Abstract
We previously found that intravenous injection of extracellular vesicles (EVs) from human adipose tissue–derived stem cells (hADSC) could ameliorate allergic rhinitis (AR) in mice through immunomodulatory effects. In clinical trials, nasal delivery has been an attractive treatment for AR. We sought to determine whether there are differences in the therapeutic effects between caudal injection and their combination. We treated AR mice with ADSC-EVs via caudal vein, nasal cavity, or both. After treatment, the mice were re-sensitized and the indices of behavior, nasal mucosa morphology, and cytokine secretion of the mice under different modes of administration were calculated. The resultes show that tail vein, nasal, and combined administration could effectively relieve the inflammatory infiltration of the nasal mucosa of mice, reduce the secretion of IgE, IL-4, and other inflammatory factors, and alleviate the Th1/Th2 imbalance. Injection and nasal delivery, as well as their combination, effectively alleviated the symptoms of rhinitis in mice. Nasal administration has a better therapeutic effect when the inflammatory response is mild. It could be speculated that ADSC-EVs have excellent properties in the treatment of AR, and modes of administration can be selected for different stages of treatment in clinical therapy.

Keywords
Introduction
Mesenchymal stem cell–derived extracellular vesicles (MSC-EVs) have attracted ever-increasing attention due to their high biosafety, low immunogenicity, ease of storage and transport, and their broad utilities with numerous preclinical and clinical studies conducted in diverse disease settings1–3. Drug administration strategies could have broad influences on their efficacy and implementation. For MSC-EVs, a highly heterogeneous, plastic, and versatile entity known for its context-dependent pleiotropic effects, the routes of delivery, could have even more profound impact. However, efforts remain rare to comprehensively investigated whether and how different administration strategies may impact the therapeutic mechanisms and efficacy of MSC-EVs with any complex disease settings4,5.
Allergic rhinitis (AR) is one of the most common diseases encountered by medical professionals and believed to comprise both local and systemic diseased components, potentially an ideal disease setting to test MSC-EV pleiotropic mechanisms and/or effects 6 . The prevalence of AR is difficult to measure accurately because of the “mild” symptoms and the fact that many people with AR do not seek professional help but self-medicate 7 . Surveys indicate that AR affects more than 400 million people worldwide, and in some countries, more than 40% of the population suffer from AR8–10. Pharmacotherapy and allergen-specific immunotherapy (AIT) are treatments for AR11–13. However, adherence to pharmacotherapy is poor in many patients. The adverse reactions of AIT are relatively common13,14. There is an urgent need to research more effective and safer therapies.
MSC-EVs could effectively improve the microenvironment of AR by influencing T-cell differentiation and dendritic cell (DC) function15,16 and are being considered as a potential and effective cell-free therapy for AR 17 . We have recently shown that administration of hADSC-EVs through tail veil injection could effectively restore nasal epithelium integrity and attenuate AR symptoms in a mouse model 16 . Nonetheless, for AR management, clinical treatment has shown that effective delivery to the nasal cavity remains a significant obstacle 18 . International guidelines recommend topical therapy and intranasal sprays as targeted therapies for AR 6 . Topical therapy can be delivered directly to the site of disease, reducing the likelihood of systemic side effects and improving treatment efficacy 19 . In the case of antihistamines, oral and intranasal antihistamines were selected in different therapeutic settings 20 . Clearly, different routes of drug administration may affect their efficacy in AR treatment and should be carefully investigated.
In the present study, we aim to comprehensively investigate whether and how three different administration strategies, including tail vail injection, intranasal application, and a combination of both, could impact the therapeutic mechanisms and effects of hADSC-EVs in mouse AR models. Our study shows that while all three administration strategies were useful in mouse AR treatment, intranasal application and tail veil injection of hADSC-EVs may have distinct but complementary therapeutic mechanisms and effects. Overall, intranasal application of hADSC-EVs could be preferable with mild AR for easy execution and less adverse effects and a combination of both might be advantageous for severe AR when pleiotropic mechanisms and therapeutic effects are desired.
Materials and Methods
Cell Culture
The hADSCs were cultured in DMEM/F12 medium (Gibco, New Mexico, USA) with 10% fetal bovine serum (FBS) (Gibco), 100 units/ml penicillin‒streptomycin (Gibco), and 40 μg/ml basic fibroblast growth factor (Proteintech, Wuhan, China). FBS was centrifuged at 130,000 × g for 90 min prior to use. The cells were cultured in a 5% CO2 humidified atmosphere at 37°C, and the medium was changed every 3 days. After attaining 80% confluency, hADSCs were detached from the 10-cm dish using 0.05% trypsin-EDTA (Gibco) and replated.
Identification and characterization of hADSCs were performed with fluorescence activated cell sorting (FACS) analysis, and adipogenic differentiation and osteogenic differentiation assays were performed as previously reported 16 .
