Abstract
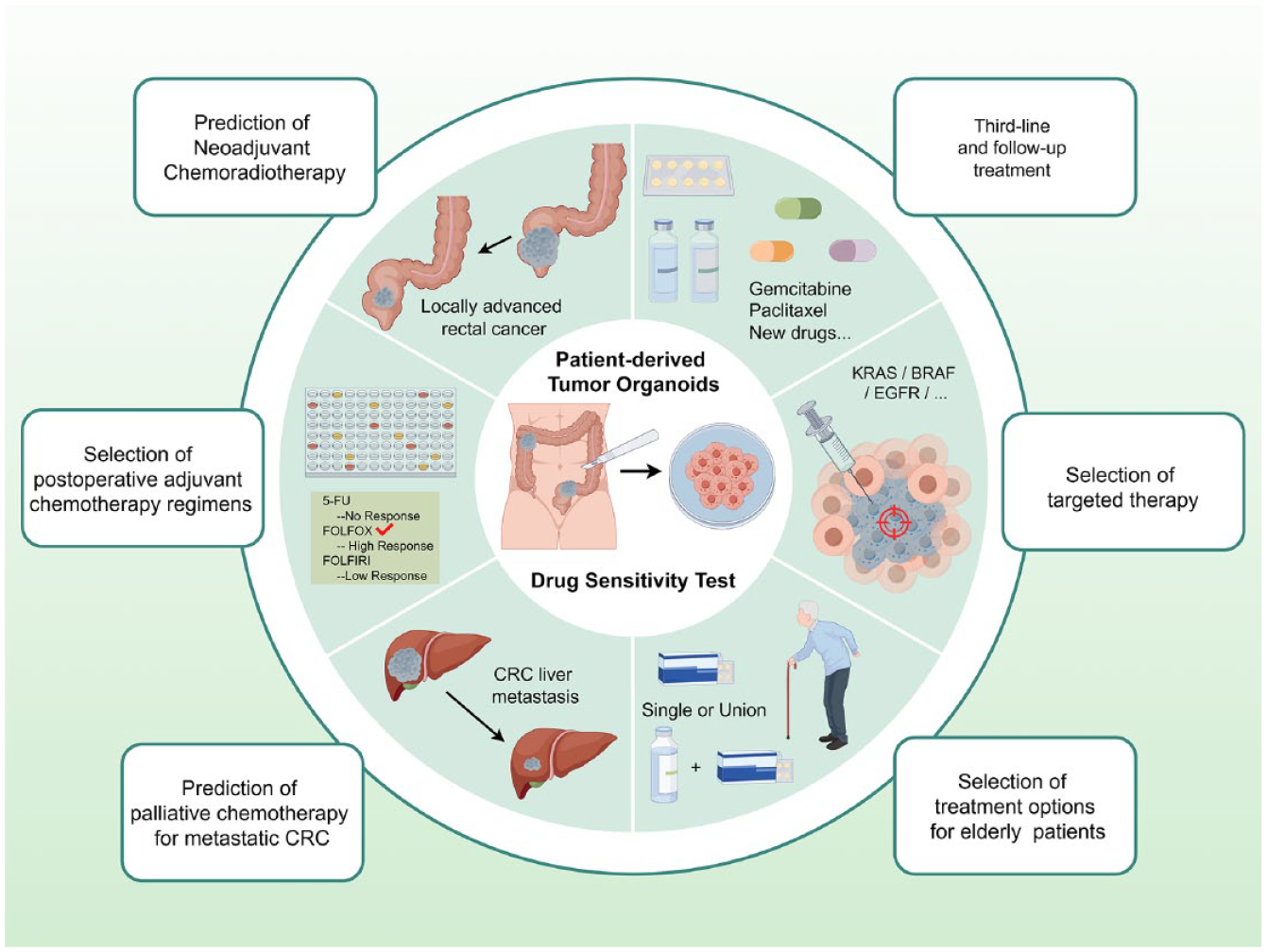
Colorectal cancer (CRC) represents a significant cause of cancer-related mortality on a global scale. It is a highly heterogeneous cancer, and the response of patients to homogeneous drug therapy varies considerably. Patient-derived tumor organoids (PDTOs) represent an optimal preclinical model for cancer research. A substantial body of evidence from numerous studies has demonstrated that PDTOs can accurately predict a patient’s response to different drug treatments. This article outlines the utilization of PDTOs in the management of CRC across a range of therapeutic contexts, including postoperative adjuvant chemotherapy, palliative chemotherapy, neoadjuvant chemoradiotherapy, targeted therapy, third-line and follow-up treatment, and the treatment of elderly patients. This article delineates the manner in which PDTOs can inform therapeutic decisions at all stages of CRC, thereby assisting clinicians in selecting treatment options and reducing the risk of toxicity and resistance associated with clinical drugs. Moreover, it identifies shortcomings of existing PDTOs, including the absence of consistent criteria for assessing drug sensitivity tests, the lack of vascular and tumor microenvironment models, and the high cost of the technology. In conclusion, despite their inherent limitations, PDTOs offer several advantages, including rapid culture, a high success rate, high consistency, and high throughput, which can be employed as a personalized treatment option for CRC. The use of PDTOs in CRC allows for the prediction of responses to different treatment modalities at various stages of disease progression. This has the potential to reduce adverse drug reactions and the emergence of resistance associated with clinical drugs, facilitate evidence-based clinical decision-making, and guide CRC patients in the selection of personalized medications, thereby advancing the individualized treatment of CRC.
