Abstract
The lung is a vital organ in the respiratory system, and there is a critical need to develop more effective methods for lung health management. Extracellular vesicles (EVs) play an important role in intercellular communication. They exhibit high bioavailability and low immunogenicity, making them essential in maintaining cellular homeostasis and in the prevention and treatment of numerous diseases. This review describes the diverse sources, isolation techniques, functions, and challenges associated with EVs, particularly exosomes. We highlight their significant role in the diagnosis and treatment of lung diseases, as well as their potential as drug delivery vehicles. By synthesizing recent advances in EVs research, this review aims to provide a theoretical foundation for future studies and clinical applications of EVs.
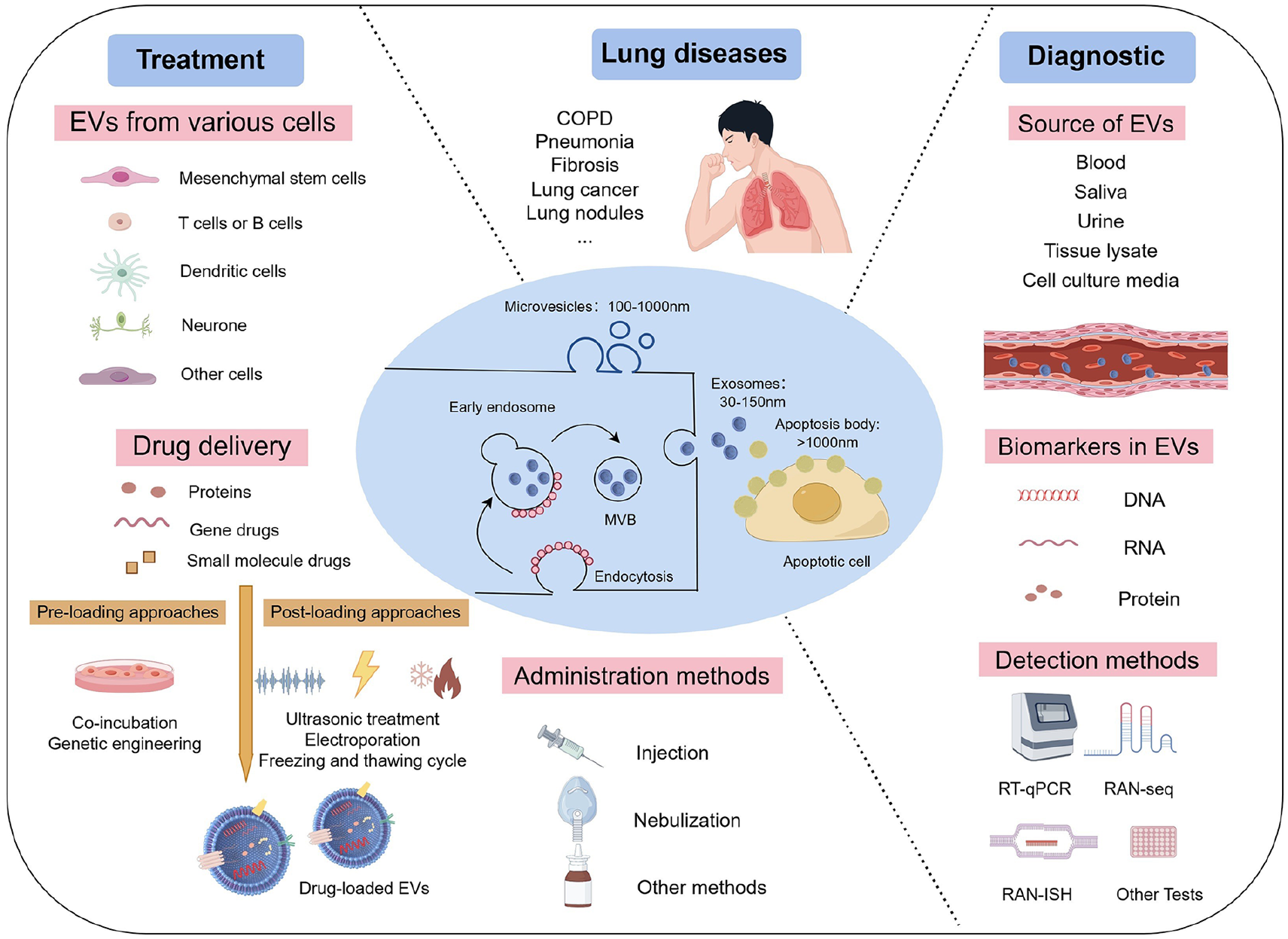
Introduction
The lung works around the clock in our bodies, providing the most basic oxygen support for life and ensuring that the organism functions normally. However, lung health is at risk due to air pollution, particularly from vehicle and industrial exhaust. Lung cancer and chronic obstructive pulmonary diseases (COPD) are more common in smokers, and secondhand smoke as a risk factor, elevates the incidence of respiratory illnesses in family members1,2. Concern over lung health has grown steadily in the last few years. The focus has shifted from seeking medical attention after the onset of disease to preventing lung diseases and managing lung health every day. Consequently, the pressing need for a healthy lifestyle is driving up demand for the creation and use of innovative technologies.
