Abstract
Hepatocyte transplantation (HTx) is a promising alternative to liver transplantation; however, poor engraftment remains a major challenge. Although co-transplantation with adipose tissue-derived stromal cells (ADSCs) or islets improves engraftment, exposure of these cells to the portal vein enhances innate immune responses, resulting in a significant loss of hepatocytes. Therefore, we investigated HTx at the liver surface as a novel approach that does not involve the portal vein. Hepatocytes were transplanted onto the liver surface of syngeneic analbuminemic rats with or without ADSCs and/or islets. Serum albumin levels and immunohistochemical staining of the transplanted hepatocytes were evaluated. Hepatocyte engraftment was compared between the liver surface and intraportal groups. To examine the detailed mechanisms behind co-transplantation, co-cultured supernatants were analyzed using multiplex assays, and inhibition tests using neutralizing antibodies were performed. Results showed that islet and ADSC co-transplantation markedly enhanced hepatocyte engraftment at the liver surface (P < 0.01), and its efficiency was comparable to that of intraportal transplantation (P = 0.35). In the co-transplantation group, cells were not necessarily in proximity, suggesting that humoral factors are important. In an in vitro study, hepatocyte function was significantly improved by co-culturing with islets and ADSCs (P < 0.01). Multiplex assays and inhibition tests revealed several important humoral factors, most notably insulin, which promoted hepatocyte engraftment. These findings suggest that HTx at the liver surface, together with crucial factors, may be a novel alternative strategy for intraportal transplantation.

Introduction
Hepatocyte transplantation (HTx) is considered a promising alternative therapy to liver transplantation (LTx)1–3. HTx is a more efficient and quicker procedure than LTx because it does not require open surgery or general anesthesia. At present, intraportal infusion is the most common and preferred site of transplantation in clinical HTx 4 . Another benefit is that it effectively utilizes donor livers such as neonatal and fatty livers, which may not be suitable for LTx. However, HTx faces a serious problem: the low engraftment rate of transplanted hepatocytes. According to previous reports, a transplant efficiency corresponding to nearly 5% of the total hepatocyte replacement is considered necessary to cure metabolic liver diseases5–8. While strategies such as repeated HTx 9 , pre-irradiation 10 , administration of vasodilators targeting hepatic sinusoids 11 , and inhalation of isoflurane 12 have been reported to improve transplant efficiency, the estimated efficiency of current HTx methods under normal circumstances remains only 0.5%13,14. Therefore, it is necessary to establish a new strategy for improving the engraftment efficiency of HTx.
Exposure of hepatocytes to the circulating bloodstream is known to induce an instant blood-mediated inflammatory reaction (IBMIR), in which exposure of tissue factors expressed on the surface of the hepatocytes to portal vein flow activates coagulation and complement cascades15,16. IBMIR was originally recognized in the field of pancreatic islet transplantation and has been shown to occur not only in islet transplantation but also in the transplantation of hepatocytes and mesenchymal stromal cells (MSCs)17–20. This reaction is thought to result in the loss of 50% to 70% of transplanted cells immediately after transplantation and is considered the main cause of low engraftment. Therefore, it is crucial to consider new transplantation sites for HTx that can be used as alternatives to intraportal transplantation.
Previous research has attempted to transplant hepatocytes into the spleen21,22, subcutaneously 23 , under the renal capsule 24 , and into lymph nodes 25 . Although certain successes have been reported in these locations, various measures are required to achieve engraftment, and there are still issues to be addressed regarding long-term engraftment efficiency. While cell transplantation to the liver surface has several advantages—(1) low invasiveness (laparoscopic approach possible); (2) possible transplantation of a large number of hepatocytes, large space for transplantation, and no need to worry about portal vein embolism; and (3) avoidance of IBMIR (no exposure to blood flow)—few studies have reported on liver surface transplantation as an alternative to intraportal transplantation. In islet transplantation studies, our group successfully cured diabetes by transplanting islets onto the liver surface and covering them with keratinocyte sheets previously 26 . However, this material causes substantial inflammation, and lot-to-lot variability is a problem in clinical applications. Thus, as an alternative covering material, we next focused on a nonwoven structured gelatin substrate called gelatin hydrogel nonwoven fabric (GHNF; KYOTO MEDICAL PLANNING Co., Ltd., Kyoto, Japan) and reported outstanding outcomes in subcutaneous islet transplantation27,28. Based on these results, we hypothesized that GHNF could be used as an effective scaffold for HTx at the liver surface.