Keywords
Introduction
Colorectal cancer (CRC) is a leading cause of cancer-related death 1 . Recent statistics show that the morbidity and mortality rates of CRC have increased significantly in China, Europe, and North America in recent decades 2 . CRC is caused by a combination of genetic and environmental factors3–5. The increasing prevalence of risk factors such as smoking, obesity, and unhealthy lifestyle habits has contributed to the increase in the incidence of CRC and a shift to a more aggressive form of the disease6–15. CRC is a highly heterogeneous disease16,17. It is staged according to the depth of local invasion (T stage), lymph node metastasis (N stage) and whether there is distal metastasis (M stage), and the treatment is determined according to the stage. Approximately 5% to 15% of patients with metastatic colorectal cancer (mCRC) have high microsatellite instability (MSI-H) due to DNA mismatch repair (dMMR) defects, which are highly sensitive to immunotherapy, especially immune checkpoint inhibitors (ICIs)18,19. The high degree of individualization of patients and the great difference in drug sensitivity present challenges for treatment. However, no tumor drug sensitivity data are available for routine clinical use.
In recent years, the patient-derived xenograft (PDX) model has become a cornerstone of cancer modeling and a powerful tool for assessing drug efficacy and sensitivity20,21. However, the PDX model has the disadvantages of a long culture time, high cost, and a low success rate of culture, which limits its rapid clinical application. Therefore, performing tumor drug sensitivity experiments quickly and effectively and screening the most suitable drugs for patients in the shortest time for precise individualized treatment are challenging issues in CRC treatment.
In recent years, three-dimensional (3D) cellular model organoid technology has undergone gradual development and maturation. Organoids are analogues of tissues produced through 3D in vitro culture using adult or pluripotent stem cells. They possess a specific spatial structure. Patient-derived tumor organoids (PDTOs) are obtained by culturing a patient’s biopsied, punctured, or surgically excised tissue in stromal gel for several weeks. The successful establishment of PDTOs has been demonstrated for various types of cancer, including bladder cancer 22 , prostate cancer 23 , liver cancer 24 , and pancreatic cancer 25 . Subsequent studies have demonstrated that PDTOs are capable of maintaining the histopathological and genetic characteristics of parental tumors in a stable manner26,27. PDTOs offer the advantages of closer proximity to physiological cellular composition and behavior, a more stable genome, and high-throughput screening as compared to traditional two-dimensional culture models28,29. Compared with animal models, organoid models are simpler and less costly to manipulate 30 . They can also be used to study mechanisms, such as the onset and progression of diseases 31 . While maintaining the heterogeneity of the source tumor and interpatient heterogeneity 32 , PDTOs exhibit fundamental homogeneity in terms of the morphology and size of individual tissue organs. In a seminal study, Van de Wetering and colleagues identified a genetic correlation between a single oncogenic mutation and the drug response in CRC PDTOs 26 . Furthermore, Melissa C. Skala, an American scientist, demonstrated that chemotherapeutic drugs such as paclitaxel and tamoxifen increased the mortality of ER(+) human breast cancer tumor carcinoid cells 33 . These findings suggest that PDTOs may serve as valuable tools for predicting tumor sensitivity to clinical drug therapy. This platform offers fast and advanced technology for researching tumor pathogenesis, regenerative medicine, drug screening, personalized medicine, and related fields. This article presents a review of the application of PDTOs in the clinical management of CRC, offering a summary of the advantages of the current application and an analysis of the challenges that lie ahead.
