Abstract
Patients receiving allogeneic hematopoietic stem cell transplantation (allo-HSCT) are typically placed in a laminar air flow room until hematopoietic reconstitution occurs. In this study, we compared the differences in clinical outcomes between patients receiving allo-HSCT in a conventional laminar flow room (n = 200) and those receiving allo-HSCT in a plasma sterilizer environment (n = 201). The overall infection rates (20.4% vs 25.5%, P = 0.224) and the sites of infection (sepsis, perianal infection, and catheter-related infection) were comparable between the two groups. Additionally, the engraftment times were comparable between the two groups in terms of time to allo-HSCT, leukocyte engraftment time, and platelet engraftment time. The 100-day posttransplantation clinical outcomes were also comparable between the two groups in terms of the probability of overall survival (98.5% vs 99.5%, P = 0.316), leukemia-free survival (96.5% vs 96.5%, P = 0.991), the cumulative incidence of relapse (2.0% vs 3.0%, P = 0.523), non-relapse mortality (1.5% vs 0.5%, P = 0.316) and acute graft-versus-host disease (23.4% vs 22.0%, P = 0.723). Thus, our results demonstrated that receiving allo-HSCT via a plasma sterilizer did not increase the risk of pre-engraftment infection, and the clinical outcomes of these patients were comparable to those of patients in a conventional laminar flow room.

Keywords
Introduction
Allogeneic hematopoietic stem cell transplantation (allo-HSCT) is effective and is often the only curative treatment for many hematologic malignancies1–5. The conditioning regimen involves high-dose chemotherapy and/or radiotherapy and is performed to maximize the elimination of tumor cells and create an optimal environment for the engraftment of donor hematopoietic stem cells. During the time period before granulocyte engraftment (which occurs for 2–3 weeks), patients experience severe granulocytopenia and are highly immunocompromised, thus rendering them vulnerable to various infections. A series of studies have reported a close relationship between infections and engraftment outcomes6–10. Specifically, preengraftment intestinal infections have been observed to significantly increase the risk of 100-day acute graft-versus-host disease (GVHD)11–14, and the overall mortality rate for patients with preengraftment bloodstream infections was observed to be as high as 21% within 120 days posttransplantation. These findings underscore the critical impact of preengraftment infections on patient prognosis. Therefore, the maintenance of a highly clean and sterile environment during the granulocyte-deficient preengraftment period is essential to prevent infection and improve outcomes.
A conventional laminar air flow room (LAFR) can achieve 100-level air cleanliness by using primary, medium- and high-efficiency filters to remove pathogenic microorganisms and airborne particles. Clinical practices have demonstrated that this clean technology is an effective measure for preventing various infections and for specifically significantly reducing nosocomial infections15,16. Recent advancements in science and technology have led to the development of plasma sterilizers (also known as high-efficiency disinfectors), which ionize the air via corona discharge, thus generating plasma that inactivates and filters out pathogenic microorganisms. Compared with traditional laminar flow rooms, plasma sterilizers offer several advantages, including enhanced patient comfort, energy efficiency, ease of maintenance, high stability, quantifiable sterilization efficacy, and the ability for the adjustment or relocation of the sterilizer based on clinical needs. These benefits not only improve the conditions of patients but also reduce both patient health care costs and hospital maintenance expenses (Supplementary Table 1). Several studies in China have demonstrated that, in immunocompromised populations (such as adult and pediatric hematology patients), the use of the abovementioned plasma sterilizers can effectively prevent infections during the granulocyte deficiency stage17,18. However, it remains unclear as to whether allo-HSCT performed in a plasma sterilizer environment can achieve clinical outcomes comparable to those in conventional laminar airflow rooms.
Thus, this retrospective study was conducted to compare the incidence of infection, engraftment, and prognosis between patients who received allo-HSCT in plasma sterilizer environments and those who received allo-HSCT in conventional laminar flow rooms.
