Abstract
Tissue repair is an extremely crucial part of clinical treatment. During the course of disease treatment, surgery, chemotherapy, and radiotherapy cause tissue damage. On the other hand, Normal tissue from accidental or therapeutic exposure to high-dose radiation can cause severe tissue damage. There is an urgent need for developing medical countermeasures against radiation injury for tissue repair. Tissue repair involves the regeneration, proliferation, differentiation, and migration of tissue cells; imbalance of local tissue homeostasis, progressive chronic inflammation; decreased cell activity and stem cell function; and wound healing. Although many clinical treatments are currently available for tissue repair, they are expensive. The long recovery time and some unavoidable complications such as cell damage and the inflammatory reaction caused by radiotherapy have led to unsatisfactory results. Extracellular vesicles (EVs) derived from mesenchymal stem cells (MSCs) have similar tissue repair functions as MSCs. In tissue damage, EVs can be used as an alternative to stem cell therapy, thereby avoiding related complications such as immunological rejection. EVs play a major role in regulating tissue damage, anti-inflammation, pro-proliferation, and immune response, thus providing a diversified and efficient solution for the repair of disease- and radiotherapy-induced tissue damage. This article reviews the research progress of mesenchymal stem cell-derived EVs in promoting the repair of tissue including heart, lung, liver, intestine, skin, blood system, central nervous system, and tissue damage caused by radiotherapy, thereby aiming to offer new directions and ideas for the radiotherapy and regenerative applications.

Introduction
Mesenchymal stem cells (MSC) are multipotent stem cells obtained from various sources and can be detected in almost all organs and tissues of the human body, such as the bone marrow, liver, gingival tissue, placenta, bursa, adipose tissue, and umbilical cord. MSCs can also be obtained through the differentiation of PSCs in vitro. These cells have the characteristics of strong proliferation, high differentiation, good stability, and strong immunosuppression 1 . MSCs are crucial for immune regulation, hematopoiesis, tissue repair and regeneration, and anti-apoptosis 2 . MSCs used in clinical trials are frequently derived from the adult bone marrow, followed by adipose tissue, umbilical cord, and placenta. The combination of stem cell therapy and drugs has a broad prospect in clinical disease treatment and is increasingly favored in regenerative medicine. MSC-derived EVs (MSC-EVs) were found in sheep reticulocytes in 1980. They were cup or plate shaped; small vesicles having a diameter and density of 30 to 150 nm and 1.13 to 1.19 g/mL, respectively; and were also known as luminal vesicles 3 . These EVs are formed by the inward indentation of the cell membrane and budding and contain proteins (enzymes, TSG101, TGF-β, etc), DNA, miRNA, and mRNA 4 . They are enriched with tetraspanin (CD9, CD63, CD81, and CD82) that play a crucial role in facilitating EVs transport. Membrane proteins such as CD9, CD63, CD81, and CD82 are the most commonly used EVs marker proteins. They can be degraded by lysosomes or released to bind to receptor cells and exert their effects. (Fig. 1A). EVs were initially considered one of the ways through which cells excrete metabolites. However, on studying the source, composition, and mechanism of action of EVs, they were found to have various functions. EVs not solely a key player in the plethora of physiological events of the healthy human body, but also associated with several pathological conditions, such as cancer 5 . EVs have been demonstrated to influence a multitude of processes that in pathological conditions including cellular proliferation, angiogenesis, migration, modulate immune response and facilitate tissue regeneration 6 . EVs contents are believed to be derived from parental cells, and so their functions can, to a certain extent, be similar to those of parental cells 7 . MSC-EVs have abilities similar to those of stem cells and exert effective repair effects on tissue damage in the blood system, heart, lung, liver, intestine, and skin (Fig. 1B). The research on EVs in degenerative diseases, cardiovascular diseases, and autoimmune diseases has achieved good results. Due to the uncontrollable process of global aging, people are paying more and more attention to the degree of cure of diseases. Tissue damage caused by different diseases and radiotherapy has made identifying more effective prevention and treatment methods a hot research direction. Radiotherapy is one of the important methods in the treatment of tumor, but it can also cause adverse reactions in normal tissues within the irradiation range, such as decreased peripheral blood count, skin damage, and radiation pneumonia. Owing to their versatility, EVs provide various repair solutions for disease-induced tissue damage and have a certain prospect in the application of tissue regeneration. In this review, we explore the roles of MSC-EVs in tissue repair after injury.

Functions of mesenchymal stem cell-derived extracellular vesicles (MSC-EVs). (A) Extracellular vesicles are formed in a budding fashion by indented mesenchymal stem cell membranes. (B) EVs contain biologically active molecules including nucleic acids, proteins, lipids or membrane receptors that bind to target cells and play a role in radiation injury and regeneration and repair of heart, lung, liver, intestine, skin, blood system, central nervous system in the human body. Such as promote proliferation, suppress inflammation, attenuate apoptosis and information transfer.
Role of MSC-EVs in Myocardial Injury
The heart is a crucial blood supply organ in humans. Myocardial injury-induced heart disease is among the main causes of heart failure and death in humans. Myocardial infarction (MI), acute and chronic heart failure, atrial fibrillation (AF), pulmonary embolism, endocarditis and myocarditis, cardiac trauma (surgery or accident), chronic stable coronary artery disease, and cardiac cell damage related to cardiotoxic drug-induced effects are different types of heart diseases 8 . In addition, radiation-induced heart disease is a common complication, especially in patients receiving thoracic radiotherapy. Radiotherapy can induce cardiac tissue damage and fibrosis, thereby causing cardiac dysfunction and cardiovascular disease 9 . The coronary heart disease (CHD) risk was higher in patients who received left anterior chest radiotherapy 10 . At present, the traditional treatment of heart disease includes cardiovascular drugs and cardiac rehabilitation, but its effect is limited. Various stem cells, such as embryonic stem cells, induced PSCs (iPSCs), cardiac progenitor cells, MSCs, and cardio sphere-derived cells, can secrete EVs. EVs mediated intercellular communication is commonly involved in the pathophysiological process of various heart diseases 11 . MSC-EVs play a crucial role in myocardial injury (Table 1).
Biological Effects of Extracellular Vesicles in Heart Disease.