PDTOs in CRC Treatment
Surgical resection represents the primary treatment modality for CRC, particularly for patients with early-stage disease. Nevertheless, CRC metastasis is observed in approximately 15% to 30% of patients. In patients with intermediate to advanced CRC, surgical resection alone is frequently inadequate. Therefore, drug therapy is also one of the basic methods for treating CRC. To promote tumor shrinkage or stabilization and prolong survival, patients must undergo radiotherapy, chemotherapy, and targeted therapy. The continuous advancements in drug development, the rational use of combination therapies, and the optimization of treatment sequences have improved the survival rate of CRC patients.
PDTO models represent a novel approach to the individualized treatment of CRC, offering a means of providing diagnostic and treatment options for cancer patients within a defined time window. PDTO cultures were performed using the patient’s surgical remnant samples and evaluated for consistency with the histological characteristics of the tumor organoids and the patient’s tumor (Fig. 1). Subsequently, drug sensitivity tests were conducted, and half-inhibitory concentrations were calculated from the number of viable cells to evaluate the extent of drug resistance in the patients (Fig. 2).
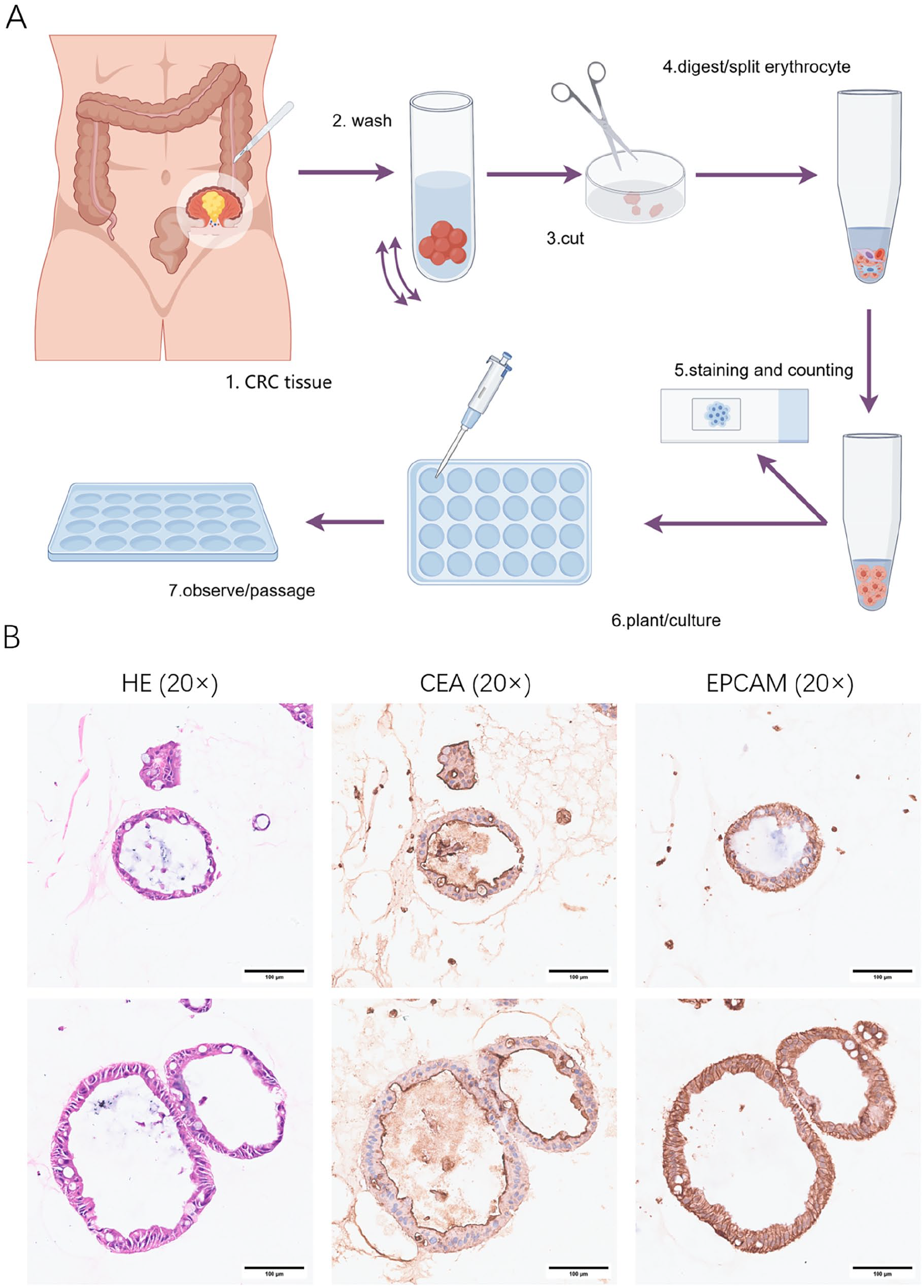
Colorectal cancer patient–derived organoid culture methods and validation: (A) 1. Fresh CRC tissue samples were placed in primary tissue preservation solution and transferred to 4°C while maintaining sterility throughout. 2. The CRC tissue was placed in precooled PBS to remove adipose tissue, necrotic tumor tissue, blood, and debris. This step was repeated until the mixture was clear. 3. The tissue was cut into 1 to 2 mm3 pieces. A moderate amount of tissue can be saved for sequencing analyses, and so on. 4. The tissue fragments were collected and added to a tissue digestion solution for a specific duration. The solution was then filtered through a strainer to collect the bowel cancer cells. If the cell precipitate remained red, the cells were resuspended in erythrocyte lysate, and erythrocytes were lysed at low temperature. 5. The dissociated cells were collected, flushed with DMEM/F12, centrifuged to collect the precipitate, and then resuspended for Taipan blue staining and cell counting. 6. The cell suspension was mixed with Matrigel, and the mixture was gently blown to avoid air bubbles. The resulting mixture was inoculated into each well and incubated at 37°C for 15 to 30 min. Afterward, prewarmed organoid medium was added, and the PDTOs were cultured in an incubator at 37°C and 5% CO2. The medium was changed every 2 to 3 days. 7. The PDTOs were observed and photographed daily to determine the initial number of PDTOs, proliferation rate, morphology, and degree of microbial contamination. (B) H&E staining and immunohistochemistry of CRC PDTOs. The scale is 100 µm.