EVs Extraction and Characterization
The culture medium of hADSCs between passages 4 and 6 was used to collect hADSC-EVs. The hADSC-conditioned medium was collected from approximately 80% confluent hADSCs. The medium was centrifuged at 300 × g for 15 min and 4,000 × g for 15 min at 4°C to further remove cells and dead cells. Then, the samples were centrifuged at 10,000 × g for 30 min at 4°C to remove cellular debris by a High Speed Bench-top Centrifuge (Neofuge160R; Shanghai Lishen Scientific Instrument Co., Ltd, China), and the supernatants were ultracentrifuged at 130,000 × g for 90 min at 4°C using an Optima XPN-100 (BECKMAN COULTER Life Science, USA). The precipitate was collected and suspended in phosphate-buffered saline (PBS) for immediate use.
EVs were then characterized by transmission electron microscopy. The concentration of EVs and their size and mass were measured by a particle matrix (Matric, Mel Busch, Germany) under 405 nm emission light. The hADSC-EVs were analyzed by Western blotting as previously reported 16 .
Animal Preparation
Female BALB/c mice (Shanghai Jiesijie Laboratory Animal Co., Ltd., Shanghai, China) were used and bred in a specific pathogen-free (SPF) animal facility. All animal care and experimental procedures were performed in accordance with National Institutes of Health guidelines and approved by the Institutional Animal Research Ethics Committee of Tongji Hospital.
OVA-Induced AR and Therapeutic Protocols
AR mice were sensitized using OVA (Sigma-Aldrich, St. Louis, MO, USA) and aluminum hydroxide (Alum) (Pierce Chemical Co., Rockford, IL, USA)21–23. Briefly, mice were sensitized using a previously described protocol by intraperitoneal (IP) injection of 200 μl ovalbumin solution (200 μg OVA emulsified in 20 mg of alum in a total volume of 200 μl)24,25. The ovalbumin solution was used to sensitize the mice 3 times on day 1 and day 15. Twenty microlitres of ovalbumin solution (20 μg OVA emulsified in 2 mg of alum in a total volume of 20 μl) was administered as an instillation through the nose. The ovalbumin solution was used to challenge the mice 7 times on days 22 to 28.
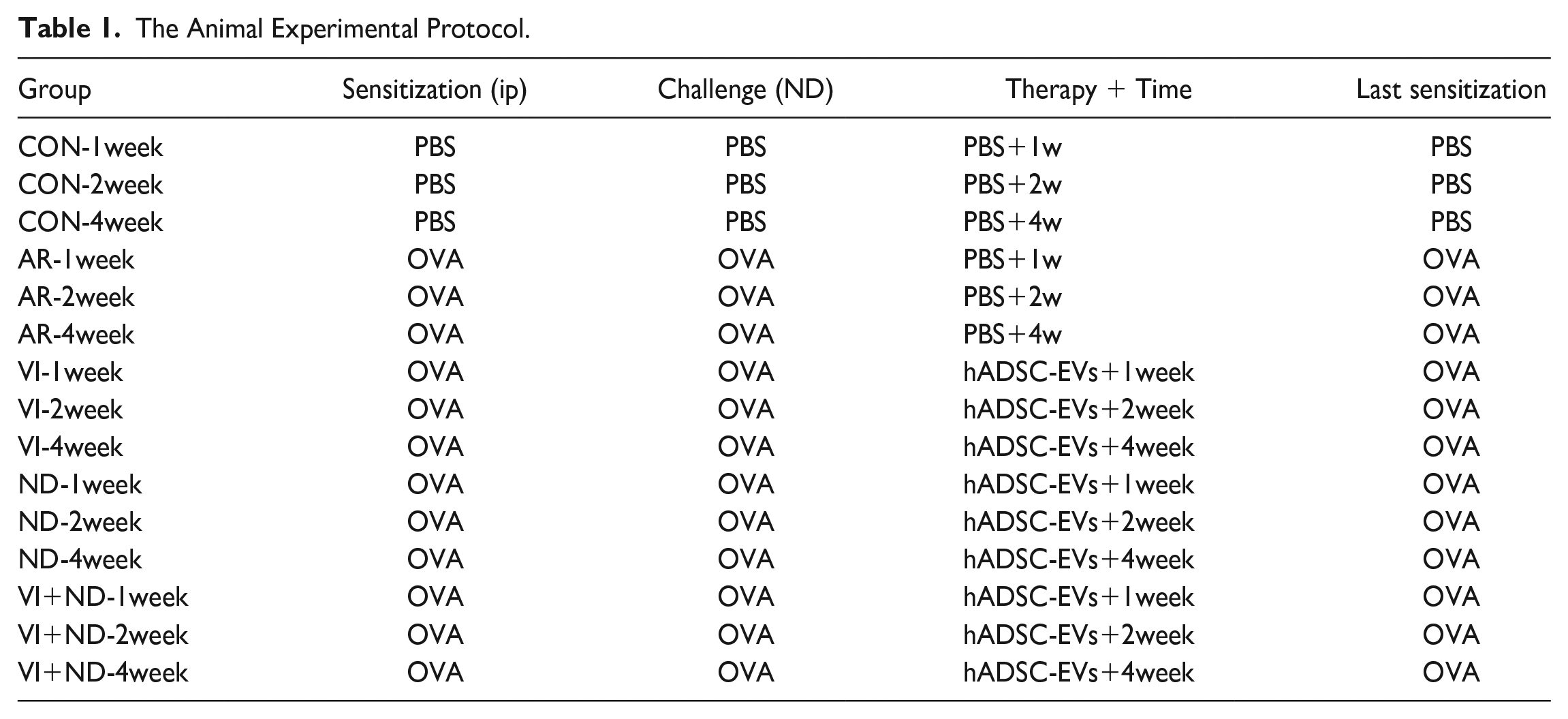
Animals in the sham group received saline instead of OVA during the sensitization and challenge steps. Forty-five female BALB/c mice were divided equally into three groups: the 1-week treatment group (n = 15), the 2-week treatment group (n = 15), and the 4-week treatment group (n = 15). The weekly treatment group was further divided into five equal subgroups: the full control group (n = 3), the AR group (n = 3), the Tail vein injection (VI) group (n = 3), the Nasal delivery (ND) group (n = 3), and the VI+ND group (n = 3).
