Abstract
Vascularized composite allotransplantation (VCA) has emerged as an innovative strategy to restore form and function in patients with severe tissue defects involving anatomical regions such as the face, hand, and abdominal wall. Composite allografts are composed of diverse tissues, including skin, muscle, bone, vasculature, nerves, and mucosal surfaces, posing unique challenges in immunological management. Clinical outcomes following VCA surgeries have been encouraging; however, a comprehensive understanding of the underlying cellular interactions and molecular pathways is still predominantly derived from studies in solid organ transplantation (SOT). Recent advances in SOT have identified mitochondria as crucial therapeutic targets capable of mediating transplant rejection, mitigating ischemia-reperfusion injury (IRI), and enhancing graft longevity. Nevertheless, the explicit role and potential therapeutic applications of mitochondria within VCA remain largely unexplored. This review aims to critically examine and elucidate the significance of mitochondria in overcoming the current limitations encountered in VCA surgery. A deeper insight into mitochondrial biology could hypothetically provide clinicians and researchers with novel, targeted therapeutic strategies to improve clinical outcomes in VCA; however, these approaches require further validation in preclinical models.
Keywords
Graphical Abstract

Introduction
Vascularized composite allotransplantation (VCA) represents a groundbreaking approach in reconstructive surgery, designed to restore both form and function through composite tissue grafts1–3. By extending beyond traditional reconstructive techniques, VCA offers transformative solutions for patients suffering severe injuries or irreversible tissue loss, such as significant facial deformities or limb amputations4,5. Since its inception in the 1990s, the frequency and variety of VCA procedures—including face and hand transplantations—have steadily increased, achieving encouraging mid- and long-term clinical outcomes6,7. VCA grafts are heterogeneous, incorporating tissues such as skin, muscle, bone, vasculature, and nerves, each carrying distinct antigenic properties and subsequently posing unique immunological challenges8–10. While past studies primarily concentrated on skin immunogenicity, recent findings have identified oral and nasal mucosa as additional antigenic sites in VCA grafts 11 .
Given the heightened immunogenic potential of composite tissues, lifelong immunosuppressive therapy is essential to prevent acute and chronic rejection (CR), maintain graft viability, and preserve functional outcomes 12 . Identifying key cellular and molecular mediators within the immune rejection cascade could provide new avenues for therapeutic interventions and improved patient management 13 .
Traditionally recognized as cellular powerhouses responsible for adenosine triphosphate (ATP) production via oxidative phosphorylation, mitochondria have increasingly been recognized for their broader roles in cellular processes such as signaling, apoptosis, calcium homeostasis, and inflammatory regulation. Recent developments in solid organ transplantation (SOT) have demonstrated that mitochondrial-targeted therapies effectively reduce ischemia-reperfusion injury (IRI), decrease mitochondrial reactive oxygen species (ROS) production, and enhance overall graft function14,15. However, targeted mitochondrial therapies remain understudied within the specific context of VCA.
This short review explores mitochondria-targeted therapeutic approaches as a means of addressing significant barriers currently faced in VCA, potentially enhancing patient outcomes and expanding clinical applicability.
Mitochondria in VCA surgery—panacea or potemkin villages?
Beyond their classical role in ATP generation, mitochondria regulate diverse biological processes, including calcium homeostasis, cell differentiation and activation, apoptosis, and inflammation, positioning them as attractive therapeutic targets across various disease states16–20. Current mitochondrial therapeutic strategies encompass promoting mitochondrial biogenesis, repairing mitochondrial DNA, modulating mitochondrial epigenetics, and altering mitochondrial dynamics through targeted antioxidants, peptides, and pharmacological modulators16,17,21. Emerging treatments, such as photobiomodulation aimed at mitochondrial cytochrome C via nitric oxide release, have further expanded the therapeutic potential of mitochondrial interventions22,23.
In transplantation, mitochondrial dysfunction is critically implicated in immune modulation and the pathogenesis of IRI24–29. During ischemic episodes, mitochondrial metabolism is profoundly disrupted, rapidly depleting cellular ATP and causing accumulation of harmful metabolites like succinate. Subsequent reperfusion results in ROS generation, activating inflammatory pathways characteristic of IRI, thus presenting opportunities for therapeutic intervention.