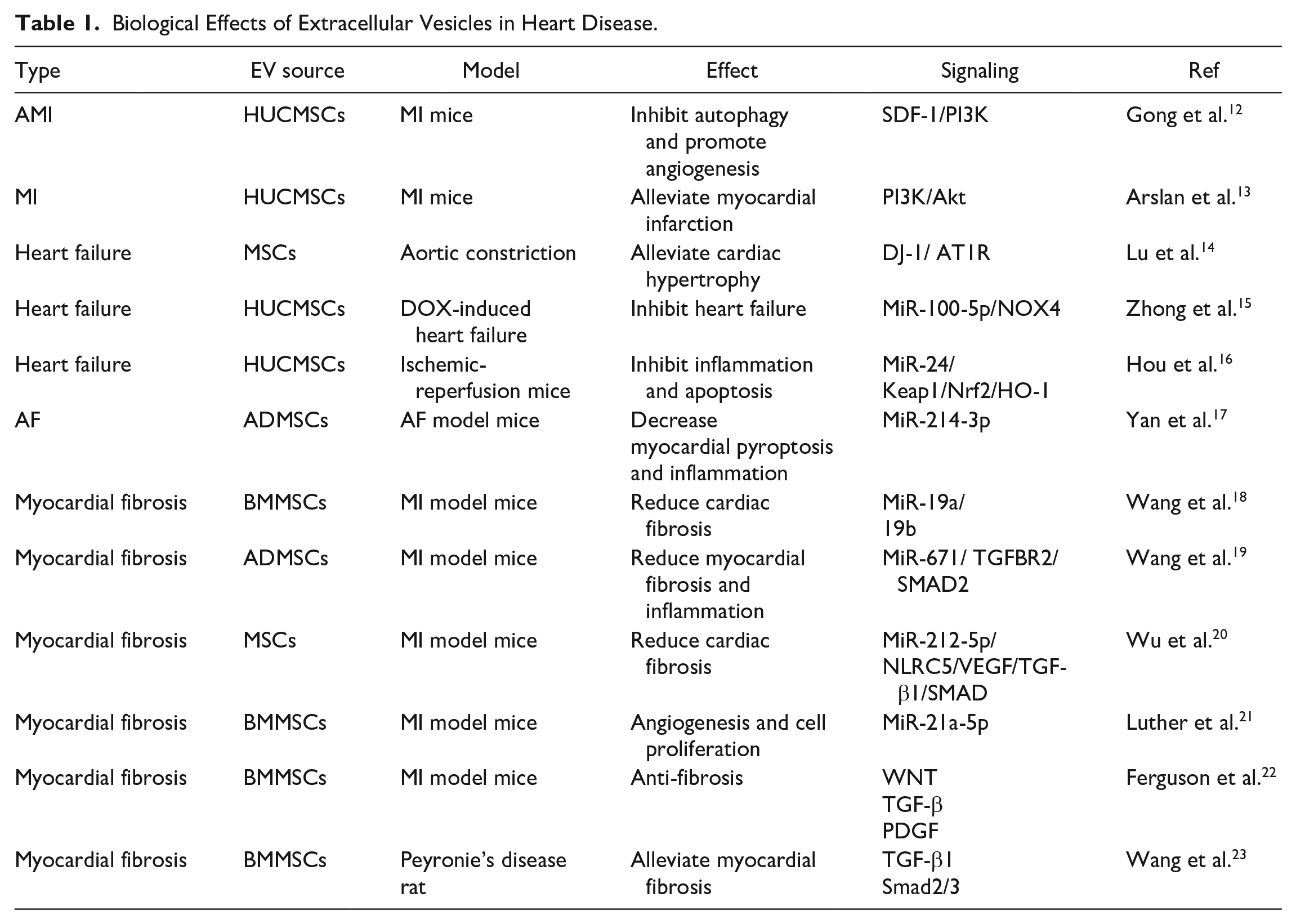
Acute MI (AMI) is caused by plaque formation on inner arterial walls, resulting in reduced blood flow to the heart and hypoxia-induced myocardial damage. When blood stops flowing normally to a part of the heart and the myocardium is injured by a lack of oxygen, MI occurs. The released EVs inhibited the apoptosis of cardiomyocytes, promoted the angiogenesis of endothelial cells, and promoted SDF-1 upregulation. SDF-1 is a crucial chemokine essential for tissue repair and angiogenesis and was found to be associated with CHD. More importantly, SDF-1 injection could attract bone marrow-derived progenitor cells in the blood, which possibly differentiated into cardiovascular cells to support angiogenesis and improved cardiac function. Compared with SDF-1-NC plasmid-transfected MSCs, interleukin (IL)-6β, IL-8, IL-1, and TNF-α expression in SDF-1-overexpressing MSCs significantly decreased, whereas vascular endothelial growth factor (VEGF) expression significantly increased. SDF-1 overexpression of MSC-EVs can inhibit the apoptosis of ischemic cardiomyocytes. SDF-1 can promote the regeneration of cardiac endothelial microvessels in MI mice, and SDF-1 overexpression in MSCs can protect the cardiac function of these MI mice 12 . Injection of MSC-EVs reduced the infarct volume by 45% and reduced systemic inflammation. Meanwhile, in the AMI rat model, myocardial infusion of MSC-EVs improved cardiomyocyte contractility and reduced infarct size 24 . This suggested that EVs play a cardioprotective role by preventing cardiac remodeling in the post-MI period. Studies have shown that LncRNA KLF3-AS1 in HUCMSC-EVs ameliorates MI through miR-138-5p/Sirt1 axis 25 . This improves our understanding of the prevention and treatment of human myocardial infarction patients. Heart failure is caused by various causes and is the result of cardiovascular disease. The heart failure-associated morbidity and mortality are higher in the elderly population. Ventricular remodeling, including cardiomyocyte hypertrophy, interstitial fibrosis, and activation of the renin–angiotensin system, is a fundamental pathological manifestation of heart failure. Parkinson disease protein 7 (PARK7/DJ-1) contained in EVs derived from hypoxic MSCs alleviates cardiac hypertrophy by inhibiting mitochondrial dysfunction and preventing AT1R-associated protein (ATRAP) degradation 14 . EVs miRNA can affect ventricular remodeling-mediating pathological processes. In a study based on the use of HUCMSC-EVs, HUCMSC-EVs inhibit doxorubicin-induced heart failure through the miR-100-5p pathway 15 . Moreover, miR-24 delivered by HUCMSC-EVs can promote M2 polarization of macrophages and enhance the protective effect on myocardial injury 16 . AF is associated with the size of the AV block and the degree of fibrosis. Some miRNAs present in EVs can regulate myocardial fibrosis to treat AF 26 . According to a study, LncRNA XIST shuttled by mouse adipose tissue-derived mesenchymal stem cell-derived extracellular vesicles (ADMSC-EVs) suppresses myocardial pyroptosis in AF by disrupting miR-214-3p-mediated Arl2 inhibition 17 . Non-coding RNAs (ncRNAs) are critical regulators of cardiac function and disease 27 . Being a vital cargo of EVs, ncRNAs (EV-ncRNA) play a crucial role in the AF pathological process and can be used as a diagnostic marker or therapeutic method. Changes in the dynamics of myocardial collagen fibers, including excessive accumulation and increase in the fiber concentration, or changes in the fiber composition cause alterations in the normal tissue architecture, thereby leading to myocardial fibrosis. According to some studies, MSC-EVs are safer and more efficient than stem cell therapy, and they are one of the novel non-cellular biological therapies with great potential 28 . MSC-EVs has been confirmed the benefits in the treatment of myocardial fibrosis. Wang et al. 18 suggested that EVs from bone marrow-derived mesenchymal stem cell (BMMSC-EVs) can promote recovery of heart function via miR-19a/19b to decrease myocardial fibrosis in the MI model. In an animal model, ADMSC-EVs treatment significantly enhanced viability while reduced apoptosis of cardiomyocytes, and in reduced myocardial fibrosis and inflammation both in vitro and in vivo 19 . Wu et al. 20 found MSC-EVs carrying miR-212-5p in mediating NLRC5/VEGF/TGF-β1/SMAD axis to relieve cardiac fibrosis. This finding provide mechanistic of miR-212-5p-containing EVs from MSCs in reducing myocardial fibrosis.
