model) 35 . Three different approaches, principally based on the number of decellularization cycle repetitions, were tested. The main decellularization agents were 0.1% sodium dodecyl sulfate and 1% Triton X-100 (protocols 1 and 2). The third protocol also included deoxyribonuclease. Conclusively, the outcomes revealed that incubation with deoxyribonuclease was the most efficient method for decellularization in terms of preserving DNA residues and ECM. However, in the case of oviducts, several components of the ECM were decreased. Authors also tested cytotoxicity by sperm-scaffold interaction and concluded that additional washing after the decellularization process is crucial in terms of enhancing safety and biocompatibility of the material. In a broader context, collagen scaffolds have also demonstrated promising regenerative capacity for reconstructing hollow organs, owing to their biocompatibility and structural resemblance to the native ECM 36 . Moreover, a crucial biomechanical characteristic, reversible radial expansion, has been observed. Maintaining this property is particularly important in the context of uterine tube tissue engineering, as it facilitates oocyte and embryo transport, which are contingent upon the peristaltic activity of the oviductal wall and the proper functioning of the cilia.
Besides collagen, several other natural polymers, including gelatin, chitosan, alginate, and hyaluronic acid, have been explored for applications in uterine and ovarian TERM 37 . These materials have been investigated as potential carriers for drugs, exosomes, or anti-inflammatory molecules 38 . For instance, gelatin/polycaprolactone (PCL) electrospun membranes have been evaluated in an animal model for the restoration of damaged endometrial structure and function. Histological analyses, gene expression of estrogen receptors and tumor necrosis factor (TNF), uterine morphology, and fertility outcomes were assessed. The results demonstrated successful regeneration of the endometrium, characterized by increased thickness, enhanced proliferation of epithelial and stromal cells, neovascularization, elevated expression of estrogen receptor α, and reduced TNF expression. The uterine cavity morphology was preserved, and comprehensive tissue regeneration resulted in improved fertility in female rats 39 . Another experimental study identified chitosan oligosaccharides as effective immunomodulators with a protective effect on ovarian tissue 40 . Similarly, alginate-based and hyaluronic acid–based hydrogels exhibited pronounced anti-inflammatory properties, particularly in the prevention of adhesion formation following surgical interventions41,42.
Restoration of fertility ultimately depends on the proper function of the reproductive organs as an integrated system. Therefore, research focused on other reproductive tissues can provide valuable insights applicable to the regeneration and functional recovery of damaged uterine tubes.
Synthetic biomaterials for uterine tubes regeneration
Scaffolds based on synthetic polymers are also widely utilized in TERM due to their high tunability, reproducibility, and mechanical strength 43 . Compared to natural polymers, synthetic materials, such as polylactic acid (PLA), polyglycolic acid (PGA), and PCL, offer precise control over biodegradation, porosity, and mechanical properties, making them well suited for applications that require structural integrity or long-term support 44 . In addition, their consistent quality and ease of large-scale production reduce batch-to-batch variability and facilitate regulatory approval processes 45 .
PLA is a thermoplastic aliphatic polyester derived from renewable resources such as corn starch or sugarcane 46 . Among its most notable characteristics are excellent biocompatibility, well-controlled biodegradability, and high mechanical stability 47 . PLA has been extensively employed as a scaffolding material for both hard- and soft-tissue engineering applications 48 . However, its inherent hydrophobicity remains a major limitation, as it negatively affects cellular attachment and overall biological performance. To address this drawback, surface modification of PLA scaffolds or blending with natural and other synthetic polymers is commonly utilized to enhance hydrophilicity and bioactivity 49 . PGA is a biodegradable aliphatic polyester well known for its good biocompatibility 50 . It has been successfully used in clinical practice, particularly in resorbable sutures, and represents a promising candidate for applications in skin and neural tissue engineering51,52. Due to its hydrolytic instability, which leads to a relatively fast degradation rate, PGA is frequently copolymerized with PLA to improve its mechanical properties and to achieve a more controlled degradation profile 53 . PCL is a semi-crystalline, biodegradable polyester characterized by high flexibility, slow degradation rate, and excellent processability 54 . These properties make PCL particularly suitable for use in additive manufacturing techniques, including 3D printing and bioprinting, thus enabling its application across a wide range of TERM fields 55 .