Expanding the donor and recipient pool
The limited availability of suitable donors significantly restricts the widespread adoption of VCA. According to analyses by Mendenhall et al., less than half of all brain-dead donors in the United States meet the inclusion criteria for VCA donation, underscoring the substantial gap in donor availability 30 . In addition, public awareness and understanding of VCA remain markedly limited, further exacerbating donor shortages and constraining the growth and acceptance of this transformative surgical approach 1 . Another significant obstacle lies in the allocation and distribution of VCA grafts, which initially occurs through organ procurement organizations (OPOs). Organ distribution staff typically possess limited familiarity with VCA procedures compared to SOT, potentially introducing allocation biases and suboptimal graft distribution 31 . Unlike SOT, where matching primarily relies on tissue compatibility and blood type, VCA introduces additional critical aesthetic considerations such as skin color, limb size, and facial symmetry, essential for ensuring seamless physical and psychological integration of the transplanted tissues into the recipient’s body32–35. While these aesthetic factors significantly contribute to successful outcomes, they markedly complicate the matching and allocation process, thus further constraining donor-recipient compatibility36,37. Consequently, expanding the donor pool has emerged as an urgent priority within VCA to advance clinical feasibility and patient access. Recent clinical milestones underscore the expanding boundaries of VCA practice. Notably, the first full-face transplantation in a Black patient illustrated both the surgical feasibility and the unique immunologic and aesthetic considerations in racially diverse populations, including challenges in rejection monitoring and donor matching based on skin tone 38 . Moreover, advanced age remains an exclusion criterion in VCA, mainly due to increased perioperative complication risks in elderly recipients, emphasizing the need for strategies to safely include older donor populations 34 . In parallel, successful facial retransplantation after irreversible graft failure has demonstrated that reoperation is a viable option in select cases, though technically demanding and immunologically complex 39 .
Beyond VCA-specific constraints, a substantial number of potentially viable organs across transplantation medicine are discarded annually due to mitochondrial dysfunction and ischemic injury, underscoring the critical need for improved preservation techniques 40 . Machine perfusion methods, capable of extending preservation beyond the typical 6- to 12-hour timeframe associated with static cold storage, offer promising potential to expand donor availability and facilitate therapeutic graft modifications prior to transplantation41,42. Numerous studies have shown that ex vivo machine perfusion significantly reduces IRI, mitigates inflammation, and enhances graft viability and functional recovery43–46. Specifically, mitochondrial transplantation during ex vivo lung perfusion has demonstrated efficacy in reducing ischemic damage and improving subsequent graft performance14,47. Haug et al. have also demonstrated the feasibility of normothermic ex situ machine perfusion of VCAs with oxygen microcarriers, enabling real-time mitochondrial redox quantification and supporting graft viability over extended periods 48 . Similarly, kidney model studies have shown that mitochondrial transplantation effectively alleviates ischemic injury and positively modulates metabolic pathways related to energy production and mitochondrial health 14 . Mitochondrial transfer also promotes mitophagy in recipient cells, a cytoprotective process that enhances cellular survival under ischemic and reperfusion stress conditions, further highlighting the therapeutic potential of mitochondria-focused interventions 49 . In addition, perfusion solutions containing mitomycin C have successfully extended heart graft viability, illustrating the versatility of ex vivo perfusion strategies 50 . Collectively, these findings suggest that targeted mitochondrial interventions could considerably improve preservation outcomes, potentially increasing preservation times and substantially broadening the donor pool for VCA transplantation 51 .
Mitochondrial dysfunction has been widely recognized as a hallmark of aging and age-related disorders, emphasizing the potential of mitochondria-targeted therapies to improve tissue quality and increase lifespan52–54. Approaches aimed at modulating ROS, NAD+, and AMP through targeted mitochondrial therapies hold considerable promise in mitigating age-associated functional decline 55 . Investigations into dietary compounds, metabolic modulators, and pharmacological agents designed to enhance mitochondrial biogenesis, function, and mitophagy have provided varying degrees of success, with mixed outcomes reported in both animal and human trials56,57. Further research into the intricate relationships between mitochondrial health, aging processes, and tissue quality is critical for future advancements in donor tissue optimization, potentially further expanding VCA donor eligibility and improving long-term graft outcomes.