Although the evidence for the significance of EVs in cardiovascular diseases is expanding, the clinical use of EVs is still strongly limited, particularly by technical difficulties. In fact, most current studies have used in vitro and/or animal models because RNAs and proteins carried by MSC-EVs are diverse and complex, and studies investigating the characteristics of EVs from cardiovascular patients are still scarce. Additional studies are warranted to optimize the efficacy, safety, and practical application issues related to EVs. In summary, MSC-EVs have great therapeutic potential to mediate cardioprotection in myocardial injury. At present, when we apply EVs in myocardial injury, how to expand their advantages and make up for their defects is a crucial consideration in the future.
Repair Effects of MSC-EVs in Lung Injury
Histologically, the lung is a very complex organ, and its main function is to deliver oxygen from the atmosphere into the blood circulation, while the lung tissue must be kept relatively free of pathogens. The alveolar epithelium is the mechanical barrier protecting the lungs from the environment. It is actively involved in the lung’s immune response and helps maintain fluid balance at the alveolar surface 29 . Lung injury is a common complication associated with thoracic radiotherapy30,31. MSC-EVs can participate in the repair of lung injury through various mechanisms. Recent studies have focused on the role of EVs in regulating normal and pathophysiological cellular activities, including radiotherapy-induced inflammatory and injury responses. MSCs and their EVs can attenuate lung epithelial cell senescence and stimulate apoptosis inhibition. MSC-EVs administered to the tail vein of mice having LPS-induced acute lung injury restored the proliferative capacity of alveolar epithelial cells and lung function through mitochondrial delivery 32 . In the LPS-induced acute lung injury (ALI) mouse model, MSC-EVs were given via tail vein significantly reduced inflammatory cell infiltration and inhibited inflammatory cytokines, repaired the pulmonary endothelial–epithelial barrier, and improved pulmonary edema within 48 h of instillation. MSC-EVs could also alleviate lung injury caused by pulmonary fibrosis 33 . In the current study, EV-miR-466f-3p derived from mouse MSCs prevented radiation-induced epithelial–mesenchymal transition (EMT) by inhibiting AKT/GSK3β via c-MET to treat pulmonary fibrosis 34 . MiR-29b-3p overexpression by BMSC-derived EVs effectively inhibited the proliferation, migration, differentiation, and invasion of fibroblasts by inhibiting FZD6 expression, thereby contributing to the anti-fibrotic effect of lung fibroblasts 35 . Various harmful factors induce damage and apoptosis of alveolar epithelial cells, often accompanied by mitochondrial dysfunction. Elevation of AFC in alveolar epithelial cells (AECs) is a consequence of mitochondrial dysfunction that leads to further injury 36 . Direct co-culture of ASMC airway smooth muscle cells (ASMC) with induced pluripotent stem cell-derived mesenchymal stem cells (iPSC-MSCs) protected the former from cigarette smoke medium (CSM) induced production of mitochondrial reactive oxygen species (ROS), mitochondrial depolarization, and apoptosis 37 . EVs can inhibit inflammatory responses and reduce cell apoptosis and injury. MSC-EVs and their miRNAs can reduce the expression of inflammatory cytokines associated with radiation-induced lung injury 38 . MSC-EVs can also mitigate the lung cell dysfunction- and injury-associated inflammatory response to preserve or restore cell function, thus promoting cell proliferation and tissue regeneration. Damaged and dysfunctional cells frequently secrete EVs into the serum or bronchoalveolar lavage fluid with altered cargo. These EVs can be used to diagnose and predict ALI/ARDS development. HUCMSC-EVs exert protective effects against lung injury in neonatal mice 39 . The soluble keratinocyte growth factor and angiopoietin-1 secreted by MSCs have repair and regenerative properties. They protect the alveolar epithelium and endothelium from injury. MSC-EVs reduce IL-6 and TNF-α levels in acute lung injury by transferring miR-124-3p and inhibiting the expression of the purinergic receptor P2X7 40 . MSC-EVs can alleviate ischemia/reperfusion injury-induced pulmonary edema and dysfunction by transferring miR-21-5p. MSC-EVs can also participate in the repair of lung injury by regulating the immune system, promoting angiogenesis, and inhibiting pulmonary fibrosis. Human BMMSC-EVs can modulate the activity of immune response-related cells. In Shentu et al.’s 41 study, BMMSC-EVs inhibited TGF-β1-induced myofibroblast differentiation through a mechanism dependent on blocking the Thy-1–β-integrin interaction and preventing the inhibition of myofibroblast differentiation. In another study, Hyperoxia-exposed mice presented with pronounced alveolar fibrosis and pulmonary vascular remodeling, which was effectively ameliorated by MSC-EVs suppressing the pro-inflammatory “M1” state and augmenting an anti-inflammatory “M2-like” state 42 . In addition, MSC-EVs have good stability and low immunogenicity, and they avoid some side effects that may prevail when using whole-cell therapy. MSC-EVs can improve radiation-induced complications in normal lung tissues 43 .
As nanoscale particles, MSC-EVs are less immune reactive than pluripotent stem cells, and have no potential to differentiate into somatic cell lineages or tumor formation. They are convenient to collect, low immunogenicity, and no risk. MSC-EVs participate in the repair of lung injury through a variety of mechanisms and reduce the inflammatory response related to lung cell dysfunction and injury. In the coming years, MSC-EVs have broad application prospects in lung injury repair.
