Abstract
Myelodysplastic syndrome (MDS) is a clonal disorder that affects hematopoietic stem cells (HSCs), primarily occurring in the elderly population. Lower-risk MDS is characterized by a decrease in blood cells, whereas higher-risk MDS is associated with an increased risk of transformation to acute myeloid leukemia (AML). Currently, the treatment of MDS is still unsatisfactory, although demethylating agents, azacitidine (AZA), and decitabine (Dec) have been successfully used to treat MDS and improve survival rates. However, hematopoietic stem cell transplantation (HSCT) remains the only curative treatment for MDS patients, effectively increasing patient survival and quality of life. Nevertheless, treatment-related toxicity, graft-versus-host disease, infectious complications, and relapse are still major post-transplant issues. In this review, through a retrospective analysis of past and present HSCT for the treatment of MDS, we provide insights for the future.
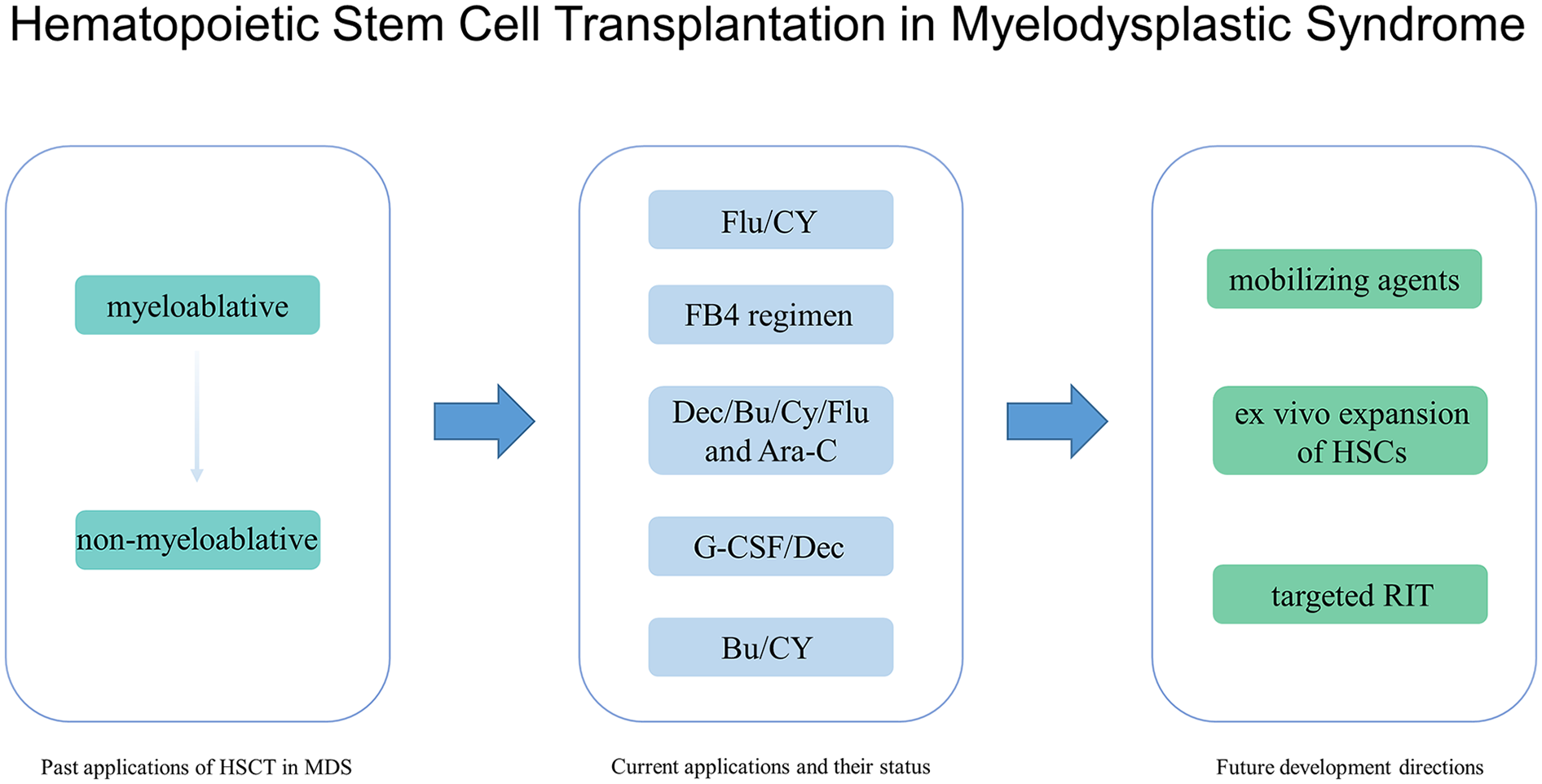
Understanding Myelodysplastic Syndrome
Myelodysplastic syndrome (MDS) are a group of disorders characterized by varying degrees of cytopenia, morphological dysplasia, and a risk of progressing to acute myeloid leukemia (AML) 1 . The concept of MDS can be traced back to 1953 when it was initially referred to as preleukemia 2 . It was not until 1982 that the term MDS was coined 3 , it is now also referred to as myelodysplastic neoplasms (MDS). The World Health Organization published classifications for MDS in 2001 and 2008 and revised the 2008 version in 2016. The most recent classification method is the 5th edition released by the World Health Organization in 2022 (Table 1) 4 . The aim of MDS classification is to identify entities with shared genetic underpinnings and molecular pathogenesis, and the specific subtype can inform clinical decision-making alongside prognostic risk categorization. The current MDS classification schemes incorporate morphologic features (bone marrow and blood blast percentage, degree of dysplasia, ring sideroblasts, bone marrow fibrosis, and bone marrow hypocellularity) and also recognize