Mediating acute and CR episodes
VCAs inherently carry significant immunological complexities due primarily to the pronounced immunogenicity of their skin components and the general absence of major histocompatibility complex (MHC) matching 58 . This immunological mismatch predisposes recipients to acute rejection (AR), one of the most frequent complications following VCA surgery. Studies consistently demonstrate that approximately 85% of VCA patients experience at least one AR episode within the first postoperative year, with more than half of recipients encountering multiple rejection events, highlighting the critical need for innovative strategies to mitigate rejection and enhance graft survival59,60. Recent evidence shows that mucosal rejection may involve distinct immune pathways not captured by traditional skin biopsies. A study of nine facial VCA recipients revealed that while both skin and mucosa show T-cell-mediated rejection, the mucosa uniquely exhibits B-cell infiltration and occasional plasma cells, suggesting an under-recognized mucosal immune response 61 . Despite the high incidence of AR, the underlying cellular and molecular mechanisms, as well as effective therapeutic options, remain incompletely understood and require further elucidation59,62,63.
CR, distinct from AR, manifests as a progressive and irreversible process occurring later in the post-transplantation period, characterized by functional deterioration and accelerated aging of the graft. Although the precise cellular cascades contributing to CR remain under investigation, existing evidence suggests that both direct cellular immune responses and antibody-mediated mechanisms, particularly targeting the graft’s skin and vasculature, play critical roles in this phenomenon64–66. Current immunosuppressive regimens for preventing and treating rejection episodes demonstrate suboptimal efficacy and frequently present significant adverse effects, necessitating the development of novel and targeted therapeutic approaches67,68.
Emerging evidence indicates a critical role for mitochondria in modulating transplant rejection dynamics. Recent studies have shown that circulating extracellular mitochondria, present in deceased organ donors, can activate endothelial cells, thereby exacerbating allograft rejection through the induction of adhesion molecules and inflammatory cytokines 26 . Consequently, strategies aimed at reducing circulating mitochondria represent promising therapeutic avenues. One such approach involves administration of β2-glycoprotein I, which facilitates clearance of extracellular mitochondria by promoting their binding and subsequent phagocytosis by macrophages, potentially reducing their immunogenic impact 69 .
In addition, mitochondria-derived damage-associated molecular patterns (mtDAMPs) released following IRI injury significantly contribute to the activation of recipients’ immune responses, potentially exacerbating transplant rejection 29 . Cytosolic mtDAMPs have been demonstrated to activate host inflammasome complexes, leading to chemokine secretion and heightened inflammatory reactions. Extracellular mtDAMPs, released following cellular damage, directly stimulate neutrophils and T-cells, amplifying inflammatory cascades that compromise graft viability and function 70 . Consequently, mitigating the release and inflammatory impact of mtDAMPs emerges as a promising strategy for improving clinical outcomes following VCA procedures. Therapeutic interventions targeting mtDAMP reduction include pharmacological agents such as tranexamic acid and induction of PINK-1-mediated mitophagy, both demonstrating potential efficacy in preclinical models71,72.
Furthermore, transplantation of autologously derived mitochondria has demonstrated encouraging results in protecting tissues from IRI across various preclinical and limited clinical contexts, suggesting its potential applicability in mitigating rejection episodes within VCA73,74. Specifically, mitochondrial transplantation has been shown to enhance ATP production, reduce oxidative stress, and promote cellular repair mechanisms, thereby improving overall tissue resilience and recovery post-transplantation75–77. Importantly, evidence suggests minimal or no immune rejection occurs following transplantation of donor-derived (allogeneic) mitochondria, indicating that mitochondrial transplantation does not trigger conventional immune rejection responses typically associated with whole organ transplantation 78 . This unique immunological tolerance to transplanted mitochondria offers the exciting potential of using exogenous mitochondrial therapies to facilitate tissue recovery, reduce immunological injury, and significantly enhance graft survival and function in VCA recipients.
