Abstract
Wound healing is a complex process, which involves three stages: inflammation, proliferation, and remodeling. Inflammation is the first step; thus, immune factors play an important regulatory role in wound healing. In this study, we focused on a chemokine, C-C motif chemokine ligand 3 (CCL3), which is often upregulated for expression during wound healing. We compared cutaneous wound healing at the histological, morphological, and molecular levels in the presence and absence of CCL3. The results showed that the wound healing rate in the wild-type and CCL3-/- + CCL3 mice was faster than that of CCL3-/- mice (P < 0.01), and application of CCL3 to wounds increased the healing rate. In the process of wound healing, the degree of reepithelialization and the rate of collagen deposition in the wound of CCL3-/- mice were significantly lower than those of wild-type mice (P < 0.01). The number of macrophages and the expression levels of tumor necrosis factor(TNF)-α and transforming growth factor (TGF)-β1 in the wounds of wild-type mice were much higher than those of the CCL3-/- mice. Removal of macrophages and CCL3-/- mice share similar phenotypes. Therefore, we infer that the wound healing requires the participation of macrophages, and CCL3 may play an important regulatory role through recruiting macrophages to the wound sites.
Introduction
Skin is the largest organ of the human body, which directly contacts the external environment and thus acts as a barrier protection 1 . Therefore, improving cutaneous wound healing is an important achievement in medicine. Wound healing is a complex and highly dynamic process characterized by fibrosis, angiogenesis, and reepithelialization, which involve proliferation of fibroblasts, endothelial, and epithelial cells2,3. The healing process has been divided into three stages: inflammation, proliferation, and remodeling4,5. The first stage of cutaneous wound healing is the inflammation. Neutrophils are the first immune cells that reach wound sites at the stage, which is followed by macrophages. The latter help to remove cell debris and bacteria, and secrete some pro-inflammatory cytokines [eg, Interleukin (IL)-1, IL-6, and tumor necrosis factor (TNF)-α], so as to coordinate the wound repair process and prevent infection of the wound6–8. During the proliferation stage, numerous cells and connective tissue are accumulated in the healing tissue; fibroblasts gradually migrate to the wound site and produce collagen fibers, resulting in the formation of extracellular matrix (ECM). At the same time, lodging macrophages start to secrete multiple types of growth factors [eg, transforming growth factor (TGF)-β1, vascular endothelial growth factor (VEGF), and basic fibroblast growth factor (b-FGF)). These growth factors stimulate proliferation of healing-related cells and promote the interaction between capillaries, fibroblasts, and collagen to form granulation tissue for filling up the wound gap, and finally complete the entire reepithelialization9–11. The remodeling stage is an exceedingly long period of regulation. It is the balance between the apoptosis of existing cells and the production of new cells. At this stage, macrophages release matrix metalloproteinases (MMPs) to decompose the temporary ECM, type I collagen then gradually replaces type III collagen in the granulation tissue and skin strength is gradually restored12,13.
Macrophages participate in the process of wound healing not only by exerting their antibacterial and pro-inflammatory functions 14 but also play a vital role in the regeneration of damaged tissue and remodeling 15 . Macrophages play different roles in different types of tissue injuries. They can quickly repair and rebuild damaged blood vessels through secreting VEGF16,17. Macrophages mainly perform the function of phagocytosis when the bone injury occurs to ensure the orderly decomposition of damaged bone tissue and play a key role in remodeling the bone tissue in the later healing stage 18 . When muscles are injured, macrophages highly express the CCR2 chemokine receptor, quickly migrate to the wound site, and highly express the chemokine receptor CX3CR1 to promote muscle tissue regeneration through secreting growth factors, such as insulin-like growth factor 1 (IGF1)19–21. When skin injury occurs, macrophages maintain skin homeostasis throughout the wound healing process through chemotaxis, phagocytosis, and immune regulation 22 .
One of the chemokines is C-C motif chemokine ligand 3 (CCL3) and is known as macrophage inflammatory protein 1α (MIP-1α)23–25. This chemokine is produced by multiple cell types, such as macrophages, T cells, monocytes, fibroblasts, and epithelial cells, and is indispensable for the recruitment of inflammatory cells under steady-state and pathological conditions 26 . As a key regulator of the immune microenvironment, CCL3 may mediate the transportation of immune cells in inflammation through promoting the accumulation of leukocytes and angiogenesis 27 . Preclinical studies have shown that CCL3 can actively recruit monocytes and macrophages to the inflammation sites. A chemotactic effect on neutrophils and monocytes is exerted by CCL3 in vivo or in vitro 28 . In this study, we established mice deficient in CCL3 gene and studied the rates of cutaneous wound healing with or without CCL3 treatment and found that application of CCL3 significantly increased accumulation status of macrophages and related factors, thus significantly improved the rate and quality of cutaneous wound healing.
Materials and Methods
Animals and Experimental Design
All animal experiments were carried out in accordance with the guidelines and research protocols of Institute of Antler Science and Product Technology, Changchun Sci-Tech University (No. CKARI2020011). A quantity of C57/BL6 mice (6 weeks old, female, 20 ± 3 g) were purchased from Liaoning Changsheng Biotechnology Co., Ltd. (Shenyang, China). In addition, C57/BL6 mice (female, 6 weeks old) with CCL3 gene knockout (CCL3-/- mice) were purchased from Cyagen Biosciences (Jiangsu, China). Crossbreeding of C57/BL6 mice with CCL3-/- mice was done to produce purebred mice deficient in the CCL3 gene. Through nucleic acid electrophoresis detection, pure and CCL3-/- mice were screened for the follow-up experiments. In our in vivo experiment, mice were divided into four groups (10 mice/group) (Table 1).
Animal Allocation and Treatment.
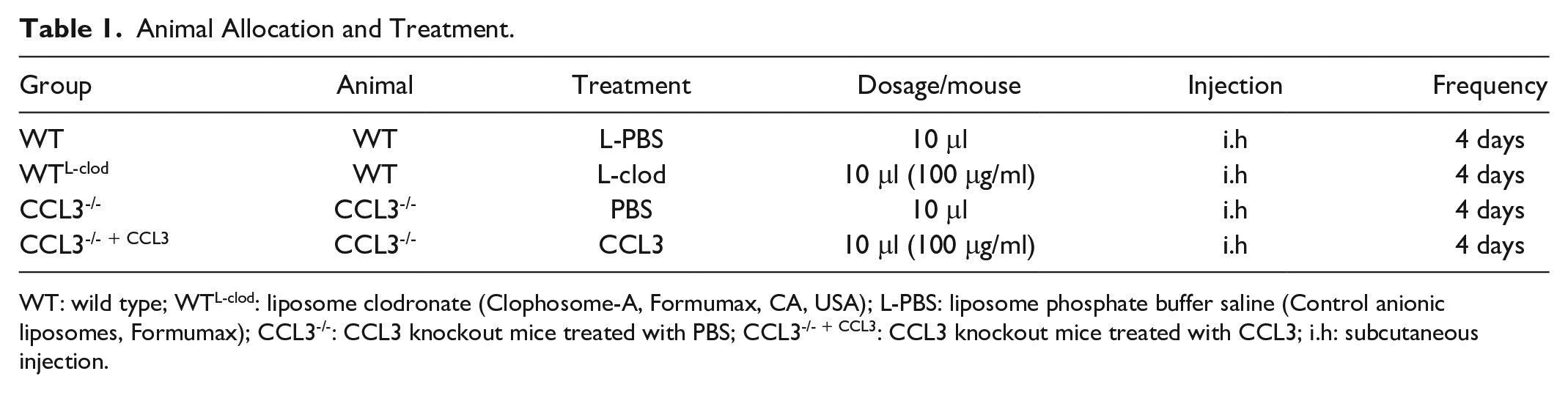
WT: wild type; WTL-clod: liposome clodronate (Clophosome-A, Formumax, CA, USA); L-PBS: liposome phosphate buffer saline (Control anionic liposomes, Formumax); CCL3-/-: CCL3 knockout mice treated with PBS; CCL3-/- + CCL3: CCL3 knockout mice treated with CCL3; i.h: subcutaneous injection.