Extracellular vesicles (EVs), which serve as carriers for intercellular substance and information transfer, offer novel opportunities for diagnosing and treating lung diseases 3 . Secreted by all cell types, EVs are classified into exosomes, microvesicles, and apoptotic bodies based on their size, biogenesis, and secretion mechanisms4,5. Exosomes are formed through the endosome system and have a size range of 30-150nm. Microvesicles are directly generated by the budding of the plasma membrane, with a size range of 100-1000nm. Apoptotic bodies are produced by membrane blebbing during the late stage of apoptosis, and their size is generally larger than 1000nm. The 2023 updated guidelines from the International Society for Extracellular Vesicles (ISEV) recommend using “extracellular vesicle” as a generic term for vesicles of all sizes 4 . This recommendation is due to the difficulty in enriching EVs produced by different mechanisms, the lack of definitive characterization of biogenesis-based subtypes, and the absence of universal molecular markers for each EV subtype.
These EVs selectively encapsulate active substances such as proteins and nucleic acids, which regulate physiological and pathological processes5,6. EVs retain the properties of parent cells, so the characteristics of EVs from various sources are significantly different. To advance biomedical technology and improve human health, it is essential to summarize recent progress in EVs research and develop innovative therapeutic strategies for lung diseases. This article reviews EVs isolation methods and related clinical trials, while also highlighting the latest advances in EV-based diagnostics and therapeutics for pulmonary diseases.
EVs from different sources
EVs derived from mesenchymal stem cells
It is well known that mesenchymal stem cells (MSCs) stimulate immuno-modulation and tissue regeneration 7 . Mesenchymal stem cell–derived EVs (MSC-EVs), especially exosomes (MSC-Exos), have high bioavailability and low immunogenicity8–10. According to studies, paracrine processes including the release of exosomes by MSCs may be related to the therapeutic benefits of MSCs. The potential application of MSC-EVs for different tissue repair and regeneration has been assessed in an increasing number of research8–10, and it is becoming a growing trend as an appropriate replacement for stem cell therapy 11 .
The therapeutic efficacy of MSC-EVs is exerted by tissue regeneration, repair of damaged tissues, and immunomodulation. Studies demonstrate that MSC-Exos exhibit potent wound-healing and angiogenesis-promoting functions, and show an effective anti-scar formation and anti-inflammatory effects 12 . Additionally, MSC-Exos modulate macrophage polarization, decreasing pro-inflammatory cytokines (TNF-α, IL-6, IFN-γ, G-CSF) and enhancing anti-inflammatory cytokines (IL-4, IL-10) 13 . In a rat model of acute graft versus host disease (aGVHD), MSC-Exos reduced the CD4+/CD8+ T-cell ratio, increased Treg cells, and decreased autophagy in the spleen and thymus 14 .