One day after the last challenge, the VI groups were injected with hADSC-EVs via the tail vein. In the ND group, hADSC-EVs were administered intranasally. In VI+ND group, hADSC-EVs were administered intranasally and intravenously, respectively. The hADSC-EVs were administered every 5 days during treatment. In the treatment group, the hADSC-EV dose was administered as follows: 0.5 × 106 cell secretions, 200 μl (Table 1).
One-week treatment group: hADSC-EVs were administered 2 times on days 29 and 34.
Two-week treatment group: hADSC-EVs were administered 3 times on days 29, 34, and 39.
Four-week treatment group: hADSC-EVs were administered 6 times on days 29, 34, 39, 44, 49, 54, and 57.
Mice in the 1-week group, 2-week group, and 4-week group were sacrificed on days 36, 43, and 57, respectively.
The Animal Experimental Protocol.
Characterization of Nasal Allergic Symptoms
Sneezing and nasal rubbing were characterized. In detail, the characterization method was as follows: On the day of the last challenge (day 28), the sum of the nasal scratching and sneezing events of each mouse was recorded within 15 min. After the above methods of treatment, the control group was stimulated by PBS once, and the AR model group and treatment group were stimulated by OVA once before sacrifice (days 36, 43, 57). The sum of nasal scratching, sneezing events, and runny noses of each mouse was observed and recorded within 15 min after stimulation. The frequencies of sneezing, nasal rubbing, and runny noses were recorded by two observers in a blind manner and scored on a behavioral rating scale.
Histologic Analysis and Immunohistochemistry
After the mice were sacrificed by cervical dislocation, the nasal mucosa was collected and washed with PBS, fixed with 4% paraformaldehyde, and embedded in paraffin wax. The paraffin blocks of nasal mucosa were sectioned serially and stained with an hematoxylin and eosin (H&E) and periodic acid-Schiff (PAS) Staining Kit (Wuhan Xavier Biotechnology Co., Wuhan, China). Finally, images were collected and analyzed on a digital section scanner (Pannoramic 250 FLASH; 3DHISTECH, Budapest, Hungary).
Measurement of Serum OVA-Specific IgE and Cytokines
After symptom scoring, blood samples of mice were immediately collected via eyeball, clotted at room temperature for 20 min, and centrifuged at 2,000 × g for 20 min to extract serum. The levels of OVA-specific IgE, OVA-specific IgG, IL-4, and IFN-γ were measured by an enzyme-linked immunosorbent assay (ELISA) kit (AMEKO, Shanghai, China) in accordance with the manufacturer’s instructions.
