Abstract
Schwann cell (SC) transplantation is considered as a promising strategy for spinal cord injury. However, SCs show less capability in assisting the regenerative axons to penetrate through astrocyte (AS)-formed scar barrier. Anosmin-1, an extracellular matrix glycosylated adhesion protein expressed in the olfactory bulb, is involved in olfactory ensheathing cells and reborn olfactory nerve axons continually penetrating the glial barrier and targeting the olfactory bulb. In this study, we employ a dorsal root crush injury model treated with anosmin-1. A vertical climbing test was used for behavioral analysis and immunohistochemical study for SC/AS interaction in regenerative axon targeting. Anosmin-1 improved rat forepaw grasping as revealed by forelimb proprioception assessment. After treated with anosmin-1, p75+ immature SCs and P0+ mature SCs mingled well with ASs at the peripheral/central glial interface, reforming the glial barrier from a tight to loose structure. Furthermore, regenerated axons traced by BDA staining revealed proper axonal targeting to the dorsal horn of the spinal cord. These results suggest that anosmin-1 can regulate SC/AS interactions at the peripheral/central boundary site to open the glial barrier for regenerating axons crossing, targeting, and establishing functional neuronal circuits. Anosmin-1 might have a potential application in repair of spinal cord injuries, particularly in combination with SCs for autologous cell transplantation.

Main Points
Extracellular matrix protein anosmin-1 is involved in olfactory ensheathing cells and olfactory nerve axons continually penetrating the glial barrier and targeting the olfactory bulb.
In a dorsal root crush injury model, anosmin-1 can regulate Schwann cell/astrocyte interactions at the peripheral/central interface to open the glial barrier for regenerative axons crossing, targeting, and establishing functional neuronal circuits.
Anosmin-1 combined with Schwann cells might have a potential application as autologous cell transplantation for the spinal cord injuries.
Introduction
Spinal cord injury (SCI) causes significant physical and psychological problems for patients, imposing a considerable financial burden on patients’ families and the healthcare system 1 . Current therapeutic strategies for SCI, including cell transplantation, bioactive substances, and biomaterial, face one of the critical obstacles that the glial scar formed by activated astrocyte (AS) impedes regenerative axons to traverse and target to the distal lesion site for the functional synapse formation. While the glial scar helps to isolate the injury site to mitigate further inflammatory damage on the existing axons, it also forms a physical barrier and secretes inhibitory molecules to inhibit axonal growth, migration and targeting that significantly hinder neuronal regeneration and functional recovery2,3.
Olfactory ensheathing cells (OECs) can reduce AS stress response after transplanted into the SCI lesion site by enhancing OEC-AS integration and assist regenerative axons to penetrate through the AS scar. Olfactory ensheathing cells as one of the specialized glial cells in the olfactory system, exhibit Schwann cell-like (SC-like) characteristics peripherally and transform into astrocyte-like (AS-like) glial cells in the olfactory bulb (OB) 4 . They can continuously support the regenerative olfactory nerve axons from the peripheral olfactory mucosa to cross the peripheral-central glial barrier into OB to form functional synaptic connections 4 . These properties make OECs as a promising candidate for cell transplantation in SCI repair5–7. However, relatively low yield and poor purity, in addition to insufficient knowledge of biological characteristics, limit OEC transplantation for SCI in humans. In contrast, peripheral SCs have been shown to promote recovery of neurological function after SCI5,8. They are easier to extract, culture, and purify9,10. However, the ability of SCs to assist regenerating nerves in penetrating the glial scar is not as effective as expected. Therefore, identifying the key factors that make the peripheral OECs capable of crossing the peripheral/central glial barrier will offer new therapeutic strategies for the use of SCs in SCI treatment.
Anosmin-1 protein may be a proper candidate among these key factors. Anosmin-1, an extracellular matrix glycosylated adhesion protein encoded by the KAL-1 11 , is predominantly expressed in GABAergic neuronal granule cells and in the outermost layer of the OB 12 . In humans, a mutation in the KAL-1 results in hypogonadotropic hypogonadism with anosmia or hyposmia, a condition clinically known as Kallmann syndrome 13 . In the absence of anosmin-1, olfactory neuron axons fail to project and target to the OB surface despite the normal generation of olfactory neuron axons and OB mitral cells 14 . This suggests that peripheral SC-like OECs may rely on anosmin-1 to cross the peripheral/central glial barrier into the OB. Indeed, we found that anosmin-1 primarily inhibits the differentiation of immature SOX10+ and BLBP+ OEC into mature GFAP+ OEC, maintaining the immature state of OECs during the early stages of OB development. This finding implicates that anosmin-1 might facilitate glial layer opening during the interaction of OECs with ASs, enabling olfactory axons to penetrate the glial layer on the OB surface 15 . Furthermore, anosmin-1 promotes OEC migration by activating the fibroblast growth factor (FGF)-related ERK1/2 downstream signaling pathway 16 . These results suggest that anosmin-1 could regulate peripheral SC-like OECs to interact with AS glial barrier on the OB surface via the FGF signaling pathway, aiding olfactory nerve axons to penetrate from the peripheral nervous system (PNS) into the central nervous system (CNS).
Based on these findings, we hypothesize that anosmin-1 may regulate peripheral SCs to cross the peripheral/central glial barrier or glial scar for regenerating neuronal targeting, thereby simulating the behavior of peripheral OECs entering the OB in the olfactory system. In this study, we employed the dorsal root crush injury model to demonstrate that anosmin-1 regulates the interaction between SCs and ASs in the peripheral/central glial interface to promote the regenerated axons to cross from the PNS into the CNS, assist axonal projection to the dorsal horn region and establish a functional neuronal circuit.
