Abstract
Basic fibroblast growth factor (bFGF) is a potent neurotrophic agent that promotes neuronal survival and outgrowth. Previous studies have shown that bFGF, administered intraventricularly or intravenously before or within hours after ischemia, reduces infarct size and neurological deficits in models of focal cerebral ischemia in rats. In the current study, we tested the hypothesis that bFGF, administered at later time points after ischemia, might improve behavioral recovery without affecting infarct size. Mature Sprague–Dawley rats received bFGF (1 μg/injection) or vehicle by biweekly intracisternal injection for 4 weeks, starting at 1 day following permanent proximal middle cerebral artery (MCA) occlusion. Animals were examined every other day using four different behavioral tests to assess sensorimotor and reflex function. At 4 weeks after ischemia, there was no difference in infarct volume between bFGF- and vehicle-treated animals. There was, however, an enhancement in the rate and degree of behavioral recovery among bFGF-treated animals, as measured by all four tests. There were no apparent side effects of bFGF treatment, except that bFGF-treated animals tended to recover body weight more slowly than did vehicle-treated animals following stroke. The mechanisms of enhancement of behavioral recovery by bFGF require further study, but may include protection against retrograde neuronal death and/or stimulation of neuronal sprouting.
Although recovery from sensorimotor, cognitive, and other disabilities after stroke is often incomplete, partial recovery often occurs. Mechanisms of recovery may include reversal of metabolic suppression in distant brain regions (“diaschisis”), unmasking of normally inhibited alternative brain pathways, and neuronal sprouting and synapse formation (Goldstein and Davis, 1990). Recent functional imaging studies in humans show new activation of brain regions both ipsilateral and contralateral to focal infarcts as patients recover motor function after stroke (Weiller et al., 1992). In animals, new neuronal sprouting and synaptic reorganization occur in both ipsilateral and contralateral cerebral cortex during motor recovery after unilateral cortical lesions or infarcts (Jones and Schauert, 1994; Stroemer et al., 1995).
Neurotrophic growth factors are likely to play important roles in the regulation of molecular and cellular processes underlying recovery after stroke. These endogenous polypeptides, acting through high affinity receptors, initiate and sustain cellular processes supporting cell survival and outgrowth. In particular, basic fibroblast growth factor (bFGF), an 18 kDa, 154 amino acid polypeptide, that is found in brain, supports the survival and outgrowth of a wide variety of brain neurons, as well as glial and endothelial cells (Baird, 1994). Endogenous bFGF expression is upregulated in tissue surrounding focal brain wounds or infarcts, and exogenous application of bFGF reduces the degree of cell death and stimulates neuronal sprouting in several models of brain injury or ischemia (Berlove and Finklestein, 1993; Finklestein, 1995). In particular, in previous studies, we showed that bFGF, administered intraventricularly or intravenously before or just after ischemia, reduced infarct volume and neurological deficits in models of focal cerebral ischemia in mature rats (Koketsu et al., 1994; Fisher et al., 1995). The mechanisms of infarct reduction may include direct protective effects on cells at the borders (“penumbra”) of focal infarcts and/or effects on cerebrovascular tone and cerebral blood flow. The infarct reducing effects of bFGF, however, are likely to occur within a relatively narrow “therapeutic window” of time after ischemia. Indeed, other investigators have shown that intracisternal administration of bFGF starting at 1 day after focal ischemia does not reduce infarct volume, but does protect against the “secondary” death of distant neurons projecting into infarcted regions (Yamada et al., 1991). Because bFGF may regulate several cellular processes contributing to functional recovery after stroke, we tested, in the current study, the hypothesis that late administration of bFGF might enhance neurological recovery without affecting infarct size after ischemia. In this study, bFGF was delivered intracisternally to gain access to intact as well as ischemic cerebral hemispheres.