Adipose tissue-derived stromal cells (ADSCs), among MSCs, can be harvested in large quantities with minimal invasiveness, and we have reported that co-transplantation with ADSCs improves hepatocyte engraftment 29 . In addition, it is known that islets also improve hepatocyte engraftment30–32. These studies suggested that several humoral factors secreted by ADSCs and islets promote hepatocyte engraftment. They also suggested that cell-to-cell contact and proximity may contribute to the maintenance of hepatocyte viability. Likewise, co-transplantation with these cells may improve hepatocyte engraftment, even in the case of liver surface transplantation.
In this study, we examined the utility of HTx at the liver surface as a non-intraportal transplantation procedure, using an albumin-free rat model to overcome the problems associated with IBMIR. We also investigated the crucial inflammatory mediators and growth factors that may affect hepatocyte function using an in vitro co-culture model to clarify the interactions among ADSCs, islets, and hepatocytes.
Materials and methods
Animals
Rat hepatocytes were obtained from male inbred F344/NSlc rats (age: 9–19 weeks; weight 195–296 g; Japan SLC, Inc., Shizuoka, Japan). Syngeneic analbuminemic rats (age: 11–18 weeks; weight 201–277 g) were bred at Tohoku University. All rats were kept on a 12-h light/dark cycle, and food and water were provided ad libitum. All animals were treated under the “Guide for the Care and Use of Laboratory Animals” published by the National Institutes of Health and the guidelines for animal experiments and related activities established at Tohoku University (approved protocol ID: 2022 MdA-080). All surgical procedures were performed under isoflurane anesthesia (Viatris Inc., Canonsburg, PA, USA), and efforts were made to minimize suffering.
Hepatocyte isolation
Hepatocytes were isolated using a two-step collagenase digestion method as previously reported 33 . Isolated hepatocytes were suspended in Dulbecco’s modified Eagle’s medium (Sigma-Aldrich Co. LLC, St. Louis, MO, USA) containing 10% fetal bovine serum (FBS) (Equitech-Bio Inc., Kerrville, TX, USA) and 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (Thermo Fisher Scientific Inc., Waltham, MA, USA). Density gradient centrifugation (50G, 20 min, 4°C) was performed using a Percoll density gradient centrifuge (Cytiva Sweden AB, Uppsala, Sweden) to obtain a highly purified cell population. Hepatocyte viability was measured using the trypan blue exclusion assay, and hepatocytes with ≥ 90% viability were used in the subsequent experiments as soon as possible after isolation.
Isolation and culture of ADSCs
Rat ADSCs were isolated and cultured as previously reported29,34.
Flow cytometry of ADSCs
Flow cytometry was performed to characterize the phenotypes of ADSCs. ADSCs at passage 3 isolated from F344/NSlc rats were labeled with fluorescein isothiocyanate-conjugated anti-rat CD29 antibody (Becton, Dickinson and Company, Franklin Lakes, NJ, USA), anti-rat CD31 antibody (Thermo Fisher Scientific Inc.), anti-rat CD34 antibody (Bioss, Inc., Woburn, MA, USA), anti-rat CD44 antibody (Becton, Dickinson and Company), anti-rat CD45 antibody (Becton, Dickinson and Company), anti-rat CD73 antibody (Bioss, Inc.), anti-rat CD90 antibody (Becton, Dickinson and Company), and anti-rat CD105 antibody (Bioss, Inc.) for 45 min on ice. Labeled cells were measured by a BD AccuriTM C6 Flow Cytometer (Becton, Dickinson and Company) and the obtained data were analyzed by BD AccuriTM C6 (Becton, Dickinson and Company).
Islet isolation
Rat islet isolation and culture were performed as previously described 35 . Before transplantation, islets were cultured overnight in Roswell Park Memorial Institute-1640 medium containing 5.5 mmol/L glucose (Thermo Fisher Scientific Inc.) and 10% FBS at 37°C in 5% CO2 and humidified air.
Hepatocyte transplantation
For transplantation onto the liver surface, hepatocyte suspensions obtained by spontaneous sedimentation and pellets of ADSCs and/or islets were mixed immediately before transplantation and seeded into GHNF. GHNF is a biodegradable three-dimensional (3D) gelatin fiber sheet originally developed for MSC culture that has strong tissue affinity36,37. The cell-containing GHNF (diameter: 11 mm; thickness: 0.5 mm) was placed on the middle lobe of the rat liver and covered with Seprafilm® (Baxter Healthcare Corporation, Deerfield, IL, USA) for fixation and adhesion prevention. For intraportal transplantation, the hepatocyte suspension was slowly injected into the portal vein through a catheter using a 25-G needle with a gas-tight syringe, as previously described 12 . Serum albumin concentration was quantified during the observation period using a LBIS rat albumin enzyme-linked immunosorbent assay (ELISA) kit (AKRAL-220; Fujifilm Wako Shibayagi, Gunma, Japan).
