Abstract
Heart failure remains the leading cause of human death worldwide. After a heart attack, the formation of scar tissue due to the massive death of cardiomyocytes leads to heart failure and sudden death in most cases. In addition, the regenerative ability of the adult heart is limited after injury, partly due to cell-cycle arrest in cardiomyocytes. In the current post-COVID-19 era, urgently authorized modified mRNA (modRNA) vaccines have been widely used to prevent severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. Therefore, modRNA-based protein replacement may act as an alternative strategy for improving heart disease. It is a safe, effective, transient, low-immunogenic, and integration-free strategy for in vivo protein expression, in addition to recombinant protein and stem-cell regenerative therapies. In this review, we provide a summary of various cardiac factors that have been utilized with the modRNA method to enhance cardiovascular regeneration, cardiomyocyte proliferation, fibrosis inhibition, and apoptosis inhibition. We further discuss other cardiac factors, modRNA delivery methods, and injection methods using the modRNA approach to explore their application potential in heart disease. Factors for promoting cardiomyocyte proliferation such as a cocktail of three genes comprising FoxM1, Id1, and Jnk3-shRNA (FIJs), gp130, and melatonin have potential to be applied in the modRNA approach. We also discuss the current challenges with respect to modRNA-based cardiac regenerative medicine that need to be overcome to apply this approach to heart disease. This review provides a short description for investigators interested in the development of alternative cardiac regenerative medicines using the modRNA platform.
Keywords
Introduction
Heart failure (HF) is one of the causes of death and hospitalization worldwide, imposing a significant economic burden1–3. When any of the coronary arteries supplying blood to the myocardium becomes narrowed or blocked, it restricts the delivery of oxygen and essential nutrients to the heart, leading to myocardial hypoxia and malnutrition. This, in turn, results in damage to heart tissue, severe shock, or HF. While heart transplantation can address end-stage HF, the limited availability of donor hearts for allotransplantation poses a challenge. Moreover, allograft rejection is a concern, necessitating lifelong immunosuppressive drug therapy for transplant recipients.
Among these, myocardial infarction (MI), also known as acute myocardial infarction (AMI), is a sudden and severe heart condition. It occurs when there is a sudden interruption in the blood flow supplying the heart muscles, resulting in damage to the heart muscle due to oxygen deficiency. MI is often primarily caused by the complete blockage of a coronary artery due to the rupture of an atherosclerotic plaque. The longer the coronary artery remains blocked, the greater the damage to the cardiac muscle, the higher the risk of HF, and the worse the prognosis for the condition4,5. While cardiac catheterization and the placement of cardiac stents can help maintain smooth coronary blood flow and improve MI, the limited regenerative capacity of damaged cardiomyocytes restricts cardiac repair. Moreover, there are numerous causes of heart attacks, and any damage to the heart leaves a lasting scar on heart tissue that is difficult to repair. This scar can also disrupt the heart’s electrical conduction, leading to irregular heartbeats. This not only reduces blood flow throughout the body but also places additional stress on the heart 6 . Consequently, ischemic heart disease remains a significant global cause of disability and healthcare expenditure 7 .
Commonly used therapeutic drugs today include beta-blockers, which initiate cellular changes and inhibit cardiomyocyte loss through multiple mechanisms; angiotensin-converting enzyme inhibitors, which reduce both blood preload and afterload, resulting in positive effects on cardiac function; and angiotensin II receptor blockers, which inhibit the stimulation of the renin-angiotensin-aldosterone system8,9. These drugs effectively enhance the quality of life for patients with heart disease. Early reperfusion can reduce myocardial injury and significantly lower mortality rates in most patients with acute coronary syndromes. Revascularization methods, such as catheter-based reperfusion, fibrinolytic therapy, and coronary artery bypass surgery, are also employed for these patients. However, HF occasionally occurs in some patients due to suboptimal anatomy or restenosis of the coronary arteries. With modern treatment modalities, current guidelines for immediate revascularization, and access to multidisciplinary heart teams, short-term mortality rates, including in-hospital mortality, have decreased to 5% to 8% 10 . The overall mortality associated with ischemic heart disease due to AMI is approximately 33% 11 . In order to better assist patients with HF, comprehensive treatment approaches may be considered.
During HF, the loss of billions of cardiomyocytes leads to the formation of nonfunctional fibrotic scar tissue, which significantly impairs the pumping ability of the myocardium 12 . The remaining cardiomyocytes have limited regenerative capacity because the lost cardiomyocytes cannot be replaced. A study reported that proliferating cardiomyocytes are mainly found in fetuses and decrease after birth13,14. Therefore, one of the primary constraints on cardiac regeneration lies in the limited regenerative capacity of cardiac cells. Cardiac cells typically exhibit heightened regenerative potential in the early stages following birth; however, this capability significantly diminishes over time. In adulthood, the heart primarily relies on repair mechanisms for damaged areas, often involving the formation of scar tissue rather than a complete regeneration of cardiac cells. Promoting cardiac regeneration through cardiomyocyte proliferation may be effective in treating ischemic heart disease. Given the various roles of RNA molecules in biological processes, including disease initiation and progression, RNA has emerged as a promising therapeutic option for various diseases 15 . The development of innovative technology platforms for the rapid, transient, and quantitative expression of proteins in vivo following heart damage in both animal and clinical settings can enhance our ability to quickly translate new findings from traditional genetic model systems to clinical applications. To achieve this goal, synthetic modified mRNA (modRNA) may serve as an alternative approach to generate functional proteins for cardiac regeneration, both in vitro16,17 and in vivo 18 . In this review, we summarize several factors that have been employed with the modRNA method to promote cardiomyocyte proliferation for the improvement of heart disease. In addition, we discuss other potential cardiac factors, modRNA delivery methods, and injection techniques using the modRNA approach to explore their potential applications in heart disease.
