Abstract
Subcutaneous transplantation is garnering attention as a potential transplantation site for pluripotent stem cell–derived islet cells to address the shortage of pancreatic islet transplant donors. However, subcutaneous transplantation of cells presents challenges related to angiogenesis, which is necessary for successful islet bioproduction. This study aimed to investigate a novel method for enhancing vascularization at the transplant site and thereby promote islet engraftment using a clinically available bioabsorbable medical device. A nonabsorbable device (agarose) or a bioabsorbable device (collagen–gelatin sheet [CGS]) loaded with basic fibroblast growth factor (bFGF) was implanted subcutaneously in C57BL/6 mice. There were two other groups of mice, one of which was implanted with CGS alone, which acted as a control, and another group that was implanted with bFGF-loaded agarose rods. Subsequently, 200 islets were transplanted into the subcutaneous pre-vascularized sites. An equivalent number of islets was also transplanted into the portal vein (IPTx) to compare transplantation efficacy. Vascularization of the graft site was evaluated before and after transplantation. bFGF significantly enhanced angiogenesis in the CGS mice. The normalization rate of blood glucose levels following islet transplantation in the bFGF-loaded CGS was group comparable to that in the bFGF-loaded agarose rod and IPTx groups. The presence of islets was confirmed using single-photon emission computed tomography (SPECT/CT), histological examination. Furthermore, it was noted that blood glucose levels rapidly increased after graft removal, showing that graft function was crucial to maintain normoglycemia. Importantly, the bFGF-loaded CGS showed a high rate of engraftment. This novel bioabsorbable medical device method exhibited remarkable efficacy in enhancing subcutaneous islet engraftment, potentially paving the way for a more straightforward and less invasive approach for islet cell transplantation in future clinical applications.
Keywords
Introduction
Cell transplantation is a minimally invasive therapeutic approach to restore impaired organ and cellular functions 1 . One of the most widely used forms of cell transplantation is islet transplantation, which serves as a β-cell replacement therapy for patients with type 1 diabetes who suffer from episodes of severe hypoglycemia 2 .
Currently, allogeneic islet transplantation involves the transplantation of islets into the liver via the portal vein. Intrahepatic exposure of islets to the recipient’s blood elicits an instant blood-mediated inflammatory reaction (IBMIR) 3 , characterized by platelet consumption and activation of coagulation and complement systems. This results in the loss of numerous transplanted islets in the liver during early transplantation, which is a significant limitation of intrahepatic portal vein transplantation and suggests that the liver may not be an optimal site for islet transplantation 4 . Importantly, allogeneic islet transplantation cannot be performed on demand as it requires pancreas donation, which is obtained from donors after brain or cardiac death. Researchers are currently exploring pluripotent stem cell–derived 5 and xenogeneic (porcine) 6 islet cell transplantation to address this limitation. However, these proposed novel islet cell sources come with concerns regarding malignant neoplasia and infection. Moreover, the difficulty in removing grafts from the liver has highlighted the need for alternative sites. Requisite factors for islet transplantation sites include proximity to the vascular network, suitability for minimally invasive graft removal, and low inflammation to suppress immunogenicity. Subcutaneous tissue has been the focus of many studies owing to its suitability for minimally invasive graft removal and capacity to accommodate many cells. It is considered safe and amenable to multiple transplantations, which has prompted numerous studies. However, subcutaneous tissue has poor blood flow, necessitating vascular induction before transplantation for islet engraftment. Various approaches have been proposed to address this, including the creation of pre-vascularized sites using vascular access catheters 7 , the use of scaffolds to promote angiogenesis 8 , the development of transplantable microvascular meshes 9 , the incorporation of device-free islet viability matrices 10 , the use of microencapsulation devices 11 , the formation of vascularized β-cell spheroids through 3D layer-by-layer cell coating technology 12 , and the delivery of exogenous growth factors using biomaterials 13 . Although pre-vascularized sites using vascular access catheters 7 and scaffolds to promote angiogenesis 8 have been proposed, these methods often face challenges such as limited long-term viability, potential adverse immune responses, and practical difficulties in clinical application. For instance, vascular access catheters 7 may lead to thrombosis over time, while scaffolds 8 can elicit an immune response that jeopardizes the graft.