Experimental groups in the in vivo model
To evaluate the effect of HTx at the liver surface, hepatocytes were transplanted with or without ADSCs and/or islets, and serum albumin levels were quantified over time. GHNF was used as a cell scaffold for transplantation. In the Hep group (n = 19), GHNF containing five million hepatocytes were transplanted onto the middle lobe of the recipient liver. In the Hep-ADSC group (n = 5), hepatocytes (five million) and ADSCs (10 million) were co-transplanted. In the Hep-Islet group (n = 11), hepatocytes (five million), and islets (1,200 islet equivalents [IEQs]) were co-transplanted. In the Mix group (n = 7), hepatocytes (five million), ADSCs (10 million), and islets (1,200 IEQs) were co-transplanted. In a comparison of the transplant efficiency between transplantation at the liver surface and intraportal HTx, two sheets of GHNF (Double Mix group, n = 9; each containing a total of 10 million hepatocytes, 20 million ADSCs, and 2,400 IEQ islets) were transplanted onto the liver surface, while 10 million hepatocytes were transplanted intraportally (Ipo group, n = 9).
Immunohistochemistry
Recipient’s livers were retrieved 8 to 9 weeks after transplantation, fixed with 4% paraformaldehyde, embedded in paraffin, and subjected to immunohistochemistry with albumin staining using anti-albumin antibodies (MP Biomedicals, Santa Ana, CA, USA) combined with the VECTASTAIN ABC system (Vector Laboratories, Inc., Burlingame, CA, USA), insulin staining using rabbit anti-insulin antibodies (Abcam plc., Cambridge, Cambridgeshire, UK) combined with the EnVision System Labeled Polymer anti-rabbit (Agilent Technologies, Inc., Santa Clara, CA, USA), hematoxylin-eosin (HE) staining, Elastica-Masson (EM) staining, and periodic acid-Schiff (PAS) staining using a PAS stain kit (MUTO PURE CHEMICALS CO.,LTD., Tokyo, Japan).
Identification of key factors for hepatocyte function in co-culture supernatants
To investigate the detailed interactions among cells, hepatocytes were cultured with or without ADSCs and/or islets in an in vitro co-culture system. In the co-culture experiments, 1 million hepatocytes, 2 million ADSCs, and 240 IEQs of islets were used for each experiment, which is one-fifth the volume of the in vivo experiments. Hepatocytes alone (Hep group, n = 12), hepatocytes with ADSCs (Hep-ADSC group, n = 12), hepatocytes with islets (Hep-Islet group, n = 12), hepatocytes, ADSCs, and islets (Mix group, n = 12) were cultured for 3 days. Each cell was seeded into a 6-well plate coated with type 1 collagen (Corning, Inc. Kennebunk, ME, USA) and the medium (5 ml total per well) was Williams medium E (WE, Thermo Fisher Scientific Inc.). As previously reported, an ammonia metabolic assay was performed to evaluate the hepatocyte function38,39. The ammonia metabolic rate was calculated as the percentage decrease from the ammonia level at time 0 to 6 h. To identify the humoral factors that were significantly elevated in the co-culture supernatants, various cytokines were measured as follows: Interleukin (IL)-1α, IL-1β, IL-2, IL-4, L-5, IL-6, IL-10, IL-12p70, IL-13, IL-17, IL-18, interferon (IFN)-γ, epidermal growth factor, monocyte chemoattractant protein-1, IFN-γ inducible protein 10, CXC motif chemokine ligand 1, vascular endothelial growth factor (VEGF), fractalkine, LPS-induced CXC chemokine (LIX), macrophage inflammatory protein (MIP)-1a, MIP-2, tumor necrosis factor α, and regulated upon activation, normal T-cell expressed and secreted (RANTES) levels in the culture supernatant (n = 12, respectively) were measured using a Milliplex MAP Rat Cytokine/chemokine Magnetic Bead Panel (Merck KGaA, Darmstadt, Germany). Glucagon levels were measured using a Milliplex MAP Rat Metabolic Hormone Magnetic Bead Panel (Merck KGaA). Hepatocyte growth factor (HGF) levels were quantified using a Quantikine ELISA Mouse/Rat HGF Immunoassay (R&D Systems, Minneapolis, MN, USA). Insulin levels were quantified using a rat Insulin ELISA kit (Mercodia AB, Uppsala, Sweden).
