Abstract
Mesenchymal stem cells (MSCs) are pluripotent stem cells with self-renewal. They play a critical role in cell therapy due to their powerful immunomodulatory and regenerative effects. Recent studies suggest that one of the key therapeutic mechanisms of MSCs seems to derive from their paracrine product, called extracellular vesicles (EVs). The EVs contain much DNA, messenger RNA (mRNA), microRNA, and protein components, which can exert intracellular communication to target cells. In clinical applications, the MSC-EVs have been widely used in tissue repair and immune disorder diseases. However, there are serval issues need to be considered such as how to accomplish the large-scale production of EVs and how to verify the exact mechanism of EVs. In this review, we summarize the current progress of MSC-EVs and discuss the challenges and future of MSC-EVs.
Introduction
Stem cell therapy has hailed as one of the most promising therapy of the 21st century 1 . Among the types of stem cells, mesenchymal stem cells (MSCs) have been widely used in clinical applications due to great regenerative potential and immunomodulatory functions2,3. Also, MSCs can be isolated from umbilical cord, amniotic fluid, bone marrow, fat, and other tissues 4 . In recent years, there are more than 10 kinds of MSC drug marketing around the world. Initially, the therapeutic mechanism of MSCs was attributed to their homing into the lesion or multiple differentiative ability, but the number of cells is extremely small and quickly being cleaned by immune cells after transplantation5,6. Therefore, more and more studies suggested that MSCs play significant role in the process of therapeutic effects through their paracrine function 7 . Studies have shown that the extracellular vesicles (EVs), one of the paracrine products of MSCs, have become key role of cell-free therapy (CFT), which can escape the immune response, not tumorigenic and safer to use compared with MSCs therapy8,9.
In 1983, EVs were discovered in rat reticulocytes by Pan and Johnstone but being reproved cellular waste 10 . In 2007, researchers found that EVs contain proteins, lipids, and RNA, such as mRNAs and microRNAs9,11,12 (Fig. 1). At present, a number of in vitro and in vivo experiments have claimed that the nano-sized vesicle plays a crucial role in cell-to-cell communication, thereby promoting proliferation, simulating vascularization and regulating immune responses 13 . It is worth noting that this CFT based on EVs has functions similar to MSCs and other stem cell types. Therefore, a series of clinical practice was carried out to verity the efficacy, and the indication was extensive, including immune disorders, cardiovascular disease, COVID-19, and wound healing14–20. The purpose of this review is to sum up the current development of MSC-EVs in biomedical and discuss the current challenges and future directions.

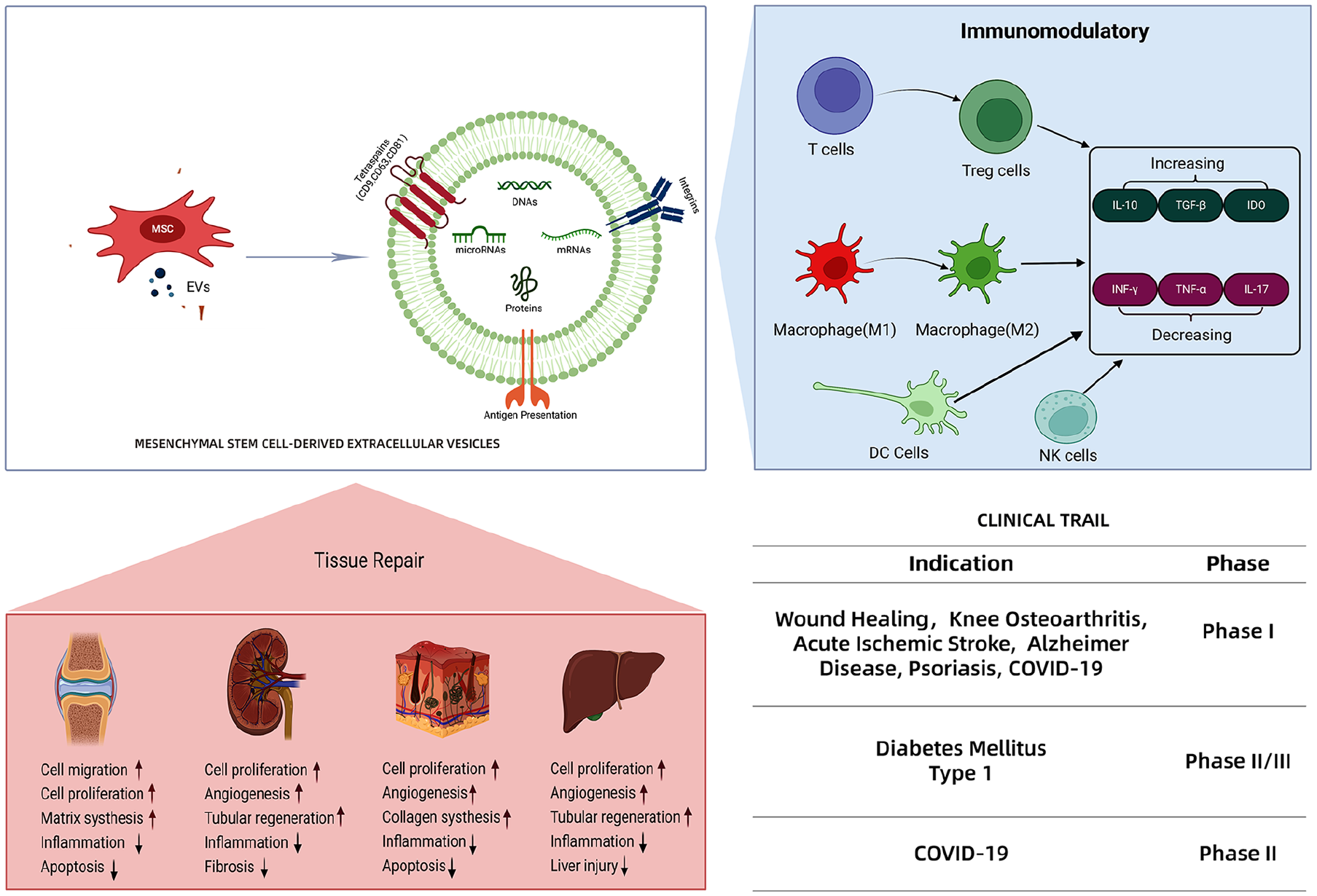