Repair Effects of MSC-EVs in Liver Injury
The liver is an important organ in humans, and its rapid and abundant regenerative capacity allows nearly two-thirds of the liver to be resected surgically and the remaining liver to be restored to its original size rapidly 44 . The incidence of liver diseases is high. Stimulants such as drugs, alcohol, and radiotherapy-associated complications can cause chronic/acute liver injury and inflammation, which can subsequently lead to liver failure and cirrhosis, and even hepatocellular carcinoma. Orthotopic liver transplantation is the only effective treatment for cirrhosis and liver failure. Therefore, novel and effective therapies are urgently required to address these medical problems 45 . Various factors, including the role of MSC-EVs, affect liver injury repair, a highly coordinated process 46 . MSC-EVs are associated with intercellular communication, and fluctuations in the levels of MSC-EVs may constitute a new diagnostic tool 47 . MSC-EVs can participate in liver injury repair in various ways. Firstly, MSC-EVs inhibit the inflammatory response during liver injury and reduce the release of inflammatory mediators and cell apoptosis. In a study, MSC-EVs significantly improved the liver function (reflected through ALB, ALT, AST, ALP, and γ-GT levels), repaired injured liver tissue (reflected through the damaged area and Ishak score), and reduced the expression and levels of inflammatory factors (TNF-α, IL-1β, IL-6, and interferon (IFN)-γ). Moreover, they increased the anti-inflammatory factor (IL-10) levels 48 . In the subgroup analysis of liver disease models, MSC-EVs exerted therapeutic effects against acute (ALI, I/RI) and chronic (liver fibrosis, NAFLD, AIH) liver diseases 49 . AML12 cells (a mouse liver cell line, in vitro) and D-GalN/LPS-induced fulminant hepatic failure (FHF) mice (in vivo) were treated with EVs derived from human menstrual blood mesenchymal stem cells (MenSC-EVs), which migrated to the injury site. The results revealed that MenSC-EVs significantly improved liver function, increased survival, and inhibited hepatocyte apoptosis at 6 h after transplantation. MenSC-EVs also reduced the number of hepatic monocytes and the level of the active apoptotic protein caspase-3 in the damaged liver 50 . BMMSC-EVs prevent hepatocyte apoptosis, reduce biochemical and histological liver damage, and improve the survival of mice following D-galactosamine/TNFα-induced acute liver failure 51 . In a liver injury model, higher expression of proliferative proteins (PCNA and cyclin D1) was observed in the EVs treated group than in the control group. EVs also upregulated BCL-XL protein expression, thereby inhibiting APAP- and H2O2-induced hepatocyte apoptosis 52 . In mice with CCl4-induced liver injury, HUCMSC-EVs reduced serum MDA levels, proinflammatory cytokine secretion, hepatic 8-OHdG expression, and apoptosis 53 . Moreover, MSC-EVs can stimulate hepatocyte proliferation and promote liver function recovery. After partial hepatectomy in rats, miR-124 derived from HUCMSC-EVs can downregulate Foxg1 and promote liver regeneration 54 . Tan et al. 52 suggested that MSCs and their EVs can promote liver regeneration through transplantation into the damaged liver tissue, trans-differentiation into hepatocytes, and secretion of nutritional and immunomodulatory factors. During liver failure, EVs from mouse bone marrow-derived mesenchymal stem cells can inhibit the storm of inflammatory factors, inhibit apoptosis, promote angiogenesis, and provide energy support. They also promote hepatocyte proliferation and contribute to biliary tract recovery 51 . BMSC-EVs could regulate the p53 signaling pathway through PTEN to inhibit cell apoptosis, promote hepatocyte proliferation and ameliorate liver ischemia-reperfusion injury 55 . HUCMSC-EVs alleviate rat hepatic ischemia-reperfusion injury by suppressing oxidative stress and neutrophil inflammatory response. Meanwhile, HUCMSC-EVs prevented hepatocytes from oxidative stress-induced cell death in vitro 56 . These findings suggest a potential liver regeneration mechanism that could be used for novel therapeutic approaches for acute and chronic liver diseases 57 . MSC-EVs also have immunomodulatory and anti-fibrotic effects, which can help prevent chronic liver injury and liver fibrosis. According to Li et al., HUCMSC-EVs reduced surface fibrous cysts and softened their texture, thereby reducing liver inflammation and collagen deposition in CCl4-treated cells. HUCMSC-EVs ameliorate liver fibrosis by inhibiting hepatocyte EMT and collagen production 58 . Qu et al. reported that miR-181-5p-containing EVs inhibited the STAT1/Bcl-3/Beclin 2 pathway and thus increased autophagy and reduced TGF-β4-induced liver fibrosis in HST cells and CCL1-induced liver fibrosis mouse model. The potent anti-fibrotic function of engineered adipose-derived stem cells (ADSCs) can selectively transfer miR-181-5p from the EVs to damaged hepatocytes, thereby leading to EVs mediated therapeutic delivery of liver disease-targeting miRNAs 59 . MiRNA-125b produced by human chorionic plate-derived MSCs regulates the expression of Hedgehog (Hh) signaling, promotes fibrosis regression, and ultimately contributes to liver regeneration 60 .
Unfortunately, there are few clinical studies on MSC-EVs in the treatment of liver diseases. Compared with whole cell therapy, MSC-EVs have better stability and low immunogenicity, cause no immune rejection, and alleviate various liver injury models. However, an effective treatment for restoring radiation-induced liver injury is lacking. The potential of MSC-EVs to treat liver diseases is exciting, but the difficulty is challenging. Although MSC-EVs are relatively easy to obtain, the number of in vitro passages of MSCs themselves is limited, which results in limited opportunities to obtain EVs. Future applications of MSC-EVs are most likely to be in combination with other drugs or systems. Therefore, the therapeutic mechanism of EVs intervention in hepatic disease must be further elucidated. Applying MSC-EVs as a therapeutic drug is challenging, and numerous studies on MSC-EVs are needed to explore the ultimate clinical application. Overall, the results of various studies suggest the strategy of using MSC-EVs for the treatment of liver injury has great potential.