Treatments were carried out under anesthesia (0.3% pentobarbital sodium, PANPHY, Tianjin, China). The back skin of each mouse was thoroughly shaved and disinfected. A biopsy punch sampler (5 mm in diameter; Rapid-Punch: Deluxe Sampling Tool Kit) was used to make two symmetrical full-thickness back skin holes. Tissue sampling and photographing were conducted on days 4, 8, 12, and 16 of post-surgery, respectively (Fig. 1). Adobe Photoshop CS6 was used to calculate the area of each wound. One day before tissue sampling, the mice were injected with 5-ethynyl-2-deoxyuridine (EdU) into the abdominal cavity for subsequent detection of proliferation cells. Before tissue sampling, the mice were sacrificed through neck dislocation, each cutaneous wound tissue was then removed and divided into two halves: one was fixed in 4% paraformaldehyde (Biosharp, Guabgzhou, China) for histology, and the other stored in a −80°C freezer for protein and RNA extraction.
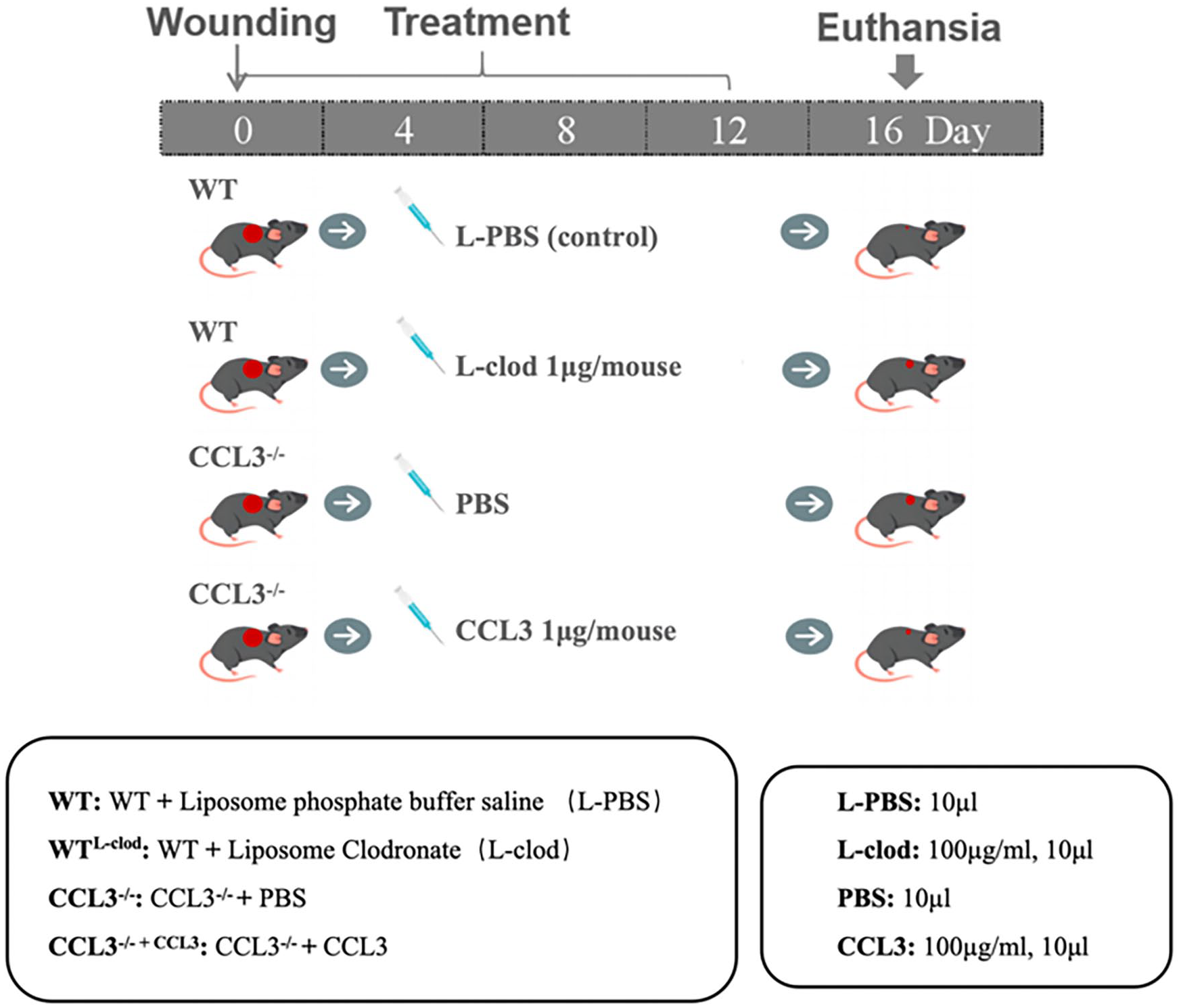
Experimental design and procedure.
Polymerase Chain Reaction Screening
The tail tip from each mouse was sampled, and the DNA was extracted from the tissue using a DNA extraction kit (Transgen Biotech, Beijing, China). Polymerase chain reaction (PCR) amplification system: ddH2O, 9.0 μl; Product primer F, 1.0 μl; Product primer R 1.0 μl; Premix Taq, 12.5 μl; DNA, 1.5 μl; Total, 25 μl (primer concentration: 10 μM). PCR conditions: Initial denaturation, 94°C, 3 min; Denaturation, 94°C, 30s (35 cycles); Annealing, 60°C, 35 s (35 cycles); Extension, 72°C, 35s (35 cycles); Additional extension, 72°C, 5 min. Conditions for nucleic acid gel electrophoresis: configure agarose gel with a concentration of 1.5%, electrophoresis solution with 10% tris-acetate-EDTA (TAE; Beyutime, Shanghai, China), set voltage at 120 V, electrophoresis for 30 min, and then the record the results. The primer sequences are listed in the additional information: Supplemental Table S1.