three entities defined by genetics: isolated del(5q) cytogenetic abnormality, SF3B1 mutation, and TP53 mutation 5 . The main clinical manifestation of MDS is anemia, which is due to ineffective erythropoiesis with a high degree of apoptosis in bone marrow erythroid progenitor cells 6 , leading to fatigue, poor quality of life, and potential instability in cardiovascular disease 7 . In addition, it may be accompanied by thrombocytopenia and/or neutropenia 8 . MDS causes functional abnormalities of hematopoietic cells in addition to low numbers of blood cells and is well known to be complicated by systemic inflammatory autoimmune diseases (SIADs) 9 . In the general population, the incidence of MDS is approximately 4.5 cases per 100,000 people per year. The incidence is only 0.1 cases per 100,000 people per year in individuals below the age of 40, whereas in the 70 to 79 age group, it rises to 26.9 cases per 100,000 people per year, and it is as high as 55.4 cases per 100,000 people per year for those aged 80 or above 10 . The median age of patients is 77 years old 11 . Most MDS patients die from complications rather than progressing to AML 12 . Infections, particularly bacterial infections, are the leading cause of death 13 . In addition, SIADs before allogeneic hematopoietic stem cell transplantation (allo-HSCT) constitute an independent risk factor for death in MDS patients undergoing allo-HSCT 14 . Therefore, prevention of infection is critically important. The pathophysiology of MDS involves a multi-step process that includes cytogenetic changes, gene mutations, or both 15 . Recurrent genetic alterations, gene mutations, and epigenetic dysregulation have been recently implicated in the pathogenesis of MDS 16 . Common etiological factors for MDS include chemotherapy, ionizing radiation, and occupational exposure to benzene 17 . In addition, congenital diseases, such as Fanconi anemia, can increase the risk of developing MDS 18 .
Classification and Defining Features of Myelodysplastic Neoplasms (MDS).
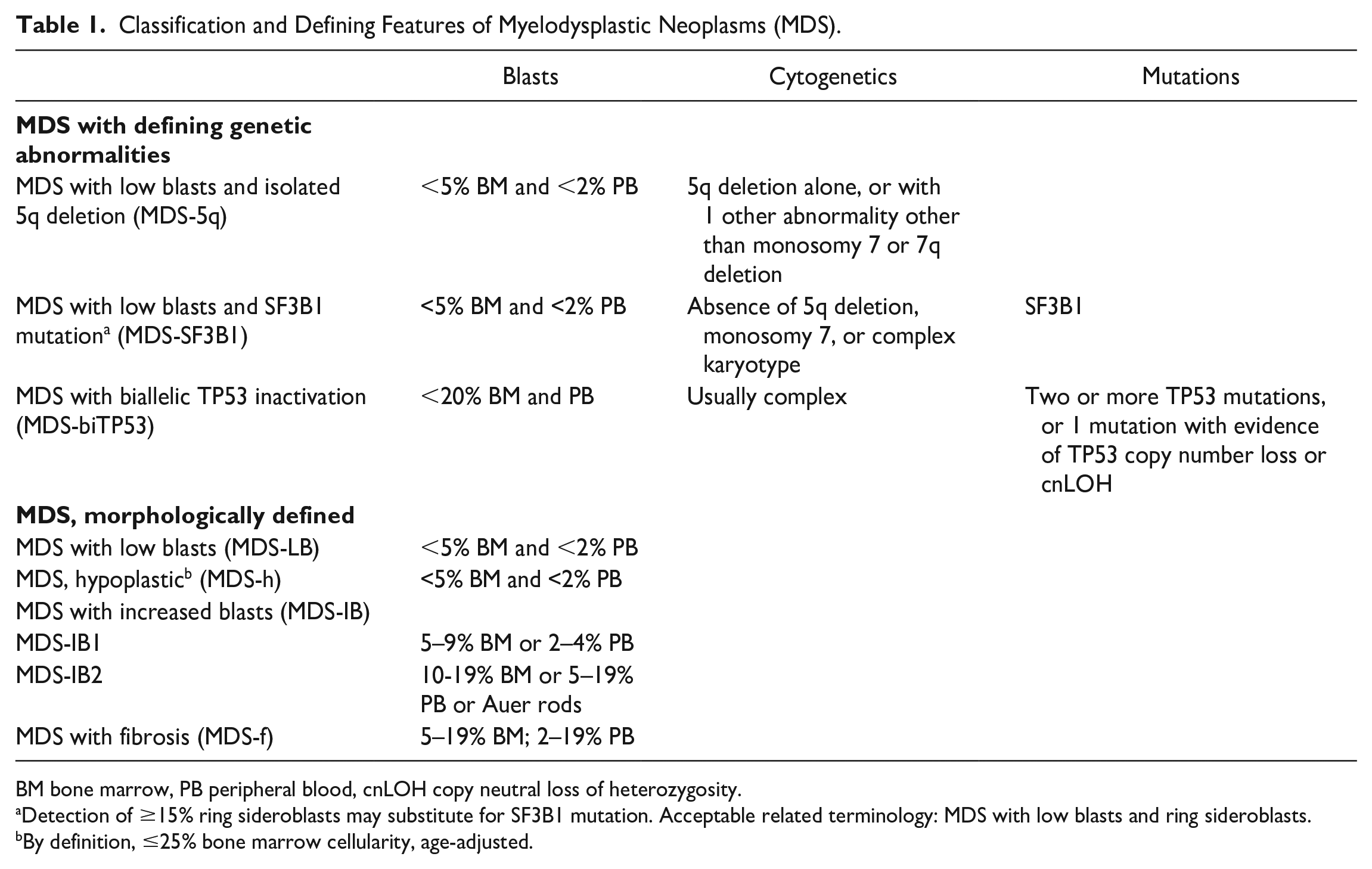
BM bone marrow, PB peripheral blood, cnLOH copy neutral loss of heterozygosity.
