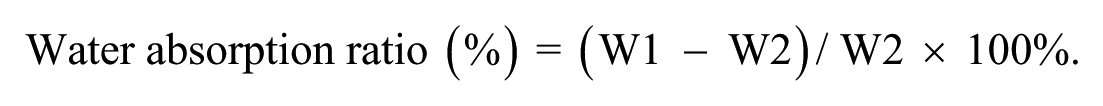Abstract
Islet transplantation represents one of the most promising therapies for curing type 1 diabetes, yet it encounters significant challenges, including early islet damage due to inflammation and hypoxia, which complicate engraftment and survival within the host. It is urgent to develop new strategies to improve islet grafts survival. In this study, we developed a microporous silk fibroin scaffold loaded with liraglutide (SF-Lira). It can provide mechanical support for the islets seeded on its surface and prevent excessive aggregation. The SF-Lira scaffold significantly protected the islets from inflammatory injury, notably enhancing islet viability. In the syngeneic islet transplantation model, SF-Lira significantly improved transplantation outcomes at the epididymal fat pad (EFP) site, with a higher percentage of mice achieving and maintaining normoglycemia compared to the control. Histological analysis revealed superior graft morphology in the SF-Lira group. Our study provides new insights into the application of SF scaffold in islet transplantation and shows potential for clinical translation in extrahepatic islet transplantation.

Introduction
Type 1 diabetes (T1D) is an autoimmune disease characterized by the specific destruction of β cells, which is caused by a combination of genetic and environmental factors 1 . Islet transplantation stands as the most promising treatment for T1D, offering patients a route to evade hyperglycemia and regain physiological blood glucose regulation 2 . In some experienced medical centers, the median graft survival is more than 5 years3,4. Beyond glycemic control, islet transplantation has been shown to stabilize hemostatic abnormalities and improve cerebral metabolism in T1D patients, mitigating diabetes-related complications 5 .
However, islet isolation causes damage to the internal vasculature and extracellular matrix (ECM), resulting in substantial islet loss and making grafts more susceptible to inflammation and hypoxia post transplantation6–8. To overcome these issues, the use of functional biomaterials to explore new transplantation strategies had gradually become an important approach to improving early engraftment and survival of transplanted islets.
One strategy is to use bioscaffold at extrahepatic sites to support islets and facilitate their survival9–11. Numerous studies have demonstrated the feasibility of improving islet transplantation outcomes by using biomaterials, such as polydimethylsiloxane (PDMS) 12 , poly(D, L-lactide-co-ε-caprolactone) (PDLLCL) 13 , polytetrafluoroethylene (PTFE) 14 , polycaprolactone (PCL) 15 , and poly(lactide-co-glycolide) (PLG) 16 . However, most current bioscaffolds mainly serve the structural support functions and play additional ECM roles for embedding the islets internally10,11,17, which might distance the islets from the recipient’s vasculature and potentially increase the risk of hypoxic injury after islet transplantation 18 . Therefore, designing a biological scaffold with pores smaller than the islets can shorten the distance between the islets and the host tissue while avoiding excessive aggregation. Silk fibroin (SF), as a natural protein derived from Bombyx mori, possess excellent mechanical property, biocompatibility, biodegradability, and low immunogenicity 19 . It can be fabricated into a porous scaffold with a three-dimensional structure. In addition to aiding the exchange of nutrients, the porous structure has been shown to facilitate the loading of active factors and enable their sustained release 20 .
Inflammation occurring in the early stage after transplantation can induce cell death and impair function of the transplanted islets21,22. Liraglutide, a glucagon-like peptide-1 (GLP-1) receptor agonist, widely used in the clinical treatment of diabetes mellitus23,24, exerts significant beneficial effects on β-cell survival by protecting cells against apoptosis25,26. Merani et al. 27 have shown that systemic injection of liraglutide after islet transplantation can significantly shorten the time to normoglycemia and improve glucose tolerance in diabetic animals. In our previous study, we have demonstrated that liraglutide can ameliorate damage induced by oxidative stress and prevent islet apoptosis during cold preservation or hypoxia28,29. Additionally, pretreatment or systemic use of liraglutide can enhance the outcomes of islet transplantation by mitigating the instant blood mediated inflammatory reaction (IBMIR) following intrahepatic transplantation30,31. Nevertheless, it remains uncertain whether liraglutide can augment the efficacy of islet transplantation by attenuating inflammatory responses through localized sustained-release delivery at the epididymal fat pad (EFP) site.
