Abstract
Although notable progress has been made, restoring motor function from the brain to the muscles continues to be a substantial clinical challenge in motor neuron diseases/disorders such as spinal cord injury (SCI). While cell transplantation has been widely explored as a potential therapeutic method for reconstructing functional motor pathways, there remains considerable opportunity for enhancing its therapeutic effectiveness. We reviewed studies on motor pathway regeneration to identify molecular and ultrastructural cues that could enhance the efficacy of cell transplantation. While the glial scar is often cited as an intractable barrier to axon regeneration, this mainly applies to axons trying to penetrate its “core” to reach the opposite side. However, the glial scar exhibits a “duality,” with an anti-regenerative core and a pro-regenerative “surface.” This surface permissiveness is attributed to pro-regenerative molecules, such as laminin in the basement membrane (BM). Transplanting donor cells onto the BM, which forms plastically after injury, may significantly enhance the efficacy of cell transplantation. Specifically, forming detour pathways between transplanted cells and endogenous propriospinal neurons on the pro-regenerative BM may efficiently bypass the intractable scar core and promote motor pathway regeneration. We believe harnessing the tissue’s innate repair capacity is crucial, and targeting post-injury plasticity in astrocytes and Schwann cells, especially those associated with the BM that has predominantly been overlooked in the field of SCI research, can advance motor system restoration to a new stage. A shift in cell delivery routes—from the traditional intra-parenchymal (InP) route to the transplantation of donor cells onto the pro-regenerative BM via the extra-parenchymal (ExP) route—may signify a transformative step forward in neuro-regeneration research. Practically, however, the complementary use of both InP and ExP methods may offer the most substantial benefit for restoring motor pathways. We aim for this review to deepen the understanding of cell transplantation and provide a framework for evaluating the efficacy of this therapeutic modality in comparison to others.
(A) Four components of the glial scar: the core of the glial scar (cGS), the basement membrane (BM), the layer of scar-forming astrocytes (SFA), and the outermost layer (OML). Glia limitans perilaesonis (GLP) corresponds to the basal portion of SFA (bSFA) and the BM. X indicates the area of normal tissue (NrT). (B) A sprout (Spr) from a severed neuron in the corticospinal tract (CST, asterisk; severed point) regenerates on the pro-regenerative BM (blue arrow). (C) A host-derived sprout (Spr, blue) forms a synapse with a neuron derived from the transplanted motor neuron progenitors (MNp, green). This MNp neuron (green) then establishes a synapse with a host propriospinal neuron (PSN, blue), which, in turn, connects to the lower motor neuron pool (LMNP). This is an example of discomplete spinal cord injury (refer to the text).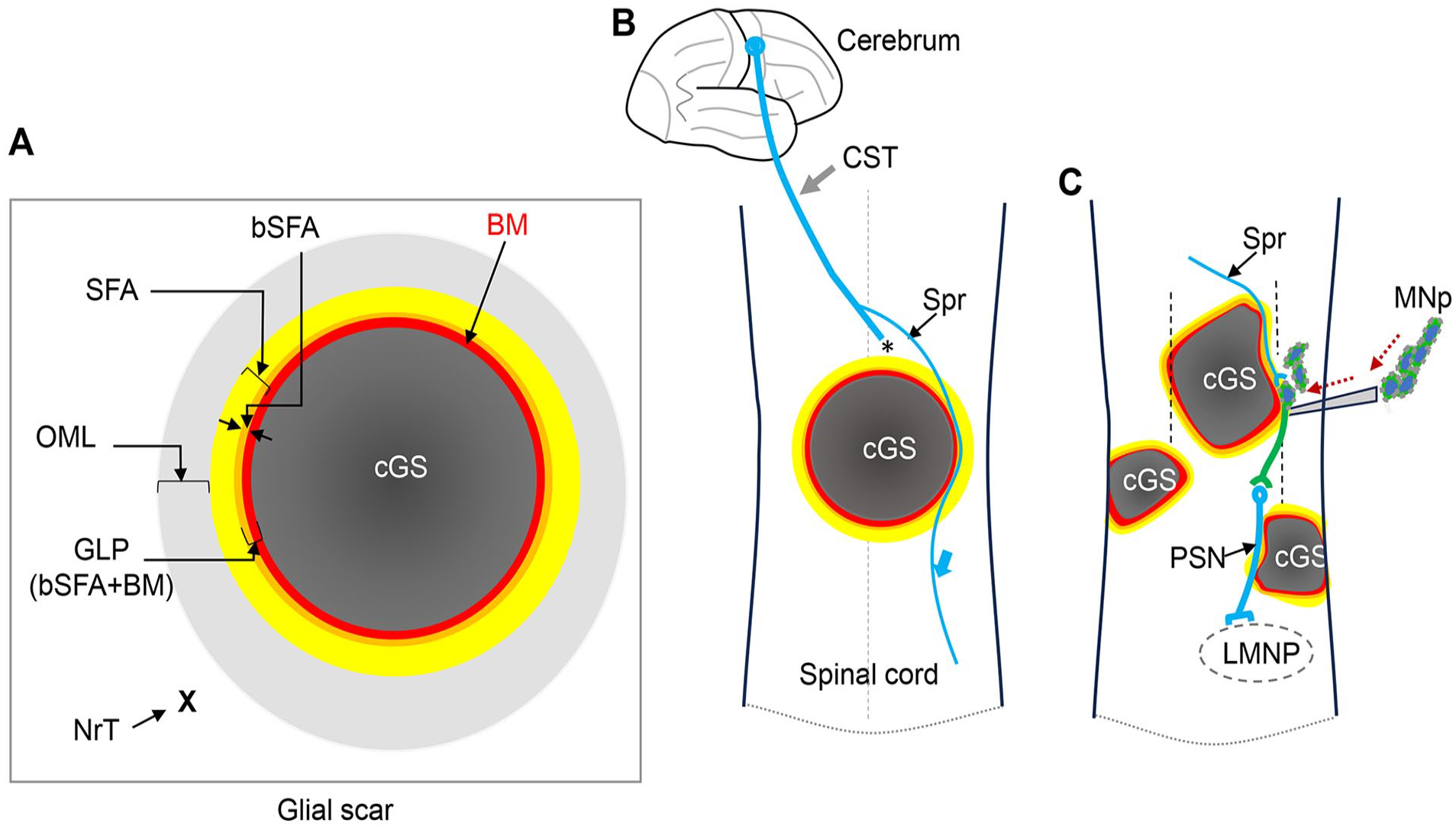
Introduction
The corticospinal tract (CST) consists of upper motor neurons (UMNs) and lower motor neurons (LMNs) and either or both of them may be damaged by a plethora of insults, resulting in motor neuron diseases/disorders (MNDs;

The Motor Pathway.
Cell transplantation has been extensively studied as a promising therapeutic approach for rebuilding functional motor systems but there is still significant room for improvement to optimize its therapeutic efficacy1–3,5–12.
Our review of previous studies revealed that the glial scar exhibits a “duality,” comprising distinct components: an anti-regenerative core and a pro-regenerative surface. Some studies explicitly indicate that the surface permissiveness of the glial scar is attributed to pro-regenerative molecules, such as laminin, expressed in the basement membrane (BM) encasing the glial scar core. Others, while not explicitly stating this, can be interpreted as supporting the notion, as it offers a plausible explanation for phenomena observed at the glial scar surface13–20.
Opinions on the permissiveness of the glial scar for axon regeneration have been in conflict since the 1970s21,22. Introducing the concept of “duality,” based on ultrastructural and molecular data accumulated over the past 50 years, may help resolve the ongoing debate regarding the role of the glial scar. Until now, the concept of the BM has largely been overlooked in the field of spinal cord injury (SCI) research but it allows us to assess more accurately the roles of various molecules that appear in the SCI lesion epicenter and its surrounding areas in terms of axon regeneration.
Current studies present conflicting results on whether removing components of the glial scar in SCI is beneficial for regeneration20,23–30. These contradictory findings may arise at least in part from various factors, such as differences between in vitro and in vivo experimental settings and the ablation techniques employed. However, a more critical issue lies in the uncertainty pertinent to the extent of glial scar ablation, specifically regarding which components of the glial scar were removed, which remains unclear due to a lack of ultrastructural evidence. When an ablation procedure removes both the BM and the astrocytes adjacent to the glial scar core, the exposure of active fibrotic tissue and cell types, including macrophages, within the lesion core may impede axon regeneration 22 . Conversely, if the BM is preserved while the surrounding astrocytes are removed, the BM’s pro-regenerative property may be retained. Immunostaining has been widely utilized as a research tool in studies on glial scar formation and CST functional regeneration. However, most results obtained through this research method have provided information at the light microscopy level. In this review, we emphasize that this alone is insufficient, and we highlight the importance of simultaneously incorporating the ultrastructural data, including those related to the BM, obtained through electron microscopy.