Materials and methods
Surgery
Establishment of the dorsal root crush injury model
Under isoflurane anesthesia, the prominent C2 and T2 spinous processes were identified, and the incision site was marked on the skin. A midline dorsal incision was made using sterile instruments to expose the muscles. Hemilaminectomies were performed from C5 to T2 to expose the left dorsal roots of C6, C7, C8, and T1. Fine forceps (DUMONT 0209-55-PO) were then employed to crush the dorsal roots as close to the spinal cord as possible, ensuring complete crushing. The crushing was sustained for 10 s and repeated 3 times. Finally, the overlying muscle and skin were closed layer by layer with absorbable sutures. The animals used in this study were female Sprague-Dawley rats, aged 6 to 8 weeks, which were obtained from Beijing Charles River Laboratory Animal Breeding Co. Ltd.
Reagents application
A 33G pointed needle was used to create a small puncture near the spinal cord of the injury site. Subsequently, 2 μl of the reagent was slowly injected along the puncture using a Hamilton microsyringe. The needle was left in place for approximately 1 minute before being withdrawn upon completion of the injection. The experimental groups were as follows: group C (sterile normal saline), group F (250μg/ml FGF2 (Thermo Fisher Scientific, USA)), group A (2.5μg/ml anosmin-1), group AF1 (2.5μg/ml anosmin-1 and 1mg/ml FGF2 mixed solution), and group AF2 (2.5μg/ml anosmin-1 and 250μg/ml FGF2 mixed solution). Anosmin-1 protein is homemade as described in previous publications 17 .
Nerve tracing
Four weeks after modeling, 2 µl of 10% biotinylated dextran amine (BDA, Thermo Fisher Scientific, USA) was injected into the dorsal root ganglia (DRG) of C7 and C8, the two intermediate segments, to track the regeneration of dorsal root axons. Two weeks following the tracer injection, the rats were sacrificed and perfused for sampling.
Postoperative care
After surgery, the rats were placed in warm cages to allow them to recover from anesthesia before being returned to their previous rearing cages. During the first postoperative week, they were provided with a paste diet and soft cotton bedding. Because the dorsal root crush injury might result in sensory impairment and mild pain in the operative forepaw, some rats might self-mutilate. To manage pain and prevent infection, appropriate analgesics (5 mg/kg carprofen) and antibiotics (5 mg/kg enrofloxacin) were injected subcutaneously once daily for 3 days postoperatively. If any rat was observed self-mutilating, it was removed from the experiment. The rats’ rearing room was maintained at a temperature between 20°C and 22°C, with a 12-h light/dark cycle. The rats received food and water ad libitum.
Behavioral test-vertical climbing test
Rats were placed on a 1.4-meter near-vertical grid (30° inclination) and allowed to climb daily during the preoperative week, on the third day postsurgery, and weekly throughout the six postoperative weeks. Each climbing session was filmed and analyzed in slow motion. Thirty consecutive vertical movements were recorded, consisting of 15 upward and 15 downward movements. The grasping behavior was assessed based on the extent of grasping and the distance the operative forepaw extended along the grid. All analyses were performed with two independent researchers unaware of the experimental group allocations to ensure unbiased results.
Perfusion sampling and immunofluorescence staining
Six weeks after modeling, rats were deeply anesthetized with carbon dioxide and then perfused transcardially with 60 mL PBS, followed by 250 mL 4% paraformaldehyde (PFA). After fixation, at least four spinal cord segments (C6-T1) were carefully dissected under a microscope, ensuring that the dorsal root nerves were preserved as long as possible and maintaining the continuity between the spinal cord and the dorsal roots. The tissues were postfixed in 4% PFA at 4°C for approximately 2 days, then sequentially placed in 10% and 20% sucrose solutions to sink for use. Tissue blocks were embedded in pre-cooled OCT cryoembedding agent (SAKURA-4583, USA) and frozen in a cryostat chamber (LEICA CM1950) at −20°C until fully hardened. The tissues were cut into serial sections of 15 μm thickness along the coronal or horizontal plane, as required.
For immunohistochemical analysis, sections were blocked and permeabilized for 1 h at room temperature using 3% bovine serum albumin (BSA, BioFroxx, Germany) containing 1% Triton X-100 (BioFroxx, Germany). Between each step, sections were washed in PBS (3 × 10 min). For double immunostaining, sections were incubated overnight at 4°C with primary antibodies, as detailed in Table 1, diluted in 3% BSA. The corresponding secondary antibodies (Table 1), were diluted in 3% BSA and incubated for 1.5 h at room temperature in the dark. To detect BDA labeling, sections were incubated in streptavidin conjugated with Alexa Fluor 546 (1:1,000, Thermo Fisher, USA) for 1.5 h at room temperature, protected from light. Finally, an anti-fluorescence quencher containing DAPI (Abcam, USA) was added to seal the slides. Fluorescent images were visualized and captured using a widefield microscope (THUNDER DMi8 LEICA).
List of antibodies.

GFAP: glial fibrillary acidic protein; P0: Protein Zero; p75: p75 nerve growth factor receptor; ms: mouse; rb: rabbit; gt: goat.
Quantitative data analysis
We measured the SC migratory distance by calculating the straight distance from the spinal cord PNS/CNS boundary site to the farthest point of SC migration. For the quantification of punctate cross-sections, we used ImageJ software to select an appropriate threshold and count the punctates in the cross-sections within the dorsal column region of the spinal cord. Two sections from each rat were selected for analysis.
Statistical analysis
Results are expressed as mean ± standard error of the mean (SEM). GraphPad Prism 9.3 was used to analyze the experimental data and create statistical charts. Statistical comparisons between groups were conducted using one-way analysis of variance (ANOVA) or two-way ANOVA to assess significance, and p < 0.05 was considered statistically significant.