The structure of EVs. EVs consist of lipid bilayer membrane, rich in DNAs, mRNAs, microRNAs, and proteins, such as tetraspanins (CD9, CD63, CD81, etc).
Biology of MSC-EVs
EVs have a cup-shaped morphology under an electron microscope and have a diameter ranging from 30 to 120 nm 21 . These vesicles can be concentrated in the 1.10–1.21 g/ml section of a sucrose density gradient22–24. Multi-omics have shown that the contents of EVs are rich, including various microRNAs, mRNAs, and proteins. Compared with stem cells, the stem cell–derived EVs have more stable biological characteristics due to special lipid bilayer membrane structure, which can prevent the degradation of its contents and maintain the function of internal proteins and genetic materials25–27. Also, EVs can be stored long term at −80°C but stem cells should be stored at −196°C liquid nitrogen28,29. Different types of MSCs can secrete EVs under normal and pathological conditions. In addition, there are some differences between the types of MSCs. Studies have shown that EVs derived from human umbilical cord MSCs (hUC-MSCs) have higher activity than EVs derived from human bone morrow MSCs (hBM-MSCs) 30 . Compared with hBM-MSCs, the yield of EVs secreted by human amniotic fluid MSCs (hAF-MSCs) is higher30,31. However, the EVs of hUC-MSCs and hBM-MSC, sharing 60% of protein components related to cell growth and antioxidant stress, have been confirmed that can inhibit the growth and apoptosis in tumor cells32,33. Significantly, no substantial differences were found in EVs among hUC-MSCs and hBM-MSC, and generally, these EVs have similar function9,34–37.
EVs are largely originated from multivesicular bodies (MVBs). When MVB is fused with MSC membrane, the inner vesicles are secreted as EVs38,39. The formation process of EVs is as follows: first, intraluminal vesicles (ILVs), the precursors of MVBs and EVs, are formed under cellular maturation process; second, MVBs merge with the lysosomal membrane and then the inner vesicles are released into extracellular matrix as EVs 40 . As a paracrine product of MSCs, EVs have two ways to enter the target cell: one is through endocytosis by target cell, and the other is through membrane fusion between EVs and target cell 38 . And then, the various biological substances contained into target cell are released to finish signal transduction, for example, the mRNA contents can translate the corresponding protein when it binds to target cell, the microRNA contents can regulate the expression of target protein by inhibiting the process of mRNA translation or degrading mRNA directly41,42.
Clinical Significances of MSC-EVs
Currently, there are more than 150 clinical trials involving EVs as listed in www.clinicaltrails.gov. These trials not only focus on using the EVs as a diagnostic biomarker to predict the outcome of treatments, but also concentrate on using the EVs as CFT in various clinical applications, especially for tissue repair and immune disorders (Table 1).
Clinical Trials of MSC-EVs.