In this study, we aim to design a SF scaffold loaded with liraglutide (SF-Lira) for islet transplantation. The design of this small-pore scaffold loaded with liraglutide offers new islet transplantation strategies and holds significant potential for application.
Materials and methods
Fabrication and characterization of SF scaffolds
The 10 wt% SF solution was prepared according to previously reported method 20 . The solution was then transferred into the tubes and cooled to −20°C at a rate of 1°C per minute using a cell freezing container. After that, the cooled solution was freeze-dried to prepare the SF scaffold. The SF scaffold was subsequently immersed in methyl alcohol to induce crystallization and then rinsed with deionized water. For comparison, an SF scaffold with a 5 wt% concentration was also prepared using the same method.
The microstructure of the scaffold was examined using scanning electron microscopy (SEM, Phenom Pro, Eindhoven, Netherlands). The pore size was determined using the Image-J software.
The water absorption ratio of the scaffold was measured using a weighing method 32 . The water-saturated scaffold was initially weighed (W1), followed by weighing the scaffold again after water removal (W2). The water absorption ratio was calculated using the following formula:
Preparation and characterization of SF-Lira scaffold
Liraglutide was loaded in the scaffolds through physical adsorption according to the following steps 33 . The 10 wt% SF scaffolds were directly cut into appropriate sizes as needed and washed with phosphate buffered saline (PBS). They were then irradiated under ultraviolet (UV) light for 30 min. After the scaffolds dried naturally, liraglutide solution was added onto them and incubated at room temperature for 1 h. The volume of the added liraglutide solution was determined based on the water-absorption capacity of the 10 wt% scaffold.
Fluorescence microscopy was used to detect the effective loading of liraglutide in the scaffold (5 mm diameter, 0.5 mm height). Liraglutide was labeled with a Cy3 NHS eater (IC8220; Solarbio Science & Technology Co., Ltd, Beijing, China) and purified using PD Minitrap G-10 columns to remove unbound Cy3 (18918010, Cytiva, Uppsala, Sweden). Fluorescence images were acquired by a confocal microscope (A1+, Nikon, Tokyo, Japan).
The mechanical properties of the cylindrical SF-Lira scaffold (diameter of 10 mm, height of 12 mm) were tested at 20%, 40%, and 60% strain using a texture analyzer (TMS-pro, FTC, Virginia, USA) with a speed of 50 mm/min. The elasticity and mechanical stability of the cylindrical scaffold were evaluated using a cyclic compression test. The compression strain was set at 60%, and the compression speed was 50 mm/min.
In vitro release of liraglutide from the SF-Lira scaffold
The dialysis method 34 was used to analyze the release features of liraglutide from the SF-Lira scaffold. Release kinetics were assessed in PBS (pH 7.4) at 37°C with 100 rpm shaking—conditions simulating physiological temperature, pH, and osmolarity. The Lira-loaded SF scaffold (diameter of 10 mm, height of 12 mm), which had been added with 500 μL of liraglutide solution (600 μmol/L), was placed into a dialysis bag (molecular weight cutoff of 8000–12,000 Da, MD1444, Biotopped, Shanghai, China) and submerged in a beaker containing 20 mL of PBS. Release samples (500 μL) were collected at specific time points: 0.5, 1, 2, 3, 4, 5, 6, and 7 days. Fresh PBS was added to the beaker to maintain a constant volume. The experiment was repeated three times. Released liraglutide was quantitated by a UV/Visible spectrophotometer at a wavelength of 201 nm.