Result
Dorsal root crush injury model
The highly complex structure of the spinal cord makes the detailed analysis of mechanistic processes and the validation of therapeutic targets complicated. To address the effect of anosmin-1 on SCs interaction with ASs at the peripheral/central interface, we employed a simplified SCI model, the dorsal root crush injury model (Fig. 1a). Crushing the central branch of the dorsal root allows for an in-depth analysis of the regenerative processes in sensory neurons, as well as the glial response at the dorsal root-spinal cord interface, without direct injury to the spinal cord. One advantage of this model is that it allows for the severing of the dorsal root nerve while preserving its overall anatomical structure. Complete injury to the dorsal roots is indicated by the presence of a characteristic clenched fist in the ipsilateral forepaw (Fig. 1b), a phenotype that is absent with incomplete dorsal root crush injury 18 . Histological analysis further confirmed the disruption of nerve axons at the entry zone of the injured dorsal root (Fig. 1c, d, e).

Dorsal root crush injury model. (a) Experimental design diagram. The dorsal roots of the C6-T1 segments on the left side were individually clamped close to the spinal cord surface, and reagents were injected at the injury site near the spinal cord. For nerve tracing, BDA was injected into the dorsal root ganglion 4 weeks after modeling. (b) Postoperatively, the left forepaw showed a characteristic of fist clenching, which was notably different from the right forepaw (uninjured side). (c) A representative image showing a distinct lesion at the entry zone between the dorsal root ganglion and the spinal cord following the crush injury. 68 kDa-NF staining labeled small-diameter axons. (d, e) High-magnification image of the boxed area in Figure c, the crushed notch is marked with a white arc (horizontal section. Red: 68 kDa-NF; blue: DAPI).
Vertical climbing test– Anosmin-1 improves grasping ability
Vertical climbing test was used for grasping ability analysis (Fig. 2a). The score ranged from 0 to 4: (0) the paw grasps the bar with full functional grasp; (1) the paw reaches the grid but does not grasp; (2) the paw extends to the level of the wrist; (3) the paw extends to the level of the elbow; (4) the paw extends to the level of the axilla (Fig. 2b).

Behavioral test—vertical climbing test. (a) Example of normal grasp; (b) error scoring scheme for the left forepaw locating and grasping the grid bars. 0, successful grasp; 1, paw reaches the grid but does not grasp; 2, paw extends to the level of the wrist; 3, to the level of the elbow; 4, to the level of the axilla. Figure b adapted from Minkelyte et al. 7 (c, d) Anosmin-1 improves grasping ability in vertical climbing test. (c) The obvious error rate of forepaw positioning for grasping the bar in the climbing task [time: F(5.521, 209.8) = 167.5, P < 0.0001; group: F(4, 41) = 24.37, P < 0.0001; interaction: F(28, 266) = 5.586, P < 0.0001]. **P < 0.01, ****P < 0.0001, group C versus group AF2; ##P < 0.01, ###P < 0.001, ####P < 0.0001, group C versus group A. (d) Mean error scores for positioning the forepaw to grasp the bar in the climbing task [time: F(3.887, 144.9) = 329.6, P < 0.0001; group: F(4, 40) = 14.49, P < 0.0001; interaction: F(28, 261) = 4.141, P < 0.0001]. ***P < 0.001, ****P < 0.0001, group C versus group AF2; #P < 0.05, ##P < 0.01, group C versus group A. Groups C, A, and AF2 (n = 10); groups F and AF1 (n = 7). Scores during the 6-week test period. Mean ± standard error. Two-way ANOVA.
Analysis of the recorded videos revealed that normal rats were able to accurately position all four paws before surgery, with minimal misplacement of the horizontal bar, and subsequently executed a functional grasping motion by closing their fingers. The percentage of obvious errors and the error grade in forepaw grasping were both approximately 0%. Due to the abundant blood supply near the lamina, we performed a “sham surgery” on four rats, in which the lamina was opened without crushing the dorsal root. By day 3 postsurgery, rats in the sham-operated group were able to regain functional grasping, excluding the potential impact of hemorrhage on the behavior test.
In terms of grouping, recovery of forepaw grasping on the operated side was shown in the order: group AF2 > group A > group AF1 > group F > group C. All groups, except the control group, showed gradual week-by-week improvements in grasping ability until the approximate fourth week.
The percentage of obvious errors
We defined the rat’s left forepaw positioned beyond the bar level during grasping as an obvious error, corresponding to error scores of 2, 3, and 4. On postoperative day 3, the rate of obvious grasping errors in group C was as high as 90.0% ± 2.7%, indicating that nearly all grasping attempts were unsuccessful. This error rate decreased to 67.5% ± 5.1% after 2 weeks, with an error rate of 69.5% ± 5.8% by the sixth week. Group F exhibited a similar grasping error rate of 87.5% ± 1.6% on day 3, comparable with group C. However, improvement was more pronounced after 2 weeks, with the error rate decreasing to 55.0% ± 4.4%, and further declining to 47.9% ± 4.8% at 6th week. The error rate in group AF1 was not significantly different from that of group F at any time point. In contrast, there was a notable improvement in the positioning and grasping ability of the operative lateral forepaw in groups A and AF2. The error rate in group A was 51.1% ± 7.2% on day 3, indicating that only approximately half of the grasping attempts exceeded the bar and gradually improved with further decreasing to 22.5% ± 3.3% by week 6. Group AF2 showed a better grasping performance than group A, with the error rate decreasing from 57.5% ± 5.4% on day 3 to 20.0% ± 2.7% at week 6, indicating that only about one-fifth of the grasping attempts were obviously incorrect (Fig. 2c).