Methods
Patients
Patients who received allo-HSCT in the conventional laminar flow room from January to December 2022 (200 patients) and those who received allo-HSCT in a plasma sterilizer environment from May to December 2023 (201 patients) were consecutively enrolled in this study. The last follow-up was December 2024.
Data collection
The investigators utilized the institutional electronic medical records of clinical databases to obtain the required information. The collected data included information on patient demographics, diagnosis, disease status before allo-HSCT, transplant regimen, donor type, occurrence of infections (including pathogens, sites of infection, and time of occurrence, among other data), engraftment, GVHD status and clinical outcomes (including relapse, mortality, and survival).
Environmental monitoring methods
Sampling timing of new wards
Prior to testing, all of the rooms were thoroughly cleaned and disinfected, after which they were sealed off to prevent personnel entry. The laminar airflow clean room purification system was checked to ensure normal operation for 24 hours before the procedure was conducted. During the first three months after the operation, cleanliness and dynamic bacterial concentration tests were performed on a monthly basis in room 4. Static air quality was tested using the sedimentation method in rooms 2 and 3, as well as on ultraclean tables and in biosafety cabinets. Microbial sampling and culturing were conducted across all types of environments. Once the ward’s operation was stabilized, testing was conducted on a quarterly basis.
Sampling method
The environmental monitoring points were established according to the ISO 9000 “Quality Management and Quality Assurance” standards and were based on the guidelines provided by the Infection Control Department of our institution. To monitor the cleanliness of room 4, three sampling points were established and positioned at diagonal corners of the room. Air quality was assessed using the sedimentation method. In room 4, one Petri dish was placed at the head of each bed, whereas in room 3, at least three Petri dishes were placed. In room 2, at least 18 Petri dishes were placed along the corridors. For ultraclean tables and biosafety cabinets, a minimum of one Petri dish was placed at each unit. Surface monitoring involved sampling tests at various surfaces, with at least three surfaces sampled in room 4, three surfaces sampled in room 3 (with a focus on high-frequency contact areas such as ultraclean tables, monitors, infusion pumps, infusion racks, and infusion carts), and at least 10 surfaces sampled in room 2 (such as nurse station countertops, computer mice, keyboards, telephones, and treatment room refrigerators).
Statistical analysis
Frequencies and percentages were used to describe the characteristics of the patients. The Mann-Whitney U test was used to compare continuous variables, and the chi-square test and Fisher’s exact test were used to compare categorical variables. The Kaplan–Meier estimator was used to calculate the probabilities of overall survival (OS) and leukemia-free survival (LFS). The cumulative incidence function was used to calculate the incidence of relapse and non-relapse mortality (NRM) via competing risk analysis. Univariable and multivariable Cox regression analyses were performed to determine the impacts of potential prognostic factors on infection status. A two-sided statistical test was utilized, and P < 0.05 was considered to be statistically significant. Tests were performed using SPSS (2023) and R software.
Results
General information
A total of 401 patients were enrolled in this study; moreover, 201 and 200 patients were categorized into the plasma sterilizer and conventional laminar flow room groups, respectively (Table 1). A total of 31 plasma sterilizers in this ward were monitored in terms of particulate matter, microorganisms and object surfaces. A Class-100-standard laminar standard flow cleanroom met the Class 100 cleanliness standard, which stipulates that the maximum particle concentration of airborne particles ≥ 0.5 μm in diameter should not exceed 100 particles per cubic foot (approximately 3,520 particles per cubic meter). Thus, the number of particles ≥ 0.5 μm in diameter per cubic foot of air should be limited to 100 particles. The monitoring results of both laminar flow chambers met the Class 100 standard (Table 2).
Patient characteristics.

HCT-CI: hematopoietic cell transplantation-specific comorbidity index.
Pathogen microorganisms and particulate matter monitoring results.