We have previously reported a method of pre-vascularization via the subcutaneous insertion of agarose rods loaded with basic fibroblast growth factor (bFGF), considering the inflammatory response during the induction period 14 . bFGF and agarose rods were used as implantation site–forming agents. bFGF, which primarily contributes to angiogenesis, has a short half-life 15 ; however, cross-linking with agarose gel results in a sustained release of bFGF at the site 16 . Importantly, agarose lacks an established safety profile in humans and is not approved for clinical use. Therefore, an appropriate alternative to agarose needs to be found. Given concerns regarding bleeding and inflammation during removal of implants, a clinically approved bioabsorbable material that does not require removal from the implantation site would be an ideal candidate for enabling sustained bFGF release in clinical applications. To induce vascularization at the subcutaneous site and create an ideal environment for islet engraftment, a “simple” and “reliable” method should be employed. The use of non-bioabsorbable materials in islet transplantation has been associated with prolonged inflammatory responses and necessitates surgical removal, posing additional risks to patients. A clinically approved bioabsorbable material, which would obviate the need for surgical removal while minimizing inflammation, represents a critical unmet need in enhancing the safety and efficacy of islet transplantation. This study presents the application of a bioabsorbable material, which is clinically utilized for skin wound healing and is hypothesized to promote angiogenesis, to establish a graft site for subcutaneous pancreatic islet transplantation. By exploring this approach, we aim to provide a more effective, safer, and clinically feasible alternative for subcutaneous islet transplantation.
Materials and methods
Animals
For all experiments, we used male C57BL/6 mice aged 6–8 weeks, obtained from either Shimizu Laboratory Supplies (Kyoto, Japan) or Japan SLC Inc. (Shizuoka, Japan). The Institutional Animal Care Committee at Kyoto University approved all animal protocols (C23-79). Mice were maintained under standard conditions and provided unrestricted access to food and water, except during fasting periods. Appropriate anesthesia was administered during all surgical procedures.
Pancreatic islet isolation
Pancreatic islets were isolated from mice using a modified protocol derived from previously established methods 17 . C57BL/6 donor mice were anesthetized via isoflurane inhalation, and the distal common bile duct was clamped. To distend the pancreas, Hanks balanced salt solution (HBSS) containing 0.15 mg/ml collagenase P (Roche Diagnostics, Indianapolis, IN, USA) was carefully introduced into the common bile duct. The pancreas was then excised and subjected to an 18-min incubation at 37°C to facilitate tissue dissociation. Subsequently, the dissociated tissue was washed with HBSS and purified using discontinuous density gradient centrifugation, employing layers with densities of 1.110, 1.103, 1.096, and 1.070 g/ml. Islet fractions were collected from each gradient layer and further purified by centrifugation. Before transplantation, the isolated islets were cultured overnight in RPMI1640 medium supplemented with 10% fetal bovine serum (Thermo Fisher Scientific, Waltham, MA, USA) under controlled conditions (37°C, 5% CO2, and a humidified atmosphere with 95% air).
Induction of diabetes and recipient preparation
Diabetes was induced in C57BL/6 recipient mice by a single intraperitoneal injection of streptozotocin (STZ, 150 mg/kg; Wako, Japan) administered 3–6 days before transplantation. Blood was collected from the tail vein, and glucose concentrations were measured using an Accu-Chek glucose meter (Roche Diagnostics K.K., Tokyo, Japan). Diabetes was confirmed when non-fasting blood glucose readings were 400 mg/dl or higher in two successive measurements.
Preparation of the bFGF-loaded collagen–gelatin sheet and agarose rod
To prepare the bFGF-loaded CGS, a circular piece of collagen–gelatin sheet (CGS) (PELNAC Gplus, PNG-SD82060; Gunze Medical Limited, Osaka, Japan) with a diameter of 1 cm was cut. Next, a solution containing 12.5 μg of bFGF (Fibrast Spray; Kaken Pharmaceutical Co., Tokyo, Japan) was evenly applied on to the CGS, resulting in a final concentration of 16 μg/cm² of bFGF. Before subsequent experiments were conducted, the bFGF concentration on the CGS was reconfirmed using previously described protocols18,19.
To prepare the bFGF-loaded agarose rods, we followed a protocol adapted from a previous study 14 . Briefly, a 4.5% agarose solution was prepared by dissolving 450 mg of agarose (Seakem GTG agarose; Cambrex Bio Science Rockland, Inc., Rockland, ME, USA) in 10 ml of double-distilled water. The solution was sterilized by autoclaving and poured into a tube with an inner diameter of 4 mm. To promote gelation, the tubes were placed on ice. After solidification, the gel was cut into rod-shaped segments, each 1.7 cm in length. These segments were frozen at −20°C overnight and subjected to freeze-drying for 24 h under reduced pressure. Then, a solution containing 50 μg of bFGF and 25 μg of heparin (Mochida Pharmaceutical Co. Ltd., Tokyo, Japan) was evenly applied onto the surface of the freeze-dried agarose rods. The treated agarose rods, containing both bFGF and heparin, were stored at 4°C overnight to ensure stability before use.