The growth process and drug sensitivity test results for colorectal cancer patient–derived tumor organoids: (A) Growth process of CRC PDTOs. (B) Results for the sensitive and resistant groups at 72 hours postdosing. The scale is 20 µm.
Postoperative Adjuvant Chemotherapy
Chemotherapy treatments currently include monotherapy and multiagent regimens, such as fluoropyrimidine (5-FU), capecitabine (CAP), oxaliplatin (OX), and irinotecan (IRI). The combination regimens FOLFOX (5-FU + OX), XELOX or CAPOX (CAP + OX), and FOXFIRI (5-FU + IRI) remain the primary treatment options for first-line therapy34–36 (Table 1). In recent decades, the survival rate of patients with advanced CRC has improved as a result of the widespread use of chemotherapy regimens. However, chemotherapy has several significant drawbacks, including high toxicity, innate and acquired resistance, and low selectivity for tumor heterogeneity. These limitations significantly diminish the efficacy of chemotherapeutic agents and may ultimately result in the cessation or interruption of chemotherapy37,38.
CRC postoperative adjuvant chemotherapy treatment.

Table summarizing the Standard for Diagnosis and Treatment of Chinese Colorectal Cancer (2023 Version) and the NCCN Clinical Practice Guidelines in Oncology, Colon Cancer.
PDTOs have the potential to predict individual drug responses39,40. In a blinded study, Ting Wang et al. 41 assessed the predictive accuracy of PDTOs for the response to chemotherapy regimens in stage IV CRC patients. This study demonstrated that PDTOs were effective in predicting the response of individual patients to available chemotherapy regimens. The sensitivity, specificity, and accuracy of the PDTOs in predicting the response to chemotherapy regimens were 63.33%, 94.12% and 79.69%, respectively. In a prospective clinical study, Ooft et al. 42 demonstrated that PDTO can prevent cancer patients from receiving ineffective IRI chemotherapy. In a study by Geevimaan et al., an in vivo biobank was established using samples from patients with advanced CRC to evaluate the sensitivity of PDTOs and mouse organ xenograft tumors to OX. The results substantiated the utility of these models in informing decisions regarding OX-based chemotherapy regimens 43 . In a cohort study, Ting Wang et al. reported that resistance to PDTOs was linked to shorter progression-free survival. To enhance the prediction of progression-free survival, it may be beneficial to include drug testing for PDTOs in current clinicopathologic models 44 .