Quantitative Real-Time Polymerase Chain Reaction Analysis
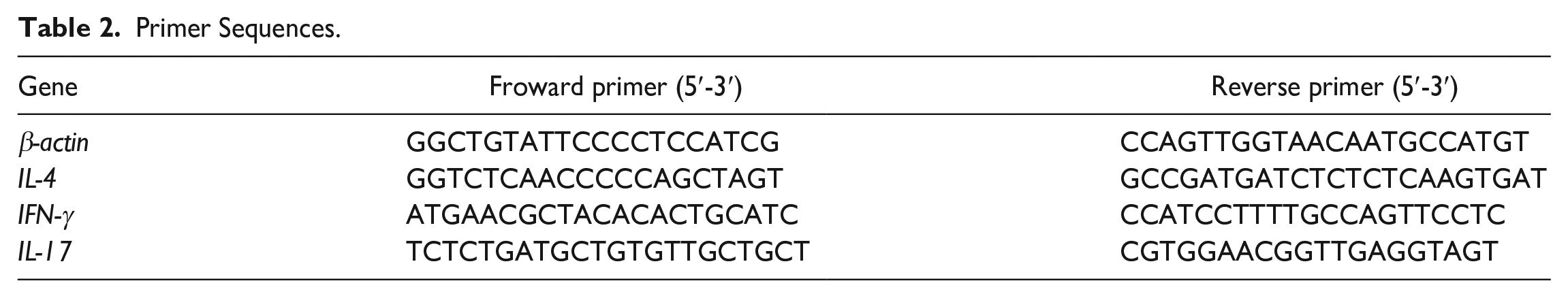
Fresh spleens of mice were separated under aseptic conditions after sacrifice. Total RNA was extracted from the spleen using the TRIzol reagent kit (Invitrogen, Carlsbad, CA, USA). Equivalent amounts of RNA were reverse transcribed using the one-step First Strand cDNA Synthesis SuperMix (Novoprotein, Suzhou, China). The mRNA expression analysis was performed using the QuantStudio 5 Real Time PCR System (Thermo Fisher Scientific Inc., MA, USA). The corresponding primers are shown in Table 2. The average transcript levels of genes were normalized to β-actin levels. The relative expression levels of target genes were calculated by the 2−ΔΔCT method.
Primer Sequences.
Flow Cytometry Analysis
Mouse spleen lymphocytes were isolated with mouse lymphocyte isolation solution (Dakewe Biotech Co., Ltd., Shenzhen, China) and stimulated with Cell Activation Cocktail (Biolegend, San Diego, CA, USA). Then, lymphocytes were blocked with anti-mouse CD16/32 and stained with FITC anti-mouse CD4 antibodies, and membrane rupture was performed with buffer. Finally, APC-IL-4 and Brilliant Violet 421-IFN-γ were added for intracellular staining. Data were acquired using a Cytoflex LX flow cytometer (Beckman Coulter, Brea, CA, USA) and analyzed using FlowJo software (Becton, Dickinson and Company, Franklin Lakes, NJ, USA)
Treatment Effectiveness Index Analysis
The Treatment Effectiveness Index (TEI) is a novel parameter that has been developed based on data standardization methods (maximum-minimum normalization) and z-score. The purpose of its development was to facilitate more effective comparison of differences in treatment effectiveness26–28. The TEI is calculated as the ratio of the difference between the measured values in the AR group and the treatment group to the difference between the measured values in the AR group and the control group.

Statistical Analysis
The results were shown as the mean ± standard deviation (SD). Multiple groups were compared with analysis of variance (ANOVA) or Student’s t test using SPSS software 26.0 (SPSS Inc., Chicago, IL, USA). Results with P value of <0.05 were considered significant. The statistical results were performed by using GraphPad Prism version 8 (GraphPad Software, San Diego, CA, USA).
Results
Tail Vein and Intranasal Administration of hADSC-EVs Potentially Had Complementary Therapeutic Effects on AR Mouse Symptoms and Tissue Damage
The hADSCs and hADSC-EVs were characterized as previously reported 16 , both conformed to classical characteristics of ADSC and EVs (Supplementary Figure 1)29,30. Although we have recently showed that hADSC-EVs via tail vein injection could alleviate symptoms in AR mice 16 , clinical experience and data suggested that AR patients are more compliant with intranasal sprays and drops. It is important to consider if topic administration could have a same/better therapeutic efficacy with fewer side effects 10 . The patients seeking treatment share a common goal of symptom relief and continued administration of treatment can further reduce symptoms31–33. Therefore, we designed and investigated the therapeutic effect of intranasal delivery of hADSC-EVs and compare it with tail vein injection. We also tested the combinatorial therapy of the two routes with the dose of each was halved.
As shown in Fig. 1A, the mice were sensitized and then challenged for a period of 28 days. Subsequently, they received tail vein injections (VI group), intranasal drips of hADSC-EVs (ND group), or combinations of both (VI+ND group) for 4 weeks. We began our analysis with evaluating changes in nasal symptoms, the primary concern for AR patients, through various AR relevant behavioral assays a week after the final treatments. All three groups exhibited a significant improvement in behavioral symptoms, such as sneezing, nose rubbing, and/or runny nose, compared with the AR group (P < 0.05) (Fig. 1B). Closer examination suggests that intranasal application was effective in treating running nose but not sneezing and/or rubbing. In contrast, tail vein injection was effective in treating sneezing and rubbing but not running nose. When combined, tail vein injection together with intranasal application had higher efficacy on reducing the symptoms than either one alone, strongly suggesting that tail vein and intranasal administration of hADSC-EVs potentially had complementary therapeutic mechanisms and effects on AR mice. Eosinophilic infiltration and destruction of nasal mucosa are main characteristics of AR34,35. As determined by H&E and PAS staining, the three strategies all reduced eosinophil infiltration and goblet cell production (P < 0.05) with the combinatorial strategy having strongest improvement in the tissue integrity (Fig. 1C, D).