Transplantation protocol
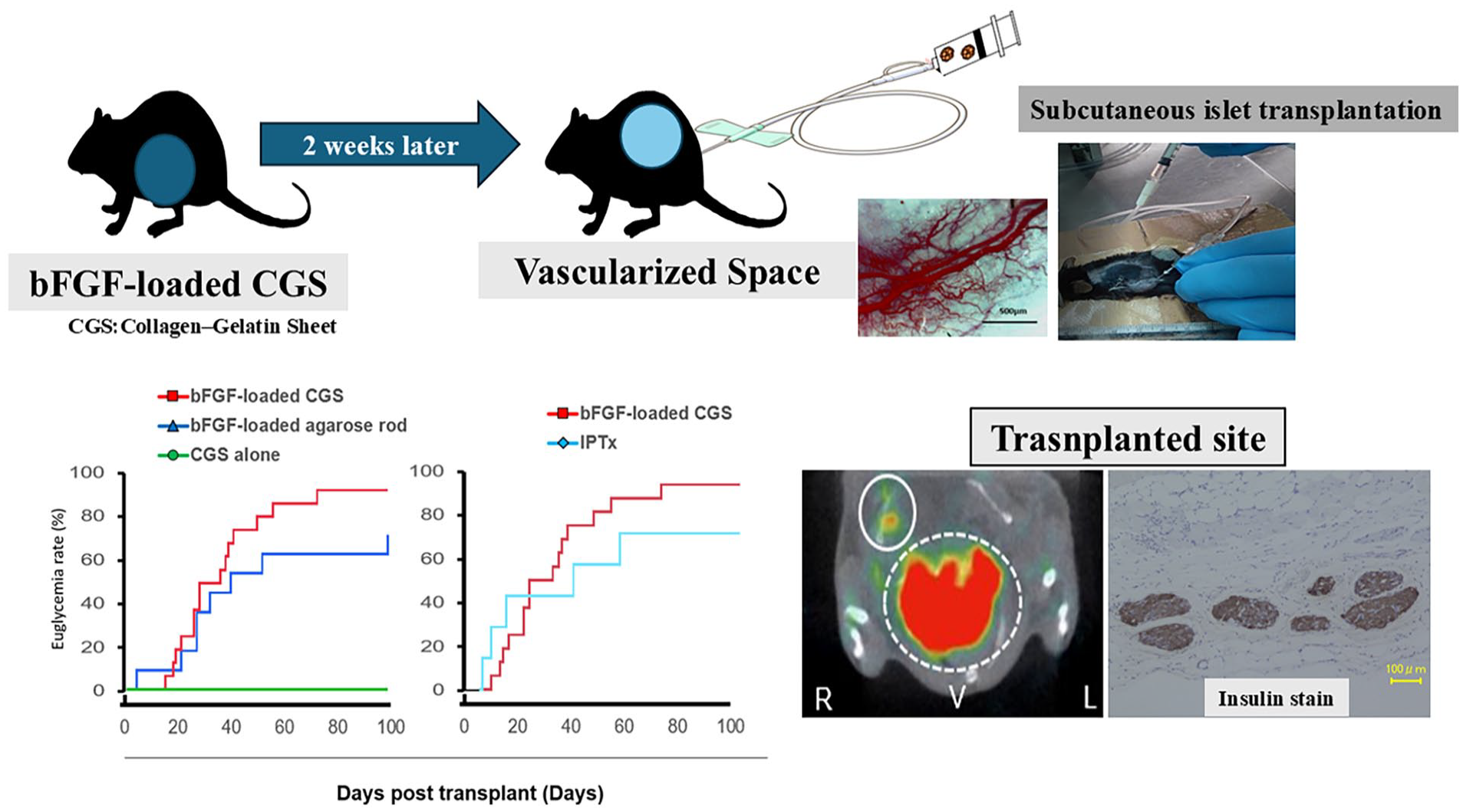
The recipient mice were divided into four experimental groups: (1) bFGF-loaded CGS, (2) bFGF-loaded agarose rod, (3) CGS alone, and (4) transplantation into the portal vein (IPTx). Table 1 summarizes the characteristics of the experimental groups. To induce neovascularization, either an agarose rod or CGS was surgically placed under the skin on the backs of recipient mice 2 weeks before islet transplantation 14 . In the bFGF-loaded agarose rod group, after neovascularization, the rod was extracted, and 200 pancreatic islets from C57BL/6 mice were transplanted into the newly vascularized dorsal pockets of mice STZ-induced diabetes. However, in the bFGF-loaded CGS group, the CGS remained in place, and 200 islets were directly transplanted into the slightly biodegraded, vascularized CGS in recipient mice with STZ-induced diabetes. Transplantation involved injection of 200 islets in a 150-µl volume into the slightly biodegraded CGS through the skin using a 27-gauge Surshield (Terumo, Inc., Tokyo, Japan). IPTx was performed as described previously17,20. Briefly, 200 islets were injected into the portal vein to engraft islets in the liver.