The error score
In group C, the grasp score of the operated forepaw was 2.4 ± 0.1 on postoperative day 3, and decreased to 2.0 ± 0.1 after 6 weeks of training. This indicated that the forepaw of control rats on the operated side failed to achieve a functional grasp, typically extending beyond the bar and positioning between the wrist and elbow. Group F had a score of 2.2 ± 0.1 at 3 days and 1.6 ± 0.1 at 6 weeks after surgery, showing slight improvement, with the forepaw reaching only to the wrist on average, but not to the elbow. Group AF1 performed slightly better than group F, with scores of 2.2 ± 0.2 on day 3 and 1.5 ± 0.2 at week 6 postoperatively. Compared with group C, rats in group A showed significant improvements in grasping ability in the operated forepaw and were occasionally able to complete purposeful grasping movements. The score for group A was 1.9 ± 0.1 on day 3 and decreased to 1.3 ± 0.1 at week 6, which was significantly different from that of group C at all time points. The most substantial improvement in grasping ability was observed in group AF2, where the score decreased from 1.6 ± 0.1 on postoperative day 3 to 1.1 ± 0.0 at week 6. The forepaw on the injured side barely extended to the wrist, positioned the crossbar more accurately, and exhibited more purposeful grasping movements. Although group AF2 had overall lower error scores than group A, the difference between the two groups was statistically significant only at week 6 (Fig. 2d).
Anosmin-1 modulates SCs and ASs interaction in the peripheral/central boundary site
We further investigated the effect of anosmin-1 ± FGF2 on SCs and ASs interaction in the peripheral/central boundary site by labeling mature myelinated SCs with P0 (Fig. 3), immature nonmyelinated SCs with p75 (Fig. 4), and ASs with GFAP (Fig. 3, 4). Observations of coronal sections from rats at 6 weeks postoperation revealed interrupted GFAP+ AS boundaries in groups A (Figs. 3c and 4c) and AF2 (Figs. 3d and 4d), with P0+ and p75+ SC penetrating inside the spinal cord close to the dorsal root-spinal cord junction. Most of the P0+ mature SCs were oval in the periphery dorsal root area, while few of them became round on the peripheral side near the spinal cord boundary. As migrated inside the spinal cord, these cells became uniformly round and were primarily accumulated within 100 µm away from the spinal cord border, showing a mixed distribution with ASs (Fig. 3c, d). p75+ immature SCs appeared elongated and spindle-shaped in the dorsal root, while they exhibited morphological diversity within the spinal cord. Within the central part, most p75+ SCs were short, round spindle-shaped, with a smaller proportion displaying longer spindle shapes as seen in the peripheral SCs. As compared with the mature SCs, these p75+ cells migrated further deeply inside the spinal cord, mingling well and partially overlapping with AS (Fig. 4c, d). The farthest migration distance of P0+ SCs inside the spinal cord was 230.9 ± 29.9 µm in group A and 107.7 ± 22.0 µm in group AF2, both of which were significantly higher than that in group C (4.3 ± 2.8 µm) (Fig. 3f). Similarly, quantitative analysis showed that p75+ SCs migrated even further inside the spinal cord in groups A and AF2 than that in group C (Fig. 4f). However, there was a statistically significant difference in p75+ SC migratory distance between groups AF1 and AF2, but no significant difference in P0+ SC migration distance (Fig. 3f and 4f).

Anosmin-1 modulates mature SCs and ASs interaction in the peripheral/central boundary site. Tight boundary formation between mature SCs and ASs at the peripheral and central interface in group C (a, a’) and group F (b, b’); (c, c’) group A. Mature SCs were seen in two consecutive segments near the central border, with striated AS in the peripheral side; (d, d’) group AF2. Several clusters of mature SCs were observed at the peripheral-central junction, and the AS in the PNS migrated farther than in group A; (e, e’) group AF1. Scattered mature SCs were present in the spinal cord, while ASs migration to the peripheral side was observed. (a’, b’, c’, d’, e’) High-magnification images within the white boxes in the corresponding figures. (f) Quantitative analysis showed that the farthest migration distance of P0+ SCs inside the spinal cord was statistically much further in groups A and AF2 as compared with group C and there was no significant difference between groups AF1 and AF2. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001. Groups C, A, and AF2 (n = 6); groups F and AF1 (n = 4). Mean ± standard error. One-way ANOVA (coronal section. “*” indicates the side of the spinal cord. Red: P0; Green: GFAP; Blue: DAPI).