Overall infections
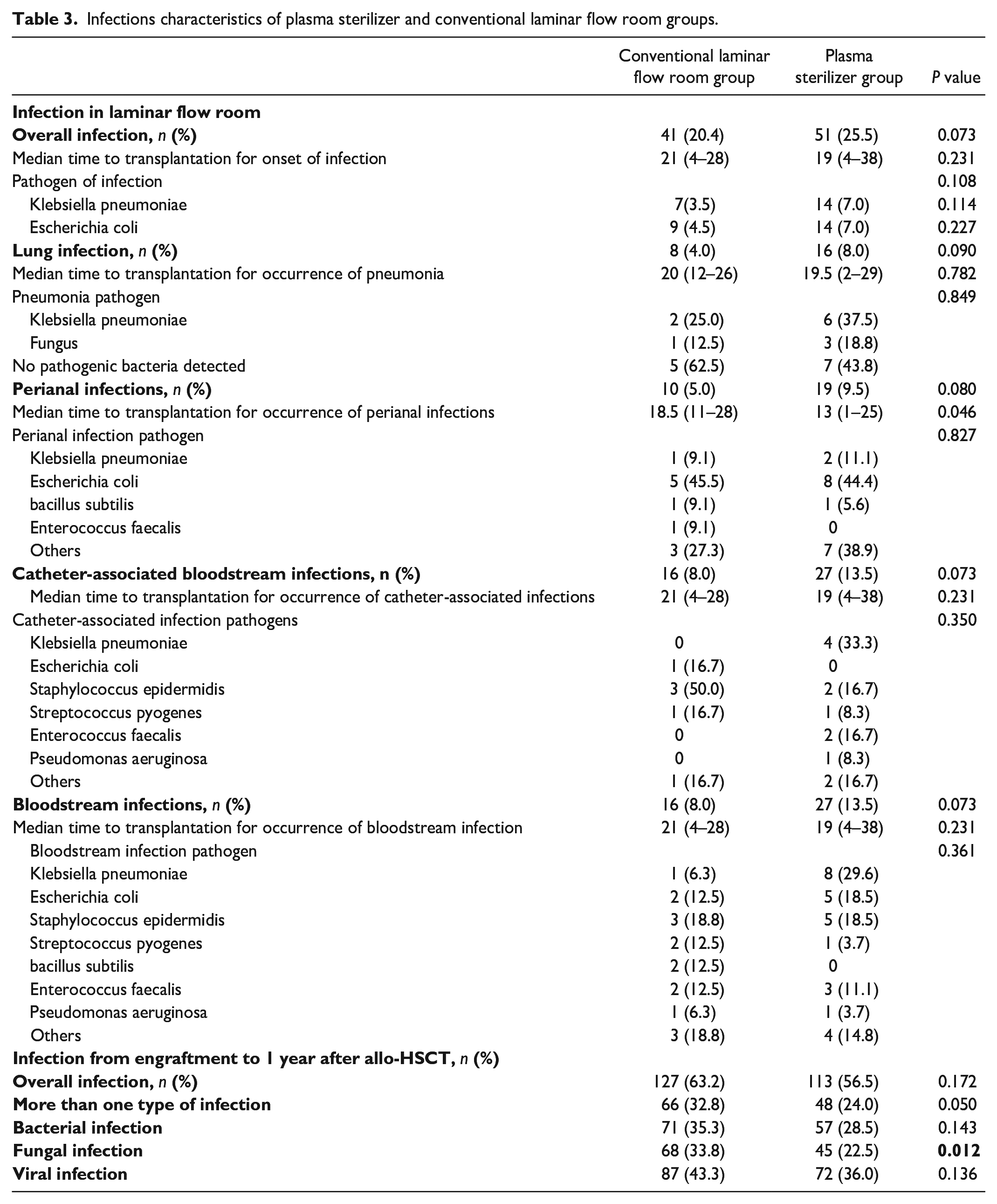
During the period when the patient was residing in the laminar flow room, there was no significant difference observed between the two groups of patients in terms of the incidence of infection, the time of occurrence of posttransplantation infections, or the types of detected pathogens (Fig. 1). Moreover, when the incidence of each major infection site was analyzed (such as the incidences of lung infection, perianal infection, catheter-related bloodstream infection and sepsis), the time of occurrence was not significantly different, except for the time of occurrence of perianal infection to the time of transplantation (P = 0.046); additionally, the other sites were not significantly different (Table 3). According to the multivariate analysis, ABO-mismatched grafts increased the risk of infection after allo-HSCT in the plasma sterilizer group, and MNC counts ≤ the median increased the risk of infection after allo-HSCT in both the plasma sterilizer group and the total cohort. Female donors to male recipients demonstrated increased risks of more than one type of infection in the conventional LAFR group. MNC counts ≤ the median also increased the risks of bacterial infection in the plasma sterilizer group and total cohort. HCT-confidence interval (CI) score ≥ 3 increased the risks of more than one type of infection and fungal infection in the plasma sterilizer group. The absence of chemotherapy before allo-HSCT was associated with an increased risk of fungal infection in the total cohort (Tables 4 and 5).