Experimental groups.
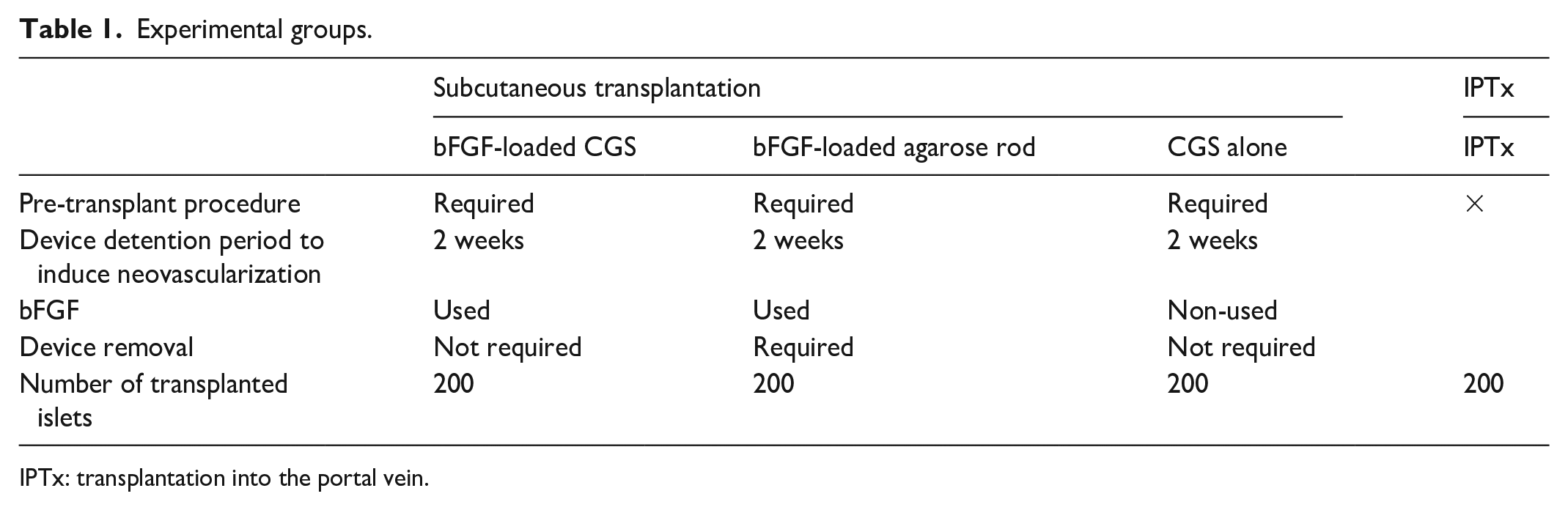
IPTx: transplantation into the portal vein.
After the transplantation procedure, random blood glucose measurements were performed three times a week. Normal blood glucose levels were considered to be achieved when glucose readings fell below 200 mg/dl for 2 days in a row.
Intraperitoneal glucose tolerance test
A period of 30 days after transplantation, transplant recipients underwent an intraperitoneal glucose tolerance test (IPGTT). Mice were fasted for 14 h and then administered a glucose solution (2 g glucose/kg body weight) via intraperitoneal injection. Subsequently, blood glucose levels were monitored at several time points (0, 15, 30, 60, and 120 min) after glucose administration.
Macroscopic vascular imaging
Pre-vascularization of the subcutaneous graft site was evaluated using video capillaroscopy (GOKO-Bscan-Z GOKO Imaging Devices Co, Japan) in conjunction with macroscopic observations.
Histological analysis and capillary area measurement
Before transplantation, subcutaneous tissues surrounding the implanted CGS, with or without bFGF, were extracted for pre-vascularization analysis. At 100 days post-transplantation, the transplanted sites were harvested for further examination. The tissue samples were preserved in 10% formalin neutral buffer solution, embedded into paraffin blocks, and stained using standard hematoxylin and eosin (H&E) or Masson’s trichrome protocols. Immunohistochemical staining was performed according to previously established methods 14 . The tissue sections underwent overnight incubation at 4°C with primary antibodies, including mouse anti-insulin (1:2000 dilution, 66198-1-Ig; Proteintech Japan, Tokyo, Japan) and anti-CD31 antibodies (1:100 dilution, clone: D8V9E; Cell Signaling Technology, Danvers, MA). To assess neovascularization, ImageJ software 21 was used to quantify the area fraction of capillaries formed at the CGS margins. The analysis focused on vascular-rich regions measuring 500 µm in width and height located at the CGS margins in each specimen.