Tail vein and intranasal administration of hADSC-EVs had complementary therapeutic effects on AR mouse symptoms and tissue damage. (A) Experimental scheme. (B) Quantification of symptom scores, including sneezing and rubbing in 15 min after final OVA sensitization. (C) H&E staining and quantification of eosinophils in the nasal mucosa. (D) PAS staining and quantification of goblet cells in the nasal mucosa. *P < 0.05; **P < 0.01; ***P < 0.001. Significant difference analysis was performed between AR group and control group, treatment group (VI, ND, and VI+ND group) and AR group. n = 6 or 24.
hADSC-EV Treatment Improved Inflammatory Environment in AR Mice
Then, we decided to determine the serum IgE (sIgE) and IgG4 (sIgG4) levels, usually elevated in AR patients36–38. All three treatments significantly reduced sIgE level. Although only the ND group had a significant reduction in sIgG4 level, the other two groups also showed a strong trend toward reduction when compared with the AR one (Fig. 2A, B). A disbalance between Th1 and Th2 cells plays a major role in AR development and progression. IL-4 can be produced by various cells, for example, T cells, and considered a major Th2 cell cytokine39,40. IFN-γ can also be secreted by various cells, for example, T and natural killer (NK) cells, and functions as a key Th1 cell cytokine. The level of IL-4 and IFN-γ closely related to AR development and pathology. As determined by ELISA analysis, all three treatments attenuated the increased secretion of IL-4 and IFN-γ (P < 0.05) caused by AR (Fig. 2C). Quantitative real-time polymerase chain reaction (qRT-PCR) analysis further confirmed that both IL-4 and IFN-γ transcripts were reduced in the three treated groups compared with the AR group (P < 0.005 and P < 0.05, respectively, Fig. 2D).

hADSC-EVs ameliorated inflammatory environment in AR mice. (A-C) Levels of sIgE (A), IgG4 (B), IL-4 (C), and IFN-γ (C) in the serum measured by ELISA. (D) Relative expression of IFN-γ and IL-4 in the serum by qRT-PCR. *P < 0.05; **P < 0.01; ***P < 0.001. Significant difference analysis was performed between AR group and control group, treatment group (VI, ND, and VI+ND group) and AR group. n = 9.
hADSC-EV Treatment Restored the Homeostasis of Th1, Th2, and Th17 Cells
Consistent with the above results, flow cytometry analysis indicated that all three strategies substantially restored the balance between Th1 and Th2 cells (P < 0.01) (Fig. 3A, B). Besides disbalance between Th1 and Th2 cells, an imbalance between regulatory T cells (Treg) and Th17 also contributes to AR 41 . Patients with allergic fungal rhinosinusitis exhibited increased secretion of IL-17 and a shift in the Treg/Th17 balance toward Th1742,43. Therefore, we investigated Th17 differentiation and found significant reduction in Th17 cells after treatment in the VI and the combinatorial groups (P < 0.05 and Fig. 3A, C). The ND group showed a decreasing trend, albeit the difference was not significant. Furthermore, as determined by qRT-PCR analysis, nasal delivery of hADSC-EVs resulted in a significant decrease in IL-17 expression compared with the AR group (P < 0.05), albeit the other two groups showed a downward trend (Fig. 3D).

The hADSC-EV treatment improved inflammatory environment by restoring the balance Th1/Th2 cells. (A) Flow cytometry analysis of splenic Th1, Th2, and Th17 cells. (B and C) Ratios of Th1/Th2 cells and Th17 calculated according to the results from (A). (D) Relative expression of IL-17 mRNA by qRT-PCR. (E) The radar chart of TEI comparison between the three administration strategies. *P < 0.05; **P < 0.01; ***P < 0.001. Significant difference analysis was performed between AR group and control group, treatment group (VI, ND, and VI+ND group) and AR group. n = 9.
Since some biological indicators alone showed no significant difference in the therapeutic effects of the three delivery routes, we hoped to utilize the TEI indicators proposed in the preceding study to provide a comprehensive comparison of the differences between them 16 . After homogenizing TEI for each group under a same indicator, we conducted an overall comparison and found that the ND group had overall higher TEI scores with more top-ranked indicators (Fig. 3E), suggesting that with 4-week repetitive administration strategies, intranasal delivery could represent an effective option, albeit combinatorial strategy might have better efficacy in symptom reductions. The findings of the above indicate that intranasal drug delivery is more efficacious when employed in conjunction with a 4-week dosing strategy or when utilizing repeated dosing regimens.