Western Blot Analysis
Proteins from the sampled tissues and cells were extracted using a protein extraction kit and separated by running a 12% SDS-PAGE gel, and transferred to polyvinylidene difluoride (PVDF) membranes (Amersham Biosciences, Piscataway, NJ, USA). With 5% skim milk powder to block nonspecific bindings for 2 h on the shaker [room temperature (RT)], primary antibody was then used to immunoblot the membrane (4°C overnight incubation). After washing with tris-buffered saline tween-20 (TBST), goat anti-rat or goat anti-rabbit secondary antibodies conjugated with horseradish peroxidase (HRP) were used (incubation in a shaker at RT for 1–2 h). The bands were analyzed using an enhanced chemiluminescence (ECL) substrate (Thermo Fisher Scientific, Waltham, MA, USA) and a fully automated chemiluminescence imaging analysis system (Tanon5800, Shanghai, China). Primary antibodies used in this experiment were CCL3 (R&D, MAB4501, 1:1000, Minneapolis, MN, USA), GAPDH (Transgen Biotech, HC301, 1:5000, Beijing, China).
Histology
The healed tissue of the mouse wounds fixed in formalin was embedded in paraffin wax and cut into 5 μm thick sections. After deparaffinization and rehydration, hematoxylin and eosin (H&E) and Masson staining were performed according to the manufacturer’s procedures (Sigma-Aldrich, San Francisco, MO, USA). The tissue sections were observed and examined under a microscope (Precipoint M8, Freising, Germany); angiogenesis, collagen deposition, reepithelialization, and hair follicle formation were evaluated.
Immunohistochemistry
The 5 μm thick tissue sections were also used for immunohistochemistry (IHC) staining. After dewax and rehydration, antigen retrieval solution (MVS-0101, MXB Biotechnologies, Fuzhou, China) was used for the sections. Based on the instructions of the immunohistochemistry kit (Maixin KIT-9710, MXB Biotechnologies), the following primary antibodies were applied onto each tissue section and incubated at 4°C overnight. After washing, the sections were incubated with biotinylated goat anti-rabbit second antibody for 2 h. Thereafter, DAB staining and hematoxylin staining were performed to visualize the specific staining, and finally the tissue sections were examined under a microscope (Precipoint M8). Primary antibodies used in this experiment: CCL3 (R&D, MAB4501, 1:200), TGF-β1 (Bioss, bs0086R, 1:300, Beijing, China), F4/80 (Bioss, bs11182R, 1:300), TNF-α (ab183218, 1:200, Cambridge, UK), and IgG [Santa, MTUST(Q-16), 1:500, USA].
Immunofluorescence
The macrophages were seeded at a density of 10,000/cm2 in 24-well plates. After 24 h of incubation, cover slips with adhered cells were rinsed with PBS and fixed in 4% formaldehyde for 30 min. After washing with PBS for three times, cells were incubated in blocking solution (5% BSA in PBS) for 1 h. For reaching intracellular proteins, cells were permeabilized with blocking solution containing 1% TritonX-100. Cells were incubated with primary antibody F4/80 (Bioss, bs11182R, 1:500) for 1 h at room temperature (RT), and isotype-matched rabbit or mouse IgG served as the negative controls. Cells were washed three times using PBS to remove excess primary antibody, followed by incubation with the second antibody conjugated with fluorescein for 1 h at RT in the dark. The nuclei of cells were counterstained with DAPI for 5 min at RT after removing the excess second antibody. The sections were washed three times with PBS and examined under a fluorescent microscope (EVOS Life Tech, CA, USA).
EdU Labeling of Proliferating Cells
Two hours before tissue sampling, each mouse was injected with EdU (100 μl, 1 μg/μl) into the abdominal cavity for detection of proliferating cells. After fixation, dehydration, and embedding in paraffin, each tissue block was cut into 5 μm sections, which were rehydrated through gradient concentrations of xylene and alcohol. Antigen retrieval process was conducted according to those used in the EdU cell proliferation detection kit (Cell-Light EdU Apollo488 In Vitro Kit, Ribobio, Guangzhou, China) and finally sealed with a fluorescence quencher sheet. The results were observed under a fluorescent inverted microscope (EVOS Life Tech).
Cell-Proliferation Assay Using Cell Counting Kit-8
Mouse skin fibroblasts (prepared by our laboratory) were seeded in 96-well plates at a density of 4,000 cells/well. The cells were divided into three groups: The wild-type (WT) group (Dulbecco’s modified Eagle’s medium [DMEM], Gibco, Grand Island, NY, USA), the IGF1 group (10ng/ml, Gibco), and the M-CM group. Cells in each group were incubated for 24, 48, 72, and 96 h, respectively, and cell proliferation status was measured using cell counting kit-8 (CCK8) cell growth assay kit (Sigma-Aldrich, St. Louis, USA). Briefly, 10 μl of CCK8 solution were added to each well in a 96-well plate and mixed gently, then incubated for 2 h at 37°C in a 5% CO2 incubator. The absorbance was measured using a microplate reader (Bio-Rad, Hercules, CA, USA) at the wavelength of 450 nm.
Extraction, Identification, and Culture of Immune Cells
After neck dislocation, each mouse was transferred to the cell culture room. The abdomen of each mouse was shaved and disinfected, and then 3 ml of precooled DMEM was injected into the abdominal cavity of each mouse. The abdomens were gently massaged for 2 to 3 min and stood for 5 min (repeat the operation procedure twice). The fluid in each abdominal cavity was collected using a 5-ml syringe, and centrifuged at 1,000 rpm for 5 min. The cell pellets were resuspended in the complete culture medium (DMEM + 10% FBS) and the cells were counted and plated (The cell density: 2 × 106/ml) for cell culture. Based on the method of immunofluorescence (IF) staining, the specific marker F4/80 for macrophages was used to determine whether the extracted cells were macrophages.
Mouse macrophages and fibroblasts were cultured, respectively, in DMEM medium containing 10% fetal bovine serum (FBS), 100 mg/ml of streptomycin, and 100 units/ml of penicillin (Gibco, Grand Island, USA) in a humidified incubator with 5% CO2 at 37°C.
Quantitative Real-Time PCR
The total RNA from each sampled tissue was isolated using Trizol reagent (Sigma-Aldrich) and reversely transcribed into cDNA using a cDNA synthesis kit (TaKaRa, Tokyo, Japan). SYBR premix (Roche, Basel, Switzerland), primers, and cDNA were combined, and then detection in quantitative polymerase chain reaction (qPCR) Detection System (qTOWER 3G, Germany) was performed. Furthermore, qPCR was performed using the miScript SYBR Green PCR Kit (Qiagen, Germany), following the manufacturer’s instruction. The primer sequences are listed in the additional information: Supplemental Table S2. All reactions were in triplicates, and the data were analyzed using the 2−ΔΔCt method.