Notably, the concept of “duality” presented here fundamentally differs from the conventional usage of the term “dual” which has been commonly employed in previous literature22,31,32. Prior studies have used the term “dual” to describe temporal changes in injured astrocytes, which sometimes contribute to neuro-degeneration but at other times exhibit regenerative properties22,31-33. However, in this paper, we focus on the “duality” of one critical chronic sequela in neuro-degeneration: the glial scar itself, viewed as an end phenomenon that encompasses both pro-regenerative and anti-regenerative characteristics.
While both donor cell selection and delivery methods are crucial for the successful restoration of motor pathways, the latter has received disproportionately less attention1–3,5–8. We critically evaluated the traditional intra-parenchymal (InP) cell injection into the host nervous tissue, a widely used method for donor cell delivery3,5–8,34,35 and concluded that a paradigm shift from the traditional InP route to the extra-parenchymal (ExP) route, in which donor cells are transplanted onto the pro-regenerative BM, could represent a significant and unprecedented advancement in neuro-regeneration research. Specifically, the BM-targeting ExP cell delivery, recruiting propriospinal neurons (PSNs), may facilitate seamless and unified regeneration of motor pathways by bypassing the glial scar core.
We aim for this review to deepen understanding of cell transplantation in the context of the glial scar, a “double-edged sword” in neuro-regeneration, which at first glance may appear anti-regenerative but could potentially be pro-regenerative if harnessed effectively, and to establish a framework for assessing its therapeutic potential alongside other interventions for motor pathway restoration.
While we did not employ a meta-analytic approach, we performed a comprehensive and impartial selection of relevant literature, prioritizing studies that substantially enhance understanding of the topic and ensuring balanced representation of the field. We scrutinized the literature, starting with the personal library of the first author, which comprises approximately 33,000 articles. We further collected relevant papers extensively using generative artificial intelligence tools, as well as other platforms. PDFs of all cited papers were downloaded and reviewed.
The CST
The UMNs and LMNs comprise the CST 4 (Box 1). Motor pathway damage resulting from UMN injuries can occur when the CST within the brain is affected, as well as when the CST within the spinal cord is impaired. However, the majority of previous studies have predominantly focused on restoring the lost function of the CST within the spinal cord1,3,5–10,35. This focus is largely attributable to the profound impact of sensorimotor paralysis caused by SCI, particularly traumatic SCI, on patients’ daily lives1,3,5–10,35. Accordingly, this review primarily centers on motor neuron (MN) regeneration in the context of SCI, while also addressing MN damage due to brain injury where relevant. Such an approach is justified because the findings from studies on MN regeneration in the context of SCI may be often applicable to motor pathway regeneration following brain damage.
Methods for Motor Pathway Restoration Other Than Cell Transplantation
Various methods, beyond cell transplantation, have been explored to restore lost motor function resulting from central nervous system (CNS) and peripheral nervous system (PNS) damage. These methods such as listed below are investigated both as standalone treatments and in combination1–3,5,7–10,34–38. In the following sections, they are reassessed particularly in relation to cell transplantation.
Cell-intrinsic manipulation
Multiple techniques and numerous biological factors and pharmacological agents have been used to manipulate cell-intrinsic properties and other molecular mechanisms to enhance the regeneration of motor axons and other axons39,40. Molecular genetic interventions are among the techniques used to treat neurodegenerative diseases41,42. However, this approach may not currently be feasible for addressing complex conditions 43 , including SCI. The results of experimental manipulation of cell-intrinsic pathways vary. A representative example to manipulate a cell-intrinsic pathway is deletion of PTEN (phosphatase and tensin homolog), an inhibitor of the mTOR (mammalian target of rapamycin) pathway for cell growth and metabolism44,45; PTEN deletion upregulates mTOR pathways and can enhance axon regeneration in damaged nervous tissue, including the optic nerve and CST neurons46–48. However, genetic deletion of three key components of the myelin inhibitory pathway, Nogo, myelin-associated glycoprotein (MAG), and common receptors NgR1 (Nogo receptor 1), was insufficient to promote spinal serotonergic axon regeneration in mice with complete lesion of the spinal cord 49 .
From the perspective of cell transplantation, it is particularly intriguing that these studies, which involved manipulations of cell-intrinsic mechanisms, consistently demonstrated that regenerating axons crossing the lesion were surrounded by a scaffold of glial fibrillary acidic protein (GFAP)–positive astrocyte processes or bridges47,49. On the other hand, other studies revealed that the same phenomenon—axons regenerating in close association with GFAP-positive astrocyte processes or bridges—spontaneously occurred post-injury without any manipulation of cell-intrinsic factors or mechanisms 50 . Embryologically, neuroblasts migrate within a longitudinally oriented tube-like structure, ensheathed by GFAP-positive astrocytes, the glial tube, in the rostral migratory stream 51 . Collectively, these observations suggest that GFAP-positive astrocyte processes are crucial not only for development but also for the post-injury reorganization of neuronal networks. Furthermore, the interactions between astrocytes and regenerating axons may represent a common and fundamental mechanism underlying axon regeneration.
Neural circuit manipulation
Modulation of neural circuitry is a promising tool to facilitate locomotion in SCI patients21,52. Externally controlled sustained electrical stimulation enables the spinal cord, isolated from the brain, to generate locomotor activity via the lumbosacral central pattern generator 53 . Neuromodulation with electrical stimulation of spinal cord such as epidural spinal cord stimulation is an effective and beneficial treatment for chronic SCI, particularly when combined with intensive physical rehabilitation54–56. Discomplete spinal cord injury (dSCI) is clinically complete but accompanied by residual trans-lesional muscle–brain connectivity evidenced with various neurophysiological examinations, including electromyography and evoked potentials57,58. The prevalence of dSCI has been reported in approximately 20–50% of all SCI patients57,59,60. In these patients, electrically excitable neurons may remain across the injury site, though they are insufficient to elicit motor function below the lesion. Electrical stimulation can enhance spinal cord excitability, potentially inducing functional motor activity below the injury site 52 . Electrical neuromodulation not only facilitates the recovery of functional motor activity but also shows promise as an adjunct therapy when combined with cell transplantation 61 , potentially by increasing the number of excitable neurons 62 . In the case of SCI shown in Graphical abstract, C, three distinct glial scars are scattered throughout the spinal cord. Given motor pathways run parallel to the axis of the spinal cord, the combination of these three glial scars would effectively result in a clinically complete SCI (vertical dotted lines). However, viable tissue remains between these glial scars. In other words, this SCI case is morphologically incomplete. Hence, from the perspective of cell transplantation, it may be possible to reconstruct a regenerating neural pathway by effectively utilizing the spaces between these glial scars, potentially leading to the restoration of motor function (Graphical abstract, C). Consequently, patients with dSCI represent an important target population for therapeutic intervention using cell transplantation.
Neuronal relay formation with endogenous neurons
In humans, spontaneous recovery of motor function has been observed in Brown-Sequard syndrome, a spinal cord hemisection syndrome63,64. In the recovery process, PSNs may play a central role as relay neurons to connect between the sprouts from upper neurons and LMN pools65–71. In an experimental study to facilitate the growth of intrinsic PSNs, a combined and intricate approach was taken via a temporally controlled and spatially targeted delivery method 72 . Two weeks before spinal cord transection in rats, adeno-associated viral vectors expressing osteopontin, insulin-like growth factor (IGF)-1, and ciliary-derived neurotrophic factor were injected into the spinal cord, rostral to planned locations of the SCI lesions. Two days after the SCI, hydrogel containing fibroblast growth factor 2 and epidermal growth factor (was injected into the SCI lesion core to enhance axon growth. One week later, hydrogel containing glial-derived growth factor (GDNF) was injected caudal to the SCI lesion to attract PSNs distally. PSNs are known to express GDNF receptors. As a result, intrinsic PSNs regrew robustly across anatomically complete SCI lesions, 2 mm past the lesion center, with a significant electrophysiological conduction capacity 72 .
Degenerated neuron replacement with endogenous neurons
An approach to mobilize endogenous cells to the lesion site has been investigated to replace degenerated neurons in various diseases, including stroke, neurodegenerative disease, and inflammatory disease73–75. In response to the injury, neural stem cells in the subventricular zone (SVZ) and subgranular zone neurogenic niches are activated to undergo proliferation. They then differentiate into neuroblasts, which migrate toward the injured area, deviating from their default migration routes as observed in SVZ-derived neuroblasts in stroke 74 . The neuroblasts further differentiate into neurons and potentially contribute to the improvement of functional outcomes 76 . Currently, however, this method has not been fully proved to be clinically beneficial in restoring lost neural function clinically. Further investigation is required to address the discrepancy between the subtypes of neurons required for repair and those that are naturally generated within the CNS 77 . The number of newborn neurons is too small for recovery of neurological functions and cell proliferation is relatively transient 77 . In humans, especially in adult brains, endogenous neurogenesis is limited compared to non-human primates and rodents78,79. Hence, caution is warranted when extrapolating results obtained from non-human primate and rodent models in translational studies78,79.