Islets isolation and implantation on the surface of scaffold
Mouse islets were isolated from C57BL/6J using collagenase P, as previously described 35 . Briefly, the pancreas was perfused with an enzyme solution containing 0.5 mg/mL collagenase P (11213865001; Roche, Basel, Switzerland), 1% 4-(2-Hydroxyethyl) piperazine-1-ethanesulfonic acid (HEPES) (15630130; Gibco, Carlsbad, CA, USA), and 1.27 mM CaCl2 in D-hanks solution (H1045; Beijing Solarbio Science & Technology Co., Ltd, Beijing, China). The pancreas was gently removed, and the digestion was carried out at 37°C for 11 min, followed by purification by density gradient (Histopaque 1077; Sigma-Aldrich, St Louis, MO, USA).
The isolated islets were uniformly seeded on the surface of the SF-Lira or blank SF scaffolds and placed in the incubator for 1 h. All operations were performed in a sterile environment.
SEM was used to observe the distribution of islets on the surface of the scaffolds. Scaffolds (5 mm diameter × 0.5 mm height) were seeded with 40–60 islets per scaffold for SEM observation. The scaffolds seeded with islets were washed twice with PBS and then immersed in 2.5% glutaraldehyde at room temperature for 30 min. They were subsequently dehydrated in 50%, 70%, 90%, 100% (I), and 100% (II) ethanol, with each concentration used for 15 min. All scaffolds were dried overnight at room temperature, followed by sputter-coating with an Au-Pd layer. Finally, SEM images were taken and analyzed.
To evaluate the supportive role of the scaffold for islets and the cytocompatibility, 40 islets were seeded onto the surface of SF scaffolds (5 mm diameter, 0.5 mm height) and cultured in RPMI 1640 culture medium supplemented with 10% fetal bovine serum (FBS, Biological Industries, Beit Haemek, Israel). The control group consisted of islets alone placed directly in the culture medium. Islets were removed for fluorescein diacetate (FDA)/propidium iodide (PI) staining on days 1, 3, and 5 to assess viability. First, islets were transferred to a 12-well plate containing 1 ml of PBS per well. Then, 10 μL of PI (1.434 mM, Invitrogen, Thermo Fisher Scientific, Waltham, MA, USA) and 10 μL of FDA (46 μM, Invitrogen, Thermo Fisher Scientific, Waltham, MA, USA) were added to each well and incubated for 3 min. Fluorescence images were recorded using an ECLIPSE Ni-U microscope (Nikon, Tokyo, Japan) or IX73 (OLYMPUS, Tokyo, Japan). Image-Pro Plus software was used to quantify the PI (red) and FDA (green) image areas. The viability (%) of each islet was determined by calculating the ratio of FDA positive area to the total area of double staining.
Glucose-stimulated insulin secretion
The Krebs-Ringer bicarbonate (KRB) buffer used for glucose-stimulated insulin secretion (GSIS) had the following composition: 125 mM NaCl, 5.9 mM KCl, 2.56 mM CaCl₂·2H₂O, 1.2 mM MgCl₂·6H₂O, 25 mM HEPES, 1 mg/ml BSA, and 1 mM glutamine (pH 7.2–7.4). Twelve islets were randomly selected and preincubated in 1 mL of low-glucose (1.67 mM) KRB buffer at 37°C for 1 h. Subsequently, the islets were incubated in low-glucose KRB (1.67 mM glucose) and high-glucose KRB (16.7 mM glucose) for 1 h, respectively. Supernatants from both incubations were collected for insulin measurement using the ELISA Kit (MS200, EZassay, Shenzhen, China). Islets were lysed using the radio immunoprecipitation assay (RIPA) lysis buffer to determine total protein content. The glucose stimulation index (GSI) was calculated as the ratio of insulin-secretion level in the high-glucose KRB solution to that in the low-glucose KRB solution. Absolute insulin secretion was normalized to the total islet protein content per well.
Inflammatory treatment
To evaluate the potential protective effects of SF-Lira, 40–60 islets were seeded onto SF-Lira or SF scaffold (5 mm diameter, 0.5 mm height) as described earlier. They were then cultured in RPMI 1640 culture medium with tumor necrosis factor α (TNF-α; 25 ng/mL), interleukin 1β (IL-1β; 10 ng/mL), and interferon gamma (IFN-γ; 100 ng/mL) for 24 h. The control group was cultured directly in the RPMI 1640 culture medium with or without inflammatory cytokines for the same period. Afterward, the islets were removed for FDA/PI staining to assess viability.