Statistical Analysis
Statistical analysis was performed using Prism 8 (Graph Pad software). Unpaired two-tailed Student’s t-test or one-way analysis of variance (ANOVA) analysis was used to determine statistical significances. All quantitative data were given as the mean ± SD for at least three independent experiments. Differences were considered significant at *P value < 0.05, highly significant at **P value < 0.01, and ***P value < 0.001.
Results
C-C Motif Chemokine Ligand 3 Increased Wound Healing Rates in Mice
CCL3 knockout mice treated with PBS (confirmed at both of DNA and protein) were selected for this experiment (Supplemental Fig. S1). Number “2” represents the amplification product of CCL3 primer (500 bp), the absence of bands represents CCL3 knockout (CCL3-/-), and the presence of bands represents WT (Supplemental Fig. S1A). Full-thickness skin wounds were created and divided into four groups randomly. From day 1 to day 2 post-injury, the edges of the wound were regular, the wound area did not change significantly, and there was no significant difference in size of the wound area between these groups (Fig. 2A). On day 4 post-injury, scars were formed, the wound area reduced through contraction inwardly, and the wound edge became irregularly round. The nascent epithelium was migrating centripetally, and the wound area varied greatly among the four groups (Fig. 2B). The wound areas of both WT and CCL3-/- + CCL3 group were significantly reduced (P < 0.01) and the healing rates of WT group was significantly higher than those of WTL-clod group (P < 0.01) and the CCL3-/- group (P < 0.001). On day 8 post-injury, the scabs of all wounds fell off spontaneously, which revealed a thickened stratum corneum on the surface of the newly formed epithelium, and the unhealed area of each wound remained in the center (Fig. 2A). The wound areas of the WT and CCL3-/- + CCL3 group (P > 0.05) were significantly reduced, the healing rates of WT group were significantly higher than those of the WTL-clod group and CCL3-/- group (P < 0.001). On day 12 post-injury, the wound healing in the WT group was completed, the healing in the CCL3-/- + CCL3 group was almost completed; and the healing of both WTL-clod group and CCL3-/- group reached the late stage. The wound healing rates in the WT group were significantly higher than that of CCL3-/- mice (P < 0.01), demonstrating that CCL3 can promote wound healing rate in the CCL3-/- mice (Fig. 2B), thus treatment with CCL3 at least can partially rescue the deficiency of the wound healing rates in CCL3-/- mice (Table 2).
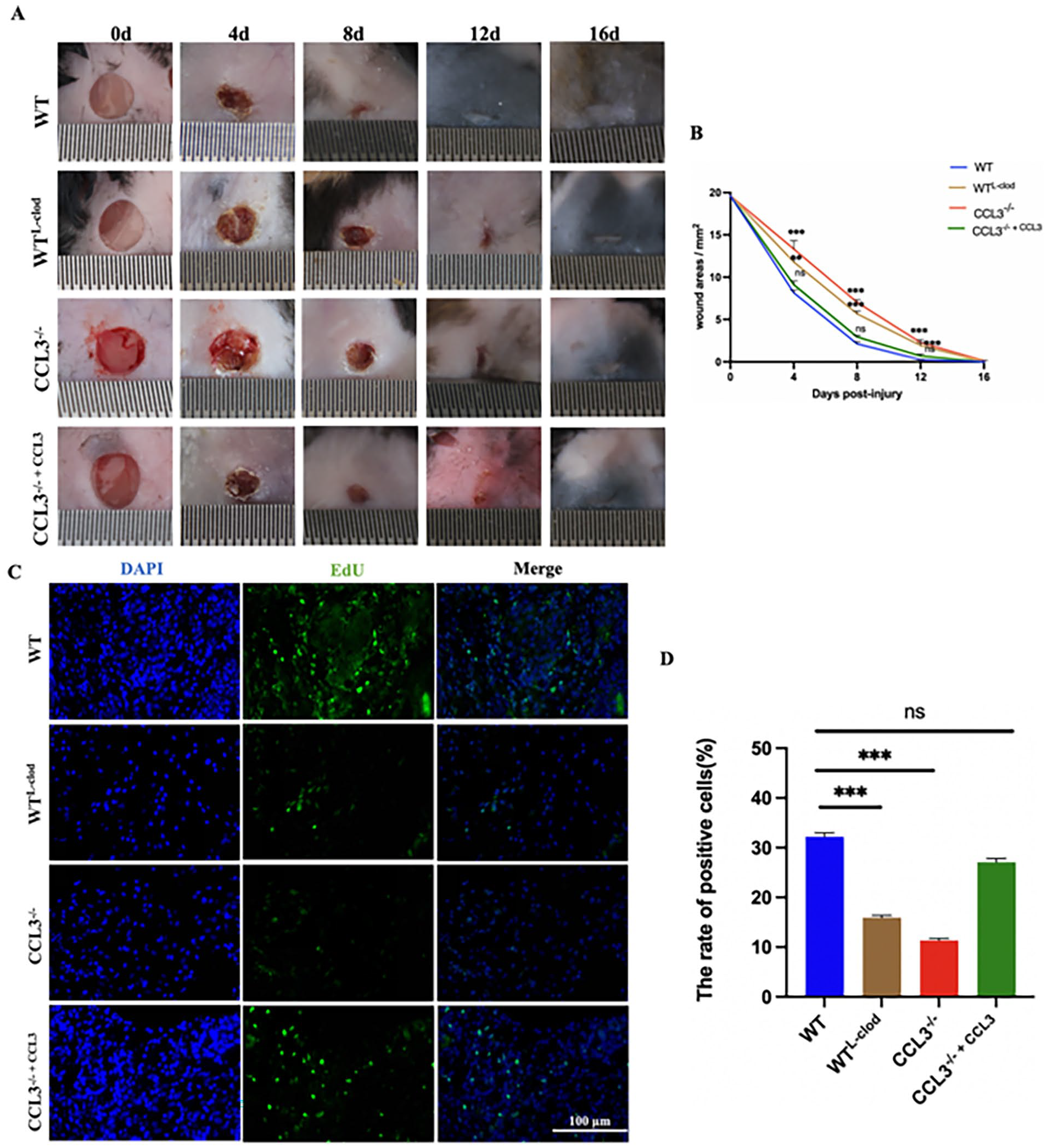
Effects of CCL3 treatment on mouse wound healing rate; (A) Overall morphological changes during the wound healing period. Scale bar = 1 mm; (B) Changes in wound area during healing period (comparison with WT group); (C) Day 8 post-injury, EdU-labeled proliferating cells (Green); (D) Bar graph of the proportion of proliferating cells; Scale bar = 100 μm, n = 10/group; *P < 0.05; **P < 0.01; ***P < 0.001.
Changing of Skin Damage Area in Mice/mm2.