Another idea is to replace degenerated neurons with endogenous cells in reverse-directed reprogramming to produce neurons from mature astrocytes 80 . This idea is theoretically supported by evidence arising from the process of mammalian forebrain development, in which radial glial cells generate both neurons and astrocytes 81 . Hence, it was expected that even postnatal adult astrocytes continue to retain latent radial glial cell characteristics and hence adult astrocytes could be instructed to generate neurons by re-expressing neurogenic transcription factors that were expressed in developmental stages 82 . Indeed, postnatal astrocytes adopt a neuronal fate when PAX6 is re-expressed in vitro 80 . In recent in vivo studies, viral vectors containing various transcription factors successfully convert astrocytes to neurons in the CNS 82 . This method may become an attractive clinical approach with further refinements because cells with astrocyte lineages are abundant in and around degenerated areas of the CNS. Nevertheless, several problems remain to be tackled82–84. One of them is to produce suitable neuronal subtypes specific to particular regions within the CNS82-84. Currently, the formation of a glial scar in response to injury is unpredictable in terms of its spatial distribution in the CNS. For instance, mechanical damage to the spinal cord does not exclusively trigger localized responses at the initial site of injury, but rather induces diffuse effects throughout the spinal cord 85 . The long-term survival and stability of sufficient numbers of induced neurons remains a challenge. It is also necessary to generate safe and effective vectors suitable for human use. Another significant concern with this approach is that converting astrocyte-lineage cells into neurons may diminish the pool of reactive astrocytes, which, as discussed above and further elaborated below, are essential for axon regeneration and elongation13–18,20,86,87. One should be cautious about mechanical injection of transcription factor–loaded viral vectors into the already compromised, delicate, and vulnerable neural tissue within the parenchyma82, 88 (see below).
Cell Transplantation of Exogenous Cells via the Traditional InP Route
Replacement or replenishment of degenerated or lost neurons can be achieved through the transplantation of exogenous cells via the traditional InP route, the most commonly used method for introducing these cells into host nervous tissues1,5,10,34,36,61. These efforts are focused on the ultimate goal of bridging the neural gap caused by SCI or enabling axons to traverse the glial scar7,21,36,61,89,90.
To penetrate the glial scar core in SCI, the potential of olfactory ensheathing cells (OECs) has been investigated91–95. During development, OECs interact with the astrocytic coverings of the olfactory glomeruli, which house the mitral cell dendrites of the CNS. Through these interactions, OECs appear to create “doors” in the astrocytic covering, facilitating the entry of olfactory axons into the glomerulus 87 . This developmental mechanism, in which OECs support axonal elongation by enabling newly formed axons to extend into the CNS, has inspired their proposed application in SCI repair91,93. However, the feasibility of translating this mechanism to SCI repair remains uncertain.
The potential of glia-restricted progenitors (GRPs) has also been investigated96–98. Astrocytic processes, particularly those of reactive astrocytes, can serve as a scaffold for axon elongation during neural regeneration, as described in previous studies16,47,49,50,99–102. GRPs give rise to both astrocytes and oligodendrocytes 96 . Hence, transplanting GRPs into the lesion site of an SCI may allow GRP-derived astrocytes to perform similar functions as astrocytes, potentially providing a scaffold for host neurons to penetrate the glial scar core 96 . In addition, GRP transplants may provide a supportive environment following SCI thorough reduction of glial scar formation, thus aiding axon growth into the lesion96–98. As with OECs, however, the regenerative potential of GRPs is currently unclear.
Transplantation of neural progenitor cells (NPCs) via the InP delivery route has been more successful1,5,7,10,34,36,61,103. In an experimental study on SCI, NPCs were grafted into the lesion site, and the neurons within the graft successfully received regenerated axons from the host CST 104 . An average of 9.7% of all host corticospinal axons quantified at 0.5 mm proximal to the lesion/graft site penetrated the graft and these axons crossed the distal host–graft interface 104 . In another study in hemisected monkey spinal cord, host neurons extended axons into the graft of human NPCs with a partial restoration of motor function. Host corticospinal axons penetrated up to 0.5 mm into the grafts 105 .
As implied by the studies mentioned above, the inhibition of axon regeneration at the interfaces between donor cells and host nervous tissue is a critical challenge in exogenous cell transplantation methods 106 . To address the non-permissiveness at the host–graft interface, transplantation of Schwann cells (SCs) has been proposed as a potential solution 106 . In experiments involving complete spinal cord transection, SCs were transplanted into the gap created by the lesion, forming an SC bridge. The transplanted SCs then migrated into the adjacent gliotic host tissue. Subsequently, host axons, scaffolded and guided by the processes of reactive astrocytes and SCs, crossed the interface between the SC bridge and the spinal cord 107 . Specifically, the combinatorial use of SCs and other cell types warrants further investigation to enhance axon regeneration following SCI 108 .
In reconstructing the human CST, the regenerated axons must span distances on the order of centimeters rather than millimeters. This point is crucial when translating results from small animal experiments to human therapies because the axonal growth achieved to date falls far short of centimeter scale.
Cell Transplantation of Donor Cells Onto the BM: An ExP Approach
We examined earlier research on motor pathway regeneration to uncover molecular and ultrastructural determinants that might improve the effectiveness of cell transplantation therapies. As a result, we concluded that focusing on the BM of the glial scar could lead to significant advancements.
The Glial Scar: A Barrier or Not for Axon Regeneration?
The glial scar is frequently cited as a significant barrier to axon regeneration, especially when considered as a whole21,89,90. Its presence indeed acts as a robust inhibitor to axon regeneration, particularly when axons attempt to penetrate through the scar’s “core” to reach the opposite side7,36,61,89,90. However, one of the key points emphasized in this review is the importance of distinguishing between the “core” and the “surface” of the glial scar (Graphical abstract, Fig. 1). The relationship between the glial scar, reactive astrocytes, and axonal regeneration is complex and not yet fully understood. However, numerous in vitro and in vivo studies suggest that the surfaces of the glial scar and scar-derived reactive astrocytes can exhibit permissive properties for axonal regeneration13–20,86,101,109–111. The surface permissiveness of the glial scar can be interpreted as primarily derived from the presence of the pro-regenerative BM of the glia limitans perilaesonis (GLP) as elaborated in the following sections.

Temporal changes of peri-lesional astrocytes in spinal cord injury and ultrastructural anatomy of the glial scar. Panels (1)–(6) illustrate the sequential changes of astrocytes around the lesion in spinal cord injury. Approximately, 1–4 days post-injury (PI), the processes of astrocytes (As) around the lesion center (LC) are gradually orienting toward the LC and continue to proliferate as shown in (1, normal state) to (4). After 2–4 weeks PI, the proliferation stage is complete and the processes of astrocytes align in parallel, while their processes overlapped with each other, forming a mature glial scar forms, (5) and (6). Magenta circle and arrow in panel (5) show that proteins such as laminin and collagen IV are produced to face the core of the glial scar (cGS) by the scar-forming astrocytes (SFA). This process finally leads to the formation of the BM surrounding the cGS, red circle and arrow in (6) and (6′). Thin arrow in (6) indicated the epi-BM compartment (the area highlighted in green) that may serve as a conduit for axon regeneration on the pro-regenerative BM. Panel (6′) shows the ultrastructural anatomy of the glial scar. The glial scar formed in SCI consists of four components: the core of the glial scar (cGS), the basement membrane (BM), the layer of scar-forming astrocytes (SFA), and the outermost layer (OML). Glia limitans perilaesonis (GLP) corresponds to the basal portion of SFA (bSFA) and the BM. NrT with x indicates normal tissue. Note that the panels in this figure are based on the reports described in the text; however, some elements, including the shapes of astrocytes, are theoretical extrapolations. The time frames indicated at the top of certain panels are approximate, as discrepancies inevitably arise due to variations in species, experimental conditions, and observational methods across studies. The epi-BM compartment (6) is not shown in panel (6′) to enhance the clear understanding of the layers. Panels (6) and (6′) depict mature glial scars at corresponding PI stages (double-headed dotted arrow). The right-directed arrow in panel (6′) indicates components associated with non-scar tissue. The thickness of each layer is not depicted to scale, with some portions intentionally exaggerated for clarity and ease of interpretation.