METHODS
Male Sprague–Dawley rats (250–300 g) (Charles River, Wilmington, MA, U.S.A.) were maintained on a 12-h light/dark cycle and allowed free access to food and water. Rats were anesthetized with 2% halothane in 70% NO2/30% O2 (vol/vol). The tail artery was cannulated for monitoring blood gases and blood glucose. Body temperature was maintained at 37 ± 0.5°C using a rectal probe and heating pad. The proximal right middle cerebral artery (MCA) was occluded permanently using a modification of the method of Tamura et al. (1981). Briefly, the proximal MCA was exposed transcranially without removing the zygomatic arch or transecting the facial nerve. The artery was then electrocoagulated using a bipolar microcoagulator from just proximal to the olfactory tract to the inferior cerebral vein, and was then transected (Bederson et al., 1986). Rats were observed until they regained consciousness and were returned to their home cages. Cefazolin sodium (40 mg/kg, body wt i.p.) was administered on the day before and just after stroke surgery.
Recombinant human bFGF was obtained as concentrated stock (2 mg/ml) as a generous gift from Scios Inc. (Mountain View, CA, U.S.A.), and stored at −80%C before use. Stock solution was then diluted into a solution containing 0.9% saline with 100 μg/ml bovine serum albumin (Boehringer-Mannheim, Indianapolis, IN, U.S.A.), pH 7.4, to give a final bFGF concentration of 20 μg/ml. Control animals received vehicle solutions with no bFGF, but containing all other components at the same final concentrations.
For intracisternal injections, animals were reanesthetized with halothane in 70% NO2/30% O2 (vol/vol) and placed in a stereotaxic frame. bFGF (1 μg/injection, n = 14 animals) or vehicle (n = 8 animals) was introduced by percutaneous injection (50 μl/injection) into the cisterna magna using a Hamilton syringe and 26-gauge needle under aseptic technique (Yamada et al., 1991). Before each injection, 1–2 μl of cerebrospinal fluid (CSF) was drawn back through the Hamilton syringe to verify needle placement in the subarachnoid space. We found in preliminary studies that dye (1% Evans blue) delivered in this fashion diffused freely through the basal cisternae and over the cerebral cortex within 1 h of injection. Intracisternal injections were made biweekly for 4 weeks, starting 24 h after stroke (i.e., on poststroke days 1, 4, 8, 11, 15, 18, 22, and 25). The total bFGF dose delivered was 8 μg/animal. Animals were assigned to bFGF or vehicle treatment in a random fashion.
Rats were handled for 10 min each day for 3 days before stroke surgery. Following surgery, they were housed in individual cages. Four behavioral tests were used to assess sensorimotor and reflex function after infarction. Full details of these tests have been described elsewhere (Bederson et al., 1986; DeRyck et al., 1992; Markgraf et al., 1992; Alexis et al., 1995). Briefly, the forelimb placing test is comprised of three subtests. Separate scores are obtained for each forelimb. For the visual placing subtest, the animal is held upright by the examiner and brought close to a table top. Normal placing of the limb on the table is scored as 0, delayed placing (<2 s) is scored as 1, and no or very delayed placing (>2 s) is scored as 2. Separate scores are obtained first as the animal is brought forward and then again as the animal is brought sideways to the table (maximum score per limb, 4; in each case, higher numbers denote greater deficits). For the tactile placing subtest, the animal is held so that it cannot see or touch the table top with its whiskers. The dorsal forepaw is touched lightly to the table top as the animal is first brought forward and then brought sideways to the table. Placing each time is scored as above (maximum score per limb, 4). For the proprioceptive placing subtest, the animal is brought forward only, and greater pressure is applied to the dorsal forepaw; placing is scored as above (maximum score per limb, 2). These subscores are added to give the total forelimb placing score per limb (range, 0–10). The hindlimb placing test is conducted in the same manner as above for the hindlimbs, but involves only tactile and proprioceptive subtests (maximal scores 4 and 2, respectively; total score range, 0–6). The modified beam balance test examines vestibulomotor reflex activity as the animal balances on a narrow beam (30 × 1.3 cm) for 60 s. Ability to balance on the beam is scored as follows: animal balances with all four paws on the top of beam, 1; animal puts paws on side of beam or wavers on beam, 2; one or two limbs slip off beam, 3; three limbs slip off beam, 4; animal attempts to balance with paws on beam but falls off, 5; animal drapes over beam, then falls off, 6; animal falls off beam without an attempt to balance, 7. Animals received three training trials before surgery; the score of the last of these was taken as the baseline score. The postural reflex test measures both reflex and sensorimotor function. Animals are first held by the tail suspended above the floor. Animals that reach symmetrically toward the floor with both forelimbs are scored as 0. Animals showing abnormal postures (flexing of a limb, rotation of the body) are then placed on a plastic-backed sheet of paper. Those animals able to resist side-to-side movement with gentle lateral pressure are scored as 1, while those unable to resist such movement are scored as 2. All behavioral tests were administered just before stroke surgery and then every other day from poststroke day 1 to day 31. At each session, animals were allowed to adapt to the testing room for 30 min before testing began.