Insulin content assay
A 1-cm square section of subcutaneous tissue was excised from the target area, and T-per (Thermo Fisher Scientific, Waltham, MA, USA) was used to extract insulin. Insulin content within the graft tissues was measured using an ELISA kit (298-89501 FUJIFILM Wako Pure Chemical Corporation, Osaka, Japan).
Non-invasive imaging of islet grafts
Islet grafts were imaged using 111Indium-labeled exendin-4 derivative 22 (111In-exendin-4) and single-photon emission computed tomography (SPECT/CT) to monitor engraftment and insulin secretion 14 . The 111In-exendin-4 was synthesized according to previously established protocols22,23. Each mouse received a tail vein injection of 111In-exendin-4 at a dose of 3.0 MBq. SPECT/CT imaging was conducted using the Triumph Lab PET12/SPECT4/CT system (TriFoil Imaging Inc., Chatsworth, CA, USA) as described in prior studies22,23. Quantitative analysis of the SPECT signals was performed using Amira software (version 5.6.0; FEI Visualization Sciences Group, Düsseldorf, Germany). To localize and assess the transplanted islet graft signals, SPECT signals were compared with the anatomical contours of the subcutaneous regions visualized on the CT images.
Statistical analysis
Data are expressed as the mean ± standard deviation (SD). Welch’s t-test was used to compare two groups. To compare the data among groups more than three, one-way analysis of variance (ANOVA) was used for analysis. When a significant overall difference was observed, post hoc pairwise comparisons were conducted using Dunnett’s test. Kaplan–Meier curves were compared using the log-rank statistical method. Statistical significance was set at a P-value < 0.05. All statistical calculations were conducted using the JMP Pro software (version 17.0; SAS Institute Inc., Cary, NC, USA).
Results
Macroscopic evaluation of vascularization
We initially assessed angiogenesis in the dorsal subcutaneous tissue of mice with STZ-induced diabetes of all groups. Fig. 1 illustrates the macroscopic and enlarged macroscopic images of mice 2 weeks after the implantation of bFGF-loaded CGS (Fig. 1a) and CGS alone (Fig. 1b). Untreated mice (Fig. 1c) and mice at 2 weeks after implantation of the bFGF-loaded agarose rod (Fig. 1d) are also shown. CGS was observed to be partially biodegraded and reduced in size but was not completely bioabsorbed. Neovascularization and accumulation of adipose components were observed in the bFGF-loaded CGS group but not in the CGS alone or in the untreated group (Fig. 1a–c). Both neovascularization and a pronounced inflammatory response were observed in the bFGF-loaded agarose rod group (Fig. 1d). The bFGF-loaded CGS group exhibited a greater extent of neovascular invasion into the CGS than the CGS alone group or the untreated group (Fig. 1e–g), confirming that bFGF plays a crucial role in enhancing angiogenesis at the transplant site.

Macroscopic images of subcutaneous skin in each group of mice. Macroscopic images of subcutaneous skin of mice in the bFGF-loaded CGS (a), CGS alone (b), untreated (c), and bFGF-loaded agarose rod (d) groups. Black arrows indicate CGS or agarose rod. Enlarged macroscopic images of the transplantation sites created in the bFGF-loaded CGS (e), CGS alone (f), untreated (g), and bFGF-loaded agarose rod (h) groups.
Evaluation of newly formed capillaries in the subcutaneous transplant site
H&E staining revealed that the tissues formed around the CGS were thicker in the bFGF-loaded CGS group than in the CGS alone group (Fig. 2a, b). Masson’s trichrome staining demonstrated that the tissues formed around the CGS were rich in collagen fibers (Fig. 2c, d). Histological analysis revealed CD31-positive blood vessels in both the bFGF-loaded CGS and the CGS alone groups (Fig. 2e, f). However, the bFGF-loaded CGS group had a significantly greater number of these blood vessels, indicating enhanced angiogenesis at the transplantation site. The area fraction of the capillaries at the margins of the CGS was significantly larger in the bFGF-loaded CGS group than in the CGS alone group (Fig. 2g). This observation further supports the hypothesis that CGS contributes to the sustained release of bFGF and that bFGF promotes vascularization of the subcutaneous graft site, thereby enhancing the probability of islet transplant success.