Modified mRNA
mRNA is a natural molecule that efficiently and accurately converts information from DNA to protein to carry out its physiological function. In 1990, Wolff et al 19 . first reported that naked-mRNA-encoded luciferase, chloramphenicol acetyltransferase, or beta-galactosidase was separately injected into skeletal muscle and then immediately produced protein in mice. Since then, synthetic in vitro mRNA has been widely utilized in vaccinations for treating cancer and infectious diseases20–27. However, mRNA strategy has not been considered as a therapeutic approach for other diseases due to mRNA instability and immunogenicity; thus, the application of synthetic mRNA for gene replacement therapy has been delayed. When exogenous mRNA enters the cell, it binds to the Toll-like receptors (TLRs) in the endosome and then activates the innate immune system, leading to the production of type I interferon and inflammatory cytokines, and finally leading to the termination of the target protein synthesis and cell death. TLR3 recognizes the double-stranded RNA 28 , while TLR7 and TLR8 recognize the single-stranded RNA29,30. Poly-U can be recognized by TLR7 31 . Therefore, unmodified exogenous mRNA is highly immunogenic to elicit innate immunity. Unmodified exogenous mRNA-induced interferons activate intracellular RNase, which eventually degrades exogenous mRNA32,33. In addition, various RNases are abundant in plasma and mammal cells. The host itself also uses RNases to manipulate the quantity of endogenous mRNA in the cell through the degradation of mRNA 34 . Consequently, the half-life of unmodified exogenous mRNA is short in vivo and the quantity of protein expressed utilizing exogenous mRNA templates are limited. This process means that the application of unmodified exogenous mRNA has limitations in gain-of-function studies.
In 2005, Karikó et al.’s 29 research overcame the above issues and provided an mRNA therapy platform for immunotherapy, regenerative medicine, and cancer vaccination. They first used chemically modified mRNA (modRNA) to produce target proteins in mammalian cells in vitro. In vitro transcription was performed using modified nucleotides, initially replacing uridine with pseudouridine and cytosine with 5-methylcytidine (m5C). The observation was that the incorporation of one or two modified nucleotides into modRNA synthesis in vitro resulted in the secondary structure change of modRNA, allowing modRNAs to evade the host’s innate immunity while still enabling effective translation for target proteins in mammalian cells. In that study, it was found that mRNA-elicited dendritic cell activation was dramatically suppressed when modified nucleosides such as m5C, 5-methyluridine (m5U), N6-methyladenosine (m6A), 2-thiouridine (s2U), and pseudouridine (ψU) were incorporated into mRNA 29 . The authors also found that purified modRNA created using high-performance liquid chromatography (HPLC) to remove contaminants significantly produced the encoded target protein and did not induce pro-inflammatory cytokines 35 . In 2010, Warren et al. 16 first used this safe and efficient modRNA approach to transiently express pluripotent proteins including KLF4, c-MYC, OCT4, SOX2, and LIN28 using several modifications such as m5C, ΨU, 5′-UTR containing the Kozak sequence, α-Globin 3′ UTR, poly-A tail, and anti-reverse cap analogs (ARCAs), which resulted in reprogramming somatic cells into RNA-induced pluripotent stem cells (RiPSCs) in vitro. Since then, modRNA technology has been largely applied to RiPSC reprogramming and therapeutic cell transdifferentiation in vitro 36 . modRNA approach also used for protein replacement has gradually been applied to animal disease models. For example, an aerosol of surfactant protein B (SP-B) modRNA was administered twice weekly, which prevented lethal congenital lung disease caused by SP-B deficiency and achieved 71% SP-B protein expression in an animal model 18 . The modRNA used contained modifications including ARCA, poly-A tail, s2U, m5C, ΨU, and N6-methyladenosine (m6A). In other studies, the injection of erythropoietin (EPO) modRNA increased the hematocrit from 43% to 64% in mice or macaques, suggesting that modRNA-encoded functional proteins hold great promise in disease applications18,37. The EPO modRNA used in these experiments was characterized by a natural 5′ cap, a poly-A tail, and a ΨU modification. Moreover, EPO modRNA containing pseudouridine was injected into animals, and pro-inflammatory cytokines were undetected in serum, suggesting that modRNA containing pseudouridine is non- or low-immunogenic 37 . In 2013, Zangi et al 38 . first used human vascular endothelial growth factor-A (VEGF-A) modRNA to treat MI, which resulted in the improvement of cardiac function through vascular and cardiac regeneration. The modRNA utilized in this treatment contained ARCA, poly-A tail, m5C, and ΨU modifications. Since 2019, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has invaded the world, which has promoted the generation of modRNA-based vaccines such as Moderna 1273 with N1-methyl-pseudouridine modification and Pfizer/BNT 162b2 with proline mutation against the COVID-19 pandemic 39 . In addition, interleukin (IL)-10 modRNA has been used to directly produce the anti-inflammatory IL-10 protein in vivo, resulting in prolonged survival of facial allografts through the induction of mixed chimerism 40 . The modRNA for this application included modifications such as m5C, ΨU, a 5′-UTR containing the Kozak sequence, 3′ UTR, poly-A tail, and ARCA.