EVs derived from immune cells
Beyond MSC-derived EVs, immune cell-derived EVs also demonstrate therapeutic potential. They have become powerful immune response stimulators as well as potential cancer treatment medications. Dendritic cell-derived exosomes (DC-Exos) include tetraspanins and all established proteins for presenting antigenic material such as the major histocompatibility complex class I/II (MHC I/II) and CD1a, b, c, d proteins and CD86 costimulatory molecule 15 . DC-Exos have the potential to promote immune cell-dependent tumor rejection16,17. Evidence suggests that DC-Exos enhance anti-tumor immunity of NK cells in patients with advanced nonsmall cell lung cancer (NSCLC) 18 .
Recent studies have explored the role of other immune cell-derived EVs. Jung et al. 19 used engineered Jurkat T-cells expressing IL-2 at the plasma membrane to create IL-2-sEVs, which increased the anti-cancer ability of CD8+ T-cells without affecting regulatory Treg cells and down-regulated cellular and exosomal PD-L1 expression in melanoma cells, causing their increased sensitivity to CD8+ T-cell-mediated cytotoxicity. Exosomes derived from NK cells have lower immunogenicity and greater stability, and they also possess antitumor properties 20 . Kang et al. 21 demonstrated that NK-exosomes exhibit cytotoxic effects on circulating tumor cells, suggesting their potential in NSCLC therapy.
EVs from other sources
In addition to MSC-EVs and immune cell EVs, EVs from other sources have also been further investigated. Yan et al. 22 demonstrated miR-31-5p delivered by milk exosomes accelerating diabetic wound healing through promoting angiogenesis. Zhou et al. 23 identified plasma-derived exosomal miR-15a-5p has the potential as a biomarker for early endometrial cancer detection. Not only can EVs be secreted by animal cells, plant cells can also secrete EVs 24 . Plant-derived EVs, as active ingredients of natural origin, have shown high advantages in terms of safety and functionality 25 . EVs derived from ginger can improve intestinal inflammation by modulating intestinal flora and increasing the release of anti-inflammatory factors 26 . Over the years, the research on plant-derived EVs such as ginseng and grape has attracted increasing attention, but the secretion mechanism and identification of plant-derived exosomes still need to be further explored.
EVs isolation methods
EVs are a promising field of research and have wide implications for the diagnosis, treatment and drug delivery of human diseases. Generally, EVs isolation is based on biophysical characteristics such as the size, density, charge and surface composition of EVs. Each method has its own advantages and disadvantages27,28. The increasing amount of research on EVs indicates that finding a more efficient and high-yield method of extracting them is crucial to developing the study and use of EVs-related medications. Table 1 lists the most common techniques for isolating EVs. With the continuous progress of technology, it is believed that there will be more efficient and convenient methods to obtain EVs in the future.
EVs isolation methods.

Role of EVs in lung disease
EVs in lung disease diagnosis
Investigating particular biomarkers and novel treatment strategies in lung diseases is urgently needed because lung diseases have emerged as a major health concern. Currently, EVs, particularly exosomes, have achieved significant breakthroughs in early disease detection and prognostic diagnosis34–36. EVs can be readily detected in various physiological fluids including blood, saliva, and urine. As illustrated in Fig. 1, the diverse contents of EVs are being actively explored for pulmonary disease diagnostics.

EVs as diagnostic (BALF: bronchoalveolar lavage fluid).
The level of gene expression in EVs offers a novel biomarker for lung disease diagnosis and progression. Gan et al. 37 reported eleven dysregulated exosomal circRNAs from the blood of idiopathic pulmonary fibrosis (IPF) patients, with significant increases in three circRNAs in IPF patients. Vázquez-Mera et al. 38 assessed microRNAs (miRNAs) profiles in serum exosomes of asthma patients and identified immune-related miRNAs (miR-21-5p, miR-126-3p, miR-146a-5p, miR-215-5p), which can be used as asthma phenotype/ endogenous and severity clinically relevant noninvasive biomarkers. Kaur et al. 39 identified that miR-122-5p was down-regulated three- to five-fold in lung-tissue-derived exosomes of COPD patients as compared to healthy nonsmokers and smokers, respectively. Sundar et al. 40 demonstrated that plasma-derived EVs miRNA-based molecular analysis hold significant potential for use in the development of biomarkers for the diagnosis, prognosis, and treatment of COPD. Furthermore, EVs-associated proteins also play a critical role for lung disease diagnosis. Enomoto et al. 41 identified surfactant-associated protein B (SFTPB) in serum EVs as a potential biomarker for progressive pulmonary fibrosis.