Side effects of immunosuppressive regimes
Immunosuppressive therapy remains a cornerstone of successful VCA, yet it is accompanied by significant adverse effects that can negatively impact patient health and graft outcomes. Long-term immunosuppressive regimens substantially increase the risk of malignancies and metabolic disorders, including diabetes mellitus, hypertension, and hyperlipidemia, thereby compounding overall patient morbidity and mortality risks79–82. Furthermore, immunosuppressive agents impair the immune system’s ability to effectively respond to local infections and inflammatory conditions. Consequently, this heightened susceptibility predisposes patients to chronic infections at the graft site or in adjacent tissues, potentially compromising wound healing, graft integrity, and overall functional outcomes83,84. Effectively balancing immunosuppression to prevent rejection while minimizing associated side effects constitutes a major clinical challenge in managing VCA recipients. Excessive immunosuppression poses severe risks, including life-threatening infectious complications and sepsis, whereas inadequate immunosuppression markedly increases the likelihood of acute and CR episodes 81,83–86. Therefore, the lack of safe, effective, and low-side-effect immunomodulatory strategies continues to represent a significant obstacle in advancing patient care within the field of VCA.
Many of the detrimental side effects associated with commonly used immunosuppressive medications are intricately linked to mitochondrial dysfunction. For instance, mycophenolate mofetil has been shown to adversely affect mitochondrial function in T-cells, resulting in decreased ATP production and heightened cellular apoptosis, which may reduce immune system efficacy and resilience 87 . Similarly, tacrolimus has been observed to disrupt mitochondrial calcium uptake in pancreatic beta-cells, consequently impairing insulin secretion and increasing the risk of diabetes mellitus post-transplantation 88 . In addition, cyclosporine-induced neurotoxicity is believed to arise from interference with mitochondrial energy metabolism, further emphasizing the central role of mitochondrial dysfunction in immunosuppressive drug-associated adverse outcomes 89 . These mitochondria-related disruptions contribute to a range of severe side effects, including diabetes, nephrotoxicity, neurotoxicity, and other metabolic and neurological disorders, collectively impairing patient survival, graft longevity, and quality of life 90 .
Mitochondrial-targeted therapeutic strategies, particularly the use of antioxidants such as MitoQ, have demonstrated promising potential in mitigating oxidative stress and improving outcomes in preclinical models, particularly in cardiac IRI scenarios 91 . Overall, the data on this field are scarce, but this approach could potentially be applied to mitigate the side effects of immunosuppressive regimens, offering a new therapeutic strategy for transplant recipients (Figure 1 and Figure 2) 92 .

Overview of key challenges in VCA surgery. (a) The limited donor pool results from stringent medical and aesthetic matching criteria and low public awareness. (b) High immunogenicity of skin and incomplete MHC matching predispose patients to frequent acute rejection episodes and eventual chronic rejection. (c) Long-term immunosuppression, while essential, is linked to infections, malignancies, and metabolic complications; balancing effective rejection prophylaxis with minimal toxicity remains an ongoing challenge.