In vitro-transcribed (IVT) mRNA is designed to mimic the structure of mature and processed mRNA found in the cytoplasm of eukaryotic cells. Unlike natural mRNA, which is synthesized in the nucleus and transported to the cytoplasm through nuclear export mechanisms, IVT mRNA must directly enter the cytoplasm from the extracellular environment. This entry is crucial for the pharmacodynamic activity of IVT mRNA, which primarily occurs in the cytoplasm. The process for cellular uptake of IVT mRNA is a complex multistep mechanism, initially involving endocytosis, where IVT mRNA is encapsulated by the plasma membrane and subsequently internalized within endosomes. These endosomes then break down, releasing the IVT mRNA into the cytoplasm for immediate protein synthesis41,42. However, unmodified IVT mRNA might activate innate immune responses, as human TLR8 and mouse TLR7 on endosomal membranes can recognize single-stranded RNA sequences, triggering an immune response. In addition, unmodified IVT mRNA is more prone to RNase degradation. To enhance its functionality and reduce immunogenicity, IVT mRNA can be synthesized by incorporating modified nucleosides like pseudouridine, 2-thiouridine, 5-methyluridine, 5-methylcytidine, and N6-methyladenosine29,37,43–45. These modifications suppress the inherent adjuvant activity and translational inhibition associated with IVT mRNA. Furthermore, they can facilitate the escape of TLR7- and TLR8-elicited innate immunity, and specific modifications, such as pseudouridine and 2-thiouridine, render the IVT mRNA undetectable by immune sensors like retinoic acid-inducible gene I protein (RIG-I) and protein kinase RNA-activated (PKR). PKR initiates a critical mechanism in the cellular response to RNA sensing, where it phosphorylates the eukaryotic translation initiation factor 2α. This action by PKR significantly inhibits the efficiency of mRNA translation, often causing a halt in translation, and promoting RNA degradation 46 . RIG-1 is activated by short double-stranded RNA molecules with a 5′ triphosphate group and triggers interferon production 47 . The advantage of using pseudouridine-modified mRNA includes improved translation efficiency, attributed to increased stability and decreased interaction with PKR. The strategic insertion of modifications like 1-methyl-pseudouridine (1-mΨU) into mRNA significantly curtails TLR3-mediated immunogenicity and enhances resistance to RNase degradation. These modifications not only reduce immunogenicity by preventing TLR activation but also result in lower activation of innate immunity genes in cells, thereby offering a more stable and less immunogenic therapeutic mRNA construct. The removal of double-stranded RNA contaminants through HPLC further diminishes any residual immunostimulatory effects, rendering the modified IVT mRNA immune-silent 48 .
Therefore, modRNA has the potential to express functional proteins to improve or prevent many diseases through the gain of function or correction of gene deficiency. Although recombinant protein therapy can immediately administrate proteins to patients and the concentrations of functional proteins can be controlled, recombinant proteins are unable to deliver into cells and need repeat administrations due to their instability and short half-life. In addition, although adeno-associated viruses (AAVs) can deliver high expression levels of target proteins in the long term, the levels of expressed proteins are not accurately controlled in vivo. AAVs can also induce neutralizing antibodies, which can entirely nullify the vector’s transduction capability, consequently diminishing the effectiveness of gene transfer49–51. Taken together, the modRNA approach overcomes these disadvantages to transiently express high levels of target proteins in a secreted or intracellular form for the short term (average 7 days). It has very low immunogenicity and is more stable than unmodified mRNA. It exhibits safe properties due to no genomic integration. The advantages and disadvantages of modRNA and viral gene delivery systems are compared in Table 1.
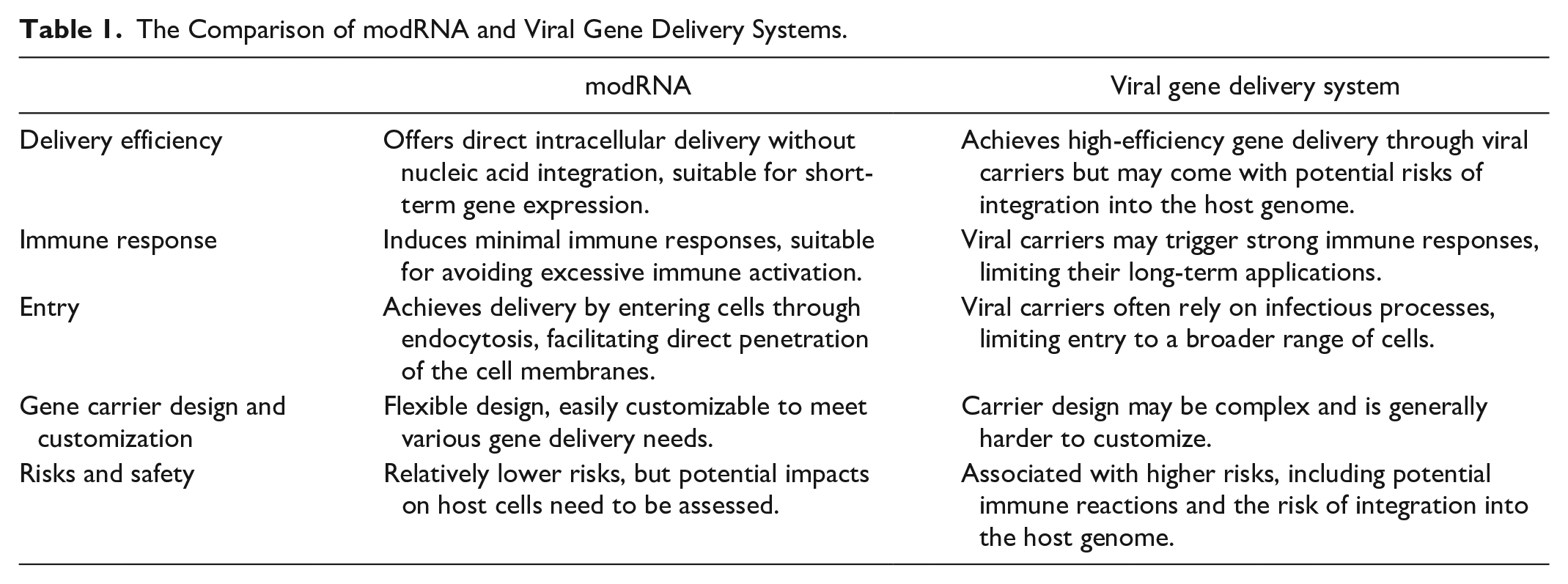
The Comparison of modRNA and Viral Gene Delivery Systems.