Inhibition of candidate factors in co-culture supernatants
In addition, experiments were conducted to verify the additive effects of the culture supernatant on hepatocyte function, followed by inhibition experiments to identify key humoral factors. Supernatants were collected after 3 days of culture and another hepatocytes (2.0 × 105) were seeded on a type l collagen-coated 24-well plate (Corning, Inc.) in 1.0 ml of the following media: basal medium (WE group, n = 8), hepatocyte supernatant (Hep sup group, n = 6), and the culture supernatant obtained from the co-culture of hepatocytes with ADSCs and islets (Mix sup group, n = 8). Thereafter, the hepatocytes were incubated for 3 days, and the ammonia metabolic assay was conducted. In the inhibition experiments, the co-cultured supernatant of the Mix group was separately combined with anti-VEGF antibody (anti-VEGF group, n = 6) (1.0 μg/ml) (R&D Systems), anti-HGF antibody (anti-HGF group, n = 6) (1.0 μg/ml) (R&D Systems), anti-LIX antibody (anti-LIX group, n = 6) (1.25 μg/ml) (R&D Systems), and anti-insulin receptor (InsR) antibody (anti-InsR group, n = 12) (2.15 µg/ml) (Bioss Inc.), respectively32,40–42. Following a 30-min reaction of the culture supernatant with specific antibodies, hepatocytes were mixed with each culture supernatant, cultured under similar conditions, and the assay was performed. Finally, the same amount of irrelevant IgG (IgG group, n = 6) (2.15 µg/ml) (R&D Systems) as the anti-insulin receptor antibody was used in the test.
Statistical analyses
All data are presented as mean ± standard error. All statistical analyses were performed using the JMP Pro 17 software program (SAS Institute Inc., Cary, NC, USA). A mixed-model repeated-measures analysis was used to analyze the serum albumin level trends in each group. For a comparison of the ammonia metabolic rate and serum cytokine levels, a one-way analysis of variance was conducted, followed by the Tukey-Kramer test for a post-hoc comparison. P was set at <0.05.
Results
Characterization of ADSCs
The cells isolated from the adipose tissues of F344/NSlc rats displayed CD antigens characteristic of ADSCs 43 . Flow cytometry confirmed positive expression of CD29, CD44, CD73, CD90, and CD105. However, CD31 (endothelial marker), CD34 (hematopoietic stem and progenitor cells marker), and CD45 (hematopoietic markers) were not expressed (Fig. 1).

Characterization of ADSCs. Flow cytometry showed strong expression of CD29 (99.1%), CD44 (98.2%), CD90 (97.4%), and CD105 (98.7%), and moderate expression of CD73 (54.4%). In contrast, markers associated with endothelial cells (CD31) and hematopoietic lineage cells (CD34 and CD45) were not detected.
Effects of HTx at the liver surface: Impact of co-transplantation with ADSCs and pancreatic islets on hepatocyte engraftment
Engraftment of the transplanted hepatocytes was evaluated by measuring the serum albumin levels of the recipient rats. Although no increase in serum albumin levels was observed in the Hep and Hep-ADSC groups, a significant increase in serum albumin levels was observed when islets were co-transplanted (P < 0.01). Furthermore, a marked increase in serum albumin levels was observed when hepatocytes, islets, and ADSCs were seeded on GHNF and implanted on the liver surface (P < 0.01) (Fig. 2).

The impact of co-transplantation with ADSCs and pancreatic islets on hepatocyte engraftment when transplanted onto the liver surface using GHNF. There was no increase in albumin levels in the Hep (n = 19) or Hep-ADSC (n = 5) groups. Serum albumin levels were significantly higher in the Hep-Islet group (n = 11) than in the Hep group (**P < 0.01). In addition, serum albumin levels in the Mix group (n = 7) were markedly higher than those in the Hep group (**P < 0.01). Hep group: transplantation of hepatocytes alone, Hep-ADSC group: hepatocyte co-transplantation with ADSCs, Hep-Islet group: hepatocyte co-transplantation with pancreatic islets, Mix group: hepatocyte co-transplantation with ADSCs and islets.
A comparison of hepatocyte engraftment between optimized liver surface transplantation and intraportal transplantation
Two sheets of GHNF seeded with a suspension of hepatocytes, ADSCs, and islets were transplanted onto the liver surface (optimized liver surface transplantation), and serum albumin levels were compared with those in the intraportal transplantation group. No significant difference was observed between the two groups during the observation period (P = 0.35) (Fig. 3), demonstrating that HTx at the liver surface showed a transplant outcome comparable to that of intraportal transplantation. Given that this is the result of using 10 million hepatocytes, which is empirically considered the upper limit of intraportal transplantation 14 , the results of optimized liver surface transplantation may surpass those of intraportal transplantation if additional GHNF sheets are prepared.