MSC-EVs for Tissue Repair
Tissue repair is a very complex process that refers to the interaction and association of different repair cells, cytokines, intercellular matrix, and proteins 43 . Ideally, the tissue damage is repaired by cells similar to injured cells to rebuild the original tissue structure and function. However, human body has limited inherent proliferation ability of tissues and cells 44 . EVs, as novel biomaterials, can be absorbed by target cells and are able to migrate long distances to lesions through the circulatory system to join in various physiological and pathological processes of tissue repair 45 . When they bind to target cells, their cargo would be released. Also, intracellular signaling pathways of tissue repair, including phosphoinositide 3-kinase (PI3K)/AKT, ERK, and STAT-3 pathway, would be triggered to facilitate and accelerate this process through downstream targets such as stromal cell–derived factor, nerve growth factor (NGF), and hepatocyte growth factor (HGF)46–48.
MSCs hold a leading post as source of EVs. Many researchers have demonstrated that MSC-EVs accelerate wound healing, relieve limb ischemia, alleviate kidney damage, stimulate cartilage tissue and bone tissue regeneration, and enhance liver and neuronal regeneration49,50 (Fig. 2). Research on the therapeutic efficacy of adipose tissue EVs are currently in clinical trials (NCT05475418), where researchers have applied adipose tissue EVs mixed with sterile hydrogel to subjects’ wounds and found to promote wound healing. A clinical study (NCT05475418) conducted by Kai Liu and team is in progress, which uses the adipose tissue MSCs (AD-MSCs)–derived EVs to treat wounds and injures. Another clinical phase I study (NCT05060107) focuses on knee osteoarthritis using the allogeneic MSC-EVs (3–5 × 1011 particles) through intra-articular injection (www.clinicaltrials.com). Besides, Nassar et al. have claimed that hUC-MSCs–derived EVs can ameliorate the progression of chronic kidney disease (CKD) from a phase II/III clinical trial. In this study, two doses (1 week apart) of EVs (100 μg/kg/dose) were used to treat 20 CKD patients. And patients intervened with EVs exhibited improved eGFRs and urinary albumin creatinine ratio 14 .

MSC-EVs accelerating great tissue repair functions for multiple organs and tissues by enhancing cell migration, stimulating cell proliferation, decreasing inflammation, and increasing regeneration.
MSC-EVs for Immune Disorders
More and more evidence have shown that MSC-EVs exert amazing immunomodulatory function similar to their parental MSCs 51 . On one hand, when the immune system is underactive, MSC-EVs can promote the maturation of immune cell and rebuilding of the immune system. On the other hand, when the immune system is over-activated, MSC-EVs can regulate the transformation of immune cells into anti-inflammatory phenotypes, upregulate the expression of anti-inflammatory factors (IL-10, TGF-β), and downregulate the pro-inflammatory factors (IFN-γ, IL-17) to avoid overactive immune diseases52–54 (Fig. 3). The immunomodulatory effects of MSC-EVs are mainly due to the presentation of antigen peptides, the transfer of miRNA, and diverse signaling pathways induced by surface ligands and inner contents 55 . For example, MSC-EVs carry PD-L1 that can bind to PD-1 on the surface of T cells to inhibit the proliferation of T cells; miRNA (miR-let-7b) of MSC-EVs could target TLR4 and p-p65 gene expression and suppress STAT3 and AKT phosphorylation, which regulate the macrophages plasticity 56 ; MSC-EVs carrying latency-associated peptide could induce downstream TGF-β/Smad2/3 signaling pathways in natural killer (NK) cells 57 .

MSC-EVs accelerating great immunomodulatory functions for multiple immune cells by transforming working patterns, increasing the anti-inflammatory factors, and decreasing the pro-inflammatory factor.
A clinical phase I study (NCT05523011), aiming at evaluating the safety and tolerability of MSC-EVs for psoriasis, has been completed the recruitment. The subjects will apply MSC-EVs ointment (100 μg MSC-EVs/g ointment) along with Vesiderm liposome cream from day 1 to day 20. Another chronic inflammatory disease, asthma, intervened by hBM-MSC–derived EVs, has been implemented by Sun Yai-Sen Memorial Hospital of China to verify whether the EVs would upregulate the expression of PD-L1 of macrophage, promote the function of Treg, and improve airway inflammation. Besides, the phase II/III study of type 1 diabetes (NCT02138331) has been conducted by Nassar team who used the hUC-MSC–derived EVs to reduce the inflammatory state and improve the β-cell mass because of restoring the balance between T helper 1 (Th1) and T helper 2 (Th2) immunological effects in early in vivo experiments. But the clinical results have not yet been published. Especially, more than 10 clinical studies for COVID-19 have been demonstrated that MSC-EVs could reduce the level of inflammation and educate the immune system to prevent the cytokines storm.