Transplantation
Mouse studies were approved by Nankai University Institutional Animal Care and Utilization Committee (approval no. 2023-SYDWLL-000620). Male C57BL/6J mice (6–8 weeks old) were purchased from Beijing Huafukang Biosciences (Beijing, China). Streptozotocin (STZ, 200 mg/kg, Sigma-Aldrich, St Louis, MO, USA) was administered via a single intraperitoneal injection to induce diabetes in C57BL/6 mice at least 1 week before transplantation. Diabetes was confirmed if blood glucose level was ≥20.0 mmol/L on 2 consecutive days, measured using contour plus blood glucose monitoring system (Ascensia, 7600P, Basel, Switzerland) and contour plus blood glucose test strips (Ascensia, 84629325, Basel, Switzerland) after tail tipping.
1.25% Tribromoethanol was administered via intraperitoneal injection at a dose of 20 μL/g body weight to the diabetic receptor mice. Once anesthetized, the abdominal hair was shaved, and the mice were fixed in the supine position. A small incision was made in the left inguinal area, the layers were separated, and the left EFP was fully exposed. The EFP was then picked out with a cotton swab and fully spread out in vitro. The scaffold (5 mm diameter, 0.5 mm height), which was seeded with 150 islets, was placed on the EFP, and the EFP was then folded and replaced within the abdominal cavity. The muscle and skin layers of the incision were sutured separately, the incision was disinfected with iodophor, and the mice were placed in a warming box to recover. The random blood glucose levels and body weight were measured at the indicated time points.
Intraperitoneal glucose tolerance test
Intraperitoneal glucose tolerance tests (IPGTT) were performed at 14 days after transplantation. Every recipient was fasted overnight and then received an intraperitoneal injection glucose dose of 2 g/kg. Blood glucose levels were measured at 0, 15, 30, 60, 90, and 120 min after injection.
Histological analysis
At 70 days after transplantation, the graft-bearing EFPs were collected, fixed, and paraffin embedded. Three mice from each group were randomly selected for immunohistochemistry to determine islet morphology and viability. The embedded EFP tissues were continuously sliced (5 μm thick per slice). Sections more than 50 μm apart were stained with hematoxylin and eosin (G1076, Servicebio, Wuhan, China) and examined for islet graft using the ECLIPSE Ni-U microscope (Nikon). After identifying the slices with the largest islet graft areas, the adjacent slices were selected and incubated overnight with anti-insulin antibody (ab63820, Abcam, Cambridge, UK), along with secondary horseradish peroxidase-conjugated goat anti-rabbit IgG (GB23303, Servicebio, Wuhan, China). Tissue sections were imaged using Olympus APX100 microscope (Olympus, Tokyo, Japan). Image-Pro Plus software was used to quantify and analyze the insulin-positive area.
Statistical analysis
All data results were expressed as mean ± standard error of the mean. A statistical analysis of quantitative data was performed using Student’s t test or one-way/two-way analysis of variance with post hoc Tukey’s test. Statistical significance was defined as a P value less than 0.05.
Results
Preparation and characterization of the SF-Lira scaffold
We prepared the SF scaffold by modified freeze-drying method and investigated the effect of the SF concentration on the pore size of the scaffold. The SEM images revealed that the pore size of scaffold decreased from 168 ± 63 μm to 69 ± 24 μm as the SF concentration increased from 5 wt% to 10 wt% (Fig. 1a, b). Studies indicated that the size of islets varies from 50 to 350 μm 36 . Thus, to facilitate the surface implantation of islets onto the scaffold rather than inside, the SF scaffold prepared by 10 wt% SF solution was used for the next study.