Partial Spontaneous Recovery Was Observed With AR Mice During 4-Week Habituation
Our results so far implied that 4-week hADSC-EV treatment could attenuate AR regardless of the delivery routes. Nonetheless, the 4-week treatment scheme had potential complications from 4-week removal of allergens and recovery after AR model preparation. To examine that, we decided to compare the control and AR mouse models between different timepoints (Fig. 4A). As expected, there was obvious difference between the control and the AR mice across all times (Fig. 4B–E). Close examination revealed that the difference for a same group of AR mice between the time of model preparation (day 28) and the time of examination (day 36—week 1, day 43—week 2, and day 57-week 4) increased over time from weeks 1, 2, to 4 in terms of sneezing, nose rubbing, and running nose, suggesting a progressive symptom improvement.

Partial spontaneous recovery was observed with AR mice during 4-week habituation. (A) Experimental schematic. (B–D) Quantification of sneezing, rubbing, running nose, and symptom scores (including sneezing, rubbing, and runny nose) in 15 min after final OVA sensitization upon 1-week, 2-week, and 4-week treatment. (E) The TEI of PBS in (B-D). (F) Level of IgE, IgG4, IL-4, and IFN-γ in the serum measured by ELISA. *P < 0.05; **P < 0.01; ***P < 0.001.
To demonstrate this difference more directly, the behavioral data of the AR group under different treatment durations were transformed by TEI. In contrast to the treatment group, which received ADSC-EVs, the AR group was administered PBS at each treatment. Consequently, this variation was designated as “the therapeutic effect of PBS..” The results demonstrated that, irrespective of the specific condition (e.g., scratching the nose, runny nose, or sneezing), the therapeutic effect of PBS exhibited a favorable trend over time (Fig. 4E). This finding underscores the notion that while the AR models with varying treatment durations have been proven to be effective, they do exhibit distinct differences in their underlying mechanisms.
We speculated that the possible reason for this phenomenon was that with the prolongation of treatment time, the time period between the establishment of the mouse AR model and the last allergic excitation before euthanasia was extended. During this period, the mice demonstrated alterations in inflammation due to factors such as the elimination of allergens and their own recovery. Consequently, mice treated for varying durations prior to the final allergic stimulus exhibited disparate levels of behavioral indices.
It is important to note that the experimental design was not sufficiently extensive to permit the collection of inflammatory factors and other indicators prior to the final allergic provocation of the mice. Consequently, it was not possible to ascertain whether there were any significant differences in the level of inflammation before the final allergic provocation of the mice across the various treatment periods.
There was no significant difference in the secretion of sIgE and IFN-γ in mice under different treatment durations after receiving the last allergic stimulation (Fig. 4F). This may indicate that the last allergic stimulation can eliminate the difference between the mice’s escape from allergens and their own recovery during the treatment cycle. It also demonstrates the feasibility of our experimental model to a certain extent.
Combinatorial Administration Manifested Higher Efficacy in AR Mice for 1- and 2-Week Treatment Schemes
To exclude differences resulted from spontaneous recovery, we then designed 1- and 2-week treatment schemes and investigated the therapeutic effects with different delivery routes (Fig. 5A). Like the 4-week observations, hADSC-EVs significantly improved AR symptoms regardless of the administration strategies (Fig. 5B). Also as observed with the 4-week treatment scheme, the ND groups generally had better improvement with running nose symptom and the combinatorial ones overall had stronger therapeutic efficacy in terms of AR symptom improvement, for both 1- and 2-week schemes (Fig. 5B). Further staining of the nasal mucosa showed significant reduction in eosinophil infiltration and goblet cell differentiation (P < 0.01 and P < 0.01, respectively, Fig. 5C, D). The levels of sIgE, IFN-γ, and IL-4 were too reduced (Fig. 5E, F). As well, the balance between Th1, Th2, and Th17 was restored, albeit overall to less extent than that for the 4-week treatment groups and some failed to reach a significance (Fig. 6A, B). When normalized and quantified, the combinatorial groups overall had the highest TEI scores with more top-ranked indicators and the ND groups had the lowest ones with least top-ranked indicators, for both 1- and 2-week schemes (Fig. 6C). In contrast to a 4-week dosing strategy, combined dosing may be particularly effective under a 1-week repeated dosing regimen. This finding suggests that intranasal drug delivery achieves optimal efficacy when used in conjunction with a repeated dosing strategy, whereas combined dosing is more advantageous for single-dose or short-term dosing regimens.