The findings of these studies substantiate the assertion that PDTO models can be employed as benchmarks for the evaluation of the efficacy of 5-FU, OX, and IRI in the screening of commonly utilized chemotherapeutic agents for CRC. PDTOs have significant potential for preclinical assessment and can serve as an important reference for clinicians in selecting the most effective postoperative chemotherapeutic regimen for surgical patients.
Palliative Chemotherapy for Metastatic CRC
Most advanced CRCs are aggressive. After initial treatment, approximately 20% to 50% of patients with localized CRC will develop stage IV mCRC45,46. The liver is the most common site of metastasis, followed by the lungs, peritoneum, and distant lymph nodes34,47. The prognosis for patients diagnosed with mCRC is typically unfavorable. The 5-year survival rate for patients diagnosed with stage I CRC is greater than 90%, whereas the 5-year survival rate for patients diagnosed with stage IV CRC is only 10% 48 . CRC liver metastasis PDTOs have been successfully constructed in several studies and have been shown to capture intra- and interpatient heterogeneity, demonstrating that CRC liver metastasis PDTOs can be effectively used for drug screening and the determination of chemotherapy resistance49–51. These findings suggest that PDTOs can predict the chemotherapy response and clinical prognosis of patients with liver metastases. Tao Tan et al.’s group collected tumor tissue samples from 169 CRC patients at four hospitals in Australia. They confirmed that the predicted drug response to PDTOs was highly correlated with patients’ clinical outcomes. They also identified hepatic metastases as the optimal site for sampling, with a 75% success rate for PDTO culture 52 . Treatment of peritoneal carcinomatosis (PC) secondary to CRC is equally challenging because of the heterogeneity of the tumor, which results in varying degrees of resistance to systemic and intraperitoneal chemotherapy. PDTOs may be able to inform clinical treatment decisions for individual patients with CRC-PC53,54.
Patients diagnosed with metastases are typically considered unresectable. However, in cases of limited metastases, surgical resection may be feasible after tumor shrinkage. Chemotherapy should be strongly considered in such patients to reduce metastases and convert them to resettable foci. Importantly, some patients may experience disease progression during chemotherapy, thus losing the opportunity for surgical resection. Predicting chemotherapy efficacy via PDTOs has the potential to improve the success of translational therapies. This enables the patient’s tumor to shrink within a relatively short timeframe, thereby facilitating surgical removal.
Neoadjuvant Chemoradiotherapy
Neoadjuvant chemoradiotherapy (nCRT) followed by tumor resection is the standard treatment for locally advanced rectal cancer (LARC) and provides excellent local disease control in patients who respond well. The goal of nCRT is to decrease local recurrence, increase the likelihood of surgical resection, improve anal preservation, and prolong disease-free survival in patients. Predicting the degree of tumor regression after nCRT for LARC is important for tailoring treatment strategies. A clinical study conducted by Hsu et al. suggested that analyzing the tissue radiosensitivity inherent in PDTOs may provide metrics to predict the response to radiation in patients receiving neoadjuvant therapy for rectal cancer. This approach could allow for pretreatment stratification of patients who may benefit from it 55 . In a small collaborative clinical trial, Park et al. 56 showed that patient radiosensitivity was positively correlated with PDTOs and that a radiosensitivity model based on PDTOs could lead to more accurate treatment of rectal cancer patients. Yao et al. 57 generated a living organoid biobank from LARC patients treated with nCRT, suggesting that PDTOs can predict the response of LARC patients in the clinic. Notably, not all patients respond well to nCRT, and postoperative adjuvant chemotherapy (AC) is the most common recommendation for treating a poor response to nCRT. Xue et al. established PDTOs using irradiated rectal cancer specimens that did not respond well to nCRT. They tested the sensitivity of the PDTO to chemotherapy regimens and confirmed that the drug testing ability of the PDTO predicted the benefit of postoperative AC in LARC patients who did not respond well to nCRT 58 .
Currently, the therapeutic concept of nCRT is widely accepted. However, its clinical application is constrained by various factors, including the lack of a uniform dosage and implementation program. In addition, there is no gold standard for evaluating the efficacy of neoadjuvant chemotherapy, and barium contrast and computed tomography (CT) scans are still not accurate enough for this purpose. The utilization of PDTOs in clinical practice has the potential to address these challenges. PDTO can assess the efficacy of radiotherapy and chemotherapy before and after nCRT, thus facilitating accurate and effective treatment and personalized treatment planning.