Detection of ≥15% ring sideroblasts may substitute for SF3B1 mutation. Acceptable related terminology: MDS with low blasts and ring sideroblasts.
By definition, ≤25% bone marrow cellularity, age-adjusted.
Treatments are tailored to the patient’s disease characteristics and comorbidities and range from supportive care with or without erythropoiesis-stimulating agents for patients with low-risk MDS to hypomethylating agents, such as azacitidine (AZA) or decitabine, for patients with higher-risk MDS. Hematopoietic cell transplantation (HCT) is potentially curative and should be considered for patients with higher-risk MDS at the time of diagnosis 8 . Although treatment with several drugs has shown improved disease control, allo-HSCT remains the only curative treatment for MDS 19 . Furthermore, VEXAS syndrome is a monogenic disease of adulthood caused by somatic mutations in UBA1 in hematopoietic progenitor cells, which is closely associated with MDS. VEXAS may be a prototype for a new class of “hematoinflammatory” diseases 20 . MDS patients with UBA1 p.Met41 mutation probably have a poor prognosis regardless of a low IPSS-R score and are unlikely to improve with immunosuppressive or hypomethylating drugs and should be considered for alternative therapies 21 .
Hematopoietic Stem Cell Transplantation
Hematopoietic stem cells (HSCs) used for transplantation can be derived from three sources: bone marrow, mobilized peripheral stem cells, and umbilical cord blood. Bone marrow is obtained through multiple punctures and aspirations of the posterior iliac crest, mobilized peripheral stem cells are obtained by leukapheresis after the administration of granulocyte-stimulating factor, and umbilical cord blood is collected from the placental blood through the umbilical cord at the time of delivery 22 . Bone marrow transplantation (BMT) was the first established stem cell transplantation (SCT) method and was first conducted in clinical trials by E. Donnell Thomas in 1957 23 , and subsequently served as the only source of HSCs. It was not until the successful peripheral blood hematopoietic stem cell transplantation (HSCT) in Heidelberg in 1985 24 that another source became available. Subsequently, in 1988, the first successful umbilical cord blood transplantation was performed in Saint-Louis Hospital, Paris, France, on a 5-year-old boy with Fanconi anemia 25 , greatly expanding the sources of HSCs and providing more transplantation hope for patients.
HSCT can be mainly classified into two types: autologous hematopoietic stem cell transplantation (auto-HSCT) and allo-HSCT 26 . In auto-HSCT, stem cells are collected from the patient themselves. However, in allo-HSCT, appropriate donors must be selected 27 . Allo-HSCT is the treatment of choice for the majority of patients with MDS who have a histocompatible donor (sibling or unrelated donor). However, only 30% of these cases have fully matched sibling donors (MSDs). Alternatively, matched unrelated donors (MUDs) and haploidentical (haplo) donors from first-degree relatives increase the access to transplantation. A meta-analysis compared the efficacy and toxicity of MSD-HSCT and MUD-HSCT with haplo-HSCT in patients with MDS. The results showed no significant differences in overall survival (OS) and several secondary outcomes between MDS patients receiving haplo-HSCT and those receiving MSD-HSCT and MUD-HSCT. This suggests that haplo-HSCT is a reasonable alternative 28 . In addition, for some patients lacking a human leukocyte antigen (HLA)-compatible donor, chemotherapy followed by allo-HSCT may be a reasonable alternative. However, a substantial number of candidates may not be eligible for allo-HSCT due to failure to induce remission or failure to collect sufficient numbers of stem cells. Peripheral blood stem cells should be the graft source of choice in the majority of patients 29 . Among the various factors contributing to successful transplantation, the most essential factor is the compatibility of HLA between donors and patients 30 . Therefore, HLA-matched related donors are considered the optimal choice for allo-HSCT, as matching HLAs and shared non-HLA gene polymorphisms decrease alloimmune reactions, facilitate rapid immune reconstitution, and lower the incidence of graft-versus-host disease (GVHD), ultimately achieving the best outcomes 31 . In cases where a matched donor cannot be found, there are currently three alternative donor options available: partially HLA-mismatched unrelated donors, haploidentical related donors, and umbilical cord blood 32 .