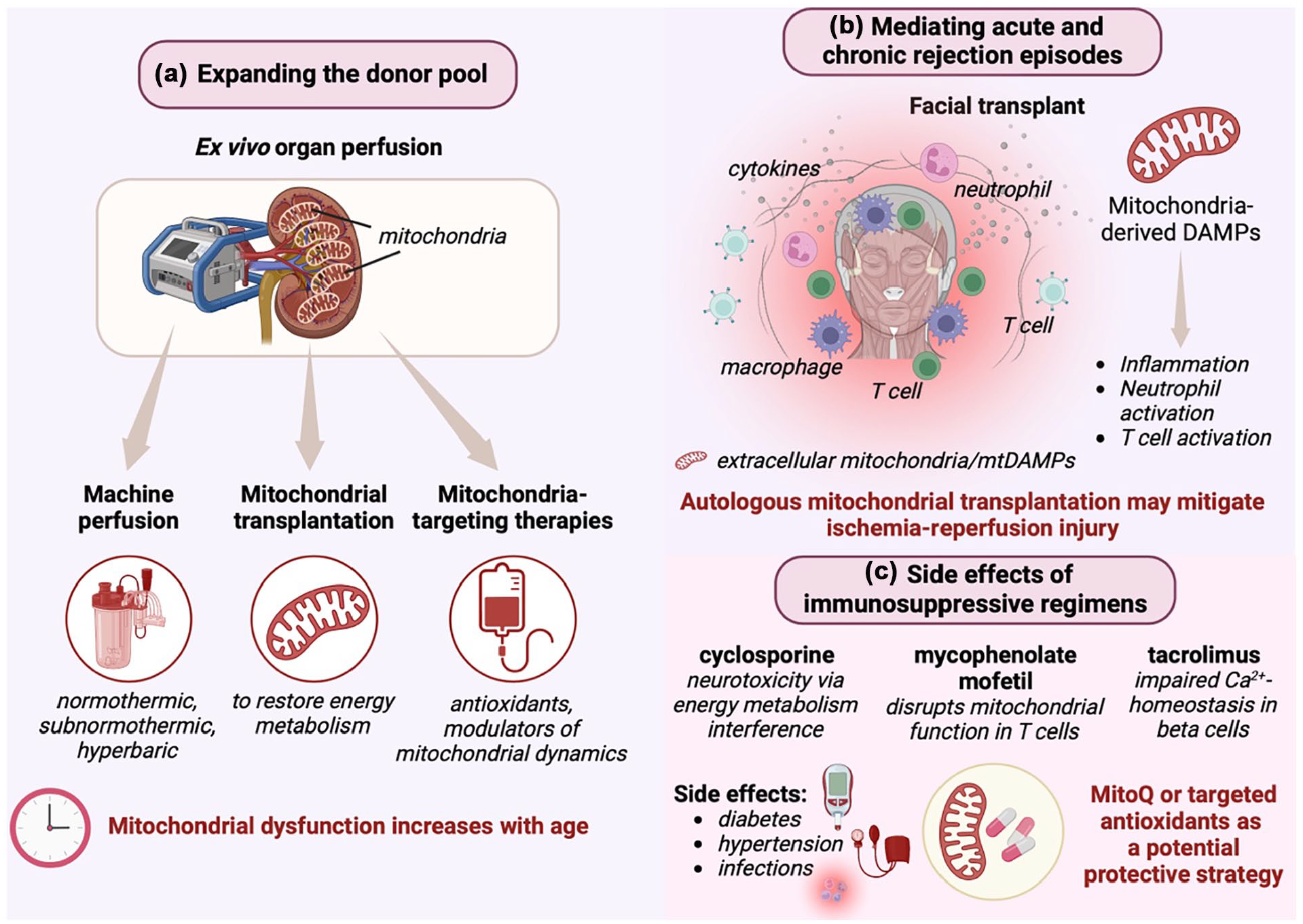
Mitochondria represent a critical therapeutic target in VCA surgery. (a) Novel strategies, including machine perfusion and mitochondrial transplantation, can rescue ischemic or suboptimal grafts and potentially expand the donor pool by improving tissue viability and even mitigating age-related mitochondrial decline. (b) Mitochondria-derived damage-associated molecular patterns (mtDAMPs) drive acute and chronic rejection; modulating mtDAMP release, enhancing mitophagy, and adopting mitochondrial transfer techniques may reduce graft inflammation. (c) Immunosuppressive medications can disrupt mitochondrial energy metabolism, leading to metabolic dysfunction, neurotoxicity, and other adverse effects. Antioxidants targeting mitochondria (e.g., MitoQ) hold promise for mitigating these complications.