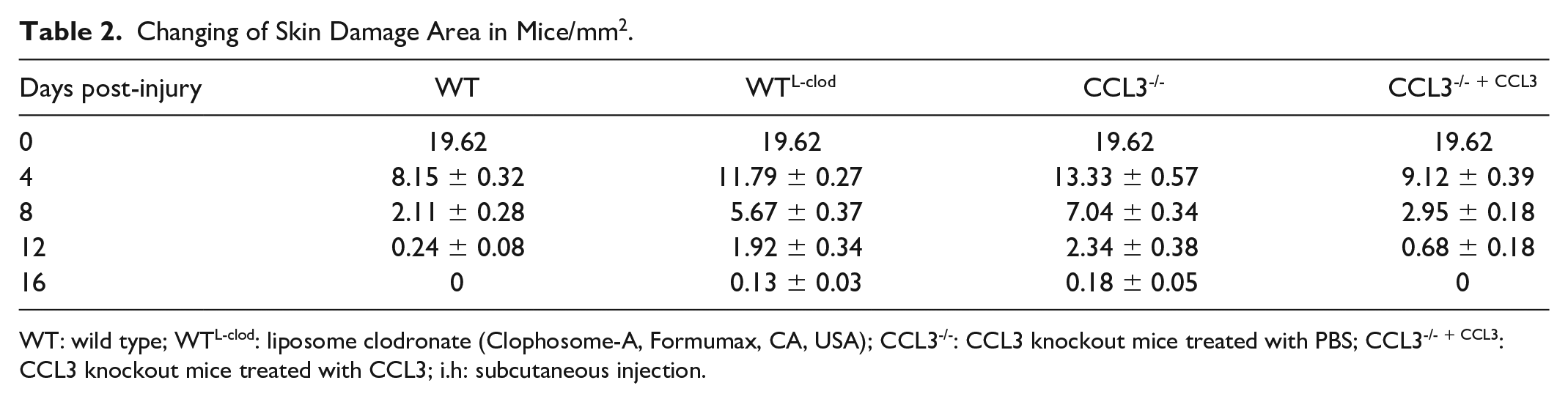
WT: wild type; WTL-clod: liposome clodronate (Clophosome-A, Formumax, CA, USA); CCL3-/-: CCL3 knockout mice treated with PBS; CCL3-/- + CCL3: CCL3 knockout mice treated with CCL3; i.h: subcutaneous injection.
Abbreviations.
We also labeled proliferating cells with EdU in vivo in mice, and the results showed that the WT group had the highest number of proliferating cells on day 8 post-injury; followed by the CCL3-/- + CCL3 group (P > 0.05; treated with CCL3 protein; Fig. 2C). The number of proliferating cells in the WTL-clod group and CCL3-/- group were significantly less than WT group (P < 0.001) (Fig. 2D).
C-C Motif Chemokine Ligand 3 Enhanced Reepithelialization and Collagen Deposition at the Wound Healing Sites
The effects of CCL3 treatment on wound healing rates in mice were evaluated at the histological level. At different time points (days 8 and 16 post-injury), there was formation of reepithelization, granulation tissue, and collagen deposition. On day 8 post-injury, reepithelialization and granulation tissue took place in all the wounds of the four groups (Fig. 3A). The degree of wound reepithelialization in the WT group was higher than that in the other three groups. On day 16 post-injury, the wounds of the four groups were basically healed completely and reepithelialization was completed. We can observe the epidermis and dermis (Fig. 3A).
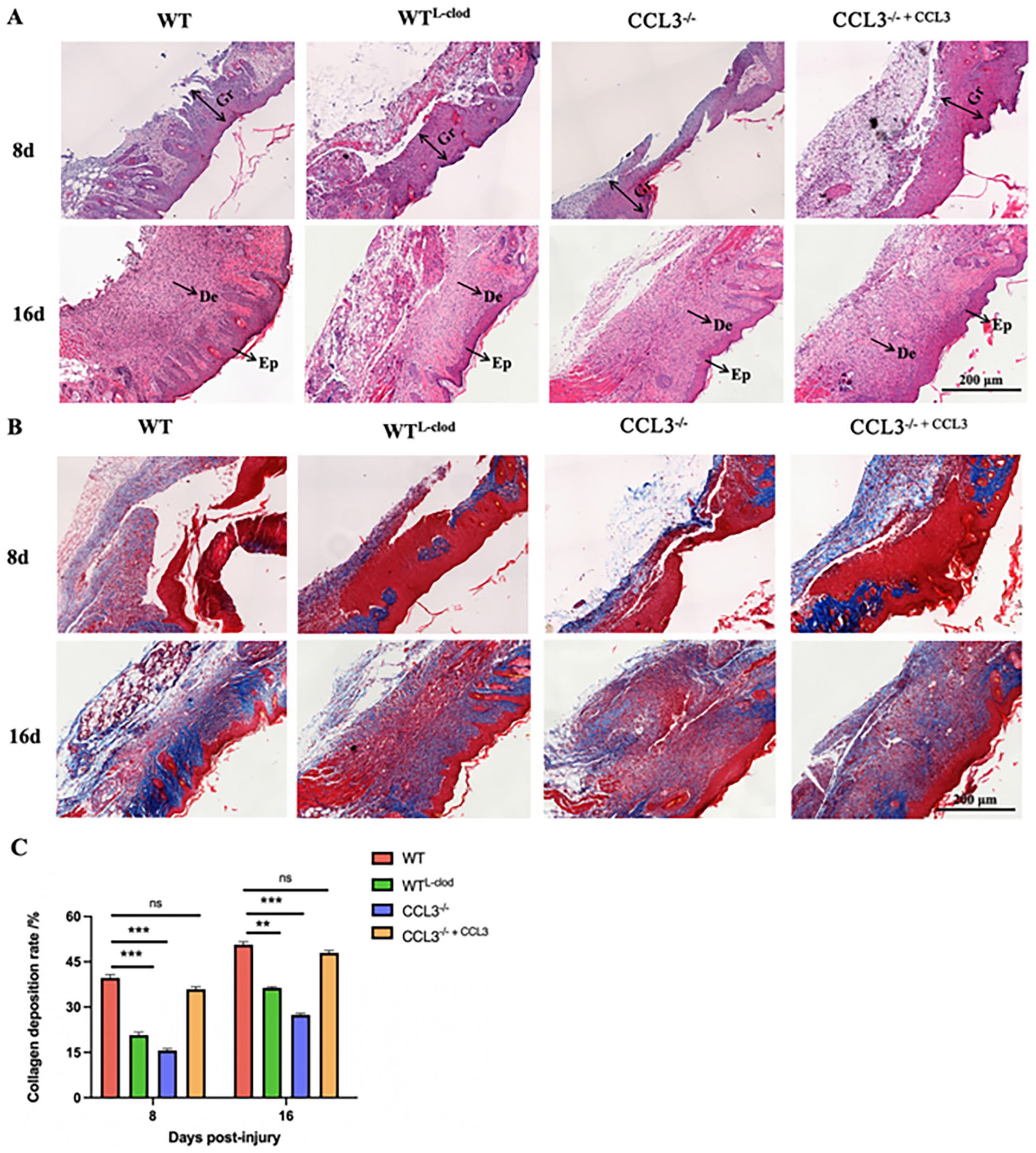
Analysis of wound healing status of the four groups of mice at the histological level using HE and Masson staining; (A) HE staining of wound tissues on the days 8 and 16 post-injury [granulation tissue (Gr), epidermis (Ep), dermis (De)]; (B) Masson staining of wound tissues on the days 8 and 16 post-injury (blue area represents the collagen fiber deposition site, red area represents muscle fibers); (C) Deposition rate of collagen (comparison with WT group); sale bar = 200 μm; *P < 0.05; **P < 0.01; ***P < 0.001.