Repair Effects of MSC-EVs in Intestinal Injury
Intestinal epithelial cells (IECs) serve as the mucosal immune barrier necessary for coordinating host–microbe interactions. MSC-EVs can treat mucosal barrier destruction-induced intestinal injury in various ways. MSC-EVs have an anti-inflammatory effect that can inhibit inflammatory response and reduce the extent of tissue damage. Patients with malignant tumors who are treated with pelvic radiotherapy develop complications such as intestinal mucosal injury and inflammation. MSC-EVs are associated with whole-body radiation-induced intestinal injury in mice 61 , and EVs coated polydatin nanoparticles reduce radiation-induced damage to intestinal cells and the expression of inflammatory factors 62 . This acts as a new therapeutic target for rescuing patients with intestinal radiation injury. Neonatal necrotizing small bowel conjunctivitis (NEC) is a life-threatening condition with up to 30% mortality in preterm infants 63 . EVs derived from AF-MSC and BMMSC have significantly reduced the incidence of NEC in experimental models 64 . MSC-EVs improve the intestinal epithelial barrier function with miR-34a/c-5p and miR-29b-3p through the Snail/Claudins signaling pathway 65 . Heme oxygenase-200 (HO-1)/BMMSC-EVs can reduce the inflammatory damage caused to IECs by reducing the high mobility group box 3 (HMGB3) through miR-200b. MiR-1b in heme oxygenase-200 (HO-1)-modified BMMSC-EVs targets the HMGB3 gene in IECs to alleviate their inflammatory response 66 . HUCMSC-EVs inhibit inflammation by regulating K48 and K63 to inhibit ubiquitin protein expression. MSC-EVs significantly attenuated inflammatory bowel disease (IBD) by upregulating IL-10 and IP10 levels, whereas by downregulating IL-1β, TNF-α, IL-6, and ubiquitin-coupling enzyme (E2M), NEDD8 activate E1 (NAe1), and ubiquitin-like modifier activating enzyme 3 (UBA3) levels. Western blotting results revealed that the expression levels of proteins such as K48, K63, and FK2 decreased in the EV-treated IBD mice compared with the control mice, indicating that EVs inhibit inflammation by inhibiting ubiquitin modification 66 . Treatment with MSC-EVs increased the levels of IL-10, a marker of M2 macrophages in colitis, downregulated inflammatory responses, maintained intestinal mucosal integrity, and alleviated intestinal fibrosis 67 . He et al. 68 found that MSC-EVs exerted their therapeutic efficacy by downregulating intestinal inflammation and apoptosis and promoting cell proliferation through the miR-455-5p/SOCS3/Stat3 pathway in radiation-induced intestinal injury. Other types of EVs also exert therapeutic effects on enteritis. BMSC-EVs injection resulted in a markedly decrease in interleukin-1β (IL-1β) and an increase in interleukin-10 (IL-10) expression in colitis model 69 . MiRNA-142-5p was highly expressed in the EVs of TBI-exposed donor mice and patients after radiotherapy. Furthermore, miR-142-5p protected IECs from radiation-induced apoptosis and death and mediated the protective effect of EVs against radiation enteritis by improving the intestinal microenvironment 70 . Furthermore, different types of MSC-EVs can alleviate intestinal injury by promoting IEC proliferation and migration, thereby stimulating intestinal regeneration and repair 71 . EVs can deliver bioactive substances through different mechanisms, thereby exerting their effects on the surrounding cells. Among them, small RNAs (including siRNAs and microRNAs) in EVs are crucial molecules involved in regulating cell proliferation. According to some studies, EVs contain specific small RNAs that can contact target cells through EVs 72 . They are delivered inside the target cells and regulate the gene expression of these target cells. These small RNAs can also affect target cell proliferation through different pathways, such as by inhibiting specific gene expression in target cells or promoting certain biological processes. Wang et al. have found that EVs from adipose-derived stem cells (ADSC-EVs) promoted lymphatic endothelial cells (LECs) proliferation, migration, and tube formation by regulating miRNA-132 pathway in the pathogenesis of IBDs. This found provide a novel target for IBDs 73 . MSC-EVs can activate colonic epithelial cell proliferation and macrophage proliferation, effectively eliminate ROS, regulate the expression of immune cytokines, and reduce inflammation, and thus, they exert therapeutic effects on mouse colitis models 74 . In addition, MSC-EVs have the ability to target areas of intestinal inflammation, interact with immune cells like macrophages, T lymphocytes, and modulate the characteristics and activities of immune cells by releasing bioactive factors, regulate immune responses and suppress inflammatory reactions 75 . Moreover, MSCs and their derived EVs can repair intestinal injury by changing the internal environment of the intestine through a modification in the level of intestinal capillary permeability, reduction in the levels of gut endogenous endotoxin, and repair of the intestinal barrier. During intestinal injury, intestinal endotoxins may be produced and enter the blood circulation, which causes an inflammatory response and injury. By regulating intestinal capillary permeability, EVs can reduce the level of gut-derived endotoxins 76 . Specifically, EVs release some bioactive substances that can enhance capillary tightness and reduce endotoxin penetration 77 . These substances possibly include cell membrane proteins, extracellular matrix proteins, and so on. Some specific types of EVs can affect vessel wall permeability through endogenous or exogenous pathways 78 . These EVs can release several bioactive molecules, such as platelet-derived growth factor, VEGF, tissue factor, and TGF-β. These bioactive molecules can regulate the function of vascular endothelial cells (VECs), thereby affecting capillary permeability. EVs can affect capillary permeability by interacting with other cells, such as inflammatory cells and leukocytes. Furthermore, MSC-EVs can reduce radiation-induced intestinal mucosal permeability as well as destruction of the intestinal mucosal barrier, thus mitigating intestinal toxicity in acute radiation mouse models 79 . Recent research shows that MSC-EVs treatment increased the proliferation and simultaneously suppressed apoptosis in radiation-stimulated Lgr5-positive intestinal epithelial stem cells 80 . BMMSC-EVs can promote wound healing of IEC-6 and change intestinal permeability. This would repair the intestinal barrier and cure necrotizing enterocolitis. Orally administered EVs can also restore intestinal barrier integrity at multiple levels, including the mucus, epithelial, and immune barriers. They have the potential to treat intestinal inflammation, and prevent endotoxin-induced colitis, thereby can become a popular trend in the treatment of intestinal diseases in the future 81 .
EVs can promote angiogenesis, improve blood circulation, and contribute to the repair of injured areas. MSC-EVs applied in intestinal injury treatment can also effectively treat radiation-induced intestinal injury. EVs, even at a small volume, can be delivered to the injury site through intravenous injection and other ways and have good biological safety and feasibility. Thus, these findings provide a new research idea for the use of MSC-EVs in the treatment of intestinal injury.