Targeted Therapy
Numerous patients with mCRC have received targeted therapies to treat their disease, with significant patient benefit 59 . However, progress in the targeted therapeutic area of mCRC has been slow because of a relative lack of biomarkers 60 . Using PDTOs, Boos et al. 61 demonstrated that a therapeutic strategy combining epidermal growth factor receptor (EGFR) pathway blockade with aurora kinase A (AURKA) inhibition may be effective in the second-line treatment of chemotherapy-resistant CRC liver metastases characterized by acquired KRAS mutations and increased expression of AURKA/c-MYC, a potential therapeutic target. AURKA may be a potential therapeutic target in liver mCRC. By analyzing a functional model based on PDTOs, Hu et al. 62 identified DACH1 as a component of BMP signaling during intestinal tumorigenesis, which may be a potential prognostic marker and therapeutic target for patients with CRC. Zaki et al. reported that NLRP12, a member of the NOD-like receptor family, is an effective negative regulator of the Wnt/β-catenin pathway and inhibits the development of CRC by inhibiting the Wnt/β-catenin pathway. The NLRP12/STK38/GSK3β signaling axis may be a promising target for the treatment of CRC 63 . These findings provide new ideas for the treatment of mCRC. In the near future, as more potential targets are identified on the basis of PDTOs, patients with advanced CRC will have broader choices of targeted agents and increased hope of survival.
Importantly, PDTOs remain deficient in pharmacodynamic testing. The efficacy of drugs that target EGFR, such as cetuximab, in the treatment of advanced colon or rectal cancer can be validated by PDTOs. Conversely, drugs that block the action of vascular endothelial growth factor (VEGF), such as bevacizumab, cannot be validated due to the lack of blood vessels in PDTOs. Regorafenib has been demonstrated to inhibit tumor growth and treat mCRC by inhibiting tumor angiogenesis, suppressing tumor cell neogenesis, and protecting the tumor microenvironment64,65. However, no studies have yet shown that regorafenib inhibits CRC PDTOs. Ramzy et al. proposed a new method for designing personalized treatment plans for CRC patients by combining organoid test results with mathematical modeling. Using their method, they reported that a synergistically optimized drug combination of regorafenib, vemurafenib, palbociclib, and lapatinib inhibited cell viability significantly better than clinical doses of FOLFOXIRI in PDTOs from patients with liver metastases 66 . The emergence of this approach has allowed more varied and optimized combinations of drugs to be tailored to the patient’s needs within the time available in the clinic rather than being restricted to drug guidelines.
Patients are not necessarily sensitive to targeted drugs derived from genetic sequencing due to the presence of drug resistance. The results of the PDTO drug sensitivity assay truly reflect the state of the tumor tissue under the effect of drugs in the 3D dimension; thus, there may be a discrepancy between the organoid drug sensitivity assay and the effective drugs shown by the gene sequencing results. The results obtained by testing the sensitivity of PDTOs to drugs can avoid the risk of blind drug administration and improve the efficacy of drugs, thereby reducing the financial burden on patients.
Third-Line and Follow-Up Treatment
Many people with stage IV CRC now have a good chance of survival owing to the current standard of care. However, some patients still progress after first-line and second-line treatment and urgently need third-line treatment. How to choose the one with the greatest survival benefit for the patient from the many drugs available is a problem that every doctor has to face. Patients are often in poorer health after frontline treatment, and safety is an important part of the decision-making process when choosing a medication. Zeng et al. created a patient-derived CRC PDTO library and used it as an in vitro model to evaluate thermotherapy synergistically with chemotherapeutic agents. They reported that, among common anticancer agents, thermotherapy enhanced the effect of raltitrexed the most 67 . Chen et al. constructed PDTOs from CRC patients. These findings indicate that curcumin primarily regulates the biosynthesis of phenylalanine, tyrosine, and tryptophan, as well as nicotinic acid and nicotinamide metabolism, and purine metabolism in CRC PDTOs. These findings shed further light on the role of curcumin in CRC 68 . Some studies suggest that gemcitabine may be suitable for CRC patients who do not respond to OX-based regimens such as FOLFOX 69 . However, we cannot guarantee that every patient with OX-resistant CRC will respond well to gemcitabine before it is used. If it is possible to use PDTO models to determine exactly how effective gemcitabine is in an individual, this problem can be solved, allowing precise dosing and avoiding waste.