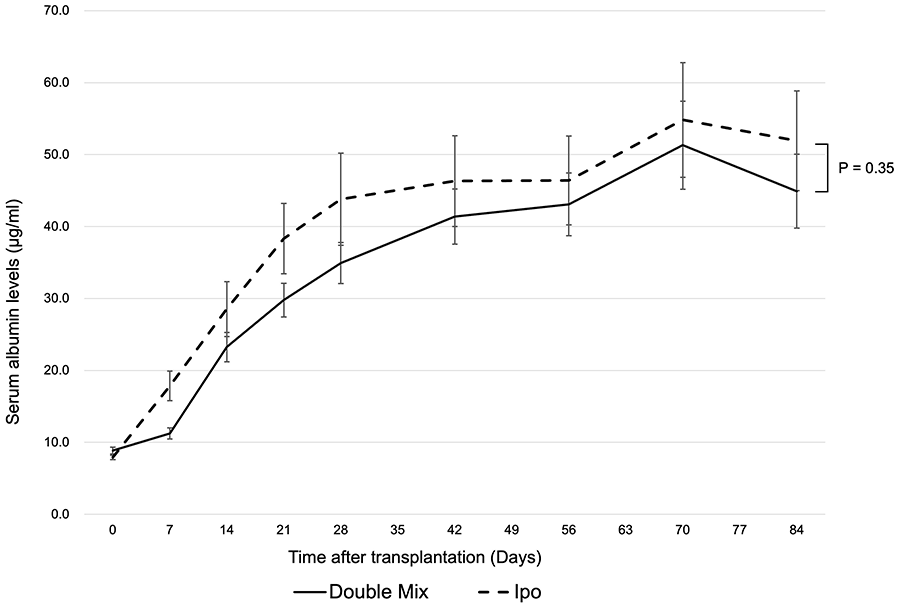
A comparison of hepatocyte engraftment between optimized liver surface transplantation and intraportal transplantation. A comparison of serum albumin levels between the group in which hepatocytes mixed with ADSCs and islets were seeded in two sheets of GHNF and transplanted onto the liver surface, and the group in which hepatocytes alone were transplanted through the portal vein. The total number of transplanted hepatocytes (10 million) in both groups was equal. There was no significant difference between the groups (P = 0.35). Double Mix group: hepatocyte co-transplantation with ADSCs and islets using two GHNF sheets, Ipo group: intraportal hepatocyte transplantation.
Immunohistochemical staining of hepatocytes, ADSCs, and islets transplanted onto the liver surface
Few albumin-positive cells were observed in the Hep and Hep-ADSC groups. In contrast, hepatocytes were observed on the liver surface in the Hep-Islet and Mix groups, in which islets were co-transplanted. In the Mix group, as we have previously observed 29 , the transplant site was covered with adipose tissue, suggesting that a portion of ADSCs differentiated into adipose tissue (Fig. 4a–c). Albumin staining confirmed that some transplanted hepatocytes were engrafted (Fig. 4d). The transplanted islets remained on the liver surface after insulin staining (Fig. 4e). Albumin-positive cells and insulin-positive cells were not always in close proximity when the observation was performed. It is suggested that islets have the potential to penetrate the organ parenchyma when transplanted under the renal capsule or on the liver surface, as observed in previous studies26,32. However, in this study, islets did not burrow into the liver parenchyma (Fig. 4f). PAS staining confirmed that the transplanted hepatocytes had glycogen storage capacity (Fig. 4g). These data suggest that mixing hepatocytes and islets can make the liver surface an effective site for HTx.

Macroscopic and microscopic findings at the transplant site in the Mix group. A, Macroscopic findings at the transplant site. The white area was thought to be due to fatty degeneration caused by the transplanted ADSCs. B and C, HE staining at the transplant site. Viable cells were concentrated in the lowermost layer of the GHNF, and the replaced adipocytes covered them. D, Albumin staining at the transplantation site. Albumin–positive hepatocytes engrafted on the liver surface are shown with red arrows. E, Insulin staining at the transplant site. Insulin-positive islets engrafted on the liver surface are indicated by yellow arrows. F, EM staining at the transplant site. The transplanted cells remained on the liver capsule, indicated by white arrows, and did not penetrate the liver parenchyma. G, PAS staining at the transplant site. The transplanted hepatocytes were positive for PAS staining. Magnification: (B) 100×, (C)–(F) 200×. Scale bar: 200 μm (all photomicrographs).
Identification of key factors promoting the hepatocyte function in the mix group using an in vitro model
The ammonia metabolic rate was significantly higher in the Hep-ADSC (52.4 ± 4.6%), Hep-Islet (64.4 ± 3.5%), and Mix groups (65.9 ± 4.1%) than in the hepatocyte alone group (32.0 ± 4.3%) (Fig. 5a). Biomarkers in the co-culture supernatants were examined using a multiplex assay and ELISA. VEGF, HGF, LIX, and insulin levels, which were significantly higher in the Mix group than in the Hep group, were identified as candidate factors for improving hepatocyte function (Fig. 5b-e). In contrast, MIP-1a, MIP-2, fractalkine, and RANTES levels, which were significantly lower in the Mix group than in the Hep group, were suggested as negative factors associated with the hepatocyte function (Fig. 5f-i).