The types of detected pathogens in the conventional laminar flow room group and plasma sterilizer group. (a) In the conventional laminar flow room group. (b) In the plasma sterilizer group.
Infections characteristics of plasma sterilizer and conventional laminar flow room groups.
Univariate analysis of infection.

CI: Confidence interval; HR: Hazard ratio; HCT-CI: Hematopoietic cell transplantation–comorbidity index; MSD: Matched sibling donor; HID: Haploidentical donor; MUD: Matched unrelated donor; NA: Not applicable.
Multivariate analysis of infection.

CI: Confidence interval; HR: Hazard ratio; HCT-CI: Hematopoietic cell transplantation–comorbidity index; MSD: Matched sibling donor; HID: Haploidentical donor; MUD: Matched unrelated donor; NA: Not applicable.
During the period from engraftment to 1 year after allo-HSCT, the rates of overall infection, more than one type of infection, bacterial infection and viral infection were comparable between the two groups, and patients in the plasma sterilizer group exhibited a lower rate of fungal infection (Table 3).
Engraftment, GVHD, relapse, and non-relapse mortality
Within 100 days after allo-HSCT, there was no statistically significant difference observed between the two groups in terms of neutrophil engraftment, platelet engraftment (Table 1). The clinical outcomes were also comparable in terms of the probability of OS (98.5% [95% CI: 86.8%–100%] vs 99.5% [95% CI: 98.5%–100%]; P = 0.316), probability of LFS (96.5% [95% CI: 94.0%–99.0%] vs 96.5% [95% CI: 94.0%–99.1%]; P = 0.991), the cumulative incidence of relapse (2.0% [95% CI: 0.1%–3.9%] vs 3.0% [95% CI: 0.6%–5.4%]; P = 0.523), the cumulative incidence of NRM (1.5% [95% CI: 0%–3.2%] vs 0.5% [95% CI: 0%–1.5%]; P = 0.316) and 100-day cumulative incidence of acute graft-versus-host disease (23.4% [95% CI: 17.5%–29.3%] vs 22.0% [95% CI: 16.2%–27.8%], P = 0.723) respectively, for conventional laminar flow room group and plasma sterilizer group (Fig. 2).