Pre-transplant conditioning regimens have been classified into high-dose (myeloablative), reduced-intensity conditioning (RIC), and non-myeloablative (NMA) 33 . High-dose or myeloablative regimens involve the administration of high doses of chemotherapy, with or without radiotherapy, followed by infusion of donor hematopoietic cells to provide blood and immune recovery 34 . To exploit GVHD effects without significant regimen-related toxicities, many investigators have reduced the dose of radiation or alkylating agents used in conditioning regimens, which are referred to as NMA or RIC regimens 35 , enabling HSCT for older and debilitated patients. However, complications such as infections, GVHD, and sinusoidal obstruction syndrome to a lesser extent persist 36 . Consequently, HSCT patients should undergo individualized, risk-adaptive, and multidisciplinary follow-up to provide an accurate diagnosis and appropriate treatment for any encountered complications 37 .
Prognostic scoring systems are also very important for MDS patients. Previously, prognostic systems for MDS have widely used WHO-classification based Prognostic Scoring System (WPSS), International Prognostic Scoring System (IPSS), and International Prognostic Scoring System–Revised (IPSS-R). In recent years, the utility of clinical-molecular prognostic model (IPSS-Molecular [IPSS-M]), which includes genetic mutation information, has also been suggested. Combining genomic profiling with hematologic and cytogenetic parameters, the IPSS-M improves the risk stratification of patients with MDS and represents a valuable tool for clinical decision-making 38 . Since these prognostic systems are not specifically designed for allo-HSCT, the Center for Blood and Marrow Transplant Research (CIBMTR) has proposed a prognostic model specifically for allogeneic transplantation in MDS patients. The scoring system uses readily available clinical data and can be calculated quickly, facilitating patient consultation with respect to allogeneic hematopoietic cell transplantation (allo-HCT), and may also be used to identify high-risk populations where interventions such as post allo-HCT maintenance therapies may be of benefit 39 . Moreover, since MDS is a disease predominantly affecting the elderly, it is necessary to assess the risk of post-transplant complications. Generally, comorbidity evaluations are performed using HCT-CI or HCT-CI/Age, but the HCT-CI was originally developed to predict the outcome of patients receiving allogeneic transplantation and is, therefore, unlikely to be equally effective in a more heterogeneous population of patients. Therefore, MDS-CI was established to better define the life expectancy of patients with MDS stratified according to the WPSS. Comorbidities have a significant impact on the outcome of patients with MDS. Accounting for both disease status by means of the WPSS and comorbidity through the MDS-CI considerably improves risk stratification in MDS 40 .
Past Applications of HSCT in MDS
The first successful utilization of HSCT in the treatment of MDS was possibly performed in 1978 by S. Bhadur et al. 41 in a case of preleukemia who underwent a BMT transplantation from identical twin. The patient received 850 rad of total body irradiation (TBI) after two and a half weeks of isolation and oral antibiotic disinfection, followed by BMT from his twin brother at a dose of approximately 2.14 × 108 cells/kg body weight. Severe Escherichia coli septicemia with fever and alkalosis occurred after transplantation, and subclinical shock was observed 7 days later, leading the patient to undergo steroid therapy. However, the patient’s peripheral granulocyte count improved significantly by day 24 after transplantation, and the complete blood count reached normal levels by day 48, indicating the feasibility of HSCT in the treatment of MDS. In 1982, Gyger et al. 42 performed an allogeneic bone marrow transplantation (allo-BMT) in a case of hematological preleukemia with Hodgkin’s disease, which was complicated by interstitial pneumonia and severe hypoxemia at day 154 post-transplantation, despite prophylaxis with methotrexate (MTX) for GVHD; high-dose steroid treatment was subsequently administered, and the patient achieved normal bone marrow function by day 316 post-transplantation. In two reports published by Appelbaum FR in 1984 and 1987, the results of allo-BMT in 30 preleukemia patients (median age of 24 years) from 1977 to 1985 were detailed. Of these patients, three who had only received cyclophosphamide (CY) before transplantation all died within 9 months after transplantation. The remaining 27 patients were treated with CY combined with TBI before transplantation. Among these 27 patients, 20 had preleukemia or severe bone marrow fibrosis unrelated to previous treatment. Thirteen of these 20 patients survived well for 9 to 56 months after transplantation. The seven treatment failures included four cases of interstitial pneumonia, two cases of candidemia, and one case of disseminated herpes zoster infection. None of the 20 patients had a relapse. All patients received immunosuppressive therapy with MTX, cyclosporine A (CsA), or a combination of both after transplantation, but GVHD still occurred43,44. In 1988, Bélanger et al. 45 reported successful implantation in seven out of eight patients (median age 34.5 years) with five of them surviving and achieving complete remission for 9 to 35 months after implantation. Prior to transplantation, the patients received CY and TBI or busulfan (Bu) and CY treatment, and after transplantation, they received immunosuppressive therapy with MTX and methylprednisolone (MP) or CsA and MP. During the non-cellular disease period, they underwent strict reverse isolation and received oral prophylactic antibiotics, but two cases died of GVHD and one case relapsed. It can be seen that bone marrow ablation protocols such as TBI and high-dose CY before transplantation and immunosuppressive therapy after transplantation can completely restore some MDS patients. Therefore, allo-BMT can eradicate the malignant clone of some MDS patients, prolong their disease-free survival (DFS), and is currently the only cure for MDS patients who can achieve long-term DFS. However, different degrees of relapse, GVHD, and interstitial pneumonia still occur after transplantation. In the same year, Bunin et al. 46 reported that three out of six bone marrow transplant patients survived disease-free for 240, 395, and 560 days post-transplant, including two unrelated donors. In 1989, Guinan et al. 47 performed matched or mismatched bone marrow transplants on eight patients, and four achieved complete remission at 10 to 44 months post-transplant. In 1993, Jeanne E. 48 Anderson et al. treated 93 MDS patients, with 65 bone marrow donors being HLA-identical siblings and 28 being other family members or unrelated donors. The prognosis of the 28 patients who received non-relative or non-HLA-matched family member donors was similar to that of the 65 siblings with HLA-identical donors. Therefore, it can be inferred that for MDS patients who have not found a sibling donor with HLA tissue completely compatible antigens, BMT from related or unrelated donors with certain matching degree may be effective, even curative treatment. In 1994, Uberti et al. 49 reported a new preparation scheme, named “BAC,” which included administration of 1 mg/kg of Bu every 6 hours, followed by 2 g/m2 of cytarabine arabinoside (Ara-C) every 12 hours for four times, and a final dose of 60 mg/kg of CY every day for two consecutive days. Among the 32 transplanted patients with a median age of 33 years, 19 achieved DFS. In 1996, Anderson et al. 50 reported that the 3-year DFS of patients treated with BU/CY/TBI was similar to that of patients treated with CY/TBI. Therefore, the outcomes of using Bu and CY with or without TBI were equivalent to those of the classic scheme of using CY and TBI.