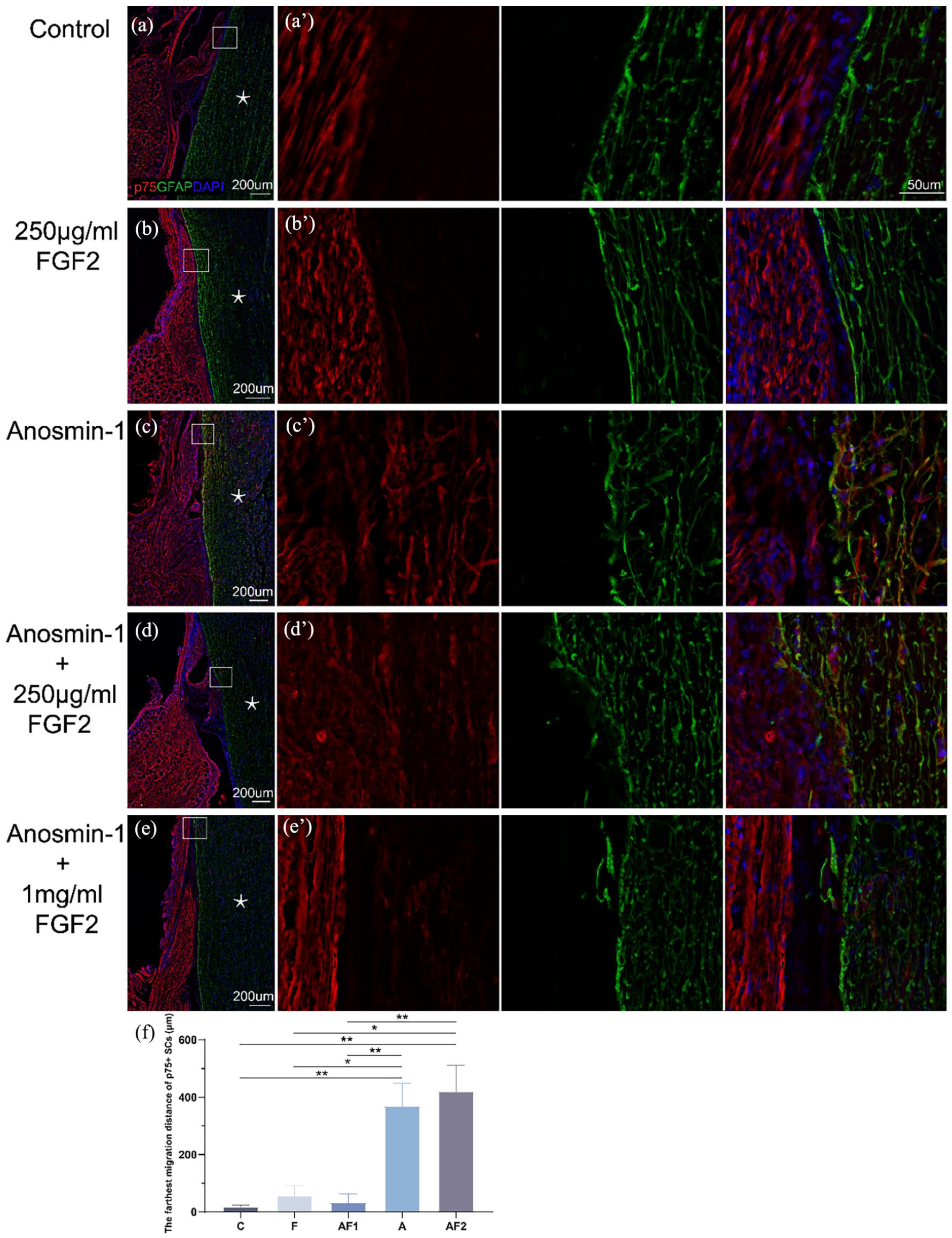
Anosmin-1 modulates immature SCs and ASs interaction in the peripheral/central boundary site. (a, a’) Group C. No interaction was observed between peripheral and central glial cells; (b, b’) group F. No interaction was observed between peripheral and central glial cells; (c, c’) group A. A large number of immature SCs were present centrally near the spinal cord, together with a sparse AS border; (d, d’) group AF2. Rounded and spindle-shaped immature SCs were observed inside the spinal cord, with scattered AS in the peripheral side; (e, e’) group AF1. Scattered immature SCs were seen in the spinal cord, with some ASs in the peripheral side; (a’, b’, c’, d’, e’) High-magnification images within the white boxes in the corresponding figures. (f) Quantitative analysis showed that the farthest migration distance of p75+ SCs inside the spinal cord was statistically much further in groups A and AF2 as compared with group C and there was a significant difference between groups AF1 and AF2. *P < 0.05, **P < 0.01. Groups C, A, and AF2 (n = 6); groups F and AF1 (n = 4). Mean ± standard error. One-way ANOVA (coronal section. “*” indicates the side of the spinal cord. Red: p75; Green: GFAP; Blue: DAPI).
At the junction of the spinal cord and dorsal roots, we observed that multiple strands of GFAP+ branching fibrous protrusions extended peripherally, as far as 800 µm to 1 mm. These branches interlaced with peripheral SCs but without significant overlap (Fig. 3c, d, e). Similar phenomena were occasionally observed in the coronal sections of rats in group AF1 (Fig. 3e and 4e). In contrast, the AS boundaries in groups C (Fig. 3a and 4a) and F (Fig. 3b and 4b) remained dense with no notable intermingle between central ASs and peripheral SCs.
Anosmin-1 promotes regenerative axon targeting to the dorsal horn of the spinal cord
We then used BDA tracing to examine whether disruption of the glial barrier induced by anosmin-1 can assist regenerative axons in crossing the glial boundary and targeting the dorsal horn of the spinal cord. The regenerating axons in groups A (Fig. 5c, c’) and AF2 (Fig. 5d, d’) were traced after they crossed the dorsal root entry zone into the spinal cord. In group A, the horizontal section revealed that some regenerating axons targeted toward the dorsal horn. High-magnification images showed these axons as filaments of varying lengths (Fig. 5c’, arrows). Multiple punctates representing cross-sectioned regenerating axon bundles (more than 60) were observed in the dorsal column of the spinal cord (Fig. 5c’, dashed box), indicating that these axon bundles ascended within the dorsal column and accumulated in the wedge bundle around the injury site. Compared with group A, group AF2 exhibited much more punctates as cross-sectioned ascending axon bundles (more than 90) within the dorsal column (Fig. 5d’, dashed box). The distinct filamentous axons targeting the dorsal horn were also obviously visible (Fig. 5d’, arrows). Conversely, groups C (Fig. 5a), F (Fig. 5b), and AF1 (Fig. 5e) showed much fewer BDA-positive signals in the central regions. Quantitative analysis showed that the number of punctates in the dorsal column in groups A, AF1, and AF2 were higher than that in group C and much more punctates were present in groups A and AF2 as compared with AF1 (Fig. 5f), further indicating that anosmin-1 could promote regenerative axons to cross the glial boundary for the central targeting.