Repair Effects of MSC-EVs in Skin Wounds
Skin wounds are caused by different types of diseases, mechanical damage, or ultraviolet and infrared radiation. These factors can damage the skin tissue structure or integrity. Unhealed skin wounds can induce inflammation, leading to several local and even systemic physiological and pathological changes such as fever, pain, redness, swelling, and loss of function. These wounds affect the psychosocial health and quality of life of affected patients 82 . The highly continuous process of wound healing involves the recovery of the skin barrier function and consists of stages, such as hemostasis, inflammation, fibroblast proliferation, and fibroblast remodeling 83 . A dynamic interplay occurs between many different skin cells and immune cells that act at specific stages to reshape wound healing 84 . Conventional methods, including various wound dressings, negative pressure suction, tissue engineering substitutes, and autologous skin grafts, are currently used in clinics to promote wound healing. MSC-EVs exhibit anti-inflammatory, anti-aging, and wound-healing properties in various in vitro and in vivo models (Table 2). EVs secreted by ADSCs, BMMSC-EVs, and HUCMSC-EVs are most commonly used for wound-healing treatment 85 . ADSC-EVs can promote wound regeneration and reduce scar formation. During the inflammatory phase, ADSC-EVs exert an immunosuppressive effect by reducing IFN-α secretion, thereby stimulating wound healing. In the proliferative phase, ADSC-EVs enhance angiogenesis by promoting VEC proliferation and migration and transfer miR-125a to VECs by inhibiting DLL4 to stimulate proliferation and promote angiogenesis. MiR-31 in ADSC-EVs contributes to angiogenesis induction by targeting FIH1 in VECs 86 . HADSC-EVs upregulate 199 miRNAs and downregulate 93 miRNAs, thereby promoting dermal fibroblast proliferation and differentiation and accelerating skin regeneration 87 . During remodeling, ADSC-EVs regulate collagen deposition and organization, thereby promoting collagen synthesis and skin barrier repair in vivo, and inhibiting scar hyperplasia. By stimulating filaggrin and increasing the expression of loricrin, a proteolytic resistance protein, and aquaporin AQP, ADSC-EVs can improve skin hydration and the epidermal permeability barrier function, promote cell proliferation, inhibit cell apoptosis, and effectively accelerate skin wound healing and skin structure remodeling 88 . ASCs-EVs can be internalized by fibroblasts and can stimulate cell migration, proliferation, and collagen synthesis in vitro. Moreover, ASCS-EVs promote N-cadherin, cyclin-1, PCNA, and collagen I, III gene expression. ASC-EVs synthesize collagen types I and III to promote collagen remodeling, while at the late stage, it reduces scar size by decreasing the collagen I/III ratio, myofibroblast differentiation, and the TGF-β1/TGF-β3 ratio in vivo. In addition, ASC-EVs can activate the ERK/MAPK pathway in dermal fibroblasts to increase the MMP3 level, thereby increasing the MMP3/TIMP1 ratio. This ensures that the extracellular matrix is reconstructed and scar formation is reduced in wound healing 89 . ASC-EVs can promote skin wound healing by optimizing fibroblast characteristics 90 .
Biological Effects of Extracellular Vesicles in Skin Wound Healing.

BMSC-EVs are another critical source of cell-free skin regeneration. Human BMSC-EVs can inhibit the TGF-β/Smad signaling pathway and induce the proliferation of HaCaT cells and HDF, thereby improving wound healing. Pretreatment of BMSC-EVs with specific drugs can increase their effectiveness in wound healing. In a diabetic rat model, ATV-pretreated BMSC-EVs (ATV-EVs) promoted endothelial cell proliferation, migration, angiogenesis, and VEGF levels in vitro. ATV-EVs upregulated miR-221-3p to activate AKT/eNOS, and the pro-angiogenic effect of ATV-EVs was attenuated after blocking this pathway 91 . Pioglitazone-pretreated BMSC-EVs promote the angiogenic function of HUVECs by activating the PI3K/AKT/eNOS pathway, thereby stimulating collagen deposition and ECM remodeling to accelerate diabetic wound healing 93 . Melatonin-pretreated MSC-EVs (MT-EVs) significantly inhibited IL-1β and TNF-α (pro-inflammatory factors) expression and decreased IL-1β, TNF-α, and iNOS expression by upregulating PTEN expression, inhibiting AKT phosphorylation, and increasing the M2 to M1 polarization ratio in vitro. MT-EVs also promoted the relative expression of IL-10 and Arg-1 (anti-inflammatory factors). Similarly, MT-EVs promote diabetic wound healing by inhibiting inflammation and promoting angiogenesis and collagen synthesis in vivo 94 .
MiRNAs in HUCMSC-EVs can be delivered to skin recipient cells and promote downstream signaling changes, which then accelerate wound healing. Under hypoxic conditions, HUCMSCs transcribe hypoxia-induced miR-125b. Once internalized by endothelial cells, miR-125b targets the 3′UTR of TP3INP53 mRNA and inhibits p53 protein expression, thereby leading to cell survival and subsequent wound healing 95 . HUCMSC-EVs stimulated with 455-nm blue light can effectively promote HUVEC proliferation, migration, and tube formation by upregulating miR-135b-5p and miR-499a-3p. EV-miR-135a derived from HUCMSCs promotes rat fibroblast migration and skin wound healing by inhibiting LATS2 expression 36 . Embryonic stem cell-derived EVs miR-135a inhibits LATS2 expression and thus facilitates epithelial cell migration and promotes skin wound healing 103 . To promote corneal epithelial cell proliferation and migration, HUCMSC-EVs upregulate the PI3K/Akt signaling pathway by inhibiting PTEN through miR-21, thereby stimulating corneal wound repair and regeneration 96 . In another study, miR-150-5p overexpression by HUCMSC-EVs promoted skin wound healing through the activation of the PI3K/AKT pathway 97 . The highly expressed miR-21-5p and miR-125b-5p in HUC-BMSC-EVs inhibited TGFBR2 and TGFBR1, respectively, thereby impeding the TGF-β signaling pathway and myofibroblast differentiation and providing a new strategy for reducing scar formation during wound healing 98 .
EVs also effectively restore UV-induced skin damage. MSC-EVs counteract UV radiation-induced oxidative stress and skin aging by degrading MMPs and downregulating ROS production. According to Gao et al. 101 , miR-1246-overexpressing ADSC-EVs results in TGF-β1 upregulation, which promotes skin collagen and elastin synthesis, and reverses the UV irradiation-induced adverse effects. In the UV-irradiated mouse model, MSC-EVs treatment led to a reduction in ROS levels and radiation-induced DNA damage in vivo, which effectively promoted skin wound healing 99 .
Furthermore, biological carriers promote the repair function of MSC-EVs in skin injury 104 . The search for different types of vector-loaded EVs will be part of innovative therapeutic skin wounds, which offer a promising option for skin injury repair. Therefore, MSC-EVs play an important role in skin injury repair. They promote wound healing, regulate cell behavior, and inhibit scar formation through a variety of mechanisms, providing new ideas and methods for efficient, safe, and aesthetic skin injury repair.