Microscopic images and capillary area fraction before transplantation. HE, Masson trichrome (MT), and CD31-stained microscopic images of transplantation sites made in the bFGF-loaded CGS (a, c, e) and CGS alone (b, d, f) groups. (g) Mean area fraction of capillaries immediately before transplantation in the bFGF-loaded CGS and CGS alone groups.
Results of islet engraftment after islet transplantation
We investigated blood glucose levels following subcutaneous transplantation of 200 islets, which approximates the quantity obtained from a single donor. Changes in blood glucose levels were compared among three groups: bFGF-loaded CGS (n = 16), bFGF-loaded agarose rod (n = 11), and CGS alone (n = 4) over a period of 100 days. In addition, changes in blood glucose levels were compared between the bFGF-loaded CGS (n = 16) and IPTx (n = 7) groups. Islets transplanted in the CGS alone group did not contribute to euglycemia; however, those transplanted into the subcutaneous space prepared by bFGF-loaded agarose rods ultimately resulted in decreased blood glucose levels and euglycemia. Similarly, those transplanted in the bFGF-loaded CGS group resulted in euglycemia (Fig. 3a). Blood glucose levels in the bFGF-loaded CGS group were also compared with those in the IPTx group (Fig. 3b).

Results of islet engraftment after islet transplantation. (a and b) Blood glucose levels after islet transplantation. (a) Change in blood glucose levels of the three groups: bFGF-loaded CGS (n = 16), bFGF-loaded agarose rod (n = 11), and CGS alone (n = 4) after subcutaneous islet transplantation. (b) Change in blood glucose levels in the bFGF-loaded CGS (n = 16) and IPTx (n = 11) groups after islet transplantation. (c, d) Euglycemia rate curves after islet transplantation. The engraftment rates at 100 days after subcutaneous islet transplantation were 93.8% for the bFGF-loaded CGS group, 72.7% for the bFGF-loaded agarose rod group, and 0% for the CGS alone group. The engraftment rate in the bFGF-loaded CGS group was higher than that in the bFGF-loaded agarose rod group (P = 0.169, log-rank test); however, the difference was not statistically significant (c). The engraftment rate in the bFGF-loaded CGS group (93.8%) exceeded that in the IPTx group (71.2%) (P = 0.328, log-rank test); however, the difference was not statistically significant (d).
The engraftment rate 100 days after subcutaneous islet transplantation was 93.8% (15 of 16 mice) in the bFGF-loaded CGS group, 72.7% (8 of 11 mice) in the bFGF-loaded agarose rod group, and 0% (0 of 4 mice) in the CGS alone group. The engraftment rate in the bFGF-loaded CGS group was higher than that in the bFGF-loaded agarose rod group, although this difference was not statistically significant (P = 0.169, Fig. 3c). The engraftment rate in the bFGF-loaded CGS group (93.8%) was higher than that in the IPTx group (71.2%, 5 of 7), although the difference was not statistically significant (P = 0.328), demonstrating a transplantation efficacy comparable to that of the standard transplantation site, the portal vein (Fig. 3d).
Graft function
Islet graft function in naïve and recipient mice that achieved euglycemia was assessed using the IPGTT 30 days after subcutaneous islet transplantation. The curves representing the blood glucose levels of the bFGF-loaded CGS, the bFGF-loaded agarose rod, and IPTx groups exhibited IPGTT profiles comparable to that of the naive group (Fig. 4a), and the areas under the curves demonstrated no statistically significant differences (Fig. 4b, four mice/group, Dunnett’s test).

Intraperitoneal glucose tolerance tests (IPGTT) to evaluate graft effect. IPGTT was performed in the bFGF-loaded CGS group, bFGF-loaded agarose rod group, IPTx group, and naïve mice group 30 days after transplantation. Blood glucose levels were measured and compared among the four groups (a). The area under the curve (AUC) was calculated (b).
Notably, an immediate increase in blood glucose levels was observed upon graft removal (Fig. 5a). Adequate quantities of insulin were extracted from the grafted sites of mice with normalized blood glucose levels after transplantation, whereas no insulin was detected in the grafted sites of mice that had exhibited graft failure or in the subcutaneous tissue of the non-transplanted mice (Fig. 5b). These results indicate that normalization of blood glucose levels was achieved owing to the grafts maintaining glucose responsiveness and insulin secretory capacity in the subcutaneous environment.

Graft removal to evaluate graft effect. (a) Graft removal was performed at 107, 110, and 110 days after transplantation. (b) Graft insulin content was measured and compared among the engraftment, graft-failed, and non-transplanted groups.