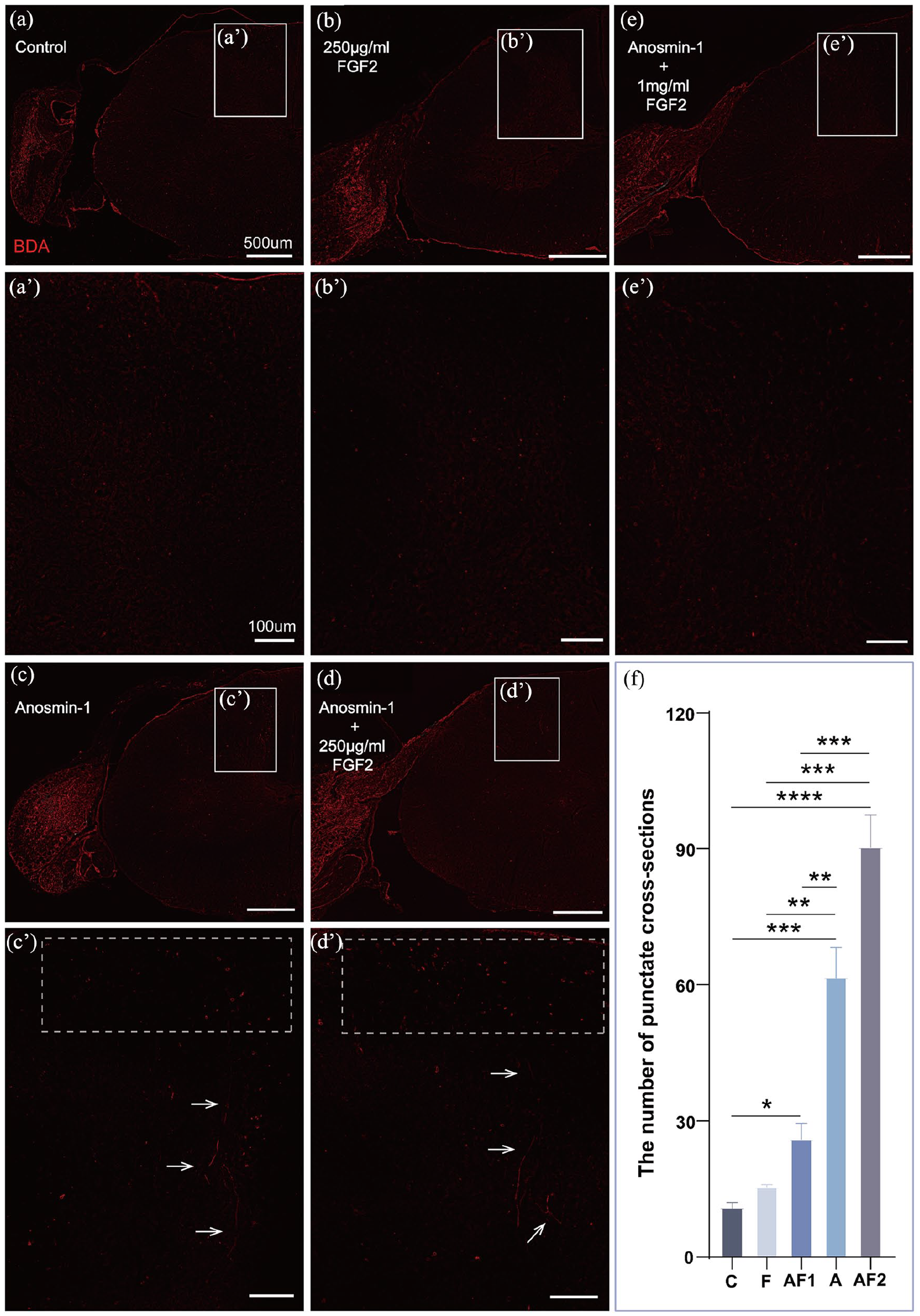
Anosmin-1 promotes regenerative axon targeting to the dorsal horn of the spinal cord. In the control group (a) and FGF2 treatment group (b), few of BDA-positive staining was observed in the spinal cord; (c) and (c’) After treated with anosmin-1, the filamentous regenerating axons of varying lengths were observed within the dorsal horn (arrows), and multiple punctates representing cross-sectioned regenerative axon bundles were seen within the dorsal column (dashed box); (d) and (d’) After treated with anosmin-1+250 µg/ml FGF2, more punctates of regenerating axon bundles by cross-section were observed in the dorsal column (dashed box), along with thread-like regenerating axons of varying lengths in the dorsal horn (arrows); (e) After treated with anosmin-1+1 mg/ml FGF2, few of BDA-positive staining was observed in the spinal cord; (f) Quantitative analysis showed that the number of punctates in the dorsal column in groups A, AF1 and AF2 were higher than that in group C and much more punctates were present in groups A, AF2 as compared with AF1. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001. Groups C, A, and AF2 (n = 4); groups F and AF1 (n = 3). Mean ± standard error. One-way ANOVA. (a’, b’, c’, d’, e’) High-magnification images of the regions within the white boxes in the corresponding figures (horizontal section; red: BDA).
The low-magnification image revealed p75+ cells inside the injured part of spinal cord as compared with the uninjured side (Fig. 6a). High-magnification images (Fig. 6a1, a2) further revealed that the distribution of p75+ SCs entering the spinal cord was diffused (Fig. 6a2, arrows) and these centrally located SCs did not exhibit the typical characteristic of wrapping regenerating axons that is commonly seen in OECs.