On day 31 after MCA occlusion, animals were anesthetized deeply with pentobarbital and perfused transcardially with heparinized saline followed by 10% buffered formalin. Brains were removed, cut into three pieces, and stored in 10% buffered formalin before dehydration and embedding in paraffin. Coronal sections (5 μm) were cut on a sliding microtome, mounted into glass slides, and stained with hematoxylin and eosin. The area of cerebral infarcts on each of seven slices (+ 4.7, +2.7, +0.7, −1.3, −3.3, −5.3, and −7.3 mm compared to bregma) was determined using a computer-interfaced imaging system (Bioquant, R&M Biometrix, Inc., Nashville, TN, U.S.A.). Total infarct area per slice was determined by the “indirect method” as area of the intact contralateral hemisphere — area of the intact ipsilateral hemisphere, to correct for brain shrinkage during processing (Swanson et al., 1990). Infarct volume was then expressed as a percentage of the intact contralateral hemispheric volume. The volumes of infarction in cortex and striatum were also determined separately using these methods.
The experimenter performing intracisternal injections, behavioral testing, and histological analysis was blinded to the treatments assigned until all data had been collected. Data were expressed as means ± SD and were analyzed by repeated measures analysis of variance (ANOVA) followed by appropriate unpaired two-tailed t-tests, with the Bonferroni correction for multiple comparisons.
RESULTS
During stroke surgery, there were no differences in blood gases or glucose among animals that were to receive bFGF or vehicle treatment. Seventeen of 22 animals prepared initially survived for 1 month after infarction, including nine of 14 bFGF-treated and all of eight vehicle-treated animals. Surviving animals were killed at day 31; these animals had large infarcts in the right lateral cerebral cortex and underlying striatum in the territory of the MCA, as has been described previously using these methods (Tamura et al., 1981; Bederson et al., 1986; Markgraf et al., 1992) (see Fig. 1). Brain regions severely damaged by infarcts included parietal cortex, areas 1 and 2; and granular insular cortex. Regions partially damaged by infarcts included frontal cortex, areas 1,2, and 3; agranular insular cortex; temporal cortex, areas 1 and 3; lateral occipital cortex, area 2; cortical forelimb area; and caudoputamen (Paxinos and Watson, 1986). The cortical hindlimb area was generally spared by infarcts. There was no difference in total infarct volume between bFGF- (n = 9) and vehicle-treated (n = 8) animals (31.1 ± 5.9 versus 30.0 ± 5.3% of intact contralateral hemispheric volume, respectively, t = 0.4, p, not significant). Moreover, there was no difference in cortical or striatal infarct volume among bFGF versus vehicle-treated animals when these volumes were calculated separately. Inspection of hematoxylin/eosin stained sections showed no evidence of abnormal cell proliferation in the brains of bFGF-treated animals.