Lung cancer was the most frequently diagnosed cancer in 2022, responsible for almost 2.5 million new cases 1 . NSCLC and small cell lung cancer (SCLC) are the two primary types into which lung cancer can be generally divided from a histological and therapeutic standpoint 42 . Current and previous smoking, passive smoking, occupational exposure, and chronic obstructive pulmonary disease or pulmonary fibrosis are the primary risk factors for lung cancer 43 . Apart from low-dose spiral CT scanning, there is no effective technique for early detection of lung cancer.
Exosomal nucleic acids such as tRNAs, mRNAs, miRNAs, and long noncoding RNAs (LncRNAs) have been the subject of an increasing number of in recent years 44 . miRNAs play a significant role in the posttranscriptional control of biological processes. Cazzoli et al. 45 studied exosomes extracted from the blood of lung adenocarcinoma patients and found that the expression levels of four miRNAs carried by exosomes were higher than those of healthy smokers. It was discovered that exosomes from recurring tumors had significantly higher levels of miR-21 and miR-155, indicating that alterations in exosomal miRNAs might accurately reflect the clinical characteristics of lung cancer 46 . The proteins and DNA of EVs have been evaluated by an increasing number of researchers as potential diagnostic biomarkers for non-small cell lung cancer47,48. Through investigations of exosomal proteins, Sandfeld-Paulsen et al. 49 discovered that the exosomes of CD151, CD171, and tetraspanin eight differed significantly between the histological subtypes of cancer patients and noncancer patients. Studies have shown that estimated glomerular filtration rate (EGFR) mutation analysis of bronchoalveolar lavage fluid (BALF) or plasma EVs can obtain accurate, specific and rapid results50,51. To improve the prognosis of patients with lung cancer, further research into biomarkers and novel treatments is necessary.
EVs treatment of lung disease
EVs exhibit special benefits when used to treat lung diseases. In addition to lung diseases such as lung cancer and COPD, a large number of patients are also suffering from diseases like pulmonary fibrosis, interstitial pneumonia, and asthma. With age, the declining regenerative capacity of cells slows down lung injury repair52,53.
Mesenchymal stem cell–derived EVs have beneficial effects in the treatment of lung injury. The potential mechanisms of MSC-EVs in reducing lung injury have been investigated in many studies (Fig. 2). Studies have shown that MSC-Exos can regulate macrophage immune function, reduce pulmonary fibrosis and improve pulmonary hypertension in experimental mice54,55. Xu et al. 56 showed that MSC-Exos prevented excessive collagen formation and extracellular matrix deposition in the lungs by inhibiting the expression of TGF-β1, connective tissue growth factor (CTGF), and COL1A1 genes in fibroblasts via Let-7i-5p. Liu et al. 57 delivered miR-17-5p to receptor cells, via exosomes from human embryonic stem cells to target platelet-responsive protein-2 (Thbs2) to prevent pulmonary fibrosis. In addition, MSC-Exos can improve fibrosis by regulating fibroblast differentiation, as well as inducing apoptosis and senescence in fibroblasts and myofibroblasts 58 . Compared with cell therapy, MSC-EVs have a more flexible mode of drug delivery, and the use of nebulization enables the therapeutic drugs to reach the airways or lungs directly, requiring a smaller dose. In preclinical lung damage models, researchers present the biological distribution and effects of nebulized cells-derived extracellular vesicles (Hamsc-EVs) and examine the safety of nebulized administration 59 .