All of the above are treated through high-dose chemotherapy-radiation combination therapy, supplemented by autologous or allogeneic BMT with HSCT from blood or bone marrow sources. However, transplantation is only limited to a few relatively young patients below the age of 55. Since most MDS patients are elderly, this treatment method has significant limitations. In 1997, Sergio Giralt et al. 51 treated 15 patients with a median age of 59 years using the method of transplanting allogeneic peripheral blood or bone marrow stem cells after NMA chemotherapy containing purine analogs. Among them, eight patients received fludarabine (Flu), idarubicin, Ara-C, or melphalan(Mel), and seven patients received 2-chloro-deoxy-adenosine and Ara-C. Thirteen patients received allogeneic peripheral blood stem cell treatment after chemotherapy, one patient received bone marrow treatment, and one patient died of multiple organ failure before receiving stem cell treatment. GVHD prevention included CsA and MP. Ultimately, eight patients achieved complete remission. In 1998, Shimon Slavin et al. 52 performed non-myeloablative stem cell transplantation (NSTC) on 26 patients using Flu, anti-T-lymphocyte globulin, and low-dose Bu, with standard-dose CsA as the only GVHD prophylaxis. Among the 26 patients who received allo-NMA SCT, 22 survived and 14 did not develop GVHD. Thus, this method can be used to treat patients who are not eligible for allogeneic transplantation due to age or comorbidities. Although this approach can significantly reduce the risk of acute graft-versus-host disease (aGVHD), there are still significant secondary toxicities such as infections and chronic graft-versus-host disease (cGVHD) 53 .
Through continuous research and exploration, the cure of MDS has become possible. The use of TBI and immunosuppressive agents is also being optimized, transitioning from myeloablative regimens initially to NMA regimens that can be applied to older individuals. However, there are still several challenges in HSCT for MDS treatment, including lower cure rates and higher risks of relapse, infection, and GVHD.
Current Applications and Their Status
Entering the 21st century, the treatment of MDS with HSCT has evolved from conventional treatment regimens to RIC regimens, and subsequently to NMA regimens 54 . Although these regimens have successfully reduced the toxicity associated with SCT, disease relapse and GVHD remain significant challenges. New treatment approaches continue to emerge, aiming to achieve effective anti-leukemia effects while limiting toxicity, especially in patients who are not eligible for standard myeloablative conditioning (SMA). In 2004, Jochen Casper et al. 55 employed a novel, less toxic allo-HSCT conditioning regimen, combining treosulfan (Treo) with Flu, to treat 30 patients. Estimated OS was 73% and event-free survival (EFS) was 49% at 7.4 to 33.4 months after treatment. In 2007, Shimoni et al. 56 similarly treated patients who were not suitable for SMA with Flu and Treo, and among 24 patients, the 2-year DFS rate was 60%. In 2011, Tapani Ruutu et al. 57 performed allo-HSCT using Treo and Flu for 45 patients with primary MDS. Following the transplantation, 80% of the patients experienced infections, and 22% had gastrointestinal symptoms. aGVHD of grades II–IV occurred in 24% of the patients, while chronic extensive GVHD was observed in 28% of the patients. The estimated OS rate and DFS rate at 2 years were 71% and 67%, respectively. The 2-year cumulative nonrelapse mortality (NRM) rate was 17%, and the encouraging aspect was the low 2-year relapse rate of only 16%. In 2020, Wedge E et al. 58 compared patients treated at Copenhagen University Hospital from 2000 to 2018 using NMA (n = 124), SMA (n = 36), and FluTreo (n = 55). Post-transplant immunosuppression included short-course MTX and CsA for SMA patients, tacrolimus and mycophenolate mofetil for NMA patients, and MTX and tacrolimus for FluTreo patients. The 1-year OS rates for FluTreo, NMA, and SMA were 84.0%, 68.3%, and 58.3%, respectively. The cumulative incidence of aGVHD at 100 days post-transplant was 55.6% for SMA, 32.0% for FluTreo, and 29.8% for NMA. The cumulative incidence of cGVHD at 3 years post-transplant was 62.2% for NMA, 50.0% for SMA, and 38.2% for FluTreo. In 2022, Dietrich W. Beelen et al. 59 analyzed the results of 570 randomized patients who completed long-term follow-up and underwent Flu/Treo or Flu/Bu conditioning. The Treo-based regimen showed superior EFS compared with the Bu-based regimen: 36-month EFS was 59.5% and 49.7%, respectively. Similarly, the Treo-based regimen had better OS compared with the Bu-based regimen: 36-month OS was 66.8% and 56.3%, respectively. Therefore, the Flu/Treo conditioning regimen can be considered a highly intensified and improved marrow-ablative regimen with effective anti-leukemic effects and acceptable toxicity. FluTreo expands the application of myeloablative conditioning, reducing the risks of aGVHD and NRM, without a significant increase in relapse rates.