Collagen deposition at the healing sites were observed using Masson staining, and tensile strength of the healing tissues could be enhanced through collagen deposition and remodeling, which would produce better healing effects. On days 8 and 16 post-injury, the WT group had the largest amount of collagen deposition, followed by CCL3-/- + CCL3 group (P > 0.05; Fig. 3B, C). Although there was certain level of collagen deposition observed in the WTL-clod group and CCL3-/- group, the amount of collagen was significantly lower (P < 0.001; Fig. 3B, C). The results showed that the deposition rate of collagen increased with the prolonged healing time. When the wound was healed completely, the content of collagen fibers essentially remained unchanged.
CCL3 Enhanced Macrophage Recruitment and Cytokines Secretion
Expression levels of CCL3 protein, macrophages markers (F4/80), and cytokines (TNF-α, TGF-β1) in the sampled tissue sections of the four groups on day 8 were immunohistochemically stained. C-C Motif Chemokine Ligand 3 was expressed in the WT group, WTL-clod group, and CCL3-/- + CCL3 group, but not in the CCL3-/- group (Fig. 4A); the WT group and CCL3-/- + CCL3 groups were higher than that of the WTL-clod group (P < 0.001; Fig. 4B). Macrophages in the healing tissues in the CCL3-/- group and WTL-clod group were barely detectable (Fig. 4A). The number of stained macrophages in the healing tissues of both WT and CCL3-/- + CCL3 groups were higher than CCL3-/- group and WTL-clod group (Fig. 4A). The number of stained macrophages in the CCL3-/- + CCL3 group was lower than that of the WT group, but not significantly different (P > 0.05).
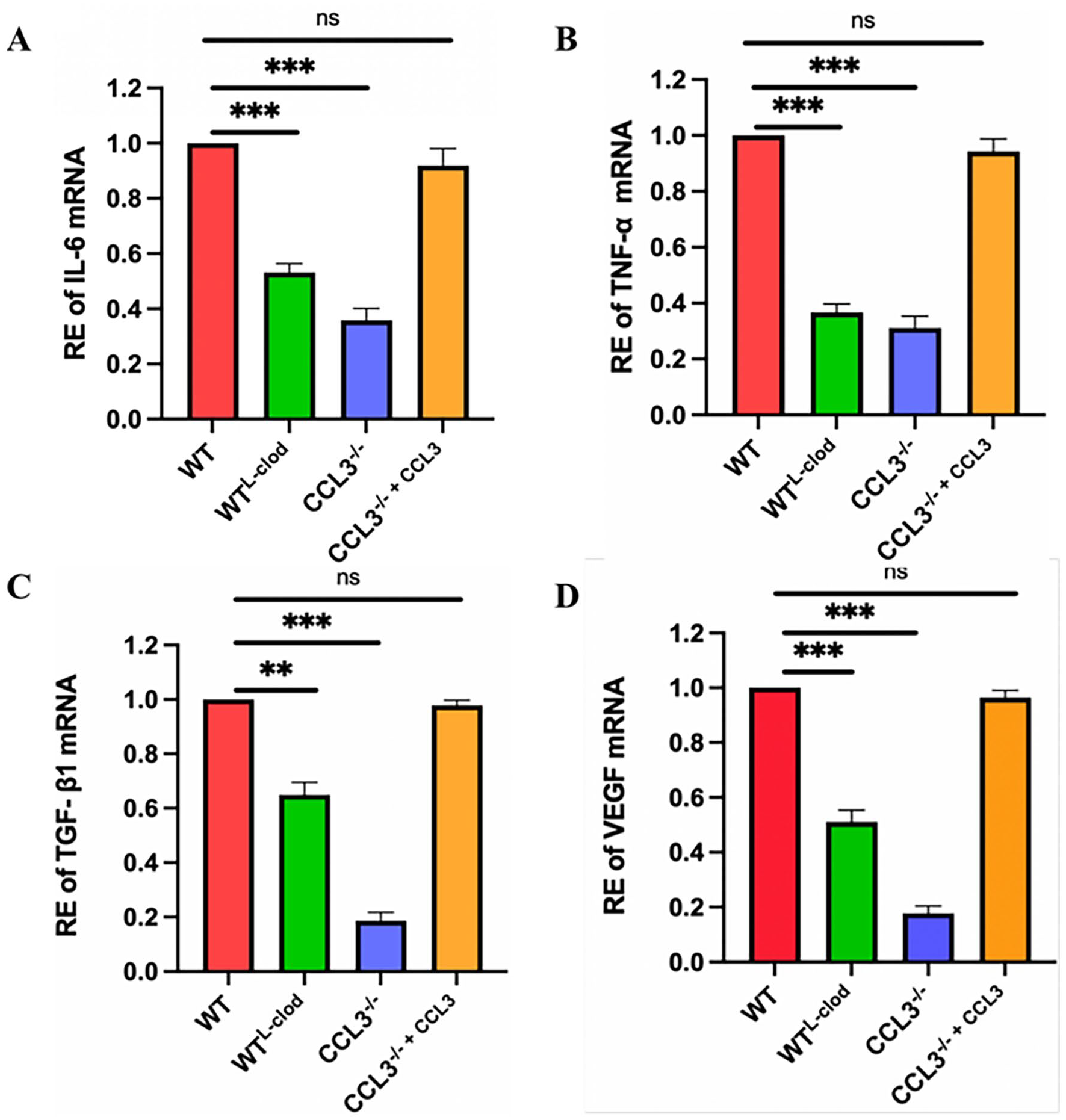
IHC staining (day 8 post-injury); (A) IHC localization of the expression of CCL3, macrophages (F4/80), TNF-α, and TGF-β1 in the wound healing tissues; (B–E) Bar graphs: number of CCL3; (C) number of macrophages; (D) number of TNF-α; (E) number of TGF-β1; (F) Cell proliferation rate via CCK8 (Comparison with DMEM). Scale bar = 100 μm; *P < 0.05; **P < 0.01; ***P < 0.001.
As macrophages are the source of many cytokines, we speculate that the presence of more macrophages may promote a higher cell proliferation rate. Therefore, we collected macrophages from the peritoneal cavity of mice (mouse peritoneal cells, Supplemental Fig. S2), collected macrophage conditioned culture medium, and then cultured fibroblasts with macrophage conditioned culture medium. The results showed macrophage conditioned medium (M-CM) significantly promoted proliferation of the mouse skin fibroblasts in vitro (Fig. 4F). The cell proliferation rate of the M-CM group was comparable to that of the IGF1 group (P > 0.05). Therefore, the macrophages would play an important role in promoting the healing of mouse skin wounds.