Mechanisms of MSC-EVs treat lung injury: (a) Nebulization is used to transfer EVs to the lungs for healing; (b) EVs enter target cells by three primary pathways: direct membrane fusion, receptor-mediated endocytosis, and direct endocytosis; (c) MSC-EVs are involved in the treatment of lung diseases via related signaling pathways54–57. A downward arrow indicates inhibition of the signal pathway, while an upward arrow indicates activation of the signal pathway; (d) EVs can attenuate the early inflammatory response by reducing the inflammatory response, differentiating macrophages from a pro-inflammatory M1 phenotype to an inhibitory inflammatory M2 phenotype, and reducing inflammatory cytokine release54,55; (e) EVs reduce lung fibrosis by inhibiting myofibroblast differentiation 56 ; (f) EVs deliver miR and protein to other receptor cells, such as alveolar epithelial cells. 58
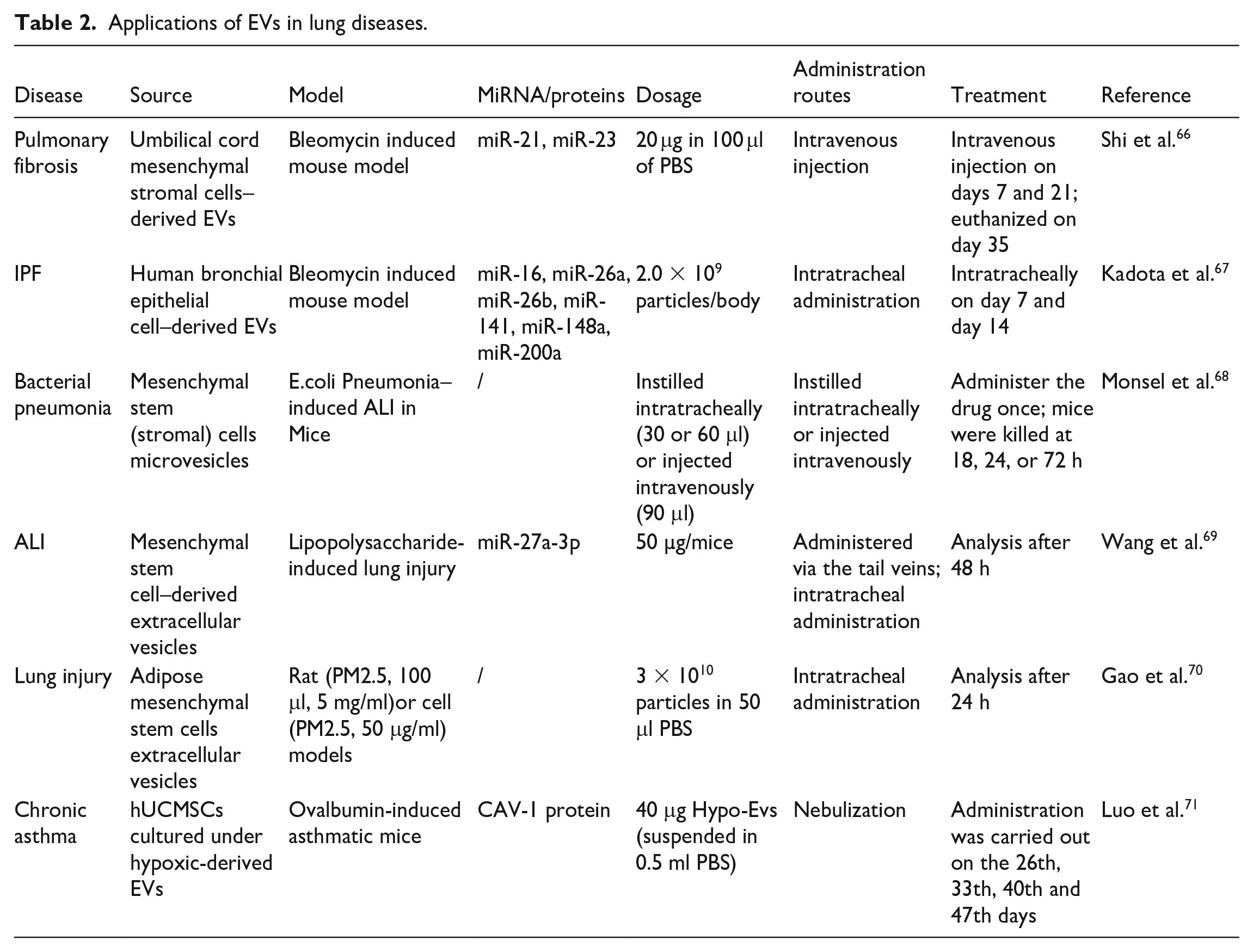
In addition, other types of vesicles have been actively studied in the treatment and prevention of lung diseases. Human bronchial epithelial cell-derived EVs were found to treat pulmonary fibrosis and lung epithelial cell senescence by inhibiting TGF-β-WNT crosstalk 60 . Fibroblast exosome-derived microRNA-22 regulates fibroblast differentiation to myofibroblasts to ameliorate fibrosis through the ERK1/2 pathway by inhibiting TGF-β1 induced α-SMA expression 61 . Analysis showed that inhalation of lung spheroid cell secretome (LSC-Sec) and exosomes (LSC-Exo) could reestablish normal alveolar structure, reduce collagen accumulation and myofibroblast proliferation to attenuate and resolve pulmonary fibrosis 62 . Previous studies demonstrated that ACE2-expressing human LSC-Exo could function as a prophylactic therapy to bind and neutralize SARS-CoV-2, protecting the host against SARS-CoV-2 infection 63 . Zhou et al. 64 found that endothelial progenitor cell exosomes carrying miR-126-3p and miR-126-5p could increase the expression of tight junction proteins and maintain the integrity of the alveolar epithelial barrier. Recent studies have revealed that M2-like macrophage-derived EVs play a protective role in the pathogenesis of acute lung injury/acute respiratory distress syndrome (ALI/ARDS), partly mediated by miR-709 65 . This offers a potential strategy for assessing disease severity and treating ALI/ARDS. With the discovery of increasingly diverse EVs functions, it provides more possibilities for the application of EVs in the treatment of lung diseases. Table 2 collects some of the studies on EVs in the therapeutic field of lung disease.
Applications of EVs in lung diseases.