Non-invasive imaging of pancreatic β-cells transplanted into the subcutaneous space
Fig. 6 presents a representative SPECT/CT fusion image of mice administered 111In-exendin-4 to detect pancreatic β-cells. In mice exhibiting normalized blood glucose levels 100 days post-transplantation, the accumulation of radiopharmaceuticals was observed at the subcutaneous transplant site. This observation constitutes one of the findings indicative of the functionality and viability of the subcutaneous islet grafts.

Non-invasive imaging of islet grafts using 111In-exendin-4 and SPECT/CT. Image of pancreatic β-cells transplanted into the subcutaneous prepared-transplanted site. Coronal (a) and axial (b) views.
Macroscopic and microscopic findings of subcutaneous space 100 days after transplantation
Fig. 7 presents the macroscopic and microscopic images of the harvested engraftments obtained from mice of the bFGF-loaded CGS group at 100 days post-transplantation. The macroscopic images show that the CGS had completely absorbed and was no longer visible. In addition, a greater accumulation of adipose tissue was observed at the subcutaneous site 100 days after transplantation than pre-transplantation (Fig. 7a). Fig. 7b shows the transplanted sites stained with H&E. Fig. 7c depicts the immunohistological staining of the subcutaneous site 100 days post-transplantation. HE staining revealed the engraftment of islets at the subcutaneous transplant site. CD31 staining demonstrated abundant vasculature in and around the engrafted islets. Masson’s trichrome staining revealed collagen layers surrounding the engrafted islets, which functioned as scaffolds. Insulin staining confirmed the presence of insulin in the engrafted islets.

Macroscopic and microscopic images 100 days after transplantation. (a) Macroscopic image 100 days post-transplantation. The transplantation site is indicated by a red circle. (b) Transplantation sites were stained with hematoxylin and eosin (H&E). Islet grafts are indicated by a red dashed circle. (c) Histological images from microscopic analysis.
Discussion
This study demonstrated the feasibility of subcutaneous islet transplantation using bioabsorbable medical devices containing bFGF, employing only 200 islets (the quantity obtained from a single donor) to maintain normoglycemia in recipient mice with diabetes. The transplantation outcomes were comparable to those of portal vein transplantation, which is the standard transplantation site, and suggest that this method may be efficacious in the transplantation of islet cells by subcutaneous induction of blood vessel formation. In addition, we showed that the diabetic condition recurred rapidly following the removal of the subcutaneous transplant site, indicating that the maintenance of normal blood glucose levels was contingent upon the functioning of the graft. This subcutaneous transplantation approach presents a simpler and more efficient alternative to conventional methods that use nonabsorbable devices 13 . The significance of this study lies in elucidating the potential utility of this method for transplanting islet cells through the induction of blood vessels in subcutaneous tissue.
CGS, the material that yielded favorable transplantation results in this study, is a novel scaffold material containing 10 wt% of acid gelatin that can release positively charged growth factors, such as bFGF, for more than 10 days in vivo via ion complex formation between bFGF and gelatin 24 . Human bFGF, with an isoelectric point (IEP) of 9.6, forms ionic complexes with acid gelatin with an IEP of 5.0 24 , 25 . bFGF impregnated into CGS is released during biodegradation19,24. Pre-vascularization has been identified as a critical factor for successful subcutaneous islet transplantation, as demonstrated in several previous studies. Kuwabara and Iwata 26 utilized gelatin as a sustained-release agent for bFGF to create a bioabsorbable device and reported that the outcomes of subcutaneous islet transplantation in rats were comparable to those of conventional nonabsorbable devices (agarose), suggesting that this method is an effective approach for inducing pre-vascularization. Based on these findings, we verified the formation of a vascular network at the graft site in a mouse model using CGS. This material exhibits optimal properties for the sustained release of bFGF and possesses the advantage of biodegradability, thus eliminating the need for removal after blood vessel induction. Saito et al. also reported that pretreatment with bioabsorbable scaffolds combined with adipose tissue–derived stem cells promoted neovascularization of the subcutaneous space and improved the outcome of subcutaneous islet transplantation. However, there are hurdles to its clinical application 27 . In contrast, CGS has been successfully implemented in clinical practice, and its efficacy in facilitating skin ulcers 18 and skin reconstruction is well documented 28 . The demonstrated clinical efficacy and safety profile of this CGS present advantages for its potential application in subcutaneous islet transplantation.