Many in vitro studies reveal the pro-regenerative properties of the glial scar surface. For example, rat hippocampal neurons were seeded onto both glial scar explants and neonatal rat cerebral cortex. Although neurite extensions were less pronounced compared to those on neonatal tissue, they were still observed on the glial scar without being entirely inhibited 111 . Another study replicated similar in vivo phenomena, demonstrating that the upper surfaces of reactive astrocytes, grown as a monolayer on beta-amyloid—a potent inducer of reactive astrogliosis—are pro-regenerative and can support neuronal outgrowth 86 . Dorsal root ganglion cells readily extended their axons on the surface of astrocytes subjected to mechanical strain, but not on unconditioned astrocytes; the mechanically strained astrocytes (reactive astrocytes) exhibited increased permissiveness 110 . Additionally, in an in vitro model by co-culturing astrocytes and meningeal cells to mimic the astrocyte–meningeal cell interface observed after CNS injuries, a glia limitans formed between aggregates of two co-cultured cell types: astrocytes and meningeal cells, similarly as the GLP formed in SCI (see above). When dorsal root ganglion cells were plated onto this mixed monolayer, neurites preferentially extended on the glia limitans structure that had already developed 112 .
In vivo studies have demonstrated that the surface of the glial scar, specifically the BM, is permissive to and guides regenerating axons, allowing them to circumvent the glial scar core (Fig. 2). In the studies where avulsed roots were experimentally re-implanted into the host spinal cord to address motor function loss due to brachial plexus avulsion, axons grew from residual ventral horn cells toward the re-implanted roots. Remarkably, direct implantation of the ventral root into the spinal cord was not essential, as axons from superficially implanted grafts successfully regenerated along the cord’s surface, indicating that the gliotic spinal cord surface is a regeneration-promoting, preferential site for axon regeneration15,16. In another in vivo study of SCI, dystrophic axons were rarely found at the outermost edges of the lesion and regenerating axons were seen to circle around on the glial scar, emerging on the surface of the opposite side 50 . During neural development, the BM plays a crucial role in guiding axonal growth cones by providing both physical and chemical cues113,114.

Glia limitans perilaesonis in spinal cord injury and basement membrane—targeting cell transplantation. (A) Two sprouts (Spr 1, 2) from a severed neuron in the CST (asterisk) are shown, one sprout (Spr1) entering the plane of the basement membrane (BM) at a 90° angle collides with the surface of the BM and becomes to be dystrophic (blue dot on the BM). The second sprout (Spr2) entering at an oblique angle can readily take a detour course on the surface of the pro-regenerative BM (see B). (B) A region of the core of the glial scar (cGS) adjacent to the spinal cord surface may serve as a portal for donor cells (indicated by bracket) such as motor neuron progenitor (MNp) into the host spinal cord. This process depends on chemoattractive influence of laminin from the BM (four red dotted lines with leftward arrowheads). In addition, laminin–integrin interactions on the BM (upper enlarged panel) facilitate axon elongation. A sprout from the host CST (Spr 3) travels along the dorsal surface of the glial scar before emerging on the ventral surface (upper horizontal hollow arrow). This host-derived sprout then forms a synapse with a neuron derived from the transplanted motor progenitors (lower horizontal thick arrow), which subsequently establishes a synapse with a host propriospinal neuron (PSN, thick oblique arrow). Finally, this pathway connects to the lower motor neuron pool (LMNP). Blue, host-derived and green, donor cell-derived neurons, respectively. (C) When the BM of the glial scar develops deep in the spinal cord (double-headed arrows), an alternative method, “Intra-parenchymal (InP) microinjection,” would be feasible. After inserting an infusion device (thin black arrow), MNp are delivered on the BM (red dotted line) so that the cells are placed within the range of chemoattractive influence of laminin in the BM. A single puncture of the spinal cord may suffice, as the chemoattractive and soluble properties of laminin could facilitate the dispersion of donor cells from the initial transplantation site toward the periphery (two thick arrows). Some layers have been enlarged for clarity and ease of understanding.
The glial scar: Ultrastructural anatomy and associated cellular and non-cellular components
The glial scar comprises four major structural components revealed by transmission electron microscopy (TEM): the core of the glial scar, the BM, layers of scar-forming reactive astrocytes (SFAs) 115 , and the outermost layer of reactive astrocytes116–120. The innermost core consists of non-neural stromal cells and fibrotic extracellular matrix, collectively constituting the “true” scar 121 . Adjacent to this core is the BM, which delineates the glial scar surface122,123 (Graphical abstract, Fig. 1). The BM appears concurrently with the emergence of SFAs, approximately 2–3 weeks post-SCI, although precise temporal dynamics may vary depending on the injury model122–124 (Fig. 1, refer to Table 1 below). The BM’s protein components, such as laminin and collagen IV, are predominantly secreted by SFAs adjacent to the BM following SCI115,116,120,125 (Fig. 1(5), (6), (6′)). The outermost layer comprises non-proliferating astrocytes, which maintain their structural integrity and contain functional, viable neural tissue8,126,127. Over time, reactive gliosis within this layer gradually subsides, blending seamlessly into the surrounding normal tissue8,126,127.
Trend of Cellular and Molecular Changes Following Spinal Cord Injury (Refer to the Text for Further Details).

This table was compiled based on a synthesis of the following articles listed in reference numbers 22,28,31,90,122,123,128,129,132,133,135,136. The post-SCI events described in the lower section of this table may not strictly correspond to the time frames indicated in the upper section. A single post-SCI event may be observed across multiple time frames. These discrepancies and overlaps, including the inability to clearly delineate events, are inevitable due to variations in species, experimental conditions, and observational methods across studies. KC, keratinocyte-derived chemokine; MIP-2, macrophage inflammatory protein-2; pSTAT, phosphorylated signal transducer and activator of transcription 3 (see the text for other terms).
This review emphasizes the role of the BM as a distinguishing boundary between the glial scar core (true scar tissue) and the non-scar SFAs, highlighting the nuanced structural and functional complexity of the glial scar.
Various Cell Types Involved in SCI
The temporal progression of events following SCI highlights a complex interplay of cellular and molecular responses22,31,59,90,128–137. Most cell types that emerge in the SCI environment undergo functional changes over time, reflecting their inherent heterogeneity22,31,59,90,128–137. Table 1 illustrates the overall trend of cellular and molecular changes following SCI, primarily compiled from review articles22,28,31,90,122,123,128,129,132,133,135,136. Representative studies on cerebral stab injuries and spinal cord crush injuries have demonstrated the temporal and spatial patterns of astrocytic responses117,131,138 (Fig. 1). At 1–4 days post-lesion, astrocyte processes close to the lesion site preferentially orient toward the lesion center117,138 and the proliferation phase of SFAs is largely completed by 3 weeks post-injury131,138. After this period, approximately 3–4 weeks post-injury, SFAs no longer orient their processes toward the lesion center. Instead, they become more parallel and overlapping, contributing to the formation of a mature glial scar during the chronic stage of CNS injury (Fig. 1). The aligned layer of SFAs is organized through STAT3-dependent mechanisms and leucine zipper–bearing kinase (MAP3K13) expression127,131,139,140. Experimental studies have demonstrated that by 20 dpi, a new GLP, composed of the BM and a portion of the SFAs adjacent to the BM, is formed117,118. The GLP “walls off” the glial scar from the surrounding nervous tissue as a defensive perimeter 119 . In a SCI experiment on mice, approximately 90% of SFAs at the border between the lesion and the surrounding viable tissue originate from proliferating local astrocytes 141 . It is intriguing that SFAs in the GLP share molecular similarities with those in the glia limitans superficialis (GLS) on the surface of the CNS [see the section below, the BM in peripheral nerve (PN) injury] and possess unique transcriptional profiles that enable them to act as wound repair astrocytes, protecting neural tissue by preventing the infiltration of stromal and immune cells into the neural parenchyma 141 .
In the layer of SFAs, NG2-expressing oligodendrocyte progenitor cells (NG2-OPCs) begin to appear at the lesion site during the 3–7 days post-injury. NG2-OPCs are among the primary producers of inhibitory chondroitin sulfate proteoglycans (CSPGs) in the glial scar, alongside SFAs 142 . NG2, also known as CSPG4, rapidly induces retraction of cell process following SCI I 143 . CSPGs are key inhibitory molecules and the degradation of CSPGs by chondroitinase ABC reduces CSPG-associated inhibitory activity, thereby promoting axonal regeneration in SCI 144 . NG2-OPCs can differentiate into reactive astrocytes, thereby contributing to glial scar formation 145 . In a recent study, 10% of SFAs at the lesion site of SCI derive from NG2-OPCs 141 . The role of NG2-OPCs in axon regrowth evolves throughout the progression of SCI127,128.