Identification of key factors promoting hepatocyte function in the Mix group using an in vitro model. A, The Hep-ADSC, Hep-Islet, and Mix groups showed significantly higher ammonia metabolism rates than the Hep group. B, VEGF levels in the supernatant of Hep-ADSC, Hep-Islet, and Mix groups were significantly higher than those in the Hep group. C, HGF levels in the supernatant of the Hep-ADSC and Mix groups were significantly higher than in the Hep group. D, LIX levels of the supernatant in the Hep-ADSC and Mix groups were significantly higher than those in the Hep group. E, Insulin levels of the supernatant in the Hep-Islet and Mix groups were significantly higher than those in the Hep group. F, MIP-1a levels of the supernatant in the Hep-ADSC, Hep-Islet, and Mix groups were significantly lower than those in the Hep group. G, MIP-2 levels of the supernatant in the Hep-ADSC, Hep-Islet, and Mix groups were significantly lower than those in the Hep group. H, Fractalkine levels of the supernatant in the Hep-ADSC, Hep-Islet, and Mix groups were significantly lower than those in the Hep group. I, RANTES levels of the supernatant in the Hep-ADSC, Hep-Islet, and Mix groups were significantly lower than those in the Hep group. A-I, *P < 0.05, **P < 0.01.
Evaluation of the hepatocyte function by inhibiting candidate factors in the co-culture supernatants
The ammonia metabolic rate of the Mix sup group (73.0% ± 4.3%) was significantly higher than that of the Hep sup group (46.5% ± 8.2%, P < 0.05) (Fig. 6a). Therefore, the supernatant of the Mix group was considered suitable for use in the inhibition assay using neutralizing antibodies. Anti-VEGF, HGF, and LIX antibodies showed no significant inhibition, whereas anti-insulin receptor antibodies significantly inhibited the ammonia metabolic rate (46.5% ± 3.0%, P < 0.05) (Fig. 6b). The same amount of irrelevant IgG did not inhibit the ammonia metabolic rate (Fig. 6c), indicating that this inhibitory effect was specifically based on insulin.

The evaluation of the hepatocyte function by inhibiting candidate factors in the co-culture supernatant. A, The effects of various supernatants on the hepatocyte function. The ammonia metabolic rate in the Mix sup group was significantly higher than that in the basal medium (William’s medium E) and Hep sup groups (*P < 0.05). B, Effects of adding each neutralizing antibody on the hepatocyte function. Anti-VEGF, anti-HGF, and anti-LIX neutralizing antibodies showed no inhibitory effects, whereas anti-insulin receptor antibodies showed significant inhibitory effects (*P < 0.05). The hepatocyte lots for each antibody group did not necessarily match each other. C, The inhibitory effect of irrelevant IgG on the hepatocyte function could not be confirmed, indicating that the inhibitory effect of anti-insulin receptor antibodies was specific for insulin (**P < 0.01). WE group: William’s medium E was added, Hep sup group: culture supernatant of hepatocytes alone was added, Mix sup group: culture supernatant of Mix group was added, N.S.: Not significant.
Discussion
This study investigated the feasibility of HTx at the liver surface as a non-intraportal transplantation procedure to overcome the problems associated with IBMIR. Indeed, we demonstrated that hepatocytes co-transplanted with islets can be efficiently engrafted and function on the liver surface. Notably, hepatocyte engraftment was further enhanced when both islets and ADSCs were combined. The effective combination of three cells (hepatocytes, islets, and ADSCs) resulted in comparable outcomes to intraportal transplantation, the current gold standard for HTx. Furthermore, using an in vitro co-culture model, we showed that several humoral factors such as insulin may play a crucial role in promoting hepatocyte engraftment in vivo.
In this study, hepatocytes were viable on the liver surface when co-transplanted with pancreatic islets (Fig. 2). Previous studies have shown that islets preserve the hepatocyte function and delay apoptosis, subsequently improving engraftment of transplanted hepatocytes30–32,44. We also reported that ADSCs similarly improved hepatocyte engraftment in intraportal transplantation and renal subcapsular transplantation 29 . In contrast, in this study, co-transplantation with ADSCs alone did not further improve hepatocyte engraftment in the case of liver surface transplantation. Nevertheless, the combination of islets and ADSCs dramatically improved the hepatocyte engraftment. Given that ADSCs are known to have immunomodulatory and anti-inflammatory properties that are effective in improving the islet function45,46, the present findings suggest that ADSCs may contribute to promoting islets, but not hepatocytes, in liver surface transplantation. Another possible explanation for these findings is that ADSCs may boost the release of crucial factors for hepatocyte engraftment from islets.