Preparation and characterization of SF-Lira scaffold. (a) Representation SEM images of different concentrations of silk fibroin scaffolds. Scale bar, 100 μm. (b) Statistical data of the pore size of SF scaffolds (n = 3). (c) Water absorption ratio of 10%SF scaffold (n = 3). (d) Representation images of Cy3-labeled liraglutide distributed within the SF-Lira scaffold. (e) Compression stress-curves of SF-Lira scaffold under different strain. (f) Statistical data of compressive stress of SF-Lira scaffold at 20%, 40%, and 60% strain (n = 3). (g) Compressive stress of SF-Lira and SF scaffold at 60% strain. (h) The recovery index of SF-Lira scaffold to its initial compressions. Data were presented as mean ± SEM, **P < 0.01; ****P < 0.0001.
The loading volume of liraglutide solution was determined based on the water absorption capacity (3.55 g/g) of the 10% SF scaffold (Fig. 1c). Then, the liraglutide solution was loaded into the SF scaffold using a physical absorption method. In Fig. 1d, we can see that the Cy3-labeled liraglutide was evenly distributed within the scaffold. The mechanical properties of the SF-Lira scaffold were further assessed through a cyclic compression test. The compressive stress of the SF-Lira scaffold increased from 37.9 ± 1.1 kPa to 108 ± 25.7 kPa as the strain increased from 20% to 60% (Fig. 1e, f). Moreover, we analyzed the mechanical properties of the SF scaffold and SF-Lira scaffold using a cyclic compression test at a strain of 60%, and results showed that SF-Lira scaffold possessed comparable mechanical properties to SF alone (Fig. 1g, h), indicating high self-elasticity and mechanical stability. This remarkable elasticity allowed the scaffold to preserve its initial structure and morphology when exposed to pressure from surrounding tissue, while simultaneously offering robust physical support for subsequent islet implantation.
Interaction between SF scaffold and islets in vitro
SEM imaging was employed to observe the morphology of the islets on the surface of the SF scaffold. Fig. 2a showed that the islets were uniformly loaded on the scaffold surface without significant aggregation. We also found islets established close contact with the scaffold surface in the SEM images, while retaining their spherical morphology. In addition, the islets were relatively independent from each other, with no clumping, and still maintained a smooth morphology after being cultured for 3 or 5 days. In contrast, islets cultured alone in the culture plates showed significant aggregation (Fig. 2b). These results suggested that the micropore SF scaffold was a suitable scaffold that not only supported islets but also provided islets with a favorable microenvironment which may preserve their native architecture and function. Moreover, the islets co-cultured with SF scaffold showed a high viability of live cells (Fig. 2c), indicating the SF scaffold has no significant cytotoxicity.

In vitro interaction of SF scaffolds and islets. (a) Representative images of islets seeded on the 10% SF scaffold or free islets through scanning electron microscopy. Scale bar, 100 μm. (b) Representative images of fluorescein diacetate (FDA)/propidium iodide (PI) staining of islets showing the viability of islets culture alone or co-incubation with the 10% SF scaffolds after 1, 3, or 5 days. Scale bar, 200 μm. (c) Quantitative evaluation of islet viability (n = 3). Data are presented as mean ± SEM.
SF-Lira protected islets against inflammatory cytokines damage in vitro
To further evaluate the effects of liraglutide on islets under inflammatory conditions, islets were cultured in an RPMI 1640 medium supplemented with TNF-α (25 ng/mL), IL-1β (10 ng/mL), and IFN-γ (100 ng/mL) for 24 h to simulate an inflammatory microenvironment. First, we verified the protective effect of liraglutide on islets under inflammatory conditions. After 24 h of stimulation, islet viability decreased significantly in the inflammation group, whereas liraglutide effectively alleviated the damage caused by inflammation (Fig. 3a, b). The glucose stimulation insulin secretion (GSIS) assay was conducted to further evaluate the protective effect of liraglutide on islet function. The results demonstrated that the GSI was significantly reduced in the inflammation-stimulated group. In contrast, the addition of liraglutide during inflammatory factor stimulation significantly improved the insulin-secretion capacity of the islets compared to the inflammation-stimulated group (Fig. 3c, d).