Synthetic scaffolds often lack inherent bioactivity and often require surface modification or the incorporation of bioactive molecules to support efficient cellular response. Furthermore, some synthetic polymers can produce acidic degradation byproducts that may induce
According to our knowledge, it appears that there are currently very few peer-reviewed studies specifically detailing the application of synthetic polymer scaffolds in uterine tubes. However, certain synthetic polymers such as PCL, PLA, and PGA have been extensively utilized in the engineering of other reproductive tissues, notably the uterus and vagina. For instance, a study by Hanuman et al. developed a PCL-based scaffold for uterine tissue engineering, demonstrating favorable mechanical properties and biocompatibility 59 . Similarly, porous poly-l-lactide-co-ɛ-caprolactone (PLCL) scaffolds have been employed in vaginal tissue regeneration, offering controlled degradation rates and structural support conducive to tissue remodeling60,61. In addition, the chemical modification of a PCL nanofibrous scaffold for uterine regeneration was also evaluated. In this study, the scaffold surface was functionalized with maltose and subsequently analyzed for its biological behavior and mechanical properties. The results demonstrated that the modified structure provided a favorable environment for the attachment and proliferation of human uterine fibroblasts. Overall, the maltose-conjugated electrospun PCL scaffold exhibited suitable characteristics for potential application in uterine tissue engineering 62 . These findings may also prove beneficial and transferable to the field of uterine tube tissue engineering within the scope of TERM.
Hydrogels for uterine tubes regeneration
Hydrogels are defined as three-dimensional (3D) hydrophilic polymeric networks with the enhanced ability to absorb and retain large amounts of water while maintaining structural integrity 63 . Their application in TERM is attractive due to their excellent biomimetic properties, biocompatibility, and also biodegradability 64 . Moreover, properties such as moldability and injectability enable their use in additive manufacturing technologies, making them promising candidates for personalized medicine 65 . The connection between hydrogels and their application in uterine tube tissue engineering lies in their ability to recapitulate the physicochemical and biological properties of the native ECM 66 . Their highwater content, tunable mechanical properties, and biocompatibility create a favorable microenvironment that supports cellular adhesion, proliferation, and differentiation of both epithelial and smooth muscle cells, which are the key components of uterine tube histology 67 . Moreover, the injectability and moldability of hydrogels facilitate minimally invasive delivery and enable the fabrication of complex, tubular scaffold structures that mimic the anatomical and functional features of the oviduct 68 . Hydrogels can also be functionalized to incorporate bioactive molecules such as growth factors, cytokines, or stem cells, thereby enhancing tissue regeneration and functional restoration 69 . These characteristics position hydrogels as highly suitable scaffolding materials for the reconstruction and repair of uterine tubes following injury, disease, or sterilization procedures.
Ribes Martinez et al. 70 evaluated a bovine decellularized ECM (dECM)-based hydrogel as a bio-ink for oviductal bioprinting. To assess cytocompatibility, oviductal cells were encapsulated within the hydrogel, and cell viability and proliferation were monitored over a 2-week culture period. The results demonstrated that the addition of alginate was crucial for achieving the desired mechanical properties. The engineered ECM-derived hydrogel exhibited suitable biocompatibility, favorable biomechanical characteristics, and effective support for cellular responses. Although a relatively low porosity was observed, the overall findings suggest that this hydrogel-based scaffold holds significant promise for advancing uterine tube tissue engineering. Increasing the porosity of hydrogel-based scaffolds improves their biomimetic properties, as well as the diffusion of nutrients and oxygen transport to cells located within the inner layers of the construct. Another critical factor is the control of hydrogel degradation mediated by the seeded cells. For instance, the addition of 1% alginate has been shown to enhance the structural stability of cell-seeded hydrogels. However, this modification often compromises cell viability. Therefore, identifying the optimal ratio of polymeric components is essential to maintain a balance between mechanical stability and biological performance.