The construct of the modRNA template generally contains a 5′ cap, 5′ untranslated region (UTR), open reading frame (ORF), 3′-UTR, and poly-A tail. The natural 5′ cap has the risk of incorporating into the reverse orientation when IVT is performed. Thus, an ARCA was designed and developed to replace a natural 5′ cap and prevent the occurrence of reverse orientation in IVT, which resulted in the stability and increased translation efficiency of modRNA 52 . Optimization of the 5′-UTR and 3′-UTR vector sequences can also enhance the stability and increase the translational efficiency of modRNA. Thus, the construction of modRNA template usually involves highly stable UTRs from α/β-globin DNA sequences. For example, a 5′-UTR containing a Kozak sequence and the α-globin 3′-UTR were used in the generation of RiPSCs via the modRNA-based reprogramming method16,36. The 5′- and 3′-UTRs of Xenopus β-globin were also constructed into the modRNA template for the generation of RiPSCs 53 . Although many modified nucleotides have been discovered, only certain modifications have specific functions. Modified nucleotides such as m5C, m5U, m6A, s2U, and ΨU were reported to be low- or nonimmunogenic. Modified nucleotides such as m5C, m5U, m6A, N1-methyl-pseudouridine (m1Ψ), and 5-methoxyuridine were reported to increase the translational efficiency and stability of modRNA18,36,41,54. A poly-A tail containing 120 to 150 nucleotides can be cloned into the modRNA template vector following the 3′-UTR. Another method involves the use of poly(A) polymerase to generate a poly-A tail when in vitro transcription is performed.
Modified mRNA Application in Heart Disease
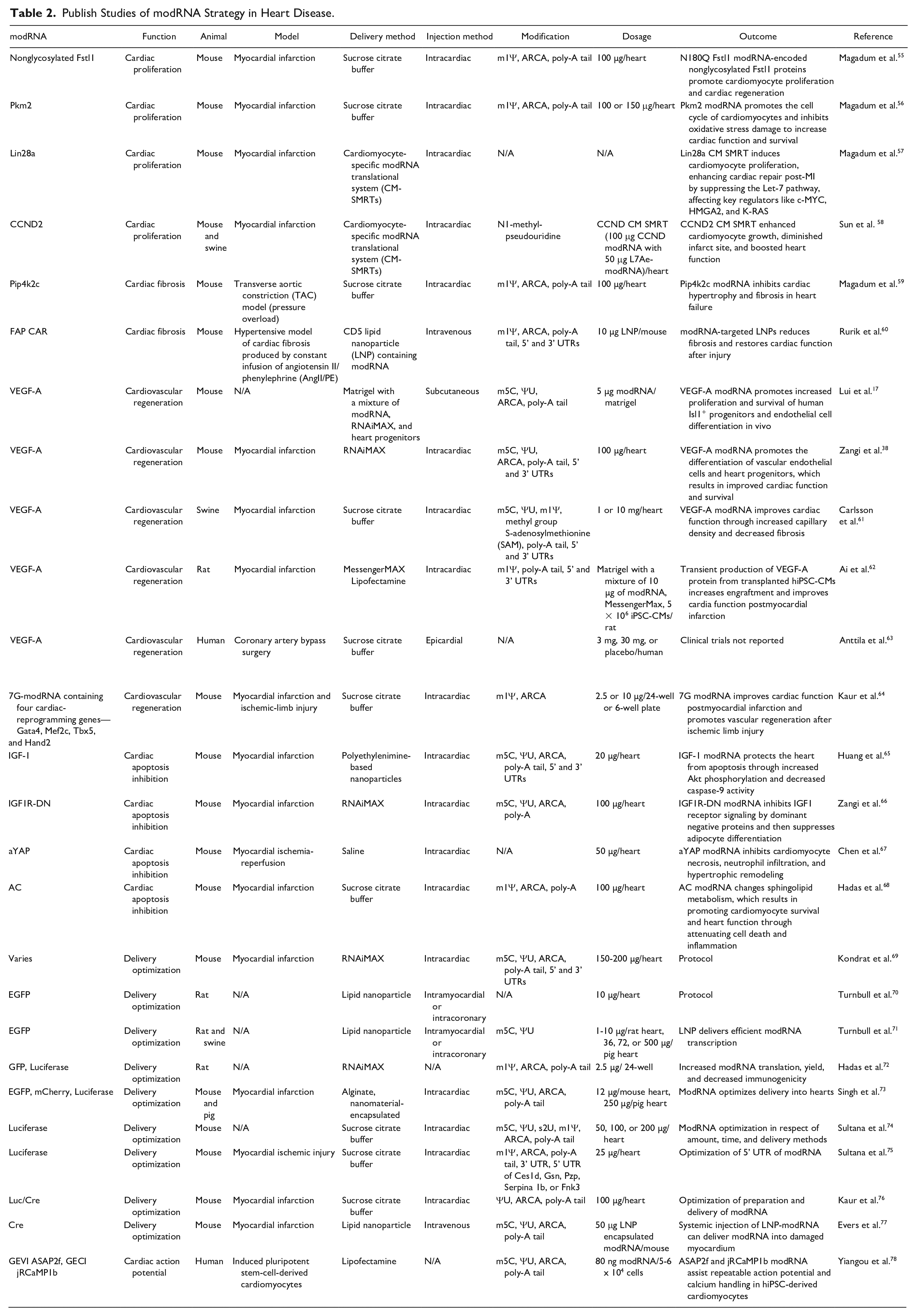
To date, 25 studies have used modRNA technology to produce target proteins in animals to prevent heart disease, including 4, 2, 6, 4, and 9 papers related to cardiac proliferation, cardiac fibrosis, cardiovascular regeneration, cardiac apoptosis inhibition, and modRNA delivery optimization, respectively. The target proteins, animal model, outcome, delivery, and injection methods of modRNA used in these studies are summarized in Table 2 and Fig. 1.
Publish Studies of modRNA Strategy in Heart Disease.