While intraportal transplantation has been widely regarded as the standard method for HTx, it poses significant challenges due to various complications. We previously reported that intraportal transplantation for HTx is the preferred procedure because of the physiological compatibility between hepatocytes and the liver 14 . However, even in this method, most grafts are known to be lost due to IBMIR, which occurs immediately after transplantation. In addition, other complications associated with intraportal transplantation, such as portal hypertension, portal vein thrombosis, and pulmonary embolism have also been reported47–49. Therefore, the amount of transplanted grafts should be strictly limited according to portal vein pressure50,51. Considering that ADSCs are well known to easily induce thrombosis and embolism due to their adherence properties52–54, particular attention should be paid when co-transplantation of ADSCs is performed intraportally. Indeed, intraportal co-transplantation of ADSCs and hepatocytes has been associated with a high mortality rate due to portal vein embolism at certain cell doses, suggesting an apparent limitation in the number of transplanted cells in this technique 29 . Cell transplantation onto the liver surface is an attractive approach to solve these problems. In fact, the present study revealed that hepatocyte co-transplantation with islets and ADSCs at the liver surface achieved equivalent outcomes while avoiding the abovementioned several complications (Fig. 3). Furthermore, since the size of the liver surface varies among animal species, even better results are expected in larger animals, including humans. This is an important topic for future studies to explore.
This study demonstrates that an appropriate scaffold structure is essential for maintaining graft survival. Hepatocytes alone in an in vitro culture rapidly lose their morphology, cell polarity, and function as hepatocytes 55 . Thus, tissue engineering methods for constructing liver tissue in vitro have been explored 56 . Various approaches, such as using extracellular matrix components24,57,58 or decellularization techniques59–61, have been studied to improve hepatocyte survival. In addition, techniques to produce uniform hepatocyte sheets from isolated hepatocytes using temperature-responsive culture dishes have attracted attention62–65. In either technique, it is important to note that biological tissue is not a two-dimensional structure but a 3D one. Thus, when transplanting cell sheets, it is essential to stack them to create a 3D structure 66 . In this context, GHNF provides suitable scaffold for forming a simple 3D structure for transplanted grafts67,68. This structure is effective as a cell carrier for transporting hepatocyte suspensions and is particularly logical for preventing the decrease in survival rates that typically occurs with centrifugation 69 . As demonstrated in this study, hepatocytes contained in GHNF were confirmed to function well in vivo.
Interestingly, immunohistochemical staining showed that hepatocytes and islets were not always in close proximity. This finding suggests that hepatocytes are beneficially affected by fluid factors from islets rather than by cell-to-cell contact. In addition, in previous studies, we observed transplanted cells infiltrating the organ parenchyma from the site of transplantation26,32. However, in the present study, none of the hepatocytes, islets, or ADSCs transplanted onto the liver surface appeared to penetrate the liver capsule into the parenchyma (Fig. 4). In contrast, the patch grafting method developed by Lola Reid et al. has demonstrated that donor cells can not only integrate directly into the liver and pancreas but also efficiently infiltrate host organs70,71. The engraftment achieved through this method has been reported to be associated with the expression of secreted matrix metalloproteinases (MMPs) produced by stem/progenitor organoids, with the maintenance of donor cells in an immature state being a critical factor. On the other hand, the present study used mature hepatocytes, which may have exhibited weaker expression of secreted MMPs. As a result, the hepatocyte grafts may not have sufficiently degraded the liver capsule, limiting their infiltration into the organ. Despite this limitation, our study achieved hepatocyte engraftment comparable to that of the intraportal approach by a simple and rapid technique that affixes a scaffold containing cells onto the liver surface. This suggests that the outcomes of liver surface HTx may surpass those of intraportal HTx if the transplanted hepatocyte grafts can successfully penetrate the liver parenchyma. Corroborating this speculation, several attempts to improve transplantation efficiency by damaging the liver capsule have been reported72–74. Among these efforts, Kawakatsu-Hatada et al. 74 recently reported that hepatocyte engraftment improved by destroying the liver capsule using an ultrasonic homogenizer.
We found that co-transplantation of hepatocytes with islets and ADSCs promoted hepatocyte engraftment through both protective and anti-inflammatory factors. In an in vitro co-culture model, multiplex assays showed that VEGF, HGF, LIX, and insulin levels were significantly elevated in the culture supernatants of the Mix group compared to those of the Hep group, indicating that these four factors are candidates for improving hepatocyte function (Fig. 5b-e). Furthermore, inhibition assays using neutralizing antibodies against each of these factors suggest that insulin may play a crucial role in the engraftment of hepatocytes, ADSCs, and islets (Fig. 6b). In contrast, inflammatory cytokines, such as MIP-1a, MIP-2, fractalkine, and RANTES, were significantly decreased in the culture supernatants of the Mix group (Fig. 5f-i). Insulin is known to support cell metabolism, growth, and survival75,76, suggesting that it may be beneficial for hepatocyte engraftment 32 . Interestingly, although there was no significant difference in insulin concentration between the Hep-Islet and Mix groups in the in vitro experiments (Fig. 5e), the in vivo model showed a noticeably greater increase in albumin concentration in the Mix group (Fig. 2). Taken together, these findings suggest that not only graft-protective factors but also anti-inflammatory factors may contribute to improving hepatocyte engraftment in islets and ADSCs co-transplantation with hepatocytes.