Combinatorial administration manifested higher efficacy in reducing AR mouse symptoms for 1- and 2-week treatment schemes. (A) Experimental scheme. (B) Quantification of symptom scores, including sneezing and rubbing 10 min after the final OVA sensitization. (C) H&E staining and PAS staining. (D) Quantification of eosinophils in the nasal mucosa and goblet cells in the nasal mucosa. (E) Levels of sIgE, IFN-γ and IL-4 in the serum measured by ELISA. (F) Relative expression of IFN-γ and IL-4 by qRT-PCR. *P < 0.05; **P < 0.01; ***P < 0.001. Significant difference analysis was performed between AR group and control group, treatment group (VI, ND, and VI+ND group) and AR group. n = 6, 24, or 9.

Combinatorial administration manifested overall higher therapeutic efficacy for 1- and 2- week treatment schemes. (A) Flow cytometry analysis of Th2 cells in splenic lymphocytes. (B) The ratio of Th1/Th2 cells and Th17 calculated according to the results from A. (C) Relative expression of IL-17 by qRT-PCR. (D) The radar chart of TEI comparison among three routes of delivery after 1-week and 2-week treatment. *P < 0.05; **P < 0.01; ***P < 0.001. Significant difference analysis was performed between AR group and control group, treatment group (VI, ND, and VI+ND group) and AR group. n = 9.
Discussion
Drug delivery strategies are expected to have profound impact on their efficacy and/or broad implementations. Lately topical therapy, for example, intranasal drug administration, has become a preferred treatment option for AR symptom management6,44. However, researchers most often choose intravenous and/or IP injections as the preferred modes of administration, and very few studies have explored and compared the therapeutic effects of other administration routes (Supplementary Table 1)16,45,46. Currently, there is only one paper that has compared the effect of intravenous and abdominal administration of MSCs 47 , calling for more comprehensive comparisons between different administration routes, especially intranasal ones. In the present study, we sought to comprehensively compare for the first time the therapeutic effects of hADSC-EVs with three different administration strategies. Our results showed that hADSC-EVs had the ability to alleviate AR mouse symptoms, regardless of the administration routes and/or treatment durations. All strategies resulted in behavioral (sneezing, rubbing, and running nose), cellular (eosinophil infiltration, goblet cell, Th1, Th2, and Th17 differentiation), and molecular (cytokine secretion) improvements.
Intriguingly no single administration strategy consistently gave better results in all the parameters examined across the three treatment durations, perhaps reflecting the underlying complexity as what is observed in numerous stem cell and/or extracellular vesicle–related clinical trials. Importantly, intranasal administration always had better efficacy in treating running nose, whereas intravenous (IV) administration was more effective in treating sneezing and/or rubbing behaviors, strongly suggesting that the two routes may have rather distinct therapeutic mechanisms and/or effects. Overall, the combinatorial administration strategy had higher efficacy in terms of symptom improvement across the three different treatment durations and a simultaneous local nasal and systematic IV application of EVs seem to be a better administration optional for AR treatment. This predication is further supported by TEI analysis. For 1- and 2-week treatment schemes, the combinatorial groups (IV+ND) showed the most optimal treatment effects with the ND manifesting the least. Nonetheless, with 4-week repeated administration strategies, intranasal delivery could still represent an effective option and the ND groups had overall higher TEI scores when partial spontaneous recovery was present or diseases were mild, even though the combinatorial ones still had better efficacy in terms of symptom improvement. It is hypothesized that intranasal drug delivery systems may achieve greater efficacy when multiple doses are administered over a repeated time period. In summary, while all three administration strategies were effective in treating AR, intranasal application of hADSC-EVs could be a viable option mild AR for its easy execution with less adverse effects and the combinatorial strategy might be advantageous for severe AR when pleiotropic effects and therapeutic mechanisms are desired.
Earlier clinical studies showed that repeated short-term oral cortisol treatment was more effective than intranasal cortisol spray at the same dose and frequency for patients with severe chronic rhinitis—sinusitis or AR with severe symptoms48–50. A similar conclusion was reached that a single dose of intranasal administration was less effective than a single oral administration 51 . In line with the above observations, we have seen that direct intranasal application alone was not as effective as the other two strategies when the symptoms and diseases were severe. Nonetheless, intranasal application could be a treatment option when the symptoms and diseases were not severe. This is indeed in line with both human and animal studies showing nasal decongestants, anticholinergics, and chromones were effective for mild AR treatment or as adjunctive therapy52–55. Together, ours and existing studies all suggest that intranasal administration may be more appropriately used as a treatment for mild AR56,57. Reports also exist showing that in the postoperative treatment of patients with more severe allergic fungal rhinitis-sinusitis (AFRS), subcutaneous injection of biologics (e.g., omalizumab, dupilumab, and mepolizumab) was effective in treating patients with resistant AFRS 58 , presumably because local treatment could achieve high-concentration and high-dose administration more safely57,58.