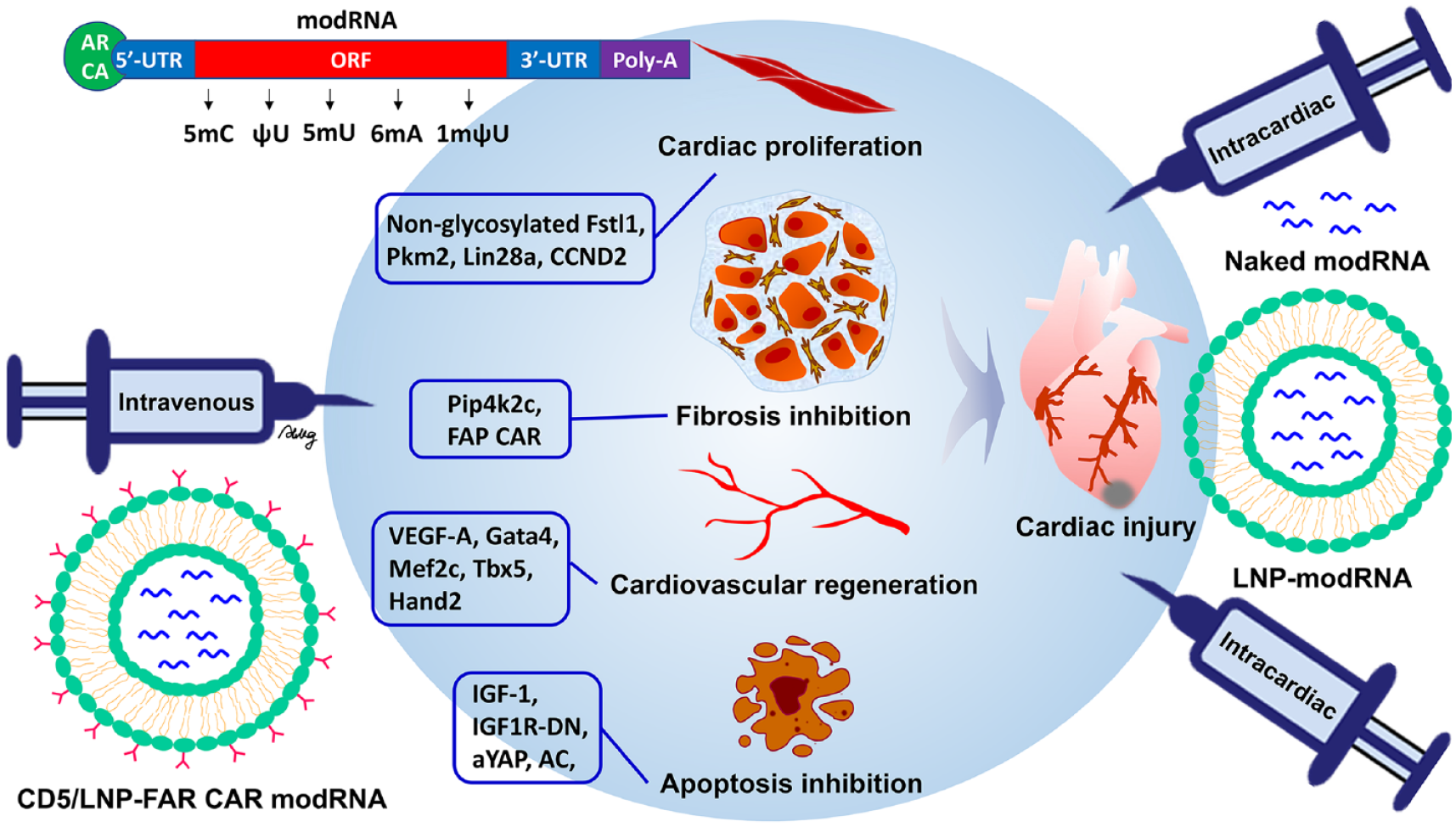
Utilizing the modRNA platform for direct cardiac repair involves delivering target genes to induce cardiac proliferation, inhibit fibrosis and apoptosis, and promote cardiovascular regeneration, as presented in Table 2.
In terms of cardiac proliferation, Magadum et al55,56. demonstrated that nonglycosylated follistatin-like 1 (Fstl1) and pyruvate kinase muscle isozyme 2 (Pkm2), encoded from their respective modRNAs, promoted cardiac proliferation and regeneration in a mouse MI model through intracardiac injection. These modRNAs were modified with m1Ψ, ARCA, and a poly-A tail. They found that glycosylated hFstl1 did not induce cardiomyocyte proliferation, whereas nonglycosylated Fstl1 did. Therefore, a mutation at a single site of N180Q was made to generate nonglycosylated Fstl1, which increased rat cardiomyocyte proliferation in vitro 55 . However, loss of function due to deleting Pkm2 specifically in cardiomyocytes showed a decrease in cell numbers, cell cycle, and myocardial size. In addition, the gain of function by Pkm2 modRNA inhibited oxidative-stress-induced damage via the β-catenin and anabolic pathways, which impacted the function and survival of the heart 56 . Recently, they also utilized the cardiomyocyte-specific modified mRNA translational system (CM SMRT) to enhance the cardiomyocyte-specific expression of the target protein and enhance cardiac repair post-MI, while other noncardiomyocyte cells do not express the target protein 57 . The CM SMRT is designed to induce cell-specific expression of Lin28a, which is known to stimulate cardiomyocyte proliferation and division. This system utilizes two modRNA constructs: one encoding Lin28a and the other encoding the L7Ae protein, which is regulated by cardiomyocyte-specific microRNAs miR-1 and miR-208, ensuring targeted expression in cardiomyocytes. This Lin28a CM SMRT targeted approach leads to effective cardiac repair and improved outcomes post-MI. The study also details Lin28a’s molecular pathway in CMs, highlighting its suppression of Let-7 microRNA, which is a negative regulator for CM proliferation, partly due to its influence on c-MYC, HMGA2, and K-RAS. This research demonstrates the potential of Lin28a CM SMRTs and modRNA technology in inducing cardiac regeneration and improving cardiac function after MI. Sun et al. also utilized the CM SMRT system to specifically deliver target proteins, such as cyclin D2 (CCND2), exclusively to cardiomyocytes, excluding non-cardiomyocytes. CCND2 is known to stimulate cardiomyocyte proliferation and aid in cardiac repair 58 . The study demonstrates that intramyocardial injections of CCND2 CM SMRTs in mouse and pig models of MI significantly promoted cardiomyocyte proliferation, reduced infarct size, and improved cardiac function. The research underlines the system’s efficacy and specificity in driving CCND2 expression within cardiomyocytes, leading to enhanced cardiac regeneration and repair while minimizing the risk of uncontrolled cell proliferation or other potential adverse effects associated with long-term gene overexpression.