The Heterogeneity of Cells Involved in SCI
Astrocytes demonstrate heterogeneity in both normal and pathological conditions, as evidenced by traditional and emerging quantitative (semi-)automatic methods121,141,146–149. In the normal brain, astrocytes vary across regions in terms of morphology, function, physiological properties, and developmental origins 121 . Similarly, astrocytic responses to diseases are highly diverse and context-dependent121,147,150. Earlier studies introduced the concept of reactive astrocyte subtypes, specifically “neurotoxic A1 astrocytes” and “neuroprotective A2 astrocytes” 151 . A1 astrocytes were reported to induce the death of oligodendrocytes and neurons in neurodegenerative disorders 151 , and their formation was driven by cytokines, including interleukin (IL)-1α, tumor necrosis factor (TNF), and complement component 1q, released by reactive microglia 151 . In an SCI model, A1 astrocytes were observed around the lesion core by 28 days post-SCI, suggesting that A1 astrocytes form a significant subset of SFAs 152 . An experimental study of SCI demonstrated that A2 astrocytes produced neurotrophic factors that supported neuronal survival and growth 153 .
The simplicity of the A1/A2 classification has been increasingly questioned154–156, despite being useful in studying astrocyte function in SCI148,157. In a recent study, astrocytic gene expression was compared across eight CNS disorders, including SCI, experimental autoimmune encephalomyelitis, neuroinflammation, Alzheimer’s disease, Huntington’s disease, amyotrophic lateral sclerosis, multiple sclerosis, and Parkinson’s disease 154 . The study revealed minimal overlap in differentially expressed astrocytic genes among these conditions. However, 61 core transcriptional regulators were identified as consistently shared across at least seven disorders, suggesting that a relatively small set of transcriptional regulators orchestrates diverse astrocytic responses, tailoring outcomes to the specific context of each disorder. It is likely that reactive astrocyte phenotypes arise from a complex interplay of transcriptional regulators and these regulators may underpin the previously reported A1/A2 phenotypes, offering a more comprehensive framework for understanding astrocytic heterogeneity in CNS diseases 154 .
Macrophages play multifaceted roles in both promoting and inhibiting tissue regeneration following SCI158–162. Within the first 24 h post-injury, monocytes migrate from the bloodstream to the lesion site and predominantly polarize into a state commonly referred to as M1-like. These macrophages, characterized by a pro-inflammatory profile, facilitate necrotic tissue and debris clearance and release inflammatory mediators, such as TNF-α and reactive oxygen species, which may contribute to secondary tissue damage158,159. Approximately, 1 week post-injury, a subset of macrophages transitions to an M2-like phenotype, associated with anti-inflammatory and tissue repair functions. These M2-like macrophages secrete cytokines such as IL-10 and transforming growth factor-β, which foster a microenvironment conducive to regeneration158,161. In the chronic phase, as the inflammatory response diminishes, macrophage numbers gradually decline. However, residual macrophages persist at the lesion site and continue to influence both inflammation and repair processes158–161. The traditional M1/M2 polarization paradigm in macrophages has faced increasing scrutiny in recent years160,163 as in reactive astrocytes (see above).
Microglia, similar to macrophages, can exhibit M1- and M2-like polarization states; however, their activation is more dynamic and context dependent161,163. Microglia in SCI initially exhibit pro-inflammatory M1-like characteristics, which contribute to inflammation and tissue clearance, and which hinder axon regeneration. Microglia transition into repair-supportive states, often termed M2-like phenotypes, during the later stages of CNS injury responses158,159. However, this classification is oversimplified due to the unique CNS-specific environment of microglia 163 . While reciprocal interactions between microglia and astrocytes are well documented22,164, the role of IGF-1 signaling in directing the organized alignment of SFAs at glial scar borders remains to be fully elucidated. This reflects the broader complexity of microglial activation states, which often diverge from the classical M1/M2 polarization framework 163 .
In summary, various cell types emerge at the site of SCI, continuously altering their pro-regenerative or anti-regenerative properties over time. Consequently, developing therapeutic approaches that target individual cell types is inherently challenging and time-sensitive. Establishing a comprehensive, effective treatment strategy that addresses the diverse range of cells involved is even more complex.
Given these considerations, conditions like SCI, which involve ongoing pathological changes, may be more effectively treated during the chronic stage—approximately 3–4 weeks post-injury or later—when the pathological state stabilizes and dynamic changes in cell populations subside (Table 1). Specifically, it may be prudent to avoid intervention during the acute or subacute stages, as these periods are characterized by rapid and unpredictable changes in cellular behavior. Building on this reasoning, we propose therapeutic strategies targeting the mature glial scar, which represents the stabilized and final pathological state of SCI, characterized by the fully developed BM at least 3 weeks after the initial impact117,118,122–124.
Our stance differs from the view that the acute and subacute phases represent the optimal windows for mesenchymal stem cell (MSC) transplantation 165 . Proponents of this timing argue that MSC transplantation during these phases effectively suppresses neuroinflammation, thereby mitigating glial scar formation and fostering an environment conducive to regeneration 165 . Furthermore, MSC transplantation during the chronic phase is considered challenging due to the presence of a well-formed glial scar, which may inhibit neural regeneration 165 . However, we contend that targeting the pro-regenerative elements of BM for cell transplantation during the chronic phase, when the BM is fully established, is more likely to yield stable and long-lasting outcomes.
The BM as a Platform for Ligand–Receptor Interactions
Laminin and collagen IV are the primary structural elements that shape the BM and they are interconnected with nidogen, perlecan, and other molecules166,167. Laminin is not only essential for the assembly and structural integrity of the BM but also demonstrates the strongest pro-regenerative capacity among BM-associated molecules. Ligand–receptor interactions, especially between laminin and integrins, are crucial in promoting axon regeneration by facilitating cell adhesion and migration168–174. However, regenerative proteins in the BM include not only laminin but also other components, such as collagen IV175,176. Collagen IV can enhance the migration of transplanted donor cells (eg, ES cells) via ligand–receptor interactions, with α2β1 integrin serving as the receptor for collagen IV 176 . This mechanism mirrors the interaction between laminin and integrin, suggesting that collagen IV similarly promotes the migration of transplanted cells177,178.
From the perspective of cell transplantation, the BM may be more accurately perceived as a broader, band-like substrate containing pro-regenerative molecules, on which various neuro-regenerative interactions occur, rather than as a simple linear structure serving solely as an anatomical boundary between cells and their surroundings. Recent measurements of BM thickness using atomic force microscopy reveal values several times greater than those previously determined through TEM 179 .
The integrin family, serving as receptors for laminin, is essential for donor cells to adhere to the BM and to extend neurites168–174. Integrin receptors are found on the surface of all cell types including neurons and stem cells, except red blood cells180,181. Experimental evidence shows that blocking integrin function results in the attenuation of laminin-dependent neuronal survival. For example, one in vitro study to measure human neural stem cell growth demonstrated that integrin β1-blocking antibodies negated the laminin-mediated increase in neurosphere numbers 182 . Conversely, in vitro experiments with dorsal root ganglion neurons cultured on laminin substrates revealed that higher levels of integrin expression on neurons enhanced their adhesion properties and neurite outgrowth 183 .
Chemoattractive Gradients of Laminin in the BM
Another crucial factor for axon regeneration is the chemoattractive gradient of pro-regenerative molecules on the host nervous tissue72,127,169,184,185 (Fig. 2B). The BM provides chemical gradients of soluble chemoattractants such as laminin along which cells can migrate169,186,187, so the likelihood of the integration of the donor cells into the host nervous tissue would be enhanced more in close proximity to the BM (Fig. 2B). Laminin–integrin interactions on the BM and the chemoattractive gradients provided by laminin are crucial for axon regeneration113,168,169.
Detour Pathways via Transplanted Cells and PSNs
The formation of detour pathways, particularly through PSNs, is recognized as a critical neural mechanism contributing to substantial motor recovery65–69,188,189. Notably, an early study observed that PSNs can spontaneously regenerate axons following SCI, even within inhibitory environments 71 . However, the precise mechanisms underlying the unique regenerative capability of PSNs remain unclear 71 . One possible hypothesis is that PSNs may exhibit a degree of resistance to inhibitory molecules, such as CSPGs in gliotic scar tissue. Disruptions in molecular pathways involving inhibitory receptors, such as PTPσ, which mediate the effects of CSPGs, have been shown to enable axonal growth in other neuronal populations, suggesting a potential avenue for PSN regeneration 190 .