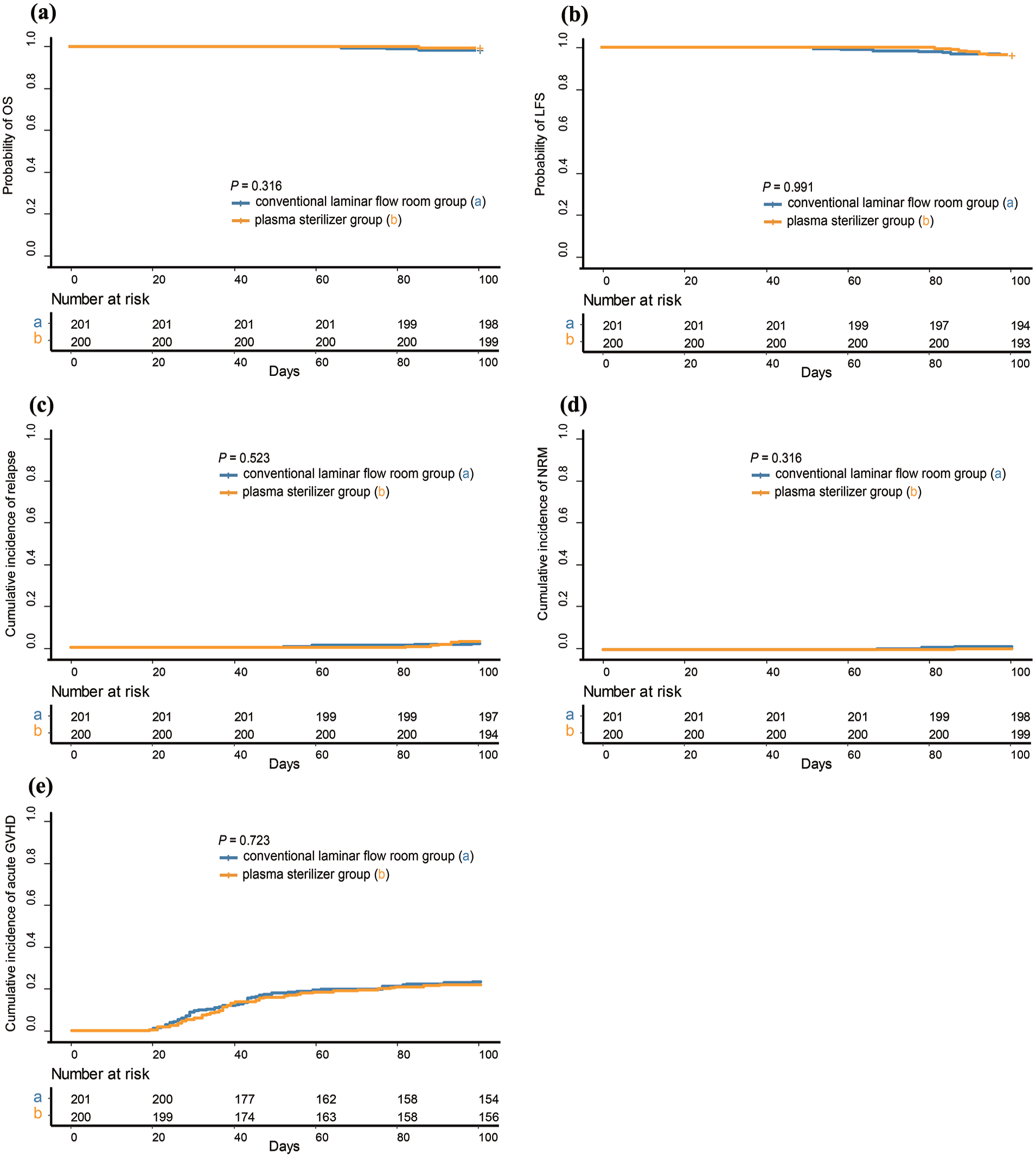
100-day clinical outcomes after allo-HSCT. (a) OS. (b) LFS. (c) Relapse. (d) NRM. (e) Acute GVHD.
At the 1-year follow-up after allo-HSCT, the probability of OS was 98.5% (95% CI: 96.8%–100%) for the plasma sterilizer group, which was better than that of the conventional laminar flow room group (91.5%; 95% CI: 87.6%–95.4%; P = 0.005). Patients in the plasma sterilizer group also exhibited a better 1-year probability of LFS (94.0% [95% CI: 97.3%–100%] vs 85.9% [95% CI: 81.2%–90.9%]; P = 0.010) and a lower 1-year cumulative incidence of NRM after allo-HSCT (1.0% [95% CI: 0%–2.4%] vs 8.6% [95% CI: 4.7%–12.5%]; P = 0.00047). The 1-year cumulative incidence of relapse (5.0% [95% CI: 1.9%–8.1%] vs 5.5% [95% CI: 2.3%–8.7%]; P = 0.851) and the 1-year cumulative incidence of chronic GVHD (36.5% [95% CI: 29.8%–43.2%] vs 30.8% [95% CI: 24.4%–37.2%]; P = 0.233) after allo-HSCT were comparable between the plasma sterilizer group and the conventional laminar flow room group (Fig. 3).