In terms of cardiac fibrosis, mTORC1 is a critical regulator for controlling cardiac hypertrophy, which usually causes HF. Type 2 phosphatidylinositol-5-phosphate 4-kinase-gamma (Pip4k2c) is a regulator for mTORC1 and TGFβ1 79 . Therefore, Magadum et al 59 . also utilized modRNA technology to transiently overexpress Pip4k2c in cardiomyocytes, which led to the reversal of cardiac fibrosis and hypertrophy in a mouse transverse aortic constriction (TAC) model. The modRNA employed contained m1Ψ, ARCA, and a poly-A tail. In contrast, Pip4k2c−/− mice showed a high frequency of cardiac fibrosis and hypertrophy, and even sudden death, after TAC. Their data indicate that Pip4k2c modRNA inhibits mTORC1-mediated cardiac hypertrophy and TGFβ1-mediated cardiac fibrosis, thereby reducing both hypertrophy and fibrosis, which results in improved cardiac function and survival following TAC injury. Furthermore, they demonstrate that the N-terminal motif (VMLLPDD) on Pip4k2c is directly responsible for its suppression of the TGFβ1 pathway. In 2022, Rurik et al 60 . advanced the use of modRNA technology to directly generate chimeric antigen receptor (CAR) T cells in vivo, targeting cells expressing fibroblast activation protein (FAP). The modRNA used in this process was characterized by modifications such as m1Ψ, ARCA, poly-A tail, and both 5′ and 3′ UTRs. FAPCAR modRNA was embedded in lipid nanoparticles (LNPs) and then conjugated with an anti-CD5 antibody, defined as CD5-targeted LNP-FAPCAR (CD5/LNP-FAPCAR). The CD5/LNP-FAPCAR particles specifically recruited T cells in the mice with HF after the intravenous injection, and then genetically manipulate T cells in vivo to CAR T cells against FAP-expressed cardiomyocytes because CD5 is a T-cell surface marker. Subsequently, these antifibrotic CAR T cells migrated to the fibrotic heart and inhibited cardiac fibrosis, which resulted in heart functional recovery.
In terms of cardiovascular regeneration, five studies used VEGF-A modRNA to examine whether it is helpful for cardiovascular disease17,38,61–63. One report demonstrated that vascular endothelial growth factor [VEGF]-A modRNA promotes the increased proliferation and survival of human Isl1+ progenitors, which then differentiate into cardiac endothelial cells in NOD/SCID mice following subcutaneous injection of a modRNA transfection-mixture-embedded Matrigel 17 . The modRNA used in this study included modifications such as m5C, ΨU, ARCA, and a poly-A tail. Isl1 was identified as a marker for endothelial cells in hearts and arteries. Four reports investigated the efficacy of VEGF-A modRNA in the improvement of MI, such as in mice 38 , rats 62 , pigs 61 , and human 63 . In mice or swine models of MI, cell-free therapies have significantly enhanced cardiac function and survival. This improvement is attributed to the increased differentiation of vascular endothelial cells and heart progenitors, and the reduction of fibrosis, following intramyocardial injection of purified modRNA38,61. The mouse VEGF-A modRNA was characterized by modifications such as m5C, ΨU, ARCA, a poly-A tail, and both 5′ and 3′ UTRs. The swine VEGF-A modRNA included m5C, ΨU, m1Ψ, a methyl group from S-adenosylmethionine (SAM), a poly-A tail, and also 5′ and 3′ UTRs. These modRNAs combined with transfection reagents, facilitated the therapeutic outcomes in the respective animal models. In addition, cell therapy using human-induced pluripotent stem cell-derived cardiomyocytes (hiPSC-CMs) transfected with VEGF-A modRNA also enhanced engraftment survival and improved rat cardiac function following MI 62 . The VEGF-A modRNA used in this therapy included modifications such as m1Ψ, a poly-A tail, and both 5′ and 3′ UTRs.
The prolonged survival of transplanted iPSC-CMs may be due to upregulating the phosphoinositide 3-kinase (PI3K)/protein kinase B (Akt) and AGE/RAGE signaling pathways in VEGF-A modRNA-transfected iPSC-CMs. Moreover, transient production of VEGF-A protein from transplanted hiPSC-CMs assisted the grafted region to form a stable vascular network, and then reduced the hypoxic environment in the infarcted area, contributing to reduced ventricular remodeling. VEGF-A modRNA strategy was also applied in a phase 2a human clinical trial in patients undergoing coronary artery bypass surgery 63 . ClinicalTrials.gov NCT03370887 was a randomized, double-blind, placebo-controlled study for evaluating the angiogenesis efficacy and safety of VEGF-A modRNA. In addition to VEGF-A, other modRNAs promoting angiogenesis were used for cardiovascular disease. A cocktail modRNA, named 7G-modRNA, including cardiac reprogramming factors such as Gata4, Mef2c, Tbx5, and Hand2, was demonstrated to reprogram cardiac fibroblasts into cardiomyocyte-like cells64,80. The 7G-modRNA cocktail can treat MI by reducing the scar area and improving cardiac function, survival, and capillary density. The cocktail also induces vascular regeneration after ischemic limb injury.