Our results clearly show an advantage of combinatorial administration strategy over the other two in 1- and 2-week treatment. We believe these likely reflect the nature of severe AR diseases as both a local and a systemic disease. Thus, to achieve optimal therapeutic efficacy, both local and systemic therapeutic mechanisms and effects are desired especially when diseases and symptoms are severe. Combinatorial treatment strategies are indeed routinely chosen in the clinics and could achieve exceptional results due to different treatment mechanisms when complex disease symptoms could not be controlled with a single drug44,59. But as several meta-analysis report, it is almost inevitable that combinatorial approach also increases the occurrence of adverse events 55 . Most of combinatorial treatment strategies utilized two or more drugs each with a distinct delivery route and rarely tested on a single entity. EVs are known to have pleiotropic functions. We suspect that delivery of hADSC-EVs through different routes help EVs to function in concert with route-specific therapeutic mechanisms. It is conceivable that intranasal application of hADSC-EVs could execute local immunomodulatory, tissue reparative, and regenerative functions, whereas tail injection could function mainly through systemic immunomodulation. When diseases and symptoms are more severe and complex, especially when involving multiple local mechanisms, for drug entities like hADSC-EVs, it may be highly desirable to consider combinatorial administration strategies such as simultaneous treatments with tail veil injection and intranasal application.
Conclusion
In this study, we aim to determine whether and how different administration strategies impact therapeutic efficacy of EVs from hADSC in mouse AR treatment. Our results show that while all three administration strategies are effective in AR treatment, intranasal application of hADSC-EVs could be preferable with mild AR for easy execution and less adverse effects and a combinatorial strategy might be advantageous for severe AR when pleiotropic mechanisms and therapeutic effects are desired. Alternatively, intranasal administration appears to exert its optimal efficacy following repeated dosing, whereas combined administration demonstrates superior effectiveness after a single dose or a limited number of administrations. Clearly, the therapeutic mechanisms and/or effects of hADSC-EVs vary contingent on the delivery routes. Thus, the administration strategies of hADSC-EVs should be differentially designed and carefully investigated for diseases (e.g., AR) and patients of different complexities, with different severity, and/or at different stages.
Supplemental Material
sj-docx-1-cll-10.1177_09636897251325673 – Supplemental material for Administration Strategy-Dependent Mechanisms and Effects of Human Adipose Tissue Stem Cell Extracellular Vesicles in Mouse Allergic Rhinitis Treatment
Supplemental material, sj-docx-1-cll-10.1177_09636897251325673 for Administration Strategy-Dependent Mechanisms and Effects of Human Adipose Tissue Stem Cell Extracellular Vesicles in Mouse Allergic Rhinitis Treatment by Wenhan Yang, Zhiyu Pan, Jiacheng Zhang, Lian Wang, Ju Lai, Kai Fan, Jingjing Zhu, Qian Liu, Yalei Dai, Jieyu Zhou, Shuhui Wu, Zhengliang Gao and Shaoqing Yu in Cell Transplantation
Supplemental Material
sj-docx-2-cll-10.1177_09636897251325673 – Supplemental material for Administration Strategy-Dependent Mechanisms and Effects of Human Adipose Tissue Stem Cell Extracellular Vesicles in Mouse Allergic Rhinitis Treatment
Supplemental material, sj-docx-2-cll-10.1177_09636897251325673 for Administration Strategy-Dependent Mechanisms and Effects of Human Adipose Tissue Stem Cell Extracellular Vesicles in Mouse Allergic Rhinitis Treatment by Wenhan Yang, Zhiyu Pan, Jiacheng Zhang, Lian Wang, Ju Lai, Kai Fan, Jingjing Zhu, Qian Liu, Yalei Dai, Jieyu Zhou, Shuhui Wu, Zhengliang Gao and Shaoqing Yu in Cell Transplantation
Footnotes
Acknowledgements
This work was supported by the National Key R&D Program of China, National Science Foundation of Shanghai, National Science Foundation of China, Clinical Research Plan of SHDC, Shanghai Oriental Talent Programme. The funders had no role in the study design, analyses, nor preparation of this manuscript.
Author Contributions
Ethical Approval
The study was reviewed and approved by the Ethics Committee for Clinical Research of Tongji University (TJAA09123101).
Statement of Human and Animal Rights
This article does not contain any studies with human subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Availability of Data and Materials
All relevant data are within the manuscript and its Additional files, and the original data are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Key R&D Program of China (No. 2022YFC2504100), National Science Foundation of Shanghai (No. 23ZR1458000), Shanghai Hospital Development Center foundation (No. SHDC12024126), Shanghai Oriental Talent Programme (WSJ2411).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