The changes in levels of TNF-α and TGF-β1 expression of the sampled tissues of the four groups were very similar (Fig. 4). The expression levels of TNF-α and TGF-β1 in the WT group and CCL3-/- + CCL3 group were higher than those of the WTL-clod group and CCL3-/- group (P < 0.001), respectively (Fig. 4D, E). It is reported that upon injury, macrophages can not only clear up the necrotic tissue, cell debris, and bacteria at the wound site but also secrete the inflammatory cytokines TNF-α, IL-6, and cell growth factors TGF-β1 and TGF-β3 29 . The interaction of these factors together would promote proliferation of healing relevant cells at the injury site, maintain the homeostasis environment, and speed up the repair process7,10.
CCL3 Upregulated Expression of the Wound Healing–Related Genes
To study the role of CCL3 in wound healing in mice, we evaluated the expression levels of cytokines in the sampled skin tissue through qPCR. Compared with the WT group, the expression levels of inflammatory factors IL-6 and TNF-α mRNA in the both WTL-clod and CCL3-/- groups were significantly lower (P < 0.001); in the CCL3-/- + CCL3 group, they were also lower but not significant (P > 0.05; Fig. 5A, B). The expression levels of growth factors TGF-β1 and VEGF mRNA followed the same trend with those of the inflammatory factors (Fig. 5C, D). Overall, similar results on the wound healing were found in either in the absence of CCL3 or in the absence of macrophages, ie, decreased cytokine expression and slowed wound healing rate.
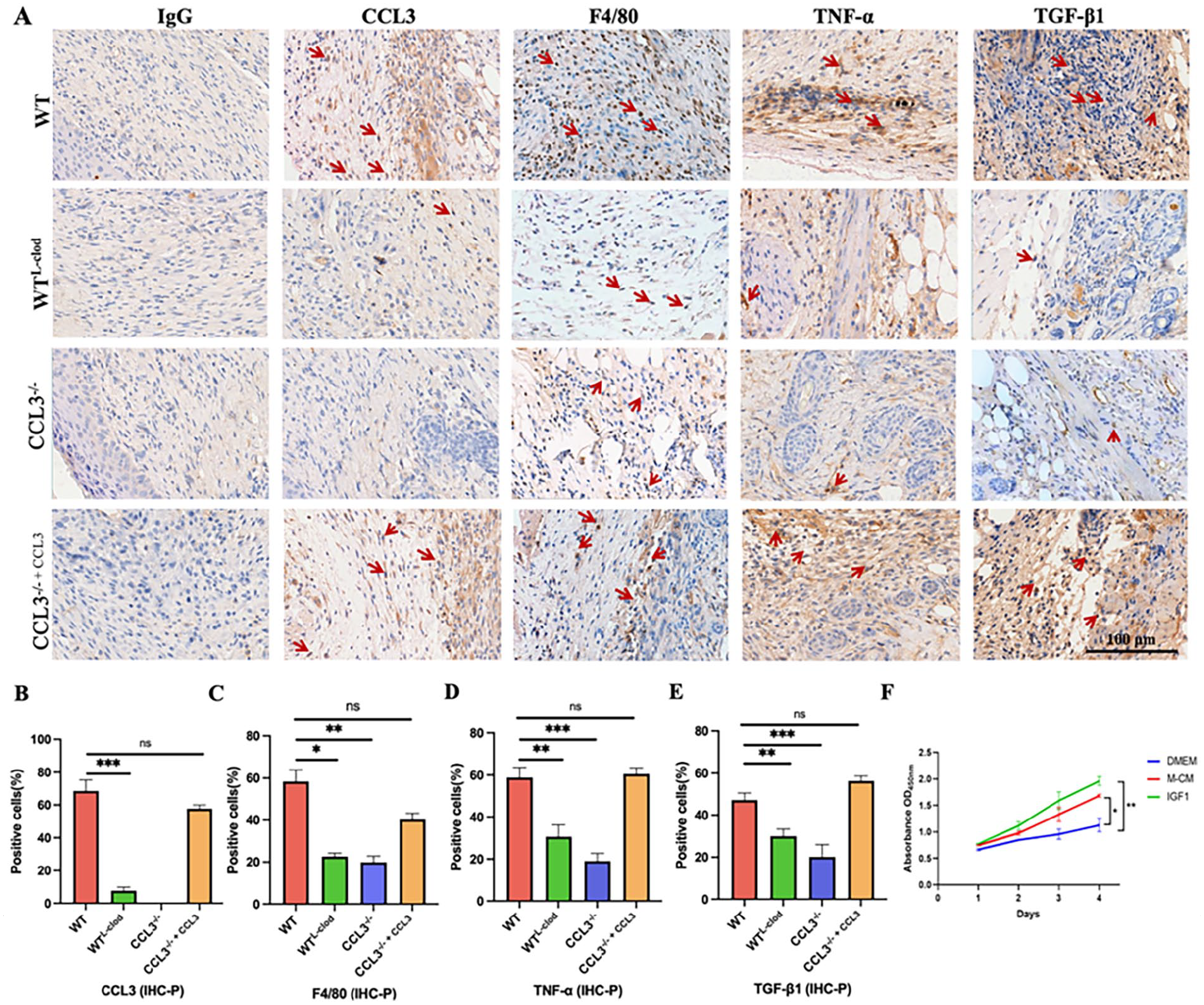
Results of qPCR of the wound healing related-genes; (A–D) mRNA expression levels of inflammatory and growth factors: (A) IL-6; (B) TNF-α; (C) TGF-β1; (D) VEGF; n = 3; *P < 0.05; **P < 0.01; ***P < 0.001. RE: Relative expression level.
Discussion
Inflammatory response to mechanical trauma is a natural process (acute) and takes place in multiple overlapping stages. These stages involve mainly cell migration, proliferation, matrix deposition, and tissue remodeling 30 . In the process, CCL3 recruits various inflammatory cells into the local wound environment. Neutrophils are the immune cells that first appear in the injured tissue, followed by monocytes and macrophages. As the inflammation subsides, many accumulated apoptotic neutrophils are removed, and macrophages begin to produce inflammatory cytokines and growth factors. The latter stimulates proliferation of fibroblasts (Fig. 6).