These studies show that CRC PDTOs can be excellent surrogates for patients, effectively testing newly developed drugs and providing feedback on drug efficacy on behalf of the patients themselves. In cases where standard treatments have been unsuccessful, in instances of rare or refractory tumors where standard treatments are currently unavailable, and in cases where patients are deemed to be at a lower level of risk according to existing guidelines and are therefore unlikely to benefit from guideline-recommended regimens, PDTO drug sensitivity testing can be employed to rapidly identify effective and safe drugs, providing a reference for their drug selection, avoiding the risk of blind drug administration, and alleviating the patient’s financial burden.
Treatment of Elderly Patients
Elderly CRC patients constitute a unique population. They experience a significant decrease in their body’s stress capacity, resulting in lower tolerance to chemotherapy, radiotherapy, and other related treatments. In addition, these patients often have more comorbidities and are more susceptible to infections. As a result, chemotherapy is less frequently used in CRC patients above 80 years of age than in younger patients 70 . Assessing whether the benefits of treatment outweigh the risks of complications or death is crucial in the treatment of older patients with CRC. Before administering chemotherapy to elderly CRC patients, it is crucial to conduct a comprehensive assessment of their tolerance to chemotherapy. The benefits and drawbacks of chemotherapy should be carefully considered, and appropriate strategies and protocols should be developed to determine a suitable chemotherapy dose that minimizes toxicity. This approach may reduce chemotherapy-related damage to organ function, increase the cure rate, prolong the survival period, and improve quality of life71,72. However, whether to reduce the dosage of a specific chemotherapy drug for each elderly patient and whether to use a single-drug or a two-drug combination regimen are mostly based on the clinical experience of doctors, and there is no uniform standard for the specific adjustment method.
As a reliable preclinical model, PDTOs can be used to assess chemotherapy tolerance in elderly CRC patients. This study provides clinicians with a valuable reference for decision-making and the selection of the most effective treatment regimen while ensuring the safety of chemotherapy. Clinical trials often exclude elderly patients, resulting in incomplete staging and treatment-related information. However, older age should not be considered a limiting factor for treatment. Instead, treatment decisions should be carefully assessed in the context of the individual patient’s situation. The use of PDTOs in clinical practice may help fill the gap in clinical research for elderly individuals.
Limitations of PDTOs
As previously stated, the PDTO model provides an efficient platform for evaluating drug sensitivity specific to a patient in vitro. These findings can aid in making personalized therapeutic decisions for patients with advanced CRC. However, there are still some challenges to overcome before the use of CRC PDTOs can be translated into clinical practice.
First, there is a paucity of uniform quality control standards in the fields of tumor organoid construction, identification, and drug sensitivity testing, which may affect the accurate assessment of tumor biology 73 . A review of the literature reveals a variety of methods for tumor-like organ culture, including the engineered matrix gel cell scaffold method, the microfluidic culture method, the rotating bioreactor method, and others. At present, the engineered matrix gel cell scaffold method is the most widely utilized among these techniques. Li et al. 74 discovered that organoids are more challenging to produce from tumors with MSI, BRAF mutations, hypodifferentiation, and/or mucinous phenotypes. This discovery may aid in identifying which CRC tumors are more likely to generate PDTOs and, as a result, benefit from PDTO drug trials. Marinucci et al. reported that adding primocin to tissue washing solutions before tissue processing eliminates the risk of microbial contamination in PDTO cultures. They also reported that the use of penicillin/streptomycin negatively affects organoid growth 75 . This result helps to improve the success rate of PDTO culture in CRC patients. Organoid scores can serve as a simulation model for determining the next therapeutic step through drug screening and aiding in the standardization of predicting cancer treatment response. Tang et al. was the first to define the IC50 threshold for PDTO drug testing to effectively differentiate between chemosensitive and nonsensitive CRC patients and predict survival benefit 76 . Data collection methods for PDTO drug sensitivity testing are also constantly being updated. At present, the commonly used methods include light microscope analysis, ATP metabolite detection, CCK8 and MTT detection, and fluorescence staining identification50,77,78. The light microscope analysis is intuitive and does not damage the PDTO and can be observed in real time; however, its flux is low, and the requirements for image acquisition machines are high and time-consuming. The detection of ATP metabolites is fast and simple and is suitable for high-throughput screening. However, after reagents are added, the cells are lysed, and real-time dynamic monitoring of the cells cannot be carried out. The detection of CCK8 and MTT is convenient and fast, and the sensitivity is high. However, the volume and color of the medium have a great influence on its determination, and the detection error of this method is therefore large. The fluorescence staining method is simple to perform, the results are intuitive, and it is convenient for statistical analysis. However, adjusting the proportion of dyes needed to achieve the best staining effect is necessary, and monitoring the growth of organoids in real time is impossible. Hence, large-scale and multicenter prospective studies are of paramount importance for enhancing the clinical application value of drug susceptibility testing of PDTO 79 . The establishment of PDTOs and the implementation of standardized drug testing procedures will facilitate their integration with translational medicine and precision medicine.