Moreover, CGS also contains collagen, which provides an effective scaffold or extracellular matrix (ECM) for islet bioproduction, thereby enhancing the efficacy of subcutaneous transplantation. The ECM surrounding islets in the pancreas is lost during islet isolation 29 . Evidence suggests that islet function improves owing to ECM compensation30,31. ECM components, including collagen IV, laminin, and fibronectin, provide structural support and biochemical signals that facilitate cellular adhesion, survival, and insulin secretion. These interactions are predominantly mediated by integrins expressed on the surface of islet cells 32 . Sakata et al. investigated the use of white adipose tissue as a transplantation site. The ECM within this tissue, comprising collagen, laminin, and fibronectin, facilitates the adhesion and engraftment of transplanted islets through integrin interactions. This microenvironment not only promotes islet attachment but also enhances insulin secretion and β-cell viability 33 .
Subcutaneous islet engraftment is considered effective when a controlled foreign body reaction forms a pre-vascularized subcutaneous site and an abundant vascular collagen network that provides a scaffold for engraftment has developed7,14. Prolonged persistence of foreign body reactions may result in the formation of avascular fibrotic granular capsules. For instance, extended implantation of a permanent encapsulation device may lead to the formation of an avascular fibrotic granular capsule and a chronic inflammatory response34,35, potentially contributing to graft failure 7 . The bioabsorbable material used in this study provides only the necessary stimulation for vascular induction to the graft site and does not induce excessive and persistent foreign body reactions or chronic inflammation, thereby enabling the creation of an optimal graft site.
Non-invasive imaging using 111In-exendin-4-based SPECT/CT technology was employed to confirm islet graft engraftment. In contrast to previous studies that required graft removal for engraftment assessment 26 , this imaging technique facilitated the evaluation of islet viability without the need for invasive procedures 36 . Our previous study demonstrated the efficacy of this method in assessing β-cell content following portal vein islet transplantation in humans 37 . The extension of its application to subcutaneous transplantation in murine models demonstrates its potential for clinical implementation in humans.
Despite these promising findings, several limitations of this study should be considered. First, while mouse and human islets exhibit similar dimensions 38 , significant differences exist in the skin structure and vascular anatomy, potentially limiting the direct applicability of the methods described in this study to human subcutaneous islet transplantation. Nevertheless, the angiogenic and wound-healing properties of CGS and bFGF in human skin 18 suggest that this method holds promise for clinical translation. Second, the dorsal subcutaneous site was used for accessibility in this study; however, alternative sites, such as the inguinal region 39 , may also be viable. Further studies are required to determine the optimal transplantation sites for humans. Third, although no adverse events were observed in this study, larger animal models are essential to validate the safety and efficacy of this approach, particularly with respect to ensuring adequate oxygenation of the transplanted islets. Fourth, although the present study was conducted in syngeneic model, it is possible that there may be differences in immune response under subcutaneous conditions in an allogeneic model. Future studies using allogeneic models are needed to fully evaluate the immunological aspects of this approach.
In conclusion, this study demonstrated that the combination of a clinically available bioabsorbable medical device composed of collagen and gelatin with bFGF constitutes a promising platform for subcutaneous islet transplantation. With further investigation, this strategy may provide a minimally invasive and efficacious alternative to the existing methods and advance clinical islet transplantation.
Footnotes
Acknowledgements
The authors thank the Center for Anatomical, Pathological, and Forensic Medical Research, Kyoto University Graduate School of Medicine for preparing the slides for microscopic observation. The CGS (PELNAC Gplus®) was provided free of charge by Gunze Medical Limited. We would like to thank Editage (![]() ) for English language editing.
) for English language editing.
Research ethics and patient consent
All animal experiments were approved by Kyoto University’s Institutional Animal Care Committee (C23-79). All surgical operations were performed under anesthesia and efforts were made to minimize suffering. Mice were housed under standard conditions with ad libitum access to food and water, except during fasting. There are no human subjects in this article and informed consent is not applicable.
Author contributions
N.E. performed the experiments, analyzed the data, and drafted the manuscript. T.A. obtained the grant, designed the study protocol, participated in the research, analyzed the data, and wrote the draft. K.Y. designed the study protocol, participated in the research, analyzed the data, and drafted the manuscript. N.F., H.F., and T.M performed the experiments and analyzed the data. C.J, S.I., K.K., A.I., S.H, K. I., and S.T. performed the experiments. K.N. drafted the manuscript. E.H. supervised the study and revised the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Grants-in-Aid for Scientific Research B (T.A., 21H02986) from the Japan Society for the Promotion of Science (Tokyo, Japan) and grants from the Japan IDDM Network.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of human and animal rights
This article does not contain any studies with human or animal subjects.
Statement of informed consent
There are no human subjects in this article and informed consent is not applicable.
Data availability statement
All data generated or analyzed in this study are included in this published manuscript.