The SF-Lira delivery system protected the survival of islets under inflammatory conditions. (a) Representative images of fluorescein diacetate (FDA)/propidium iodide (PI) staining of islets showing the viability of islets culture with or without liraglutide under cytokines stimulation after 24 h. (b) Quantitative evaluation of islet viability (n = 5). (c) Glucose stimulation insulin secretion (GSIS) analysis of islets with or without liraglutide under cytokines stimulation (n = 8). (d) Insulin secretion at basal (1.67 mM) and stimulatory (16.7 mM) glucose levels (n = 8). (e) Liraglutide release profiles of liraglutide from SF-Lira (n = 3). (f) Representative images of FDA/PI staining of islets showing the viability of islets culture alone or co-incubation with the SF or SF-Lira scaffolds under cytokines stimulation after 24 h. Scale bar, 200 μm. (g) Quantitative evaluation of islet viability (n = 5). (h) Glucose stimulation insulin secretion (GSIS) analysis of islets under inflammatory conditions (n = 3). (i) Insulin secretion at basal (1.67 mM) and stimulatory (16.7 mM) glucose levels (n = 3). Data were presented as mean ± SEM, *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001.
To clarify local delivery of liraglutide at the transplantation site, we studied its release characteristics from SF scaffolds using an in vitro dialysis method. The result demonstrated that the SF-Lira scaffold can gradually and continuously release liraglutide for more than 1 week under simulated physiological conditions (37°C, pH 7.4, isotonic PBS) (Fig. 3e).
Then, we tested the function of the liraglutide-functionalized scaffold on islets under the same inflammatory treatment. As shown in Fig. 3f, the islets exhibited significant central necrosis and a marked reduction in cell viability under inflammatory conditions. However, the SF group showed an increase in the percentage of live cells. Moreover, a more pronounced protective effect on the islets was observed in the SF-Lira group, where cell viability was significantly enhanced compared to the inflammation group, even surpassing that of the SF alone (Fig. 3g).
Further GSIS assay results showed that SF-Lira significantly upregulated the GSIS in mice islets, evidenced by the significantly higher GSI (Fig. 3h) and higher absolute insulin secretion at high-glucose condition (Fig. 3i).
In vivo co-transplantation of islets with SF-Lira improved transplantation outcomes in diabetic mice
To further understand the function of SF-Lira in islet transplantation, the syngeneic islet transplantation model was employed. The SF-Lira scaffold seeded with marginal islet (150 islets) was transplanted into one side of the EFP (Fig. 4a, b). Long-term blood glucose and body weight monitoring were conducted after transplantation. All transplanted mice survived until the end of observation period, with no major adverse events recorded. Only 66.7% of recipients achieve normoglycemia, and it took longer for these cured recipients to return to baseline values in these two groups receiving islets alone or islets on the SF scaffold without liraglutide. In contrast, the SF-Lira group exhibited a higher proportion of normal blood glucose levels and reached the normoglycemia target earlier than the other two groups. By day 15, all mice of the SF-Lira group had recovered to normoglycemia and maintained it until the graft was removed (Fig. 4c, d). By day 35, the mean random blood glucose level of the diabetic mice in the SF group had returned to the baseline but remained fluctuating, indicating highly unstable blood glucose levels. This effect was also observed in the islets alone group (Fig. 4c, d). After the implant was removed on day 70 post transplantation, all mice reverted to hyperglycemia, confirming that the normoglycemia was induced by the graft. Before transplantation, all mice had similar body weights (22.33 ± 1.70 g). After transplantation, they all showed an upward trend, with the SF-Lira group exhibiting the most significantly increase (Fig. 4e).

SF-Lira scaffold significantly improved the islets transplantation outcomes in diabetic mice. (a) Schematic diagram of diabetic mice model establishment and transplantation. (b) Photograph of SF-Lira + islets embedded in epididymal fat pad (EFP) immediately (left) and 70 days after transplantation (right). (c) Nonfasting blood glucose measurements following the syngeneic transplantation of SF-Lira + islets, SF + islets, and Islets alone, respectively. (d) The diabetes reversal time and percent recovery of diabetes mice for three groups. (e) Body weight measurements for three groups. (f) The intraperitoneal glucose tolerance tests (IPGTT) test and (g) the corresponding area under curve for three groups after transplantation 14 days. The blue asterisks represent SF + islet group versus SF-Lira + islet group, and the orange asterisks represent Islet alone group versus SF-Lira + islet group. Data are presented as mean ± SEM, *P < 0.05; **P < 0.01; ***P < 0.001.