Researchers and entrepreneurs have been tremendously excited about the development of EVs. There are already a variety of EVs-related clinical trials available on the ClinicalTrials.gov website, including several research initiatives on therapies for conditions including acute respiratory distress syndrome (ARDS), COVID-19, and cancer. Table 3 provides clinical research information on EVs used in the treatment of lung cancer, ARDS, COVID-19, and other illnesses. EVs-related treatment has been shown to be safe and effective in several of research fields.
Clinical applications of EVs in lung diseases.

Nanotechnology-based drug delivery systems provide a promising strategy for the targeted delivery of traditional chemical drugs and novel small molecule therapeutic agents to treat lung disease 72 . Various nanomaterials such as lipid nanoparticles and polymer nanoparticles have been applied in drug delivery, but these delivery mechanisms all have their own limitations73,74. Because of their small size, excellent safety, and ability to cross physiological barriers, EVs are considered to be a more effective drug delivery vehicles. EVs may carry a variety of medications, mostly small molecule medication (doxorubicin, curcumin, etc), protein medications (enzymes, transmembrane proteins, etc), and nucleic acid medications (miRNA, siRNA, etc) 75 . There are two main approaches for loading drug compounds into EVs: exogenous loading (postloading) and endogenous loading (preloading) 76 . Several drug loading strategies were compiled in Table 4. Wang et al. 77 used a simple slight sonication method to prepare PTX-M1-Exos delivery system, polarized M1 macrophage exosomes, which enhanced paclitaxel (PTX) anti-tumor effects. Zheng et al. 78 designed CAR-T-cell-derived exosome-encapsulated paclitaxel (PTX@CAR-Exos) for the treatment of a mouse model of lung cancer, and inhalation of PTX@CAR-Exos accumulated within the tumor region and reduced the tumor size. RuizdelRio et al. 79 demonstrated that fibroblast-derived EVs encapsulating the drug reduced fibrosis in profibrotic fibroblasts in vitro and also reached the fibrotic heart and lungs in vivo, improving the biodistribution of the free drugs. Exosome membrane of M2 macrophages and poly(lactic-co-glycollic) acid nanoparticles (PLGA NPs) wrapped with the smart silencer of Dnmt3aos were synthesized to treat asthma mice, which was followed by the investigation of therapeutic outcomes and the mechanism in vivo 80 . Wang et al. 81 explored M2 macrophage-derived exosomes as carriers for targeted antibiotic delivery (Antibiotics@Exos) have been used to optimize treatment strategies for bacterial pneumonia.