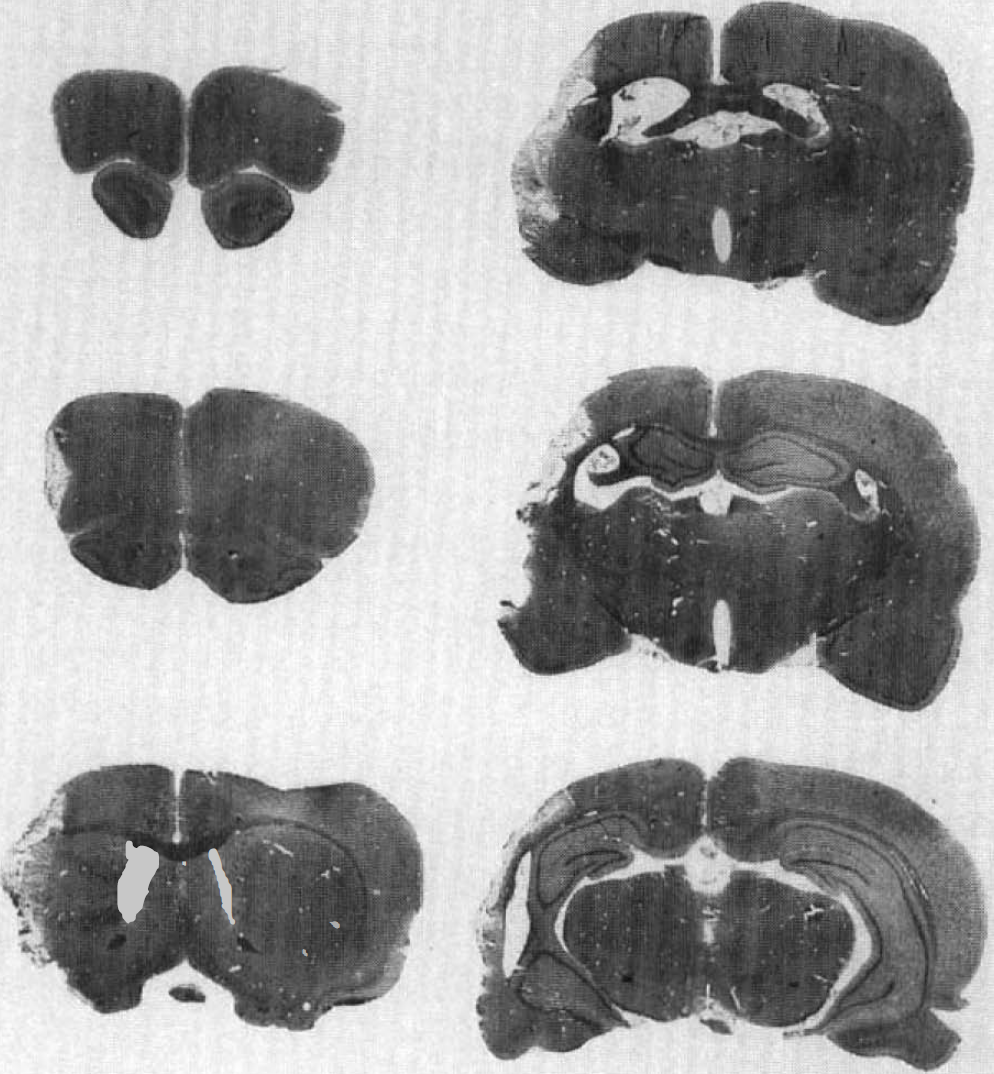
Stained brain sections showing a representative cerebral infarct following proximal MCA occlusion. Coronal sections are +4.7, +2.7, +0.7, −1.3, −3.3, and −5.3 mm compared to bregma (top left to bottom right, respectively). Hematoxylin and eosin.
Following infarction, animals showed severe disturbances of sensorimotor and reflex function on all four behavioral tests, as reported previously using these methods (Bederson et al., 1986; Markgraf et al., 1992). For the limb placing tests, deficits were confined to the contralateral (left) limbs. As described previously, animals showed partial recovery on all four behavioral tests during the first month after stroke (Bederson et al., 1986; Mark-graft et al., 1992) (see Figs. 2 and 3). Moreover, bFGF-treated animals recovered more rapidly and to a greater degree than did vehicle-treated rats. Improved recovery of surviving bFGF- versus vehicle-treated animals was most pronounced for forelimb and hindlimb placing tasks, and less pronounced, although still significant, for beam balance and postural reflex tests (Figs 2 and 3). Enhanced recovery was seen on all subtests of the limb placing tests (visual, tactile, and proprioceptive) following bFGF treatment (data not shown).