Effects of MSC-EVs in the Blood Vascular System
The blood vascular system is a vital system in humans that plays a crucial role in feeding, transportation, immunity, coagulation, regulation, and other aspects. It maintains the normal physiological function and balance of the human body. Any blood vascular system-related disease or disorder may severely affect human health. Therefore, maintaining the blood vascular system’s health is essential for the overall health of the body. Hematologic diseases usually affect the blood, hematopoietic system, and related organs and include leukemia, regenerative anemia, thrombocytopenia, and so on. Radiation-induced endothelial cell damage during cancer treatment can also aggravate these diseases. MSC-EVs have exhibited potential in the treatment of these diseases.
Leukemia is a common cancer of the hematopoietic system. Bioactive molecules released from EVs can regulate the immune response, inhibit leukemia cell proliferation, and promote their apoptosis. MSC-EVs can treat leukemia through these mechanisms. Expanded acute myeloid leukemia (AML) alters the matrix composition and restricts the hematopoietic function, AML-EVs rapidly enter hematopoietic stem cells (HSC), and its transport triggers inhibition of protein synthesis and HSC quiescence, thereby allowing for alteration of tumor cell microloops for AML therapy 105 . In a recent study, miR-425-5p-containing EVs from the BMSC of AML patients inhibited AML cell proliferation, invasion, and migration through the Wilms tumor 1-associated protein pathway. This suggests that miR-425-5p is a potential therapeutic target for AML patients 106 . Furthermore, AML-derived miR-23b-5p reduced tumor cell proliferation and induced apoptosis through the PI3K/AKT pathway 107 . BMSC-derived miR-7-5p inhibited AML cell proliferation and promoted their apoptosis through the PI3K/AKT/mTOR signaling pathway 108 . BMMSCs regulate THP-4 cell proliferation and apoptosis through the IRF1/INPP2B signaling pathway 109 . All these findings provide new therapeutic strategies for AML. Migrasome is a new type of EV that has great potential in disease diagnosis and treatment because of its size, characteristics, and biological function. These vesicles depend on cell migration and appear at the tips and intersections of contractile filaments, which are tubular structures left behind by cell migration. When cells migrate, the contractile filaments break. Then, vesicles on these filaments, called migrators, are released out of the cell or absorbed by the next cell that arrives at the location 110 . Migrasomes produced by human bone marrow-derived mesenchymal stromal cells can induce leukemic cell migration and phagocytosis, thereby mediating a new cell communication mode between MSCs and hematopoietic cells, thereby offering a new strategy for leukemia treatment 111 .
For regenerative anemia: Growth factors and cytokines in EVs can promote the proliferation and differentiation of hematopoietic stem cells (HSCs), thereby increasing blood cell production. In one study, MSC-EVs supported CD34+ cell proliferation in vitro 112 . In addition, MSC-EVs prevent HSC apoptosis by increasing the expression of the cysteine-X-cysteine motif chemokine receptor 4 and chemokines 113 . Another study exhibited that bioactive molecules in MSC-EVs can regulate HSC-related gene expression, thereby improving HSC homing in the bone marrow niche 114 . MSC-EVs can promote the proliferation of cord blood-derived HSCs in vitro. In addition, MSC-EVs added to the co-culture system of MSCs and HSCs promoted HSC proliferation 115 . Morhayim et al. 116 reported that osteoblast-derived EVs increased the proliferation of UCB-derived CD34+ cells both in vitro and in vivo. In addition, Preciado et al. found that MSC-EVs increased the colony-forming ability of CD34 cells by increasing BIRC2, BIRC3, and NF-κB expression. The expression of CASP3, CASP6, and other pro-apoptotic genes was downregulated. Moreover, CD44+, a molecule that is a crucial player in HSC homing and transplantation, was upregulated 117 . This has vital implications for the treatment of hematopoietic diseases such as regenerative anemia.
Hematopoietic cells are sensitive to radiation. Radiation may cause bone marrow failure, thereby leading to hematopoietic system damage. MSC-EVs can repair radiation-induced hematopoietic system damage, and MSC-EVs can transfer miRNAs with pro-regenerative or anti-apoptotic effects into the irradiated hematopoietic cells. For example, intravenously administered human BMMSC-EVs rapidly normalized peripheral blood cell counts in mice exposed to irradiation over their complete bodies. After treatment with MSC-EVs, miRNA-221, miRNA-451, and miRNA-644-3p contents promoted the proliferation of irradiated bone marrow cells, miRNA-210-5p, miRNA106b-3p, and miRNA155-5p prevented radiation-induced apoptosis of hematopoietic cells 118 . MSC-EVs can restore the hematopoietic function by stimulating the secretion of hematopoiesis-related cytokines. Human placental MSCs secrete human hematopoiesis-related cytokines such as G-CSF, MCP-1, IL-6, and IL-8 to effectively restore the radiation-induced hematopoietic function in mice 118 . One study revealed that MSC-EVs induce macrophages to produce hematopoiesis-related factors such as G-CSF, IL-6, IL-8, and VEGF in vitro 119 . The MSC-EVs can also remodel hematopoietic cells. For example, MSC-EVs enhance macrophage activity, and macrophages are considered key regulators of hematopoiesis 119 . MSC-EVs can directly restore irradiated BMMSCs, which effectively contribute to hematopoiesis, by activating the Wnt/β-catenin signaling pathway to alleviate DNA damage and oxidative stress 120 . This suggests that MSC-EVs can effectively treat hematopoietic diseases such as regenerative anemia.
For thrombocytopenia: Megakaryocyte production in the major bone marrow hematopoietic tissue of platelets is involved in hemostasis, arterial thrombosis, and other physiological and pathological processes Thrombocytopenia is a common disorder related to platelet production. Components in EVs can facilitate the promotion of platelet production and function, thereby improving the condition of patients with thrombocytopenia. Silachev’s research shows that MSC-EV can significantly shorten the formation time of human blood clots and significantly reduce the lag period of spontaneous clots 121 . ADSC-EVs induce coagulation via both the intrinsic and the extrinsic coagulation pathway 122 . ADSC-EVs inhibit the differentiation of T helper cell 17 (Th17) through miR-199a-5p, increase platelet count, and reduce the Th17/CD4T cell ratio in mice with immune thrombocytopenia (ITP). This study revealed the potential therapeutic role of ADSC-EVs in ITP 123 .
In addition, MSC-EVs may exert positive effects on other hematological diseases by regulating the inflammatory response, promoting angiogenesis, and regulating the immune function. However, although EVs have exhibited potential therapeutic efficacy against hematological diseases, further studies are warranted to verify their safety, efficacy, and explore their action mechanism. For the treatment of specific diseases, factors such as individual differences, disease type, and disease severity must also be considered. Therefore, careful evaluation through large-scale clinical studies is warranted before clinical application.