In 2009, van Besien et al. 60 reported a significant reduction in the incidence of aGVHD and cGVHD in MDS transplant patients by adding alemtuzumab to the RIC regimen using Flu-Mel. In 2012, they treated 72 patients with a Clofarabine-Mel-alemtuzumab regimen, resulting in a 1-year cumulative relapse incidence of 29%, 1-year survival rate of 59%, and 1-year progression-free survival of 45%. Among the 74 patients receiving phase II dose treatment, 16 cases (21%) experienced grade III–V renal toxicity. Although this regimen showed promising response rates and duration, renal toxicity posed a considerable risk, especially in elderly patients 61 .
In 2014, Mohamad Mohty et al. 62 conducted a study using a reduced toxicity conditioning (RTC) regimen consisting of Bu (130 mg/m2/d) intravenous infusion for 3 days, Flu (30 mg/m2/d) intravenous infusion for 5 days, and antithymocyte globulin (2.5 mg/kg/d) intravenous infusion for 2 days. The estimated 2-year OS rate and DFS rate were 62% and 50%, respectively. The cumulative incidence of grades II–IV aGVHD and cGVHD (all grades) was 29% and 35%, respectively. After 2 years, the cumulative incidence rates of relapse/disease progression and NRM were 44% and 11%, respectively. These results show similar NRM rates compared with another commonly used RTC conditioning regimen consisting of Flu, antithymocyte globulin, and a fully myeloablative dose of intravenous Bu (referred to as the FB4 regimen). Bredeson et al. 63 reported a 1-year NRM rate of 9% using the FB4 regimen. Russell et al. 64 reported a 2-year NRM rate of 10%. Alatrash et al. 65 evaluated the treatment outcome of the FB4 regimen without using antithymocyte globulin in elderly patients (median age 58 years) and reported a 1-year NRM rate of 21%. Shimoni et al. 66 reported a 1-year OS rate of 47% and DFS rate of 43% when using the FB2 regimen with only 2 days of intravenous infusion of Bu (130 mg/m2/d). Therefore, for medically compromised high-risk malignancy patients, using intermediate-dose Bu (3 days) appears to be an effective alternative when compared with low-dose (2 days) or high-dose (4 days) regimens.
Furthermore, in 2020, Yi-Geng Cao et al. 67 conducted a study involving the treatment of 48 patients using a novel treatment regimen consisting of decitabine (Dec), Bu, CY, Flu, and Ara-C. The median follow-up time was 522 days (ranging from 15 to 1313 days). The OS rate was 86%, with a relapse incidence of 12% and a NRM rate of 12%. The occurrence rate of severe (III–IV grade) aGVHD was 23%, and chronic GVHD was observed in 15% of the cases. The 2-year OS for high-risk and very high-risk MDS patients was 74% and 86%, respectively. In 2023, Li Xuan et al. 68 reported a randomized study involving 202 patients with MDS and AML. The patients were divided into two groups: one receiving G-CSF, Dec, and Bu–CY (n = 101), and the other receiving Dec and Bu alone (n = 101). The median follow-up time was 32.4 months [interquartile range (IQR) = 10.0–43.0]. The 2-year cumulative incidence of relapse in the G-CSF, Dec, and Bu–CY group was 10.9% (95% CI: 5.8–17.9), whereas it was 24.8% (16.8–33.5) in the Dec- and Bu-alone group [hazard ratio = 0.39 (95% CI: 0.19–0.79); P = 0.011]. Thus, the treatment regimen consisting of G-CSF, Dec, and Bu–CY is a promising therapeutic efficacy.