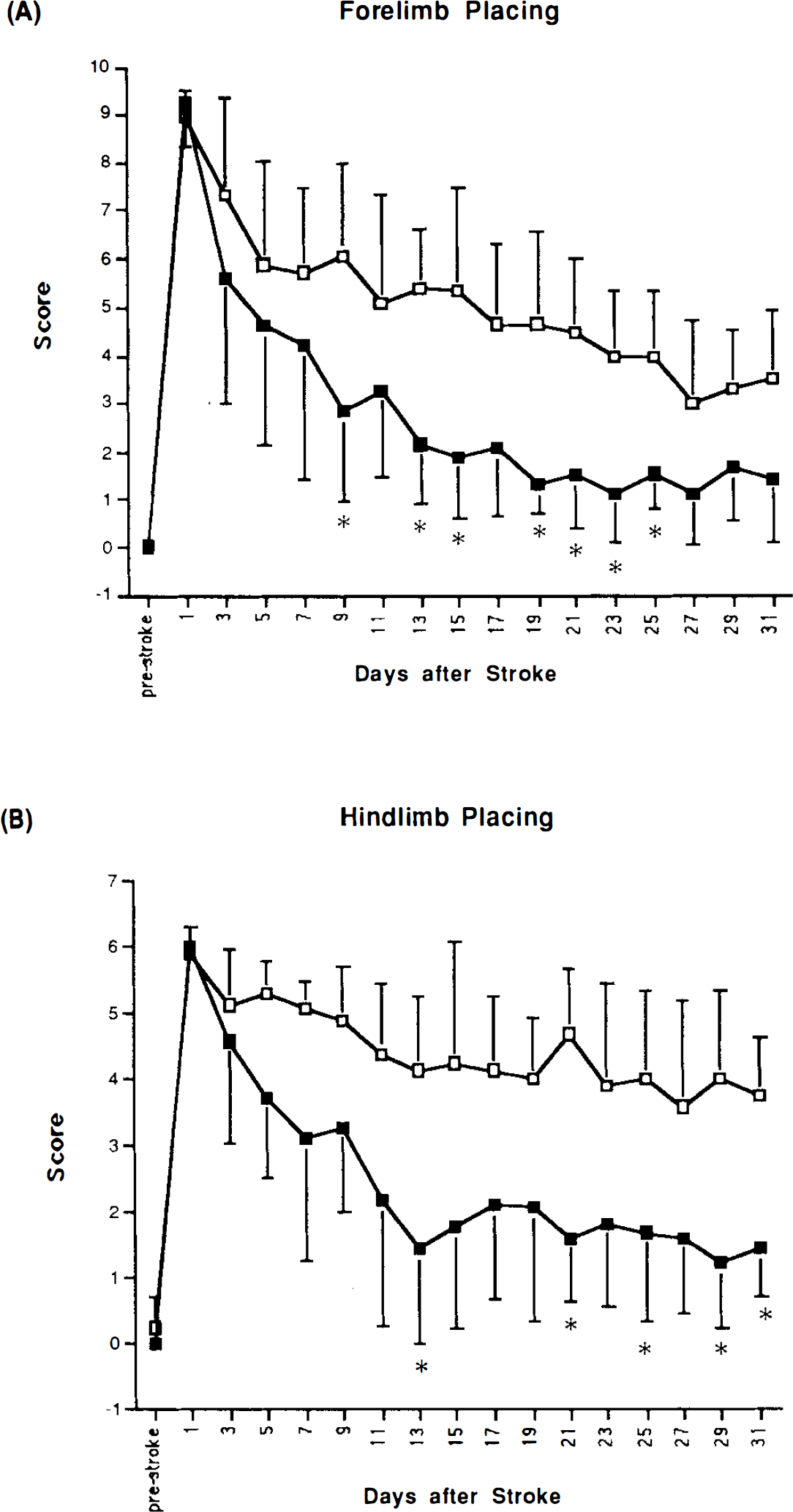
Forelimb placing
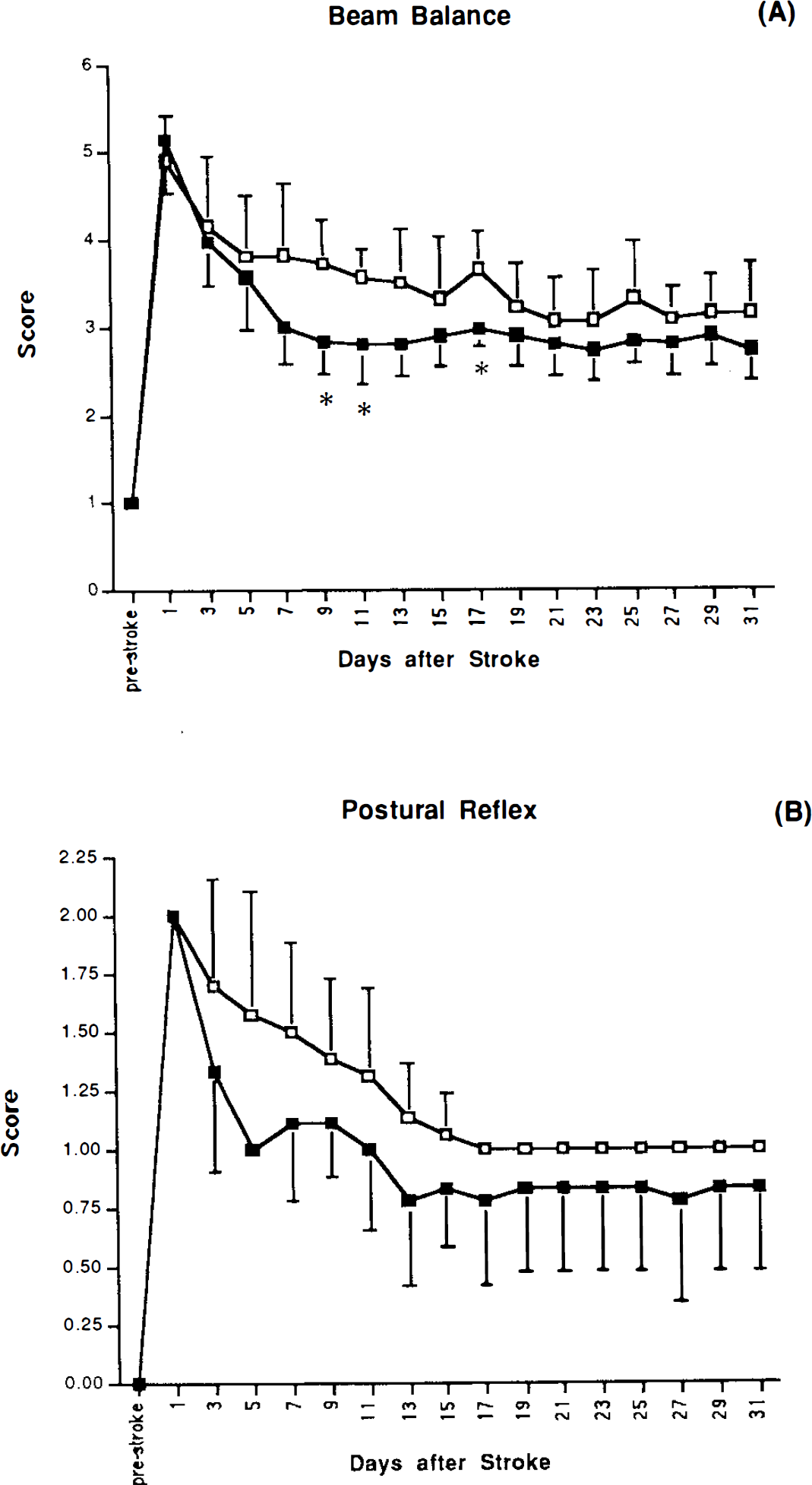
Beam balance
Five of 14 bFGF-treated animals died from severe progressive weight loss during the first month after stroke. Behavioral recovery of bFGF-treated animals that died was similar to that of surviving bFGF-treated animals until the time of death at 7–23 days after stroke. The mean weight of bFGF-treated animals that died was 165 ± 11 g on the day of death. Among surviving animals, there was a smaller degree of initial weight loss followed by a gradual recovery of body weight after stroke (Fig. 4). Surviving bFGF-treated animals tended to recover body weight more slowly than did vehicle-treated rats (Fig. 4).