To investigate the function of the graft’s response to blood glucose in each group, IPGTT were performed at day 14 after transplantation. The change of blood glucose at 30 min was the minimal in the SF-Lira group, and at 120 min, it had almost returned to the level before glucose injection (Fig. 4f). This effect was not observed in islets alone or islets-loaded SF group. Additionally, the area under curve (AUC) for the SF-Lira group was also significantly less than that of the other two groups (Fig. 4g). These results demonstrated that the graft in the SF-Lira group significantly improved the function of islet grafts in the diabetic mice.
SF-Lira improves the survival of islet graft in vivo
To further clarify the function of the transplanted islets and their potential mechanism for reversing blood glucose, grafts were collected for histological analysis at 70 days. In the control group, where islets were transplanted alone, the grafts exhibited a smaller insulin immunoreactive area and a loss of sphericity compared to the grafts in the other groups (Fig. 5a, b).

SF-Lira delivery system prevented islet loss and maintained islet morphology. (a) The representative photograph of insulin immunohistochemistry staining for three groups. Scale bar, 100 μm. (b) The mean insulin-positive area of each slide for three groups. Scale bar, 50 μm. Data were presented as mean ± SEM, ***P < 0.001.
Islets seeded on the scaffolds maintained excellent spherical morphology, and most of them were implanted on the top surface of scaffold rather than deep, with a large area in direct contact with adipose tissue (Supplemental material, Fig. S1). This demonstrated the efficacy of the microporous scaffolds we designed for surface seeding of islets. Moreover, the scaffolds have good biocompatibility, and there is no significant fibrotic encapsulation around the islet graft (Supplemental material, Fig. S2).
Discussion
This study aims to utilize functionalized biomaterials (SF-Lira scaffolds) to load islets and apply them to extrahepatic transplantation sites. The continuous release of the liraglutide improved the microenvironment of islet transplantation, thereby enhancing the outcomes of islet transplantation. Our scaffold features an interconnected microporous structure with small pores, ensuring that the islets are implanted on the surface rather than within its internal structure. This design reduces the distance between the islets and the recipient’s vascular vessels and prevents islets clumping. The liraglutide-release experiment confirmed that the scaffold can provide a sustained and gradual release of liraglutide under simulated physiological conditions. Furthermore, the SF-Lira scaffold showed significantly protective effects on islets under inflammatory conditions. Most importantly, we further confirmed that the SF-Lira and islets co-transplantation system effectively reversed diabetes after transplantation into the EFP in a syngeneic mice model.
Greater omentum is one promising alternative site for islet transplantation, having sufficient space to accommodate the islet graft, convenient for graft removal, and facilitating subsequent graft observation37,38. The EFP of mouse, which has properties similar to the human omentum, is often employed as an equivalent transplantation site 39 .
The scaffold we designed can provide islet structural support and enhance islet survival, maintaining the original round morphology, size, and structure of the islets, which have been shown to be crucial for islet survival and function post transplantation40,41. Some scaffold with large pores (>200 μm) embedding the islets internally might distance the islets from the recipient’s vasculature and potentially increase the risk of hypoxic injury after islet transplantation 18 . Moreover, the scaffold surface has been reported to be more conducive to host vascular ingrowth, due to its direct exposure to recipient tissue 42 . Therefore, in our study, we selected the SF scaffold with the smaller pore size and implanted islets on its surface. This design promotes direct contact between the islets and host tissue, enabling an even distribution of islets on the 3D structure’s surface and preventing islet aggregation. Consequently, oxygen and nutrient availability are enhanced during the critical early period post transplantation. Our research findings demonstrate the successful achievement of islet immobilization on the surface of the scaffold. This direct contact means that the pancreatic islets are closer to the recipient tissue in the early post-transplant period, and this closer proximity represents easier access for the graft to oxygen and nutrients from the recipient tissue. Our graft outcomes, including IPGTT and immunohistochemistry results in diabetic mice, supported this conclusion.