The BM of the GLP develops in tight apposition with the core of the glial scar111,117,119,120. In contrast, the SFAs adjacent to the BM (the basal SFA, orange circled band indicated by facing arrows in Graphical abstract and in Figs. 1 and 2A) are not always densely packed117,119. In one instance, the astrocytes processes in this area were loosely organized, showing “watery-appearing” 119 . Hence, the compartment above the BM, epi-BM compartment (the area highlighted in green in Fig. 1(6)), may tend to serve as a potential conduit for PSN regeneration within the inhibitory gliotic environment following SCI. Chemoattraction mediated by soluble pro-regenerative molecules in the BM such as laminin may naturally recruit and guide PSNs toward the epi-BM compartment, thereby facilitating robust regeneration. This mechanism likely underlies the sprouting observed in the severed CST. As previously mentioned, the functional regeneration of the CST through cell transplantation requires the coordinated action of various factors both within and outside the transplanted cells. However, ultimately, unless we can elucidate where the regenerated neurons integrate within the damaged host nerve tissue—in other words, the morphological framework capable of supporting regenerated nerve fibers—we cannot definitively demonstrate the regeneration of the CST. In the SFA layer, there are no functional neurons8,131, so it is not harmful when transplanted cells and their regenerating axons occupy this region.
Placement of MN progenitors onto the surface of injured spinal cord is worth exploring because it can potentially harness the pro-regenerative properties of laminin within the BM of GLP to facilitate axon regeneration (Fig. 2B). In SCI where the GLP builds near to the surface of the injured spinal cord, this site could potentially serve as a portal for donor cells to enter into the host nervous tissue (Fig. 2B). When PSNs alone are insufficient to directly establish detour pathways, transplanted cells can play a crucial role in their formation. For example, as illustrated in Fig. 2B, a sprout from the host CST (Spr3) forms a synapse with a neuron derived from the transplanted MN progenitor. This transplanted neuron then establishes a synapse with a host PNS, which connects to the LMN pool.
It may become a decisive factor how growth cones encounter the surface of the glial scar. When growth cones enter the plane of the BM at a 90° angle, they tend to collide directly with the surface, leading to the cessation of axon elongation at the surface (Fig. 2A, blue dot on the BM). A TEM study of rat SCI suggests that the growth cones of regenerating axons do not readily deflect along the BM 119 , although there is no direct evidence that the BM impedes the outgrowth of regenerating neurites 116 . Compared with such a “point encounter” at a 90° angle, a broader contact area could be gained in an “oblique-angle encounter,” leading to increased adhesion between laminin in the BM and integrin expressed on donor cells168,169. This is more likely to facilitate detour pathway formation (Fig. 2). Subsequently, regenerating axons that bypass the core of the glial scar may further extend neurites without hindrance in the outermost layer (OML) of the glial scar where each astrocyte maintains its default territory 191 . Beyond the OML, the permissiveness necessary for axon elongation would be provided by white matter tracts87,192,193 (Fig. 2B).
The BM in PN Injury
Normal anatomy of the BM at the nerve root
Normally, astrocytes at the surface of nerve roots form a dome-shaped structure with its convex surface usually directed distally (the glial dome) at the CNS and the PNS interface, known as the transitional zone194,195 (Fig. 3A). The GLS is composed of astrocyte end-feet and associated BM (Fig. 3A)194–197.

Glia limitans superficialis and BM-targeting cell transplantation. (A) Normal anatomical relationships among the basement membrane (BM) and the surrounding structures of motor nerve root. Glia limitans superficialis (GLS) consists of the BM and astrocyte end-feet (Ase) covering the spinal cord surface. The BM on peripherally convex protrusion of astrocyte end-feet (the glial dome, GD) and that on lower motor neurons (LMNs) are continuous. The transitional zone (TZ) is an interface between the central nervous system and the peripheral nervous system where central myelins (Cm) derived from oligodendrocytes are replaced with peripheral myelins (Pm) from Schwann cells (SC). (B) In peripheral nerve injury, glial scar–derived reactive astrocyte processes extend to create the astrocyte outgrowth (AO). On degeneration, reprogramed Schwann cells construct the Schwann cell column (SCC). The AO and SCC form an uninterrupted structure, the astrocyte outgrowth–Schwann cell column complex (the AO–SCC complex), connecting the central nervous system and peripheral nervous system 198 . Motor neuron progenitors (MNp) placed on the nerve root enter the nerve via laminin–integrin interactions, crossing the remodeled BM (red dotted line outside the nerve). The AO–SCC may serve as a guide and pathway for regenerating neurites (red dotted line under the BM). The BM abutting the AO–SCC is continuous with that of the GLS. Inset image at the right upper corner shows conspicuous AO (two arrows) adapted from Sekiya and Holley 266 , licensed under CC BY-NC 4.0.9. Smooth contour of normal transitional zone is shown by a curved dotted line in right upper corner of this image (immunostaining; GFAP for reactive astrocytes, green and Tuj1 for neurons, red, scale bar: 200 μm). Asterisk, compression-injured site of the auditory nerve. (C) When the glial scar (GS) forms in the anterior horn (AH) region, InP microinjection through a thin cannula close to the BM is feasible (two dotted arrows) to harness the pro-regenerative properties of the BM. MNp, motor neuron progenitors; AHC, anterior horn cell; As, astrocytes; CST, corticospinal tract; dAHC, dead anterior horn cell; LMN, lower motor neuron; MF, muscle fiber; NMJ, neuromuscular junction.
Neuro-glial plastic changes at the compromised nerve root
The normal configuration among astrocytes, SCs, and axons mentioned above transforms in LMN diseases/injuries; astrocytes are replaced with glial scar–derived reactive astrocytes that produce long extensions to form a peripherally convex, remarkably long protrusion along the longitudinal axis of the PN (Fig. 3B). This protrusion, the astrocyte outgrowth (AO) 198 , is a common feature observed in LMNDs4,199–202. In the degeneration of PNs, SCs are reprogramed and converted to repair SCs to develop the Schwann cell column (SCC, Bungner bands; Fig. 3B) 203 . The SCC plays a pivotal role as a regeneration track along which regenerating neurons elongate203,204. Another intriguing but seemingly less well-known phenomenon is that the SCC creates a naturally occurring, continuous structure associated with the AO, namely the astrocyte outgrowth–Schwann cell column complex (the AO–SCC complex) 198 , formerly called as “Schwann cell–astrocyte complex” 194 , spanning the CNS and the PNS continuously, enclosed within the same BM (Fig. 3B). Ultrastructural studies demonstrate that the BM abutting the AO–SCC is continuous with that of the GLS, covering the surface of the nerve roots and the spinal cord 194 (Fig. 3B).
BM remodeling
BM remodeling plays a pivotal role in facilitating cellular responses to the demands of host tissue, both under normal physiological conditions and in pathological states. For example, normal cells such as neural crest cells 205 and pathological cells including neutrophils during inflammatory responses 206 and metastatic cancer cells 207 rely on BM remodeling to fulfill specific functional requirements. One of the fundamental mechanisms enabling the remodeling of pores in the BM to facilitate smooth cell transmigration is the focal self-digestion of the BM at the leading edge of migrating cells by matrix metalloproteinases205,206. Studies have demonstrated that environmental cues, such as mitogens and growth factors, play a crucial role in regulating cell proliferation and survival208,209. An experimental study using the ExP approach suggests that BM remodeling may be driven by a shortage of endogenous neurons in the host nervous tissue, which are essential for maintaining the functionality of host neural circuits 210 . From this perspective, the shortage of endogenous neurons could act as an environmental cue, potentially triggering BM remodeling.
BM for PN regeneration
A preceding study has envisaged that the AO–SCC complex spanning the CNS and the PNS could guide regenerating neurites between the two 202 (Fig. 3). Later, this presumption was confirmed; donor cells placed on the surface of degenerated auditory nerve autonomously entered the nerve to be integrated in the host neural circuit 210 . The donor cells likely adhered to and subsequently crossed the laminin-rich BM, which had been remodeled to meet the host’s demands (see above). An immuno-TEM study of sciatic nerve injury revealed that regenerating axons and adjacent SCs are located within the same laminin-positive BM, supporting the notion that the BM provides a conduit for axonal regeneration 211 . Upon entering the nerve crossing the remodeled BM (Fig. 3B), donor cells exhibited various modes of cell migration, similar to those observed during development and in the compromised CNS as detailed previously210,212,213. The reactive astrocyte process–guided cell migration appeared to recapitulate gliophilic migration observed during the formation of the neocortex in mammals210-213. The surfaces of the elongated processes of reactive astrocytes are covered by a laminin-expressing BM118,120, which also extends to the surface of the AO, a structure comprising bundled reactive astrocytes200–202 (Fig. 3B).