Different techniques for drug loading.

EVs are being researched more and more as a means for carrying nucleic acids 88 . Liu et al. 87 loaded IL-12 mRNA into human embryonic kidney cell-derived exosomes (HEK-Exo) by electroporation, generating loaded IL-12 mRNA exosomes (IL-12-Exo). Research conducted on mouse models of lung cancer has demonstrated that IL-12-Exo has a lower systemic toxicity and a better biological distribution in the lung cancer tumor microenvironment following inhalation administration than liposomes loaded with IL-12 mRNA (IL-12-Lipo) 87 .
EVs that have been preloaded in vivo can be offered targeted, unique cellular regulatory, and drug-therapeutic properties. Research has demonstrated that tLyP-1 exosome can transport siRNA to human non-small cell lung cancer cells 89 . Zhang et al. 90 developed an engineered MSC-Exos with SARS-CoV-2-S-RBD- and miR-486-5p- modification that suppressed ferroptosis and fibrosis of MLE-12 cells in vitro, and alleviated RILI and long-term RIPF in ACE2 humanized mice in vivo. The use of EVs as delivery vehicles has several benefits, but there are still many obstacles to overcome, such as increasing drug loading effectiveness and precisely locating cells and tissues in vivo. Since the current technical conditions are limiting, novel technologies are critically needed to solve these challenges91,92.
Prospect
Many breakthroughs have been made in the study of EVs, especially in the field of drug delivery, regeneration and tissue repair, and specific biomarkers for diagnosis. The introduction of in vitro diagnostic products based on EVs technology further proves the development potential of EVs in the field of diagnosis. It is expected that in the foreseeable future, the diagnosis area will continue to be the main exhibition area for EVs products. Although impressive findings have been made in the study of EVs, there are still obstacles to industrial transformation. For example, rapid clearance of EVs in vivo will limit the long-term therapeutic effects. Another challenge in the industrialization of EVs is the nonuniformity caused by various cell culture conditions and cell passage. Therefore, standardized protocols for the isolation and characterization of EVs are urgently needed.
EVs can be delivered to the target organs for treatment more efficiently by atomized inhalation, nasal drops and other drug delivery technologies. The application field of EVs will be further expanded by combining with a series of biomaterials. In addition, EVs are easy to be engineered, and the function of active targeting can be realized by adding targeting molecules. These advantages expand the potential of drug delivery from EVs. With the development of cell manipulation and bioengineering technology, engineering modification can highlight the best characteristics of EVs as vehicles, and might eventually become the mainstream trend. Notably, one pressing issue that engineered EVs must address is targeted specificity.
EVs have shown significant promise for use in the three main domains including diagnostic, treatment, and drug deliveries. It is anticipated that researchers will maintain their passion for this area of study and actively investigate the field of plant-derived EVs and engineered EVs. However, close integration with other emerging technologies is necessary for the development of EVs in the future. Examples include (1) combining microfluidic methods for effective enrichment; (2) using artificial intelligence for intelligent analysis; and (3) combining with biomaterials to achieve sustained release and regulation. In the future, only by strategically integrating other technologies and continuously overcoming challenges can the advantages of EVs be fully realized.
Footnotes
Acknowledgements
The authors thank ANEXT Shanghai Biotechnology Co., Ltd, and Oriental Beauty Valley Institute of Regenerative Medicine for continuous support of this work. All the figures in the manuscript were drawn in Figdraw. The authors thank their colleagues for helpful suggestions.
Ethical Considerations
Not applicable.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
Not applicable.
Author Contributions
Wang J: manuscript writing, collection of data; Shi YN: manuscript writing, collection of data; Su YH: manuscript writing, creation of figures; Pang CY: manuscript writing; Yang YM: manuscript writing; Wang WW: conception and design, manuscript writing, collection of data, interpretation of data.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