Challenges and Opportunities
Despite the success in preclinical and clinical studies, there are several critical issues needed to be resolved such as (1) large-scale production and isolation methods, (2) quality control, (3) pharmacology and pharmacodynamics 58 . Traditional methods to acquire EVs mainly rely on a two-dimensional (2D) culture in flask by long-term MSC passaging, which is labor-intensive as well as difficult to ensure batch-to-batch reproducibility 59 . In recent years, cell bioreactors made up of hollow fibers or based on microcarrier three-dimensional (3D) culture do partly improve the yield of EVs by increasing the surface area, but the existing process is not stable and many factors, for example, cellular confluence, cell age, oxygen concentration, and media, can affect the quality and quantity of MSC-EVs60–63. Also, there are many new methods and technologies such as tangential flow filtration (TFF), molecular exclusion method to isolate large-scale EVs, and using a combination of TFF and size exclusion chromatography. Visan et al. 64 have demonstrated that TFF is a reliable and robust EVs isolation approach that surpasses ultracentrifugation in yield, reproducibility, time, costs, and scalability. Hua et al. 65 developed a double TFF-based microfluidic device for exosome isolation from cell supernatants and human serum. Although these technologies can achieve a certain volume increase, it is still difficult to meet the purification and extraction of tens or even hundreds of liters in industrial production, and more attempts and research are needed in the future. And good manufacturing practice (GMP) for isolation, as well as quality control, needs to be established urgently. Generally, ultracentrifugation has been recognized as the standard method to separate EVs 66 (Table 2). Although studies have shown that MSC-EVs share functions similar to MSCs, the therapeutic mechanism of MSC-EVs is still unclear. Also, MSC-EVs are rapidly eliminated from blood after systemic injection. It is reported that the plasma half-life of MSC-EVs is only 2–4 min 67 .
Typical Isolation Methods of EVs.

With the rapid development of technology, more and more large-scale bioreactors will be presented in future, which satisfy the GMP production and isolation processes. And more and more full-grown methods would be built up. Nowadays, there are many experiments using bio-imaging technology to trace the biodistribution and targeting of MSC-EVs to target tissues or cells, for instance, a study that used PKH26 or 5-(and-6)-carboxyfluorescein diacetate succinimidyl ester (CFSE) to label MSC-EVs has found that these nano-sized particles migrated to the infarcted brain in a rat model of stroke73,74. Smart biomaterial scaffolds are becoming a hot research area in tissue engineering due to great efficient and continuous release of MSC-EVs
75
. Several scaffold materials such as hydrogel, collagen, and fibroin have been used to load MSC-EVs aiming at sustaining release of EVs as well as extending the plasma half-life of EVs76–78. A dual-cargo smart bilayer hydrogel, loaded with the anti-inflammatory medication sodium diclofenac (DC) and EVs derived from MSCs of the bone marrow, has been engineered to alleviate early-stage inflammation and to stimulate late-stage stem cell attraction and cartilage-forming differentiation. This innovative hydrogel has been validated through both laboratory (in vitro) and living organism (in vivo) tests, demonstrating its effectiveness in reducing inflammation following injury and in boosting cartilage repair by efficiently neutralizing reactive oxygen species (ROS) and reprogramming macrophages to a pro-healing state
79
. A versatile collagen-based wound dressing (CTM) was developed by incorporating highly potent EVs-mimetic nanovesicles (
Conclusion
EVs, as paracrine product of MSCs, contain abundant mRNA, microRNA, and protein components, which act as active signal molecular to communicate with the target cell. They have powerful tissue repair and immunomodulatory functions same as parental MSC. Compared with MSC therapy, MSC-EVs have advantages that can avoid long-term abnormal differentiation and tumor formation after cell infusion. So, MSC-EVs can also be called CFT. In past decades, MSC-EVs have shown great clinical efficacy, especially in regenerative medicine, such as wound healing and knee osteoarthritis, and immune disorder diseases, such as psoriasis and COVID-19. However, for MSC-EVs, the exact mechanism of how to stimulate proliferation, promote angiogenesis, and inhibit inflammatory infiltration is not fully understood. Therefore, understanding the biological process will help researchers better design and explore the pharmacology and pharmacodynamics of MSC-EVs. In future, there is no doubt that large-scale production equipment for EVs will be invented to increase the yield and fulfilment of the GMP process. Also, more and more biomaterials or 3D bioprinting materials will be discovered to improve the half-life and efficacy of EVs significantly.
In summary, MSC-EVs technology is still in the initial stage of clinical application. With the discoveries and new technologies coming forth, they have immense potential and prospect.
Footnotes
Author Contributions
Conceptualization—C-MT; writing—MG, BZ, XW; original draft preparation—BZ and XW; funding acquisition—C-MT. All authors have read and agreed to the published version of the manuscript.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key R&D Program of China (2020YFA0112900), the Science and Technology Grant of Xiamen and Xiamen Emerging NPC project (no. X2106101), Natural Science Foundation of Fujian Province (2020J05302 and 2021J011358), Natural Science Basic Research Program of Shaanxi Province (2021JQ-780), National Natural Science Foundation of China (81802332), and Science Foundation of the Fujian provincial Commission of Health and Family Planning, China (2021GGB026), respectively.