At present, disease relapse is the main cause of treatment failure in patients with MDS undergoing HSCT, highlighting the importance of post-transplant preventive maintenance strategies. Yaara Yerushalmi et al. 69 conducted a second HSCT treatment for patients experiencing relapse after the initial transplantation. Current single-center studies indicate that approximately 25% of HSCT2 recipients achieve long-term survival. Therefore, HSCT2 may be considered as the preferred treatment option for patients experiencing relapse after HSCT1, as it is associated with longer survival. Thierry Guillaume et al. 70 reported on a cohort of 30 patients who received prophylactic post-transplant AZA plus escalated doses of donor lymphocyte infusion (DLI). The 2-year OS rate and DFS rate were 65.5%, with a cumulative relapse incidence of 27.6%. Compared with patients who did not receive post-transplant maintenance therapy, AZA showed good tolerability and reduced the risk of post-transplant relapse. Rautenberg et al. 71 through a retrospective analysis of 151 patients with myeloid malignancies who relapsed after allo-HSCT, also concluded that AZA can provide meaningful responses and long-term survival. Javier Bolaños-Meade et al. 72 compared three prophylactic regimens (tacrolimus, mycophenolate mofetil, and CY; tacrolimus, MTX, and bortezomib; or tacrolimus, MTX, and maraviroc) to a control group (tacrolimus and MTX) in reducing GVHD after RIC-HSCT. Ultimately, tacrolimus, mycophenolate mofetil, and post-transplantation CY were the only interventions to have better GVHD-free, relapse-free survival (RFS) than controls. In addition, Zheng-Ping Yu et al. 73 reported an 80.2% probability of OS for 81 patients who underwent unrelated cord blood transplantation following a conditioning regimen of high-dose CY combined with Flu. This regimen enhanced cord blood stem cell engraftment, facilitated post-transplant immune reconstitution, and improved OS.
Therefore, currently, the main pre-transplant regimens include Flu/CY, Bu/Flu/antithymocyte globulin, FB4 regimen, Dec/Bu/Cy/Flu and Ara-C regimen, as well as G-CSF, Dec, and Bu–CY conditioning. For the treatment of relapse, the main options are HSCT or reducing post-transplant relapse rates through the use of AZA. Although HSCT has reached a considerable level of maturity, challenges such as disease relapse and GVHD continue to pose significant obstacles in achieving disease cure.
Future Development Directions
HSCT remains the only curative treatment for MDS. In order to make allo- HSCT a more relevant therapeutic approach, progress must be made in alleviating three major associated issues: post-transplant relapse, regimen-related toxicity, and cGVHD. Among these, post-transplant relapse is currently the main challenge encountered. Enhanced conditioning regimens can reduce the relapse rate to some extent but may increase transplant-related mortality without improving OS. Therefore, new effective conditioning regimens are still under exploration. Several researchers have already reported on some novel approaches. Tong et al. 74 demonstrated the efficacy of a new regimen using Dec and cladribine for early measurable residual disease (MRD) clearance after allo-HSCT, resulting in low relapse rates, high survival rates, and no increased incidence of GVHD or adverse reactions. Wei et al. 75 reported the use of the chidamide and CAG (cytarabine, aclacinomycin, and granulocyte colony-stimulating factor) regimen combined with DLI for treating relapsed AML/MDS patients after allo-HSCT, which induced durable remission and prolonged survival. Samantha O’Hagan Henderson et al. 76 reported on the use of Treo/Flu/Ara-C as pre-transplant conditioning therapy in 77 patients with a median age of 54 years, achieving 1-year, 2-year, and 3-year RFS rates of 49.4%, 41.7%, and 37.6%, respectively, and OS rates of 59.3%, 49.3%, and 45.4%, respectively. The cumulative incidence of NRM was 10% at 100 days, 18.8% at 1 year, and 20.1% at 2 years. The cumulative incidence of relapse increased from 31% at 1 year to 38.5% at 3 years. Therefore, Treo/Flu/Ara-C provides a well-tolerated, feasible, and effective conditioning regimen for MDS patients, even in advanced disease stages.
In addition, Stewart et al. 77 performed the transplantation of normal male donor bone marrow into non-myeloablated female mice and compared it with 5-fluorouracil (5-FU) pretreated marrow. The results demonstrated a significantly higher engraftment rate for the normal donor bone marrow compared with the 5-FU-treated marrow. These research findings indicate that long-term engraftment can be established by transplanting normal unirradiated donor bone marrow into a normal unirradiated host mouse, suggesting that successful transplantation does not require the creation of a bone marrow niche. Omer-Javed et al. 78 reported that mobilizers create an opportunity for seamless engraftment of exogenous cells, which effectively outcompete those mobilized, to repopulate the depleted bone marrow. This competitive advantage is attributed to mitigating the detrimental effects on hematopoietic stem/progenitor cells (HSPCs) during ex vivo culture and further enhancing the transplantation effect by employing optimized messenger RNA (mRNA) delivery for transient overexpression of transplant-related factors. This may present an innovative approach to bypass the requirements of chemotherapy/radiotherapy in hematopoietic stem/progenitor cell gene therapy (HSPC-GT), providing long-term therapeutic benefits while significantly reducing patient risks and long-term toxicities. Although substantial effort has been applied to identifying HSC maintenance factors through the characterization of the in vivo bone marrow HSC microenvironment or niche, stable ex vivo HSC expansion has previously been unattainable. Wilkinson et al. 79 high levels of thrombopoietin synergize with low levels of stem cell factor and fibronectin to sustain HSC self-renewal. In addition, they replaced serum albumin with polyvinyl alcohol as a culture medium, as serum albumin has long been considered a major source of biological contaminants in stromal cell cultures. Under these conditions, functional expansion of HSCs reached 236- to 899-fold within 1 month, and a small number of expanded HSCs were successfully transplanted into recipient mice without undergoing conventional radiation-based bone marrow conditioning. This approach holds promise as a more efficient transplantation method, circumventing the limitations of requiring large quantities of HSCs and radiation therapy in traditional approaches.