Diffused distribution of centrally located SCs without wrapping regenerating axons. (a) Horizontal section of the spinal cord and connected dorsal root nerves, showing the diffused distribution of central p75+ cells on the left side as compared with the right side; (a1) High-magnification image of the boxed area in Fig. 6a, showing the irregularly scattered distribution of SCs; (a2) High-magnification image of the boxed area in Fig. 6a1. SCs (arrows) did not wrap regenerating axons (horizontal section; red: BDA; green: p75; blue: DAPI).
Discussion
Schwann cell transplantation can promote axonal regeneration, myelin regeneration, and functional recovery after SCI 9 . However, SCs showed less modulatory effects on the AS glial barrier, leading to deficient axonal regeneration within the SC grafts and regenerative axons incapable of penetrating the scar tissue into the target area19,20, thereby restricting the therapeutic potential of SCs in SCI. For the first time, we found that anosmin-1 may primarily regulate the process of SC/AS interaction to loose AS-formed tight glial barrier for regenerative axon targeting. As SCs can be easily obtained from peripheral nerves and expanded in vitro, it could be reasonable to apply anosmin-1 along with SC for autologous cell transplantation, therefore making SCs as one of the more suitable candidates for SCI treatment 10 .
In this study, we labeled mature SCs, immature SCs, and ASs using anti-P0, p75, and GFAP antibodies, respectively, to examine the reactivity of peripheral versus central glial cells and to assess whether the AS glial barrier was interrupted. Immunohistochemical analysis revealed the presence of P0+ and p75+ cells inside the spinal cord when treated with anosmin-1 alone or plus a low concentration of FGF2, indicating that SCs, which are typically confined to the PNS, were able to migrate into the CNS. Moreover, in approximately half of the coronal sections, GFAP+ ASs were observed migrating from the central to the peripheral side and mixing with the SC population, further demonstrating that anosmin-1 can open the glial barrier between the central and peripheral regions, making a dense barrier to be a loose structure. This alteration may facilitate the penetration of regenerating peripheral axons through the glial barrier into the CNS. Indeed, we observed obvious entry of regenerating axons into the spinal cord in rats treated with anosmin-1 alone or plus a low concentration of FGF2. Under the influence of anosmin-1, SCs appeared on the central side of the injured spinal cord, especially for the immature SCs migrating and extending further into the central region. However, these immature SCs were scattered in distribution and did not visibly wrap around regenerating axons as OECs do when entering the dorsal horn of the spinal cord 21 . This suggests that anosmin-1 may primarily regulate the process of SC/AS interaction and fusion, rather than directly participating in the wrapping of regenerating axons by SCs.
Mature SCs are primarily responsible for the formation of myelin sheaths, which speed up the conduction of nerve impulses 22 . They promote intercellular interactions and support axonal growth toward target tissues by remodeling the local microenvironment and secreting various neurotrophic factors 23 . After nerve injury, mature SCs dedifferentiate and reacquire some properties of immature SCs, thereby contributing to nerve repair 24 . Compared with mature SCs, immature SCs exhibit greater proliferation, regeneration, and migration, as well as higher plasticity 24 . During the early stages of nerve injury, immature SCs can rapidly migrate to the injured site, divide, proliferate, and fill the damaged area, thereby providing a crucial cellular foundation for nerve regeneration 25 . Previous studies have shown that anosmin-1 can maintain the OB-derived OECs in an immature state during chicken embryo development 15 . In this study, after treated with anosmin-1, p75+ immature SCs exhibited enhanced interaction and fusion with AS, showing even further migration toward the inner spinal cord as compared with P0+ mature SCs. In addition, the morphology of these immature cells was more diverse. These findings suggest that anosmin-1 may primarily regulate the interaction between immature SCs and ASs; however, anosmin-1 may also facilitate the conversion of mature SCs into a more immature phenotype.
Anosmin-1 exerts specific biological effects on various cell types by activating distinct FGF signaling pathways. Through FGFR1, anosmin-1 ① activates ERK1/2 and p38 MAPKs to promote axonal growth of gonadotropin-releasing hormone (GnRH) and OB nerves 26 ; ② activates the Rac/cdc42 pathway to regulate cytoskeletal rearrangement 26 ; ③ specifically regulates migration in GnRH nerves via the PI3K p110α/Akt isoform 27 ; ④ activates the ERK1/2 pathway to stimulate the proliferation and migration of oligodendrocyte precursor cells 28 ; and ⑤ promotes the proliferation of the subventricular zone (SVZ), enhances neurogenesis in the SVZ and attracts neuronal precursor from the SVZ to migrate to the OB through the rostral migratory stream, with FGF2 acting as the driving factor for this migration29,30. In addition, anosmin-1 promotes neural crest formation by enhancing FGF8 activity while inhibiting the BMP5 and Wnt3a signaling pathways 31 . These findings suggest that anosmin-1, when injected at the side of dorsal root nerve injury as shown in the present study, may regulate the transformation of peripheral SCs into central SCs, modulate SC-AS fusion, and promote the directed projection of regenerating axons toward the spinal cord via specific or multiple FGF signaling pathways.