Current challenges and future outlook
VCA represent a significant advancement in reconstructive surgery. They offer transformative outcomes for patients with devastating injuries or severe tissue loss. However, to further expand the field of VCA, several major challenges need to be addressed. Those challenges include donor scarcity, acute and CR episodes, and the side effects of immunosuppression. Our exploration highlights the multifaceted role of mitochondria as a potential therapeutic target to address these barriers. Briefly, mitochondrial therapeutics, such as antioxidants and transplantation, have shown potential in mitigating IRI, extending tissue preservation times, and improving donor tissue quality, which could help expand the donor pool and address constraints related to age or tissue quality. In addition, strategies to reduce mtDAMPs, such as promoting mitophagy or targeted antioxidant therapies, may attenuate acute and CR episodes. To counteract the side effects of lifelong immunosuppressive regimens, mitochondrial-targeted approaches like MitoQ could alleviate associated metabolic and oxidative stresses, enhancing recipient safety. Together, these advances could transform VCA outcomes by addressing both immediate and long-term challenges.
Despite their potential, mitochondrial-targeted therapies face several challenges in their future development. A key issue is the need to combine selective mitochondrial delivery with specific organ targeting 93 . This is especially problematic in VCA, since VCA grafts consist of multiple different tissues. Various strategies have been developed to deliver molecules specifically to mitochondria, including conjugation to lipophilic cations and peptides93,94. These approaches have been used to create mitochondria-targeted antioxidants and redox probes93,95. Systemic delivery of mitochondria-targeted molecules can be achieved using viral vectors, allowing organ-specific targeting 96 . The distribution of mitochondria within cells is regulated by motor proteins and adaptor molecules 97 . Oral administration of mitochondria-targeted compounds has shown promising results in mice, with accumulation in tissues affected by mitochondrial dysfunction 93 . Combining mitochondrial targeting with cell-specific delivery remains a challenge but holds potential for treating various diseases98,99. A promising arising treatment strategy is the transplantation of healthy autologous mitochondria. Animal studies and some clinical trials have shown potential benefits, but several challenges need to be addressed before widespread clinical application100,101. 4These challenges include optimizing delivery methods, ensuring long-term stability and function of transplanted mitochondria, and evaluating safety and efficacy in human trials101,102. Researchers are exploring new techniques for mitochondrial isolation, preservation, and targeted delivery to overcome these obstacles103,104. In addition, understanding the mechanisms of mitochondrial internalization and integration, as well as potential immune responses, is crucial for advancing this therapy104,105. Despite these challenges, mitochondrial transplantation remains a promising avenue for treating various diseases associated with IRI.
Conclusion
In conclusion, mitochondria represent a novel and promising therapeutic target for addressing critical challenges in VCA. By leveraging their central role in cellular energy production, immune modulation, and tissue repair, mitochondrial-targeted therapies offer potential solutions to expand the donor pool, reduce rejection episodes, alleviate immunosuppressive side effects, and combat aging-related complications. Continued interdisciplinary collaboration and translational research are essential to harness the full potential of these strategies, ultimately improving the quality of life and long-term outcomes for VCA recipients.
Footnotes
Ethical Considerations
This study was approved by our Institutional Review Board.
Author contributions
Leonard Knoedler: Conceptualization, Investigation, Writing—original draft, Writing—review & editing, Visualization, Project administration. Daniel H. Mendelsohn: Conceptualization, Investigation, Writing—original draft, Writing—review & editing, Visualization. Loïc Van Dieren: Investigation, Writing—original draft, Writing—review & editing. Thomas Schaschinger: Investigation, Writing—original draft. Cosima C. Hoch: Investigation, Writing—review & editing. Max Heiland: Writing—review & editing, Supervision. Jasper Iske: Writing—review & editing, Visualization. Felix J. Klimitz: Writing—review & editing, Visualization. Maxime Jeljeli: Writing—review & editing, Supervision. Korkut Uygun: Writing—review & editing, Supervision. Curtis L. Cetrulo: Writing—review & editing, Supervision. Alexandre G. Lellouch: Conceptualization, Writing—original draft, Writing—review & editing, Visualization, Supervision, Project administration.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of human and animal rights
This article does not contain any studies with human or animal subjects.
Statement of informed consent
There are no human subjects in this article and informed consent is not applicable.
Data Availability Statement
The references used for the writing of this manuscript are publicly available.