Repair Effects of MSC-EVs in the Central Nervous System
Diseases of the central nervous system (CNS), such as multiple sclerosis (MS), Alzheimer’s disease (AD), and Parkinson’s disease (PD), affect millions of people worldwide. The treatment of these diseases is challenging. The blood-brain barrier (BBB) restricts drug circulation to target brain areas, and surgery and radiotherapy do not effectively improve patient survival 124 . To address this problem, EVs have attracted increasing scientific interest because they can cross the BBB and are considered attractive tools for the targeted delivery of drugs across the BBB. EVs are secreted by almost every cell, and biomolecules carried by EVs are part of the intercellular communication channels between the brain and other organs. The intrinsic properties of EVs as therapeutic delivery vehicles are being investigated. For example, protecting and transferring functional cargo; developing strategies for loading therapeutic small molecules, proteins, and oligonucleotides into EVs; and targeting them to specific cell types to treat CNS diseases.
MS is a chronic autoimmune disease of the CNS. In animal models, intravenously or intranasally administered MSC-EVs improved motor function symptoms and reduced disease severity 125 . EVs exert anti-inflammatory and immunosuppressive properties, thereby significantly reducing inflammatory cell infiltration in the CNS and regulating cytokine levels. This effect is achieved because of the presence of soluble immunomodulatory factors in EVs or the ability of EVs to inactivate the NALP3 inflammasome and reduce NF-κB levels 126 . Furthermore, EVs inhibit the activation of antigen-specific T cells and microglia 127 . EVs administration reduces demyelination and can increase immunomodulation and remyelination by functionalizing the surface of EV with the LJM-3064 aptamer 125 . Finally, EVs can increase TGF-β levels and body weight to alleviate MS symptoms 128 .
Recent AD-related studies have found that MSC-EVs produced during hypoxia are more effective in rescuing learning and memory impairment than those produced during normoxia 129 . EVs can upregulate the immunomodulatory markers cyclooxygenase-2 and indoleamine-pyrrole 2, 3-dioxygenase because of pretreatment with cytokines such as TNF-α and IFN-γ to obtain an improved immunomodulatory function 130 . In addition, EVs can predict AD development. In Wang et al.’s 131 study, MSC-EVs suppresses iNOS expression and ameliorates neural impairment in Alzheimer’s disease mice. MiR-212 and miR-132 were downregulated in neurogenic plasma EVs from AD patients 132 . Fiandaca et al. extracted and quantified AD pathogenic proteins from neurally derived blood EVs. They found that the mean EV levels of total tau, P-T181-tau, P-S396-tau, and amyloid β1-42 (Aβ1-42) were significantly higher in the AD group than in the healthy control group 133 . Finally, EVs can be targeted to the brain for an increase in action. According to a study, EVs were modified using a rabies virus glycoprotein (RVG)-targeting peptide. This nerve cell-targeting peptide interacts specifically with acetylcholine receptors for the localization of EVs to hippocampal neurons, thereby allowing their entry into neuronal cells 134 . In another study, targeting EVs through the systemically injected RVG peptide resulted in siRNA delivery into the mouse brain 135 . PD studies have found that MSC-EVs contain key neuroregulatory molecules, such as cystatin C, glia-derived connexin, galectin-1, pigment epithelium-derived factor, VEGF, brain-derived neurotrophic factor, IL-6, and glial cell line-derived neurotrophic factor. These molecules are vital for neuronal function recovery and improvement 136 . These molecules cause better motor performance, reduced rotational asymmetry, and improved spatial learning. These effects are mediated by increased dopamine levels in the striatum, decreased dopaminergic neuron loss and apoptosis, and generation of new dopaminergic neurons and neuronal terminals 137 . Intravenously or intraperitoneally injected EVs can cross the BBB and reach the substantia nigra and striatum, and then play a role in PD treatment 138 . In other study, MSC-EVs loaded miR-181a-2-3p downregulated EGR1 to inhibit oxidative stress via the NOX4/p38 MAPK axis in PD mouse model 139 . These findings provide a new strategy for EV-mediated treatment of PD in the future.
In addition, EVs have potential therapeutic effects on other CNS diseases. For example, BBB destruction after ischemic stroke aggravates brain damage, and BBB aging can cause severe neurological deficits in elderly patients with ischemic stroke. EVs can prevent ischemic stroke in aged mice by partly restoring the BBB by activating the eNOS-Sirt1 axis through AKT1 and CALM delivery, suggesting that EVs therapy is an effective alternative against ischemic stroke in the aged population 140 . Glioblastoma is the most common and malignant tumor of the CNS. In patients with glioblastoma, the average survival time after treatment is 14 months 124 . The poor prognosis of this condition indicates that new therapies are urgently required. MSC-EVs can deliver antiblastic drugs to glioblastoma cells, which could inhibit the growth of glioblastoma 141 . In other study, Wang et al. 142 found that gene therapy approaches using MSC-EVs could overcome traditional CT resistance by delivering genetic material directly into glioblastoma cells, bypassing mechanisms of drug efflux. In addition, ADSC-EVs suppress glioblastoma proliferation, invasiveness and angiogenesis, can be applied as anticancer therapeutics and medicine carriers 143 . According to these findings, MSC-EVs are a promising immunotherapeutic approach against glioblastoma.
Despite great progress in EVs-based disease-related research, some open questions need to be addressed in future studies. EVs application for the diagnosis and treatment of CNS diseases and its regulatory mechanism requires more supporting evidence, especially the biomarkers of EVs isolation, EVs dose, the measurement standard of EVs, and the administration route. Side effects, immunogenic effects, and EVs heterogeneity should also be considered. Despite the obstacles, the use of EVs as potential biomarkers and therapeutics in CNS diseases is attractive, and under the limited effect of surgical treatment and radiotherapy, EVs therapy deserves more future research. MSC-EVs will surely be a part of the treatment for CNS as soon as few technical barriers are solved.
Conclusions
MSC-EVs possess therapeutic properties and can be used for regenerative applications and radiotherapy of various tissues and organs. Conducting in-depth research on the specific mechanisms of MSC-EVs in different tissues is crucial for their safety in clinical applications. Determining the biological characteristics of MSC-EVs as a cell-free therapeutic tool is of great significance for radiotherapy and regenerative applications.
Footnotes
Author Contributions
Writing of the manuscript: NW and CX. Review, editing, and supervising: FM, HS, NH, HZ, JL, QL, and CX. All authors have read and agreed to the published version of the manuscript.
Ethical Approval
Not applicable.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Natural Science Foundation of Shandong Province, China, grant number ZR2022MH011, the National Natural Science Foundation of China (32071241).