Another promising approach is to reduce pre-transplant tumor burden through targeted radioimmunotherapy (RIT). This targeted RIT, while increasing intensity, does not significantly increase toxicity. Radioactive isotopes emitting alpha particles, such as astatine-211 (211At), have gained increasing interest as they release a substantial amount of radiation within a range of several cell diameters, enabling efficient and selective killing of target cells. Laszlo et al. 80 developed a targeted RIT based on 211At directed against CD123, an antigen widely expressed on acute leukemia and MDS cells, including potential tumor stem cells. By generating and characterizing novel mouse monoclonal antibodies specific to human CD123, four antibodies were selected that all internalize in CD123+ target cells. These data support further development of 211At–CD123 RIT toward clinical application.
Research has shown that post-transplant monitoring and measurable residual disease assessment play a crucial role in early detection and prevention of relapse 81 . In patients with MDS, the detection of measurable residual disease, characterized by lineage-specific engraftment of donor cells (CD33+ or CD34+) or allo-HSCT, is associated with an increased risk of relapse82,83. In addition, the evaluation of gene mutations may help predict the survival of MDS patients undergoing allo-SCT. Lindsley et al. 84 reported that TP53 mutations are associated with shorter survival and relapse time, making them the most effective prognostic factor for post-transplant survival in MDS patients. Papaemmanuil et al. 85 reported carrying somatic mutations in genes involved in signaling pathways (JAK2, KRAS, CBL), DNA methylation (DNMT3A, TET2, IDH1/2), transcription regulation (EVI1, RUNX1, GATA2), chromatin modification (EZH2, ASXL1), and RNA splicing (SF3B1, U2AF1, SRSF2, ZRSR2) as recurrent events. According to Haferlach et al. 86 , the six most common mutated genes in MDS patients are TET2, SF3B1, ASXL1, SRSF2, DNMT3A, and RUNX1.
Besides relapse, cGVHD has always been the most challenging complication of allo-HCT. However, currently, most preventive strategies for chronic GVHD have shown adverse effects on GVHD, thereby increasing the risk of relapse. Therefore, future research will focus on confirming these preliminary findings and incorporating more participants in prospective trials.
In future, HSCT for the treatment of MDS, besides further research on optimizing pre-transplant conditioning to improve therapeutic efficacy and reduce relapse, we must explore other more effective approaches to enhance disease cure rates. These approaches may include the use of mobilizing agents, ex vivo expansion of HSCs, and the application of targeted RIT to reduce pre-transplant tumor burden.
Discussion
After decades of research and development, HSCT has achieved significant progress in the treatment of MDS. Improved prognostic scoring systems have been developed, which not only facilitate better patient selection but also provide a better understanding of post-transplant survival based on disease- and patient-related factors. In addition, the development of more effective conditioning regimens, optimal GVHD prevention strategies, and the introduction of minimal residual disease detection methods for early relapse monitoring and patient selection for maintenance therapy may further contribute to the treatment of the disease.
Footnotes
Acknowledgements
We would like to express our gratitude to the Hematology Department team at International Mongolia Hospital of Inner Mongolia for their valuable suggestions on our research.
Author Contributions
Abbreviations
211At, astatine-211; 5-FU, 5-fluorouracil; aGVHD, acute graft-versus-host disease; allo-BMT, allogeneic bone marrow transplantation; allo-HCT, allogeneic hematopoietic cell transplantation; allo-HSCT, allogeneic hematopoietic stem cell transplantation; AML, acute myeloid leukemia; Ara-C, cytarabine arabinoside; auto-HSCT, autologous hematopoietic stem cell transplantation; AZA, azacitidine; BM, bone marrow; BMT, bone marrow transplantation; Bu, busulfan; cGVHD, chronic graft-versus-host disease; CsA, cyclosporine A; CY, cyclophosphamide; Dec, decitabine; DFS, disease-free survival; DLI, donor lymphocyte infusion; EFS, event-free survival; Flu, fludarabine; GVHD, graft-versus-host disease; Haplo, haploidentical; HLA, human leukocyte antigens; HSCs, hematopoietic stem cells; HSCT, hematopoietic stem cell transplantation; HSPC-GT, hematopoietic stem/progenitor cell gene therapy; HSPCs, hematopoietic stem/progenitor cells; MDS, myelodysplastic syndrome; Mel, melphalan; MP, methylprednisolone; MSDs, matched sibling donors; MTX, methotrexate; MUDs, matched unrelated donors; NMA, non-myeloablative; NRM, nonrelapse mortality; NSTC, non-myeloablative stem cell transplantation; OS, overall survival; RFS, relapse-free survival; RIC, reduced-intensity conditioning; RIT, radioimmunotherapy; RTC, reduced toxicity conditioning; SCT, stem cell transplantation; SIADs, systemic inflammatory autoimmune diseases; SMA, standard myeloablative conditioning; TBI, total body irradiation; Treo, treosulfan.
Ethical Approval
Not applicable
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the National Administration of Traditional Chinese Medicine. This work was supported by a grant from the Youth Independent Innovation Science Fund Project of Chinese PLA General Hospital (Grant No. 22QNCZ009). This work was also supported by a grant from the High-Level Clinical Specialty Development Technology Project for Public Hospitals in Inner Mongolia.