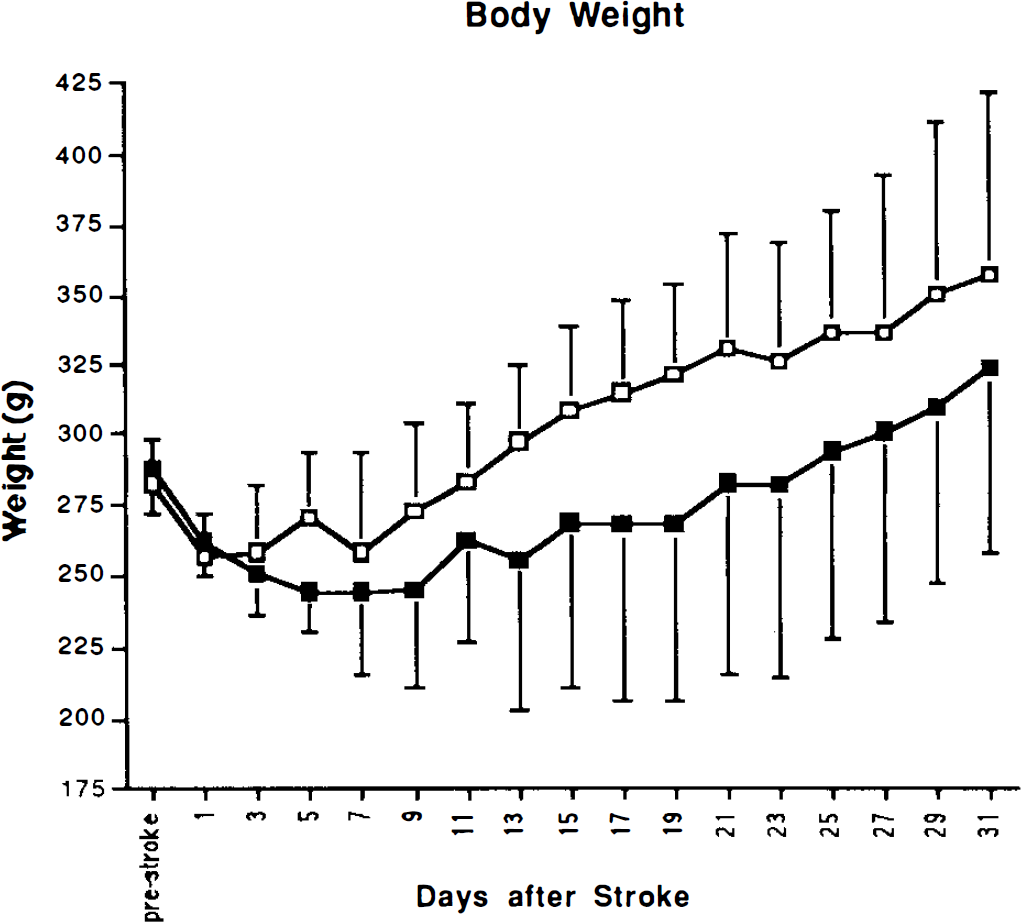
Body weight in bFGF-treated (n = 9) (solid squares) versus vehicle-treated (n = 8) (open squares) animals. Data are means ± SD. ANOVA: treatment, F(1) = 2.8, p not significant; time, F(16) = 19.4, p = 0.0001.