In terms of cardiac apoptosis inhibition, four studies used modRNA technology to transiently express functional proteins to protect the cardiomyocytes from apoptosis and necrosis through the regulation of insulin-like growth factor-1 (IGF-1), TLRs, and sphingolipid signaling pathways65–68. When the IGF-1 ligand interacts with the IGF-1 receptor, this results in autophosphorylation of the intracellular domain of the receptor and stimulates the activation of PI3K. It causes AKT activation via the phosphorylation of threonine 308 and serine 473, leading to the regulation of downstream signaling pathways, including cell proliferation and survival promotion, through increased BCL-2, FOXO1, and FOXO3a, as well as apoptosis inhibition through decreased BAD. In addition, autophosphorylation of the IGF-1 receptor drives the ERK signaling pathway for cell proliferation 81 . Therefore, certain studies have employed the manipulation of the IGF-1 signaling pathway and identified a positive correlation with cardioprotection, using cardiac-specific IGF-1 receptor transgenic mice 82 . The expansion of cardiovascular progenitors derived from human pluripotent stem cells was achieved by introducing regulated MYC expression in conjunction with IGF-1 and a hedgehog pathway agonist 83 . In particular, the intracardiac injection of IGF-1 modRNA protects the heart from apoptosis, leading to cardiac survival following MI 65 . This protection is mediated through increased Akt phosphorylation and decreased caspase-9 activity. The IGF-1 modRNA used for this purpose was modified with m5C, ΨU, ARCA, a poly-A tail, and included both 5′ and 3′ UTRs. In contrast, the IGF-1 signaling pathway had a negative effect on cardiac protection through the increase in epicardial adipose tissue (EAT) 66 . Studies reported that EAT volume and cardiovascular disease (CVD) are strongly related, suggesting that EAT has a pathophysiological role84,85. Zangi et al 66 . demonstrated that IGF1 modRNA stimulates receptor activation and epicardial adipocyte differentiation, while modRNA of a dominant negative IGF1 receptor mutant (IGF1R-DN) inhibits this signaling pathway, preventing adipocyte differentiation and ultimately resulting in cardiac protection following MI. The IGF1R-DN modRNA employed for this inhibition contained modifications such as m5C, ΨU, ARCA, and a poly-A tail. Transient expression of the transcriptional coactivator yes-associated protein (YAP) in the heart using modRNA technology demonstrated the inhibition of cardiomyocyte necrosis, neutrophil infiltration, and hypertrophic remodeling in a mouse ischemia/reperfusion model, partially through the TLR signaling pathway 67 . YAP can suppress cardiac TLR expression and inhibits TLR-induced cardiac inflammation and inflammatory cytokine production, leading to cardiomyocyte survival 86 . The accumulation of cardiac ceramide had a positive correlation with cardiomyocyte cell death and deteriorated heart function, suggesting that increased ceramide has pathophysiological significance in heart function87,88. Acid ceramidase (AC) is a well-known enzyme that hydrolyzes proapoptotic ceramide to generate sphingosine 1-phosphate (S1P) 89 . Therefore, Hadas et al. employed AC modRNA with m1Ψ, ARCA, and a poly-A to treat MI. Their observations indicated the prolonged survival of cardiomyocytes and improved heart function, achieved by attenuating cell death and inflammation 68 .
Synthetic modRNA provides a new approach to gene therapy that provides transient, safe, nonimmunogenic, and controlled mRNA delivery to cardiac tissue without the risk of genomic integration. In order to enhance its effectiveness in vivo, consistent and reliable delivery methods need to be utilized. To maximize the delivery efficiency of modRNA and the yield of modRNA in in vivo applications, several studies have focused on modRNA delivery optimization69–77. Two delivery systems, that is, lipid nanoparticles (LNPs) and sucrose citrate buffer, are the most commonly used to transfer modRNA into cardiomyocytes via intracardiac injection. EGFP or luciferase is usually used as the reporter protein to observe the modRNA-mediated biodistribution in the heart. Due to the complexity of preparing modRNA and the high cost of its reagents, an improved, cost-effective protocol for preparing modRNA is needed. The enhancement of translation efficiency in the cardiac tissue and the requirement for the reduction of modRNA have been studied, including optimization of the 5′-UTR of modRNA 75 , ratio optimization between anti-reverse cap analog and N1-methyl-pseudouridine 72 , and formulated lipidoid nanoparticles (FLNPs) 71 . In addition, the ratio optimization between ARCA and m1Ψ reduces RNA immunogenicity. These optimized protocols will make modRNA preparation more accessible and financially affordable for basic and translational applications.
Modified mRNA Candidates in Cardiac Regeneration
The studies in Table 2 prove that modRNA technology can directly express functional proteins in the heart tissue of animals to achieve cardiac repair and functional recovery. Moreover, modRNA-based vaccines have been used worldwide against COVID-19 disease in humans. modRNA technology has also been applied in several human clinical trials, and its use in basic and translational science is growing. Therefore, we list potential genes associated with cardiomyocyte proliferation in Table 3 for study and development of modNRA-based genetic medicine90–92. A preliminary screening of target gene studies related to cardiomyocyte proliferation was conducted from the PubMed database. The preliminary selected genes include skNAC, relaxin, a cocktail of three genes comprising FoxM1, Id1, and Jnk3-shRNA (FIJs), GDF11, IL-13, gp130, Nrf1, Hotair, LSD1, melatonin, IL-4, IL-6, and Caveolin 190–100. Some of these studies only focused on in vitro proliferation and did not investigate in vivo cardiomyocyte proliferation, such as Relaxin 94 . In addition, some studies solely utilized neonatal animals to explore cardiac proliferation, lacking investigations on adult animals regarding the potential for in vivo cardiac regeneration after heart injury. Therefore, Relaxin, IL-13, Hotair, LSD1, IL-4, IL-6, and Caveolin were excluded from further consideration94,96,98,99. Among the remaining potential target genes, skNAC gene deficiency in adult mice exhibited reduced skeletal muscle growth and impaired regeneration after cardiotoxin-induced injury 93 . This study primarily investigated the role of skNAC in leg muscle growth rather than cardiomyocyte proliferation. While repeated targeted delivery of the GDF11 gene through ultrasound-targeted microbubble destruction (UTMD) restored vitality to the aging mouse heart and protected it from ischemia/reperfusion damage, the study mainly demonstrated increased proliferation of cardiac stem cell antigen 1 (Sca-1+) and enhanced angiogenesis, without a focus on cardiomyocyte proliferation 95 . The study on Nrf1 confirmed its ability to promote cardiac regeneration through increased pH3+ cardiomyocytes in heart sections; however, this experiment was conducted using a neonatal MI model rather than an adult MI model 97 . Consequently, skNAC, GDF11, and Nrf1 were excluded from the potential candidates for promoting cardiomyocyte proliferation93,95,97.
Potential Candidates for modRNA-Based Protein Replacement in Cardiac Regeneration.