Oviductal dECM was utilized for
To study the morphological and physiological properties of the uterine tubes, free-floating collagen-based hydrogels were utilized
73
. Together with bovine oviductal epithelial cells, tissue-mimicking
A thermosensitive hydrogel combined with mesenchymal stem cells (MSCs) was evaluated for its potential to repair the uterine tube wall in an animal model 74 . The stem cells, isolated from dental pulp tissue, were incorporated into a PF127 hydrogel matrix. Cellular characterization confirmed that these dental pulp stem cells possessed MSC-like features, including notable immunomodulatory and pro-angiogenic properties. The hydrogel exhibited excellent cytocompatibility, and when enriched with stem cells, it significantly enhanced uterine tube repair and regeneration by modulating the inflammatory response, promoting angiogenesis, and restoring epithelial function.
The summarized results concerning reviewed polymers in the context of uterine tube tissue engineering, including their benefits, drawbacks, and applications, are detailed in the accompanying table (Table 1). Despite promising advancements, challenges such as mechanical stability, immune response, and functional integration remain. Therefore, there is significant potential and necessity for further research to optimize these materials and develop novel hybrid approaches that better mimic the complex microenvironment of the uterine tubes, ultimately improving clinical outcomes.
Summarization of the advantages, disadvantages, and applications of natural polymers, synthetic polymers, and hydrogels utilized in uterine tube tissue engineering.

dECM: decellularized ECM; ECM: extracellular matrix; OviECM: oviductal ECM; MSCs: mesenchymal stem cells; PLGA: poly(lactic-co-glycolic acid).
Organoids
Nowadays, uterine tube organoids are considered to be one of the best 3D systems providing crucial experimental models for investigating various pathologies, including high-grade serous ovarian cancer. Organoids can be derived from adult stem cells (ASCs) or induced pluripotent stem cells (iPSCs). IPSC-derived organoids have the capacity to differentiate into various cell types of the three germ disk layers (endoderm, mesoderm, ectoderm), thanks to their pluripotency. Therefore, iPSC-derived organoids are more favorable than organoids derived from ASCs
75
. Generally, the generation of organoids starts with the target differentiation of ASCs/iPSCs into specific cell lines, and subsequently, the cell culture is supplemented with specific growth and signaling factors that modulate wingless-related integration site (Wnt), Notch, tumor growth factor beta (TGF-β), epidermal growth factor (EGF), and fibroblast growth factor (FGF) signaling pathways. Under these conditions, pluripotent stem cells undergo a process similar to organogenesis in a dish, enabling them to form 3D organ-like structures
76
. Without a doubt, the key to successful organoid formation is the proper combination of the mentioned factors within the expansion medium (Fig. 2). Organoids derived from tissue-derived cells are composed mainly of epithelial tissue cells. In the case of the uterine tube, the dominant part of the organoid is made up of epithelial secretory cells and ciliated cells. Among the first models was the “
Another research group generated uterine tube organoids from the epithelial layer of the human uterine tube
79
. The samples were obtained after salpingectomy from the fimbria of the oviduct. The first step of epithelial cell processing involved their cultivation on pre-coated six-well dishes with Matrigel. After 14 days, colony formation was observed. Subsequently, the large cellular spheres were differentiated into organoid cultures, which were cultivated in a specific medium containing the aforementioned factors, including EGF, FGF-10, noggin, Wnt3a, along with the addition of nicotinamide, Y-27632, and RSPO1. As an ECM, Matrigel was used again. The organoid was cultivated in conjunction with MSCs isolated from uterine tube stroma and human umbilical vein endothelial cells (HUVEC). The primary population of human epithelial cells from the uterine tube was also used by Dai et al.
80
to establish the uterine tube organoid. These organoids served as a model of a particular malignancy carrying mutations TP53 and RAD51D, which are typical for an early stage of high-grade serous ovarian cancer. Such an organoid exhibited higher cell proliferation and an elevated IL-17 signaling pathway, typical of tumor progression, making it an applicable
As mentioned earlier, iPSC-derived organoids possess better differentiation potential than ASCs. The research group of Yucer et al.
81
generated iPSCs from healthy individuals and patients with epithelial ovarian cancer carrying pathogenic BRCA1 mutations (BRCA1
Another essential part of this process is the ECM, in which stem cells are embedded. Among the most used ECMs are the commercially available Matrigel or Geltrex, which can mimic a specific tissue microenvironment 82 .