Several limitations of the present study warrant mention. First, the non-albumin rats used were not disease models, such as cirrhosis, acute liver failure, or metabolic liver disease, and thus do not accurately reflect the pathophysiology of the disease. However, an important aim of using this transplantation model is to evaluate whether the liver surface can be used as a transplant site. In this regard, the model was found to be more than adequate. Notably, the serum albumin levels in the non-albumin rats used in this study were extremely low, making it possible to sensitively reflect hepatocyte engraftment. Therefore, this model is useful for detecting minor effects that are not detectable in conventional albumin-free rats, which can be assessed quantitatively using a commercial ELISA kit and histologically using albumin staining. Second, no scaffolds other than GHNF were examined in the present study. While we cannot completely rule out the possibility that GHNF itself has a supportive effect, the outcomes of this study are primarily attributed to the co-transplantation of cells, as no engraftment was observed in the Hep group. Our focus in this study was to evaluate the effectiveness of co-transplantation and optimize the hepatocyte engraftment process using this transplantation method. Third, in this study, the trypan blue exclusion assay was only used to ensure the quality of hepatocytes. Detailed evaluations of viability, apoptosis, or necrosis were not performed. In a previous experiment using an okadaic acid-induced apoptosis model, we attempted terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick end labeling (TUNEL) staining in vivo one month after transplantation, but by the time of evaluation, most transplanted hepatocytes had already undergone apoptosis, making accurate assessment difficult 39 . However, in vitro TUNEL staining or measuring the ADP/ATP ratio 39 were not performed in this study and may provide useful insights on hepatocyte viability, which should be considered in future research. Finally, this study evaluated hepatic-specific functions primarily through ammonia metabolism and PAS staining for glycogen storage. While these assays provide insights into some aspects of hepatic function, they are insufficient to fully assess hepatocyte metabolic activity or maturation. Additional functional assays, such as Phase I, II, and III metabolic testing, evaluation of bile acid transporters, and the presence of urea cycle enzymes, would offer a more comprehensive understanding. However, these were beyond the scope of this study and remain an important direction for future research.
In conclusion, the present study showed that the co-transplantation of islets and ADSCs resulted in hepatocyte engraftment on the liver surface. The key mechanisms of these favorable effects were inferred to be growth factors, especially insulin secreted from the islets, and the anti-inflammatory effects of ADSCs. Therefore, HTx at the liver surface using GHNF containing the crucial factors detected in the present study may be a novel strategy to avoid IBMIR and/or portal embolism.
Footnotes
Acknowledgements
The authors thank Kozue Maya and Megumi Goto (Division of Transplantation and Regenerative Medicine, Tohoku University) for their excellent technical assistance and Yuji Nishikawa (President’s Office, Asahikawa Medical University), Norihiko Shimizu (Animal Laboratory for Medical Research, Asahikawa Medical University), Hironobu Chiba (Animal Laboratory for Medical Research, Asahikawa Medical University), Chihiro Hino (Animal Laboratory for Medical Research, Asahikawa Medical University), Tomomi Kibuse (Animal Laboratory, Tohoku University), and Keisuke Nishio (Animal Laboratory, Tohoku University) for breeding and taking care of the rats. The authors also acknowledge the support of the Biomedical Research Core of Tohoku University, Graduate School of Medicine, and Tohoku Advanced Medical Research and Incubation Center (TAMRIC).
Abbreviations
3D three-dimensional
ADSC adipose tissue-derived stromal cell
ELISA enzyme-linked immunosorbent assay
EM Elastica-Masson
FBS fetal bovine serum
GHNF gelatin hydrogel nonwoven fabric
HE hematoxylin-eosin
HGF hepatocyte growth factor
HTx hepatocyte transplantation
IBMIR instant blood-mediated inflammatory reaction
IEQ islet equivalent
IFN interferon
IL interleukin
InsR insulin receptor
LIX LPS-induced CXC chemokine
LTx liver transplantation
MIP macrophage inflammatory protein
MMP matrix metalloproteinase
MSC mesenchymal stromal cell
PAS periodic acid-Schiff
RANTES regulated upon activation, normal T-cell expressed and secreted
TUNEL terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick end labeling
VEGF vascular endothelial growth factor