DISCUSSION
Repeated intracisternal administration of bFGF, starting at 1 day after ischemia, enhanced behavioral recovery in a rat model of focal cerebral infarction. Improved behavioral recovery was seen without a change in infarct volume in bFGF- compared to vehicle-treated animals. These data are consistent with those of Yamada et al. (1991), who, using a similar model of focal infarction in rats, also found that repeated intracisternal administration of bFGF (one injection/week × 4 weeks, 1 μg/injection), beginning at 1 day after ischemia, had no effect on infarct size. In previous studies (Koketsu et al., 1994; Fisher et al., 1995), we showed that intraventricular administration of bFGF starting before focal ischemia or intravenous administration of bFGF starting within the first few hours after ischemia did, indeed, reduce infarct size. Under these latter circumstances, intravenously-administered bFGF crossed the damaged blood–brain barrier and entered the ischemic, but not the nonischemic, hemisphere (Fisher et al., 1995). In the current study, bFGF was given starting at 1 day after ischemia, beyond the apparent “therapeutic window” of time during which bFGF can reduce infarct size. Moreover, in the current study, bFGF was given intracisternally in an attempt to access the nonischemic as well as the ischemic hemispheres, since cortex in the intact hemisphere appears to play an important role in recovery from brain injury or stroke (Jones and Schallert, 1994; Stroemer et al., 1995). To our knowledge, the current findings represent the first demonstration that an exogenously administered neurotrophic growth factor can enhance behavioral recovery without a reduction in infarct size in an animal model of stroke. Similar results have been reported recently for other classes of substances, including neurohormones and neurotransmitter agonists (Yamamoto et al., 1991; Yamaguchi et al., 1995).
Enhancement of recovery by bFGF was most pronounced on tests of sensorimotor function of the affected limbs and less pronounced on tests of reflex and postural function. Our infarcts did not completely damage forelimb and hindlimb cortical areas, findings compatible with recovery on limb placing tests following focal infarction in the MCA territory (DeRyck et al., 1992; Markgraf et al., 1992). Treatment with bFGF enhanced both the rate and degree of behavioral recovery during the first month after infarction. Further work will be required to determine whether this improvement persists for >1 month. Possible mechanisms by which bFGF enhances recovery include protection against retrograde cell death and/or acceleration of new neuronal sprouting and synapse formation. Yamada et al. (1991) found that repeated intracisternal injection of bFGF protected against retrograde death of distant thalamic neurons projecting into the infarcted cortex. This retrograde neuronal death occurs days to weeks after ischemia. It is possible that distant neurons in the thalamus and elsewhere, spared by bFGF treatment, might establish new functional connections, thereby enhancing recovery. Other investigators have reported new axonal and dendritic sprouting and synapse formation in cerebral cortex, both ipsilateral and contralateral to unilateral cortical lesions or infarcts in rats (Jones and Schauert, 1994; Stroemer et al., 1995). The time course of sprouting in the intact hemisphere parallels that of sensorimotor recovery of the affected limb, and blocking of sprouting (by splinting of the intact limb) inhibits recovery (Jones and Schauert, 1994). As noted above, bFGF promotes neuronal outgrowth in vitro and stimulates axonal sprouting in models of neurotoxic brain injury in vivo (Berlove and Finklestein, 1993; Finklestein, 1995). It is possible that augmentation of dendritic and/or axonal sprouting by bFGF in the poststroke brain contributes to enhancement of functional recovery.
Treatment with intracisternal bFGF was not without side effects. Indeed, five of 14 animals receiving bFGF died due to progressive weight loss, and there was a trend toward lower body weight in surviving bFGF-treated animals. These data are consistent with those previous reports showing that intracerebrally-administered bFGF lowers body weight in rats (Koketsu et al., 1994). It is possible that a reduced dose or frequency of bFGF administration might reduce effects on body weight without changing effects on behavioral recovery. It is unlikely, however, that the improved behavioral scores we observed were simply artifacts of lower body weight since all of the behavioral tests used (except the beam balance test) were done with the examiner supporting the animal. Of additional note is that in spite of known mitogenic effects of bFGF on glial and endothelial cells, we saw no gross evidence of abnormal cell proliferation in brains of bFGF-treated animals, although further studies using specific cell staining methods will be required to address this point. Indeed, further considerable study of the dose and time windows of efficacy, side effects, and molecular and cellular mechanisms of enhancement of behavioral recovery by intracisternal bFGF are required.
Footnotes
Acknowledgment:
Supported by NIH/NINDS grant P01 NS10828 and a grant from Scios-Nova Corporation, Mountain View, CA, U.S.A.