However, the widely used Matrigel poses a significant challenge for the long-term cultivation of organoids due to its instability and insufficient content, which lacks many ECM features of the uterine tube microenvironment. Therefore, sooner or later, the Matrigel as ECM for organoid formation will need to be replaced by a more suitable alternative, such as a porous collagen scaffold, artificial hydrogels, or engineered polyethylene glycol (PEG) hydrogels, which have been used in endometrial organoids83,84.
Similarly, uterine tube organoids serve as an
Three-dimensional bioprinting possibilities
Unlike organoids, 3D bioprinting offers a more precise way to organize different cell types within a biomaterial layer by layer, forming a complex structure with a microarchitecture resembling the native one. Among the first researchers to develop a 3D-printed technique combined with microfluidics to create a uterine tube model was Ferraz et al.
85
. The authors printed a U-shaped, 3D tubular-like insert using a multi-arm acrylate-based resin on a stereolithographer, enabling independent double perfusion while maintaining tubular morphology. The seeded bovine epithelial uterine tube cells were cultured in an air-liquid interference, which helped them to acquire and maintain apical and basolateral polarization for at least 6 weeks. However, further studies are necessary to investigate the ability of this construct to serve as a proper 3D uterine tube model. Most recently, Fontes et al. developed an
Generally, forming a 3D bioengineered uterine tube model is challenging due to the organ’s complexity, including its epithelial, mucosal, and muscular layers, as well as regional differences (fimbriae, ampulla, isthmus). At the histological level, achieving ciliary beating and coordinated muscular contraction is also very challenging. Last but not least, the tissue constructs of this size require a functional blood supply, which remains a significant obstacle to be addressed in future research.
Future directions
Despite extensive research and discourse on female infertility, the uterine tube remains a relatively understudied structure within the domains of TERM. On the other hand, current research on biomaterials is rapidly advancing, offering smart materials with the ability to regulate and directly influence pathological processes in affected tissues
87
. For these reasons, advanced biomaterials might provide an effective tool for oviductal regeneration and reconstruction. Current technologies enable the engineering of biomimetic hydrogels with the capacity to mimic the native oviduct’s ECM, thereby supporting epithelial cell differentiation, cilia formation, and secretory function88–90. The Oviduct-on-a-Chip approach facilitates the study of highly delicate processes, such as fertilization, gamete transport, or embryo-maternal crosstalk. In addition, a study conducted by Ferraz et al. introduces this approach as an accurate platform that better mimics the
Both natural and synthetic scaffolds can be modified by various bioactive agents, which can positively affect the restoration of damaged tissue
94
. In the context of tubal factor infertility, materials capable of regulating and healing inflammation processes seem very promising. According to recent studies, the leading cause of tubal factor infertility is blockage of the uterine tubes, most often resulting from sexually transmitted infections such as
Polymer-based scaffolds have also been studied as devices for permanent female sterilization 105 . On the other hand, loading with accurate bioactive agents, such as anti-inflammatory molecules, hormones, growth factors, antioxidants, enzymes, and matrix modulators, could enhance the function of the female reproductive tract. However, the current status of research in this area is lacking.
The integration of artificial intelligence (AI) into scaffold engineering procedures may also present a significant future direction in oviductal regeneration. This approach could improve crucial properties needed for successful
Conclusion
Biomaterial-based strategies offer a promising approach for restoring the structure and function of the uterine tubes, addressing one of the major causes of female infertility. Natural and synthetic polymers, hydrogels, and organoid systems have demonstrated encouraging results in mimicking the native oviductal environment and supporting epithelial differentiation, angiogenesis, and regeneration. However, achieving full functional restoration remains a challenge. Continued interdisciplinary research integrating advanced biomaterials, stem cell technologies, and bioprinting approaches will be essential to translate these experimental findings into clinically viable therapies that can ultimately enhance fertility outcomes.
Footnotes
Ethical Considerations
Not applicable.
Author Contributions
M.C. designed the review structure and conducted the literature review regarding natural and synthetic polymers as well as hydrogels. M.K. performed a literature search concerning the anatomy and histology of the uterine tube and organoids derived from the uterine tubes. M.K. prepared original figures. Both authors contributed to the result analysis, article writing revising, and finalization of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the EU NextGenerationEU through the Recovery and Resilience Plan for Slovakia under the project No. 09I0X-03-V04-00290.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The references used for the writing of this manuscript are publicly available.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
Not applicable.