1-year clinical outcomes after allo-HSCT. (a) OS. (b) LFS. (c) Relapse. (d) NRM. (e) Chronic GVHD.
Discussion
In this study, we observed that there were no significant differences in infection rates, engraftment rates, or 100-day posttransplantation clinical outcomes between the plasma sterilizer and conventional laminar flow rooms. At the 1-year follow-up after allo-HSCT, the laminar flow room group demonstrated superior OS and LFS, as well as a lower NRM. This is the first study reporting that allogeneic hematopoietic stem cell transplantation conducted in a plasma sterilizer environment can achieve clinical outcomes similar to those of transplantation conducted in a conventional laminar flow room. As our institution is the first in China to adopt plasma sterilizers on a large scale for allo-HSCT, the findings may serve as evidence for the potential broader implementation of plasma sterilizers in hematopoietic stem cell transplantation centers.
According to the operating instructions for both devices, conventional laminar flow rooms reduce airborne particles and pathogenic microorganisms to safe levels via primary, intermediate, and high-efficiency filters. Plasma sterilizers result in airborne pathogenic microorganisms and particles being maintained at safe standards via discharge, dissociation, and high-efficiency particulate air (HEPA) filtration. Laminar flow purification via air ventilation systems is often considered to be the safest and most effective measure for preventing respiratory infections in hematology/oncology wards. The effectiveness of HEPA in reducing the number of ambient fungal spores has been demonstrated in several studies; for example, via plasma sterilizers, the number of ambient fungal spores was reduced by 86% compared with the number of surface fungi in normal rooms, and airborne filamentous fungi were reduced by 80%. However, these fungi were not completely removed, and the monitored airborne filamentous fungi concentration was still observed at 3.2 ± 2.8 CFU/m3, which could be improved by increasing the air volume for better air purification 19 . In contrast, in our current study, repeated monitoring at multiple time points revealed that the number of pathogenic microorganisms in the plasma sterilizer group was <5 planktonic bacteria/m3, the number of settled bacteria was <1/Φ90 dish-30 min, the number of particles <0.5 µm in dust was less than 3,500 particles/m3, and the number of dust particles ≥5 µm was 0. Moreover, the rate of lung infections, the time of occurrence and the number of pathogens in patients who received allo-HSCT in the environment of the plasma sterilizer were similar to those of patients who received allo-HSCT in conventional laminar flow rooms, which also suggests that the sterile environment provided by the plasma sterilizer can effectively prevent lung infections during the granulocyte-deficient phase in allogeneic transplantation patients.
In accordance with previous domestic and international research, immunocompromised patients in the plasma sterilizer environment can be effectively protected from infections. For example, some studies have confirmed that this type of purification system can effectively reduce the occurrence of Aspergillus infections. Among hematology patients in the neutropenia stage, the infection incidence rate in the plasma sterilizer environment was observed to be 1.15%, which was significantly lower than that of patients in general wards (14.49%, P = 0.02; odds ratio (OR): 0.11; 95% CI: 0.00–0.84). They also reported a positive association between Aspergillus infection and the use of a plasma sterilizer (P = 0.03) 20 . Similarly, some authors have demonstrated that plasma sterilizers are more effective in infection prophylaxis than conventional air purification systems in burn wards. Specifically, before and after operation of the sterilizers, the use of plasma wards resulted in a significant decrease in the number of suspended particles in the air, thereby reducing the occurrence of infection 21 . Allogeneic hematopoietic stem cell transplantation recipients represent the population of patients with severe immunodeficiencies, and our study demonstrated that plasma sterilizers are effective in infection prophylaxis in preengraftment patients.