For the regeneration of LMNs, various donor cell delivery routes have been investigated, including intra-spinal cord injections and intra-PN injections214-217. In these InP approaches, a difficulty occurs when elongating motor axons reach the transitional zone (Fig. 3A). The transitional zone is a robust barrier for the movement of neuronal cells and their neurites and elongating axons cannot easily reach the muscles via the PN trunk 218 . For donor cells delivered via the InP route, the transitional zone serves as a robust barrier to the movement of neuronal cells and their neurites, preventing elongating axons from easily reaching the muscles through the intra-PN trunk. By employing the ExP approach, it becomes possible to avoid the issue of the intractable transitional zone observed with the InP approach described above. This is because the formation of the AO–SCC effectively eliminates the transitional zone, and in the ExP approach, donor cells are placed on its surface, where axon regeneration can occur on the surface of the AO–SCC. An alternative to inject donor cells into the PN trunk distal to the transitional zone5,214–216. However, with this approach the distance from the motor cortex to the site of cell injection in the PN is longer than that with intra-spinal cord injection and PN injection may directly compromise existing axons 219 .
MN Degeneration and Repair of the Neuromuscular Junction
Following MN degeneration, various adaptive/compensatory plastic changes occur220–226. One of them is that residual LMNs send sprouts to dying muscle fibers (MFs) to prevent further degeneration (Fig. 4). On MN degeneration, acetylcholine receptor (AChR) clusters on the motor endplates (MEPs) begin to fragment and the normal pretzel-like arrangement of AChRs is lost 227 (Fig. 5). Projections of terminal Schwann cells (tSCs) invade the synaptic clefts and extend processes to build a bridge (the Schwann cell bridge, SCB) to nearby vacant MEPs220–223,228. Using the SCBs, regenerating axons from surviving neurons travel to innervate contiguous vacant synapses223–226 (Fig. 4). These plastic changes can be compensatory at least in the beginning but eventually lead to a condition in which a small number of residual LMNs innervate multiple synapses. Poly-innervation places excessive demands on residual neurons229,230 (Fig. 4). If cell transplantation alleviates the deficiency of MNs in the peripheral motor pathways, it could correct the state of poly-innervation.
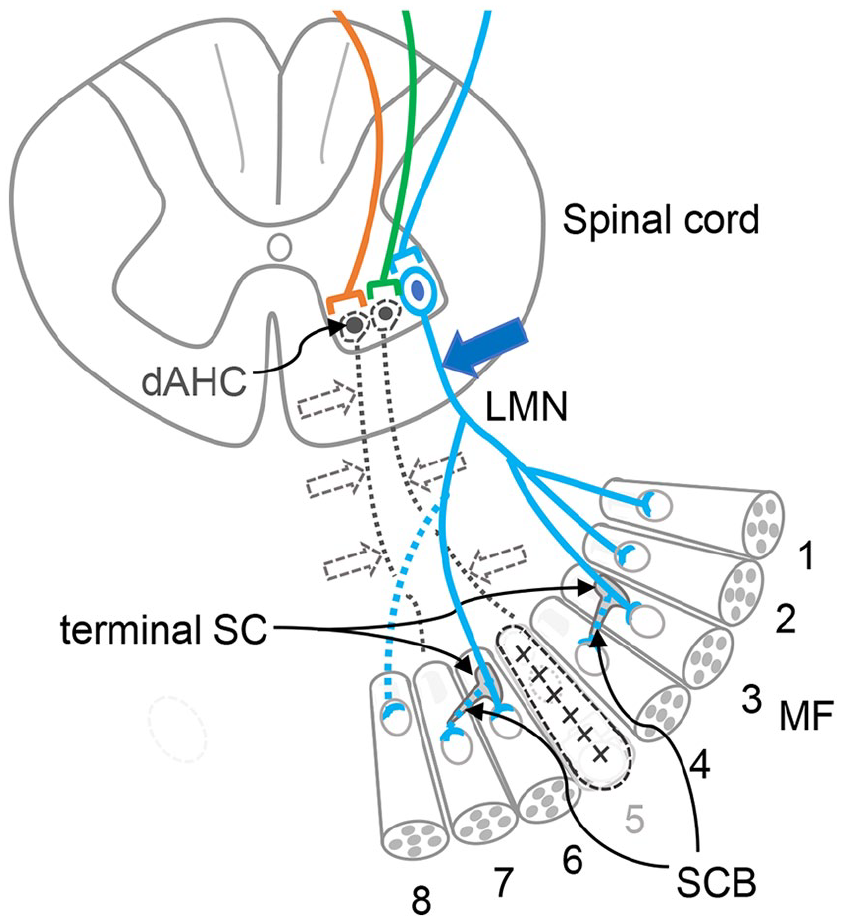
Plastic changes following death of LMNs. In the case shown here, the left-handed two lower motor neurons (LMN) degenerate (black dotted circles, lines, and arrows). No. 8 muscle fiber (MF) is rescued by sprouting from a nearby LMN (long dotted blue line). Terminal Schwann cells (tSCs) elongate tongue-like protrusions to form bridges (Schwann cell bridge, SCB, two short dotted blue lines) to adjacent MFs. Through SCBs, the sprouts travel to the degenerating No. 4 and No. 7 MFs, leading to the prevention of MF atrophy. Eventually, a single surviving LMN (blue solid line) that originally innervated four MFs (MF 1, 2, 3, and 6 in Box 1) comes to innervate seven MFs (MF 1–4 and 6–8), a poly-innervation state. No. 5 MF has degenerated and is lost marked by x. dAHC, dead anterior horn cell.
Repair of the neuromuscular junction (NMJ) requires extensive communication among presynaptic MNs, tSCs, MFs, and the synaptic basal membrane (sBM; Fig. 5) 224 . Laminin in the BM plays critical roles in both presynaptic and postsynaptic sides of the NMJs (Fig. 5). It contributes to the maintenance of the NMJs by clustering ion channels, organizing active zones in presynaptic terminals, and aiding maturation of the postsynaptic muscle membrane221,226. To prevent the permanent destruction of NMJs, it is crucial that regenerated MNs extend to the region adjacent to the synaptic cleft and secrete agrin into the sBM221,226 (Fig. 5).

Key components at the neuromuscular junction (NMJ)226,228. For the formation of NMJs, presynaptic motor neurons (MN), terminal Schwann cells (tSCs), postsynaptic muscle fibers (MFs), and the synaptic basal membrane (sBM) communicate extensively. For the clustering of acetylcholine receptors (AChRs), agrin (Agr) plays a central role, working in collaboration with molecules such as MuSK (muscle-specific kinase), Lrp4 (low-density lipoprotein receptor–related protein 4), and rapsyn. Agrin (Agr) is secreted by nerve terminals into the sBM, and hence, it is essential that regenerating axons reach the distal ends of MNs to release agrin. The lower panel illustrates the characteristic “pretzel-like” appearance of an AChR cluster at the motor endplate (MEP). Ach, acetylcholine; AChR, acetylcholine receptor; eBM, extra-synaptic basal membrane; SC, Schwann cell; SV, synaptic vesicle; SyC, synaptic cleft; MF, muscle fiber; PSM, postsynaptic membrane.
Enhancing MN regeneration through cell transplantation would help address the shortage of regenerating axons reaching the NMJ area. Agrin plays a central role in clustering AChRs at postsynaptic MEPs by interacting with the receptors MuSK (muscle-specific kinase) and Lrp4 (low-density lipoprotein receptor–related protein 4). Agrin binds to Lrp4 on the muscle membrane, forming an agrin–Lrp4 complex that activates MuSK. This activation triggers an intracellular cascade, leading to AChR clustering through the direct interaction between rapsyn and AChRs. This process facilitates the assembly of a postsynaptic scaffold that is essential for NMJ formation221,226,231–236. Regenerating motor axons tend to retrace their original pathways and re-establish NMJs on the same MFs that they previously innervated, particularly when the BM surrounding the motor axons remains intact226,237.
Advantages and Disadvantages of Various Cell Delivery Methods
Currently, the three most commonly used methods for cell transplantation are InP, intrathecal, and intravenous delivery. Each approach has distinct advantages and disadvantages, and hence, it is crucial to consider them as complementary rather than mutually exclusive strategies238,239.
InP cell delivery route
The InP route has an advantage in delivering a high concentration of donor cells directly and precisely to the target region, although it may be disadvantageous if viable host tissue is damaged by the procedure, such as insertion of the infusion device and placement of donor cell mass238–240. The clinical acceptability of a surgical approach, including InP, depends on whether the benefits substantially outweigh the risks associated not only with the surgical procedures themselves but also with the broader systemic and contextual factors241,242. A key inference from our review of previous studies is that traditional InP techniques face theoretical and inherent challenges in restoring the damaged CST, at least without modifications as pointed out above. Such considerations have led us to explore potential alternative strategies beyond traditional InP techniques.