Extensive researches indicate that inflammation significantly impacts the efficacy of islet transplantation43–45. Therefore, anti-inflammatory treatment is crucial during islet transplantation. The anti-inflammatory and immunomodulatory properties of GLP-1 receptor agonists have been extensively documented. As highlighted in a recent comprehensive review, liraglutide exerts multifaceted effects on immune cells, including suppression of proinflammatory cytokines, polarization of macrophages toward an anti-inflammatory phenotype, and enhancement of regulatory T cells 46 . So, we chose to functionalize our scaffold with liraglutide because of its anti-inflammatory properties. Finally, the combination of liraglutide and the scaffold optimizes the overall outcomes of islet transplantation. These results further validate the effectiveness of our design.
The SF-Lira scaffold holds significant promise for clinical translation in human islet transplantation. First, SF is a Food and Drug Administration–approved biomaterial with established biocompatibility and tunable degradation, minimizing regulatory barriers 47 . Second, liraglutide is clinically used for diabetes management, supporting its safety profile. Our scaffold’s localized release strategy could mitigate systemic side effects of liraglutide while targeting inflammation at the transplant site. Notably, the EFP model employed here mimics the human omentum—a promising clinical site for extrahepatic islet transplantation. Future studies in large-animal models are warranted to evaluate whether SF-Lira scaffolds can enhance not only graft survival but also systemic metabolic beyond glycemic control.
Although this study demonstrates the potential of the SF-Lira delivery system in islet transplantation, there are still some challenges and future research directions to explore. Strategies such as scaffold miniaturization, modular assembly, or layered implantation could be explored in future research. Moreover, the current design allows bilateral islet seeding, which may improve islet-loading efficiency without increasing implant size. These potential optimizations warrant further investigation in large-animal models to assess clinical feasibility. While our current study focused primarily on the functional and histological evaluation of islet grafts, future investigations are warranted to assess the immunological response state of the transplant site. In addition, to further enhance therapeutic outcomes, we plan to further optimize the design of the scaffold by incorporating various anti-inflammatory or survival-promoting factors.
Supplemental Material
sj-docx-1-cll-10.1177_09636897251374147 – Supplemental material for A microporous liraglutide-releasing silk fibroin scaffold improves islet transplantation outcomes through anti-inflammatory effect
Supplemental material, sj-docx-1-cll-10.1177_09636897251374147 for A microporous liraglutide-releasing silk fibroin scaffold improves islet transplantation outcomes through anti-inflammatory effect by Yixiang Zhan, Xinchen Du, Yan Li, Yingbo Wang, Xiangheng Cai, Runnan Yang, Tingsheng Jiang, Zhaoce Liu, Xueer Yu, Shanshan Lin, Qing Liu, Yingyi Qi, Rui Liang, Na Liu, Tengli Liu, Xiaoyan Hu, Jiaqi Zou, Xuejie Ding, Peng Sun, Houhan Feng, Jiuxia Yang, Lianyong Wang and Shusen Wang in Cell Transplantation
Footnotes
Acknowledgements
We thank NHC Key Laboratory for Critical Care Medicine for providing indispensable assistance during the study.
Ethical approval
This study was approved by Nankai University Institutional Animal Care and Utilization Committee (approval no. 2023-SYDWLL-000620).
Statement of human and animal rights
This study contains studies with animal subjects, and all animal experiments were conducted in accordance with animal ethics and safety regulations. This study does not contain any studies with human subjects.
Statement of informed consent
There are no human subjects in this article, and informed consent is not applicable.
Author Contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Tianjin Municipal Science and Technology Bureau (22JCZXJC00110), National Key Research and Development Program of China (32360233), Tianjin Municipal Science and Technology Bureau (21JCQNJC00110), Tianjin First Central Hospital (2020CM12, Youth Talent Cultivation Program to JY).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of Data and Material
Data supporting the findings are available by the corresponding authors upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.