Second, at present, CRC PDTOs are grown mainly from epithelial tissue, and there is a paucity of mature culture systems comprising suitable substrates and immune systems. The tumor microenvironment plays a pivotal role in the development of drug resistance and tumor progression. Several studies have indicated that PDTOs that grow with fibroblasts display greater tumor cell heterogeneity and more closely resemble in vivo tumor morphology than do single tumor cultures80–82. Jiang et al. 83 explored the treatment strategy for gemcitabine resistance through a macrophage‒organoid coculture model. The implementation of coculture methodologies has facilitated the approximation of PDTOs to the authentic in vivo state. However, inherent limitations are unavoidable in any model whose composition and functionality cannot attain the true in vivo level. The optimization of multitype cell coculture techniques based on tumor-like organoids is currently being investigated. The results of this research will facilitate the development of drug sensitivity assays for a broader range of drugs or drug combinations. Furthermore, the efficacy of in vitro organoid modeling varies across different tumor types, reflecting significant biological heterogeneity. For example, in vitro organoid culture of epithelial tumors has been documented with greater frequency and higher success rates. In contrast, in vitro organoid culture of nonepithelial tumors has been documented to occur less frequently. In a recent development, the research team led by Lisa E. Wagar at the University of California and Ash A. Alizadeh at Stanford University successfully constructed an organoid model for the evaluation and targeting of the immune microenvironment of follicular lymphoma (FL) 84 . This study offers a robust foundation for advancing the understanding of FL and represents a significant advancement in the construction of organoids for tumors of nonepithelial origin.
Third, the large-scale application of PDTOs still necessitates considerable capital investment. Furthermore, the technology involved is relatively complex and requires a high level of technical expertise. Pinho et al. developed the OrganoidChip, a low-cost microfluidic device suitable for culturing and amplifying organoids. Culturing CRC PDTOs in the OrganoidChip resulted in a significant increase in viability and proliferative activity 85 . OrganoidChips have the potential to perform individualized PDTO modeling and drug screening on a large scale. Li et al. 86 used Z-stack imaging to image 3D PDTOs, established a 3D PDTO imaging analysis method, and demonstrated the feasibility of using a PDTO-based platform for high-throughput drug screening analysis. These studies combine cutting-edge life science and engineering technologies to create high-throughput, highly bionic in vitro model–building platforms that can integrate a range of experimental processes, such as organoid sorting, cultivation, observation, detection, and analysis. These platforms reduce labor costs, improve the efficiency of experiments, and promise to make clinical treatment protocols more predictable.
Conclusions
Compared with traditional disease models, PDTOs offer several advantages, including rapid cultivation, a high success rate, and high throughput. The use of PDTOs in CRC allows for the prediction of responses to different treatment modalities at various stages of disease progression. This has the potential to reduce adverse drug reactions and the emergence of resistance associated with clinical drugs, facilitate evidence-based clinical decision-making, and guide CRC patients in the selection of personalized medications, thereby advancing the individualized treatment of CRC.
Footnotes
Author Contributions
All authors were involved in the conceptualization and design of the study. Yiran Li and Jun Yan conceptualized the article; Yiran Li, Wei Wu, Jiaxin Yao, and Suidong Wang conducted the literature search; Yiran Li and Wei Wu drafted the article; and Jun Yan performed the critical revision work.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Grant No. 82273360), the Clinical Research Program of Nanfang Hospital of Southern Medical University (Grant No. 2022CR003), the President’s Fund of Nanfang Hospital of Southern Medical University (Grant No. 2023B053), the Innovative and Entrepreneurial Training Program for University Students (Grant Nos. 202212121011, S202212121092, S2022121104 and 202312121318), and the Fujian Medical Innovation Project (Grant No. 2022CXA028).