For patients receiving allo-HSCT in conventional laminar flow rooms, the rates of lung infections, catheter-associated bloodstream infections, and bloodstream infections have been demonstrated to be 18.4% to 85.48%22–24, 2.8% to 11.2% 25 , and 10% to 50%26–28, respectively, during the preengraftment phase. In our present study, the rates of lung infections, catheter-associated bloodstream infections, and bloodstream infections were 8%, 13.5%, and 13.5%, respectively, during the preengraftment phase for patients receiving allo-HSCT via the plasma sterilizer. Therefore, our results suggested that the efficacy of infection prevention was comparable between plasma sterilizer and conventional laminar flow rooms.
However, the difference in the timing of enrollment between the two groups (a 12-month gap, with the patients in the conventional laminar flow room group being enrolled in 2022, whereas those in the laminar flow room group were enrolled in 2023) may have contributed to the observed differences in long-term outcomes. The changing epidemiology of COVID-19 in China29–31, which was driven by evolving government policies and vaccination efforts, led to differences in infection rates between 2022 and 2023, which may have contributed to variations in NRM during long-term follow-up. Given the multitude of factors affecting long-term prognosis, the direct impact of laminar flow rooms on extended outcomes requires further investigation.
There were several limitations in this study. For example, this was a single-center, retrospective study, which may limit the generalizability of our findings. Some patients developed symptoms such as fever and received symptomatic antiinfection treatments; however, the pathogens were not identified, which may interfere with the statistical results regarding infections. Notably, many factors can influence infections, and the differences observed in laminar flow rooms may only represent one aspect of the impact on infection occurrence in both short-term and long-term follow-ups, which could be confounded by other factors.
In conclusion, our results suggest that the conventional laminar flow room remains the cornerstone of patient treatment, whereas a plasma sterilizer may also be used to improve patient outcomes. These results should be confirmed via multicenter, randomized controlled trials in the future.
Supplemental Material
sj-docx-1-cll-10.1177_09636897251335722 – Supplemental material for Comparison of plasma sterilizer and conventional laminar flow room in allogeneic hematopoietic stem cell transplant recipients
Supplemental material, sj-docx-1-cll-10.1177_09636897251335722 for Comparison of plasma sterilizer and conventional laminar flow room in allogeneic hematopoietic stem cell transplant recipients by Ting Wang, Yi Xia, Wei Hu, Boning Liu, Xiaohui Zhang, Wenxuan Huo, Jun Kong, Yaru Ma, Wenwen Xiao, Ce Shi, Qixin Du, Leqing Cao, Dong Han, Dongyue Yao, Hongyue Yin, Daoxing Deng, Jingyu Gao, Yashu Jia, Jiating Wang, Jing Liu, Xiaoshuang Han, Junxia Wang, Ling Ma, Yunjing Xia, Shanshan Hu, Yuanyuan Zhang, Fengmei Zheng, Xiaojun Huang and Xiaodong Mo in Cell Transplantation
Footnotes
Ethical considerations
The study was approved by the institutional review board of Peking University People’s Hospital (No. 2025PHB085-001) and was conducted in accordance with the Declaration of Helsinki.
Author contributions
T.W. and Y.X. analyzed the data and wrote the manuscript; W.H. and B.N.L. contributed to the data collection and helped write and edit the manuscript. W.X.H. performed statistical analysis; X.J.H., X.H.Z., and X.D.M. designed the study, analyzed the data, and edited the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science Foundation of Beijing (Z230016) and the National Key Research and Development Program of China (2022YFA1103300, 2022YFC2502606).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of human and animal rights
This article does not contain any studies with human or animal subjects.
Statement of informed consent
There are no human subjects in this article and informed consent is not applicable.
Data Availability Statement
The data set supporting the conclusions of this article is available in the clinical data repository of our affiliation. Individual participant data were not shared. For the original data, please contact the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