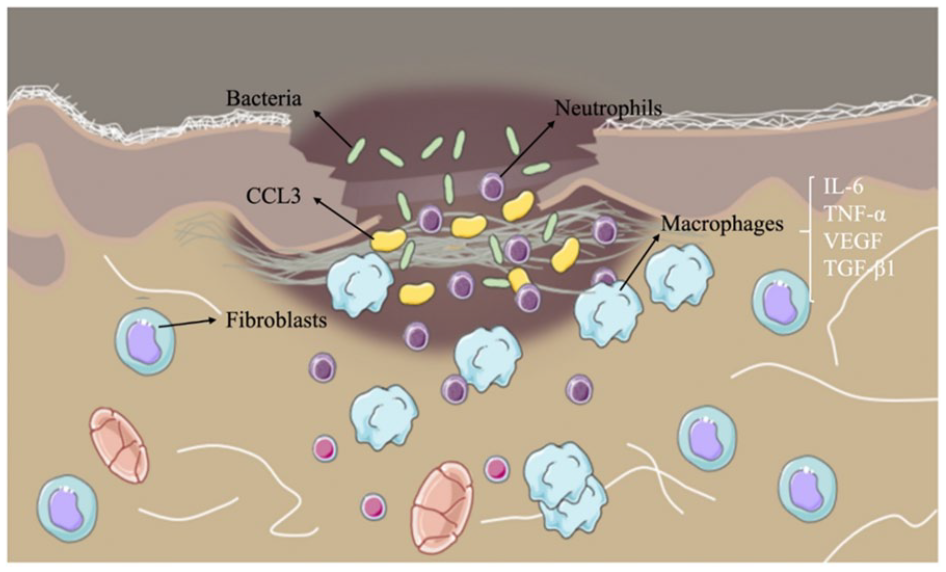
Molecular and cellular mechanisms underlying the cutaneous wound repair.
Our studies showed that CCL3 plays an important regulatory role in wound healing, possibly through recruiting macrophages. Macrophages play different roles in different stages of wound healing31,32. At the initial stage of injury (the inflammatory phase), Neutrophils first arrive at the wound site and secrete chemokines (CCL2, CCL3, and CXCL9). In turn, secreted CCL3 with other chemokines continue to recruit more neutrophils and macrophages28,31. With the aid of CCL3, neutrophils gradually differentiate into M1 macrophages, an inflammatory phenotype, recognizing the associated modified proteins on the surface of microorganisms as well as those associated with tissue injury. M1 macrophages secrete various inflammatory cytokines, such as TNF-α, IL-6, and IL-12; removes dead tissue, cell debris, and bacteria; and stimulates cell proliferation at the wound site to proceed to the next stage of wound healing33,34. The transformation of M1 macrophages to M2 macrophages implies a transition from the inflammatory stage to the proliferative stage, and M2 macrophages secrete anti-inflammatory factors as well as various growth factors such as TGF-β1, IL-10, VEGF, and they promote cell proliferation and blood vessel generation into the tissue repair stage35,36. When cutaneous wounds occur, macrophages are recruited by chemokines, such as CCL3, to the wound site to remove necrotic tissue, cell debris, and bacteria, and secrete various inflammatory and cell growth factors to promote wound healing. Compared with normal mice, the expression levels of TNF-α, IL-6, VEGF, and TGF-β1 were significantly reduced in the CCL3 knockout mice. It is known that TNF-α and IL-6 can effectively promote the proliferation of keratinocytes and epidermal cells, and accelerate the process of wound healing and reepithelialization37,38. In addition, TNF-α and IL-6 can also promote the expression of VEGF, thus indirectly stimulating angiogenesis39,40. When the expression levels of TNF-α and IL-6 in the body are low, the inflammatory response will be weakened, the process of granulation tissue formation and reepithelialization would be severely hindered, and the wound healing time of mice prolonged 41 . The reduced expression of TGF-β1 might contribute to the delayed cutaneous injury repair, as TGF-β1 is the key and potent growth factor promoting repair and regeneration following skin injury41–43. Furthermore, TGF-β1 can stimulate proliferation of fibroblasts, promote fibroblasts to synthesize and secret collagens, reduce the synthesis of collagenase, inhibit the degradation of ECM, and accelerate the remodeling of blood vessels in wound tissue 25 .
This is the first study to investigate the chemotaxis of CCL3 on macrophages using a CCL3-/- mice skin injury model. Our results indicate that CCL3 plays an important role in tissue recruitment and functions of macrophages in response to various injuries44,45. It shows that the absence of macrophages will slow down wound healing. In addition, CCL3 deficiency severely affects macrophage infiltration and impairs skin repair and regeneration. At the same time, CCL3 is very important for the phagocytic function of macrophages and the production of related growth factors. Macrophages secrete biologically active molecules to regulate the wound microenvironment, promote cell migration, and differentiation and proliferation; promote angiogenesis; restore metabolic function; and ultimately achieve the best healing effect 1 . In view of this result, we speculate that the absence of CCL3 not only stops the recruitment of macrophages to the wound but also affects the recruitment of other related cells and related factors to the wound sites, such as neutrophils, monocytes, and lymphocytes. This speculation has not yet been confirmed by relevant studies. Therefore, it is necessary to use the CCL3-/- mouse skin injury model to study the relationship between CCL3 and other inflammation-related cells.
In summary, the depletion of macrophages in the wound will delay the reepithelialization, reduce the formation of collagen and new blood vessels, and thus hinder the overall wound healing process. However, CCL3 may act like a switch that manipulates the role played by macrophages in the wound healing process. It is known that CCL3 enhances wound healing through three main pathways: (1) acting as a chemokine to recruit specific cell types to the wound site, including neutrophils, monocytes, and macrophages; (2) regulating cell migration in response to injury; and (3) stimulating proliferation of fibroblasts, keratinocytes, and endothelial cells. Absence of CCL3 in the healing tissue would abrogate the accumulation of a variety of growth factors necessary for the wound healing, and, at the same time, it interferes in the interactions between multiple growth factors, hinders the entire wound healing process, and slows down the wound healing process.
Conclusion
Macrophages play an important role in skin wound healing, and CCL3 may be a key chemotactic factor for recruiting macrophages to the wound sites. Removal of macrophages or knocked out CCL3 gene could significantly affect wound healing. Therefore, CCL3 plays an important role in the regulation of macrophages, allowing macrophages to participate in and regulate a variety of biological processes in cutaneous wound healing.
Supplemental Material
sj-docx-1-cll-10.1177_09636897241264912 – Supplemental material for CCL3 Promotes Cutaneous Wound Healing Through Recruiting Macrophages in Mice
Supplemental material, sj-docx-1-cll-10.1177_09636897241264912 for CCL3 Promotes Cutaneous Wound Healing Through Recruiting Macrophages in Mice by Wanwan Shi, Xunsheng Li, Zhen Wang, Chenguang Li, Datao Wang and Chunyi Li in Cell Transplantation
Footnotes
Acknowledgements
The authors thank their colleagues for helpful suggestions and Institutional support.
Author Contributions
C.L. and D.W. contributed to experimental design and article modification; W.S. contributed to writing—original draft; and W.S., X.L., Z.W., and C.L. performed the experiments.
Availability of Data and Material
Data are applicable after the approval of coauthors.
Ethical Approval
All animal experiments were carried out in accordance with the guidelines and research protocols of the Institute of Antler Science and Product Technology, Changchun Sci-Tech University (no. CKARI2020011).
Statement of Human and Animal Rights
This article does not contain any studies with human subjects.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the Joint fund of the National Natural Science Foundation of China (grant no. U20A20403), the National Natural Science Foundation of China (no. 32370899), and the Natural Science Foundation of Jilin Province (20240602030RC).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