Intrathecal cell delivery route
This route for cell delivery has gained attention due to its minimally invasive feature 240 . However, compared to InP transplantation, it offers less precise targeting of specific injury sites. Consequently, fewer cells may reach the lesion, which could diminish the overall efficacy of the treatment 239 .
Intravenous cell delivery route
This method may be the least invasive in comparison with others, but faces significant limitations in terms of therapeutic efficacy for SCI. A large proportion of the transplanted cells become trapped in peripheral organs, such as the lungs, which prevents sufficient numbers from reaching the CNS238–240. Consequently, intravenous administration may be more appropriate for systemic treatments rather than for targeting localized injuries in the CNS.
ExP cell delivery route
This route is minimally invasive and avoids additional mechanical stress to the spinal cord tissues that are already fragile due to injury. It ensures the preservation of naturally and plastically developed structures surrounding the glial scar core, including the BM which is crucial because it not only contains pro-regenerative molecules, such as laminin, but also serves as a scaffold for axon regeneration, as detailed above. Using the ExP route, it may be possible to place donor cells onto the surface of the spinal cord without penetrating it (Fig. 2). It is noteworthy that the proposed scar surface–based BM-targeting approach fundamentally differs from cell sheet engineering techniques used for regenerating various tissues such as cardiac muscle, liver, bones, skeletal muscles, and corneal epithelium243,244. In CNS and PNS injuries, individual donor cells should navigate their own paths to integrate into the host neural circuit. With the ExP approach, donor cells are most effectively applied with a sticky medium onto the surface of the target location which is typically small, irregular, and variable between patients. To secure donor cells at the target site, sticky materials like fibrin glue can be used as supportive matrices. Studies have demonstrated that fibrin, either alone or combined with pro-regenerative molecules, effectively delivers donor cells to the target site while promoting neuronal survival and growth245,246. It may not be feasible to apply a uniform procedure for donor cell transplantation onto host nervous tissue, given the variability in pathologies among patients with SCI. Hence, pre-manufactured cell sheets that are uniformly produced before transplantation are not adaptable in the ExP approach proposed here.
InP and ExP combined route
As described above, the BM-targeting ExP approach alone is insufficient for overcoming the glial scar when it is located deep within the spinal cord. In these cases, combining the ExP approach with InP microinjection may offer a more effective method for delivering donor cells to the host nervous tissue (Fig. 2C).
Solving the Long-Standing Questions in Cell Transplantation: When, Where, and How?
It is worth reconsidering whether past approaches, which habitually relied on empirically or personally determined timing and locations for cell transplantation 239 , might have obscured the innate potential of the host nervous tissue for motor system regeneration.
With the ExP cell delivery method proposed here, it becomes possible to precisely determine the optimal timing and location for cell transplantation. As discussed in detail above, the formation of the BM and SFAs typically occurs concurrently around 2–3 weeks post-SCI (Fig.1, Table 1)122–124. Hence, the ideal timing for cell transplantation is preferably no earlier than 3 weeks post-SCI, with a later time point potentially being more favorable. Even during the chronic stage of SCI, ExP cell transplantation may remain a viable option as long as the BM is maintained. A TEM study confirmed the presence of the BM more than 6 months after a stab injury to the rat cerebral cortex 119 .
Advancements in magnetic resonance imaging (MRI) technology, including diffusion tensor imaging, magnetization transfer imaging, and chemical exchange saturation transfer, are paving the way for more detailed comparisons between imaging data and traditional histopathological findings247–253. This progress holds promise for characterizing the glial scar, particularly the BM, in future imaging studies. Preoperative MRI data can form the basis of neuro-navigation systems, enabling precise localization of targets 254 . This precision could be further enhanced by intraoperative MRI and ultrasound255,256. Mechanical rupture of the BM capsule encasing the glial scar core could cause leakage of its contents beyond the BM, triggering harmful reactions, including the reactivation of surrounding astrocytes 22 as discussed in the Introduction section. Nevertheless, ExP and InP microinjections can be performed with minimal invasiveness using surgical techniques like micro-endoscopic laminotomy, requiring a skin incision of less than 2 cm 257 . Compared to traditional open surgery, such endoscopic approaches offer critical advantages, including shorter procedure times and reduced intraoperative blood loss, which minimize the risks of hemodynamic instability and cardiac complications—key considerations for patients recovering from the initial impact of SCI 258 .
To address the anticipated large-scale donor cell death soon after InP injection, which occurs primarily due to apoptosis and immunological rejection 259 , donor cells have traditionally been transplanted in quantities far exceeding the physiological requirement260,261. With ExP cell delivery, donor cells seem to integrate into the host tissue in an autonomously regulated manner, likely in response to the host’s demand, specifically addressing the shortage of endogenous neurons critical for the complete functionality of the host’s neural circuit (see the sections, “BM Remodeling” and “BM for PN Regeneration”).
Additionally, donor cells delivered via ExP route are immediately nourished by cerebrospinal fluid (CSF) before establishing a connection to the blood supply, as they are bathed in the CSF upon delivery 262 . The CSF is rich in pro-regenerative molecules, including brain-derived neurotrophic factor and IGF-2, which may further support donor cell survival and integration263,264.
Conclusion
Cell transplantation is a promising therapeutic approach for rebuilding functional motor systems, particularly the CST1,5,10,34,36,61 but there is still significant potential for improvement in optimizing its therapeutic efficacy1,5,7,10,34,36,61,103. The glial scar is frequently cited as an intractable barrier to axon regeneration21,89,90. However, we emphasize that introducing the ultrastructural data, especially those of the BM, obtained through electron microscopy could open new avenues for advancing regeneration studies in CNS injuries, including SCI. The glial scar exhibits a “duality,” with an anti-regenerative core and a pro-regenerative “surface.” The surface permissiveness of the glial scar is attributed to pro-regenerative molecules expressed in the BM13–20,86,109,110. Thus, transplanting donor cells onto the surface of gliotic nervous tissue, actually on the BM, may significantly enhance the efficacy of cell transplantation. A BM-targeting approach can be used alone or in combination with the traditional InP microinjection approach. We highlighted that forming detour pathways with cell transplantation, depending on the pro-regenerative BM and incorporating PSNs, could significantly enhance motor pathway regeneration by bypassing the intractable glial scar core. Another conclusion is that therapeutic interventions for SCI may be more effective during the chronic stage, avoiding the acute or subacute stages which involve rapid and unpredictable pathological changes. In clinical translation, it is a pressing and critical concern to evaluate the medico-social aspects of any proposed intervention. If a method is prohibitively expensive and inaccessible to all members of society, its widespread adoption and benefit to patients will be limited. BM-targeted InP microinjection is innovative yet clearly a low-cost option. This is because the clinical implementation of this cell transplantation method does not require significant expenditure for the development of specialized instruments or devices; it can be adequately performed by repurposing existing tools and equipment. Furthermore, should novel instruments or devices be developed in other fields in the future, their potential applicability to this cell transplantation method can be evaluated. If deemed suitable, these innovations can be adapted for use, thereby ensuring the long-term “low-cost” nature of this transplantation method. The other critical point in aiming clinical translation is the simplicity of the method. The methods that are overly complex technically are unlikely to be widely adopted in clinical settings. However, the methods proposed here may also align with this requirement. It is through the development of new techniques that new frontiers in science are opened, as noted by an eminent pioneer in biomedicine: “Progress in science depends on new techniques, new discoveries, and new ideas, in that order of decreasing importance” 265 . Further advancing research on the innovative method of cell transplantation proposed here holds significant promise for its clinical application.
Footnotes
Acknowledgements
The authors appreciate Emeritus Professor Juichi Ito and Professor Koichi Omori, Department of Otolaryngology-Head and Neck Surgery, Kyoto University Graduate School of Medicine for their support. They are grateful to Dr Yoshinori Maki, Department of Neurological Surgery, Hikone Chuo Hospital, for his valuable advice and suggestions regarding spinal cord surgery.
Author Contributions
T.S.: Conceptualization, Data curation, Data analysis, Figure creation, Funding acquisition, Writing—original draft, Writing—review & editing.
M.C.H.: Writing—review & editing. All authors read and approved the final manuscript.
Ethical Approval
Our university and hospital do not require ethical approval for any papers based on article review.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: One of the authors (T.S.) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the Japan Society for the Promotion of Science (MEXT; No. 26931051, 24931046, 13557112, 1559150, 12470281, 09557113-3, 10877209, 08457356-3, 09557113, 08457356, 07457303, 06454407, 03454341, 58770916), Univers Foundation, the General Insurance Association of Japan, the Japan Health Foundation, Osaka Gas Group Welfare Foundation, Zenkyoren, and Mitsui Sumitomo Insurance Welfare Foundation.