The regulation of AS scar barrier formation is a highly complex and multifactorial process, involving numerous factors such as cytokines 32 , signaling pathways 33 , cell types 34 , and the local microenvironment 35 . SCs overexpressing glial cell line-derived neurotrophic factor (GDNF) significantly enhanced the integration between SCs and ASs by promoting the migration of reactive AS toward the SCs-GDNF region 36 . In addition, SCs overexpressing microRNA-124 demonstrated the ability to promote SC-AS boundary integration in vitro, potentially reducing the capacity of ASs to form glial scars 37 . SCs transduced with lentiviral chondroitinase ABC that extensively digested CSPGs, thereby facilitating SC migration, adhesion, and integration in an inhibitory AS environment 38 . Furthermore, ectopic expression of polysialylated neural cell adhesion molecules in adult macaque SCs promoted SC-AS mixing and enhanced myelin regeneration potential 39 . Within these crucial factors involved in SC/AS interaction, FGF2 and FGF receptor coupling activation also modulates SC/AS boundary formation. Previous studies have shown that SCs regulate the FGF signaling pathway by secreting FGF2 and specific sulfated heparan sulfate proteoglycans (HSPGs), thereby leading to glial boundary formation with ASs. In vitro, the SC-origin secretion interfered with the interaction between OEC and AS, preventing OEC-AS integration 40 . In this study, we observed that in the presence of anosmin-1, the dense astrocytic glial barrier became sparser, and the repulsive interaction between SCs and ASs was reduced. This allowed SCs to migrate within the AS-rich CNS environment, thereby facilitating SC-AS integration. In addition, we noted that these two glial cells mixed well during migration, a phenomenon similar to the interaction between OECs and ASs6,41. It is known that anosmin-1 activates the FGF2-associated ERK1/2 downstream signaling pathway by binding to HSPGs, thereby promoting the migration of OEC and the maturation and differentiation of glial cells13,15. These findings suggest that anosmin-1 might regulate SC/AS interaction through the formation of the FGF/FGF receptor/HSPG signaling complex.
We observed better behavioral recovery in dorsal root injured rats when treated with the mixture of anosmin-1 and a lower concentration of FGF2 (250 μg/ml). Interestingly, rats injected with a higher concentration of FGF2 (1 mg/ml) in combination with anosmin-1 exhibited poorer performance in the vertical climbing test. Studies have shown that dorsal root-derived SCs secrete HSPGs, which activate the binding of FGF2 to FGFR1, triggering ASs activation and proliferation, leading to excessive glial scar formation and enhanced inhibitory effects 40 . However, OEC-derived HSPGs promote SC/AS mingle well without boundary formation, likely depending on the specific sulfation pattern of HSPGs40,42. We previous found that anosmin-1 performs dual role on FGFR1, ie. either binding to HSPG to enhance FGFR1 activation or binding to FGFR1 to inhibit 43 . Furthermore, anosmin-1 activates FGFR1 signaling to enhance OEC migration via ERK1/2 and regulate OEC maturation 16 . In the present study, anosmin-1 alone or with lower FGF2 concentration may enhance inborn and foreign FGF activation on FGFR1 signaling via HSPG binding at a dose-dependent manner, thereby amplifying capability of SC to migrate and penetrate through PNS/CNS boundary. In contrast, excess FGF2 application might consume more HSPG that leave much less HSPG and more FGFR1 available for anosmin-1 binding. High amount of FGF2 might mitigate anosmin-1 effects on SCs, partially counteracting the loosening effect of anosmin-1 on the glial barrier, thereby restricting regenerating axons from targeting the dorsal horn and impairing functional recovery. Therefore, when combined at an appropriate concentration ratio, anosmin-1 and FGF2 may synergistically promote neural recovery by modulating SC/AS interaction.
The behavioral results of this study indicate that the greatest recovery occurred during the first week following injury. A possible explanation for this is that anosmin-1 was administered only once, directly at the time of injury, which is a limitation of this experiment. Combined with biomaterials or delivered continuously via lentiviral vectors might enhance anosmin-1’s therapeutic effects and prolong its working duration44,45. However, as shown in our previous publications, anosmin-1 binds to heparan sulfate on the cell-surface and in the extracellular matrix with extremely high affinity that enable it avoid cleavage and continue to exert its biological activation on FGF signaling 17 . Therefore, anosmin-1 might exert the therapeutic effects on SC/AS interaction for a relatively long period of time.
There are other limitations to this article: (1) the specific mechanisms underlying the functional improvements observed in this study, including the involvement of FGFR-1 activation and its downstream signaling pathways has not been clearly defined; (2) the potential proliferative effects of anosmin-1 on SCs were not assessed; (3) we currently observe that anosmin-1 may regulate the process of SC/AS interaction and fusion to open the AS-formed boundary for peripheral axons entry into the CNS, its regulatory effect on the direct cellular interaction and the underlying mechanism need to be explored further.
Conclusion
In this study, we used the dorsal root crush injury model to preliminarily investigate the modulatory effects of anosmin-1 protein on SCs and ASs interaction at the peripheral/central boundary site of the spinal cord. The results demonstrated that anosmin-1 was able to disrupt the glial barrier between the PNS and the CNS, facilitating the entry of peripheral regenerating axons into the CNS and enabling them to reach their corresponding target region. This finding raises the possibility that anosmin-1 has a potential broad application in the repair of CNS injuries. Although the dorsal root crush injury model used in this study does not fully replicate the more complex and variable injury scenarios encountered in cases of spinal cord plexus avulsion or spinal cord injuries in clinical settings, the present results provide a theoretical foundation for the development of relevant therapeutic strategies for future clinical applications, especially using anosmin-1 combined with SC for SCI treatment. The following research will focus on identifying functional fragments of anosmin-1 for future drug development.
Footnotes
Ethical Considerations
All procedures strictly followed the Guide for the Care and Use of Laboratory Animals and received approval from the Institutional Animal Care and Use Committee of Nanjing Medical University, Nanjing, Jiangsu, under the approval numbers IACUC-2006022-2.
Statement of human and animal rights
This article does not contain any studies with human or animal subjects.
Statement of informed consent
There are no human subjects in this article and informed consent is not applicable.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China grant [81571193 (Y.H.)] and the Double Innovation Talent Program of Jiangsu Province grant [2017 (Y.H.)].
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