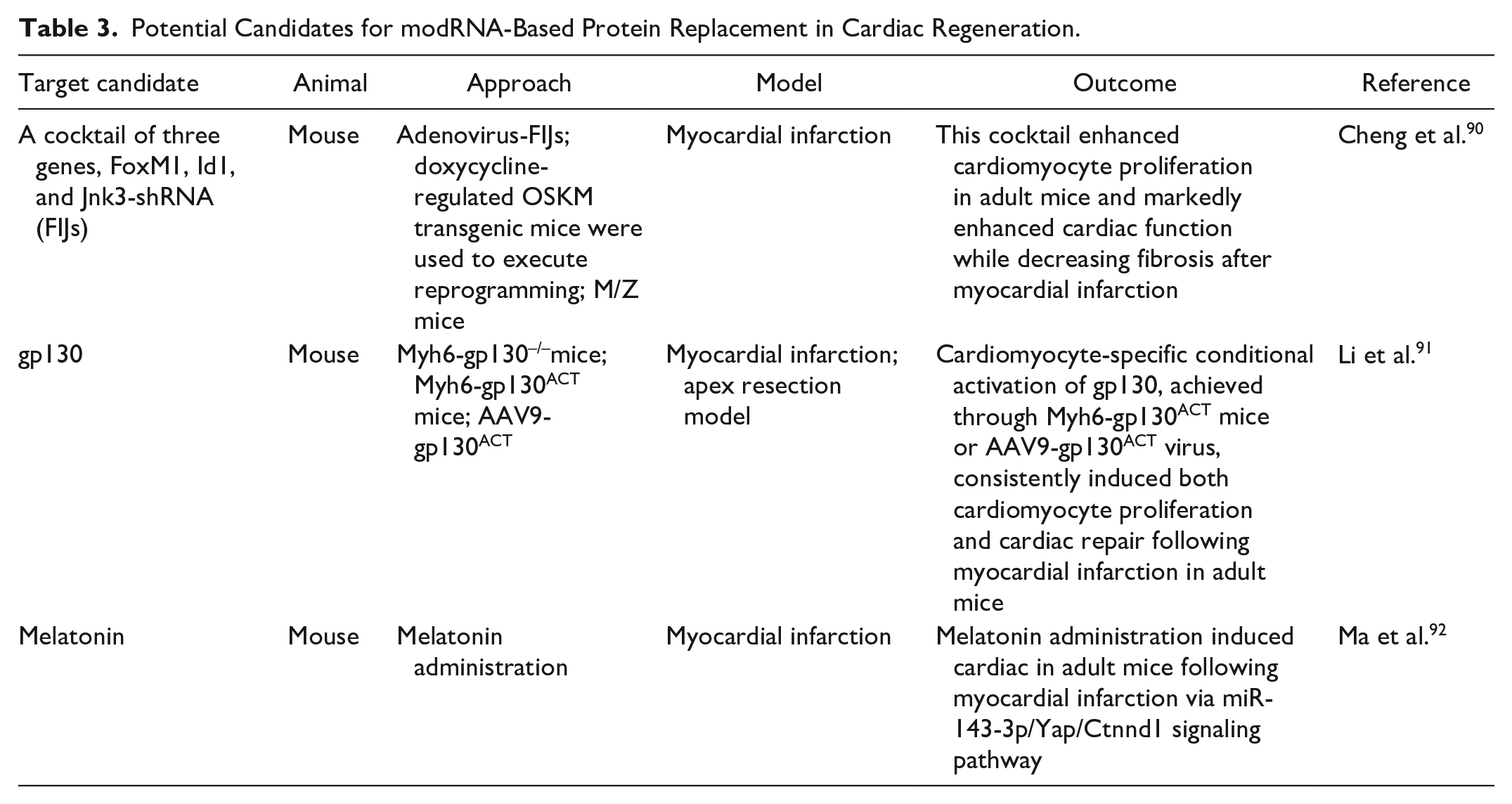
The selection criteria for potential genes were based on experiments using adult animals postheart injury to confirm cardiac functional recovery and increased cardiomyocyte proliferation, as indicated by the expression of proliferative markers, pH3, Ki67, BrdU, or Aurora B. Subsequently, three sets of target genes met these criteria: a cocktail of three genes comprising FoxM1, Id1, and Jnk3-shRNA (FIJs), gp130, and melatonin90–92. Loss- and gain-of-function studies are usually used to investigate the potential of these target genes for cardiomyocyte proliferation. In these studies, the deletion of target genes has been used in loss-of-function studies with respect to in vivo cardiomyocyte-specific knockout mice or in vitro siRNA. Gain-of-function studies using transgenic mice, AAV-target genes, and recombinant protein administration have been used to investigate the potential of target genes in respect of cardiac proliferation in vitro and in vivo. The animal model of MI is primarily used to explore the potential of these target genes for cardiac regeneration and repair in vivo.
Cheng et al 90 . introduced a gene cocktail known as FIJs, comprising FoxM1, Id1, and Jnk3-shRNA, which has demonstrated significant potential in promoting cardiomyocyte proliferation and enhancing cardiac repair after adult MI. Each gene in the cocktail plays a crucial role in regulating the cell cycle and cardiac function. FoxM1, a key component of FIJs, is known to enhance cardiomyocyte proliferation by down-regulating p21 and p27. Id1, another component, prevents senescence by reducing p16 expression. Jnk3-shRNA, the third member, stabilizes p27, leading to enhanced expression of Cdk2 for proliferation. The mechanistic basis of FIJs involves the up-regulation of mitosis markers, including Aurkb, Mad2L1, and Plk1. The combination of these genes effectively controls cyclin-dependent kinases (CDKs) and their inhibitors, promoting the expression of Cdk1, Cdk2, and Cdk4. This regulation leads to increased cardiomyocyte proliferation, as evidenced by elevated levels of proliferative markers such as H3P, Ki67, AURKB, and BrdU. In vivo studies using FIJs in animal models demonstrated improved cardiac function, reduced fibrosis, and increased ejection fraction after MI. The gene cocktail not only induces cardiomyocytes to re-enter the cell cycle but also supports complete mitosis and cytokinesis. The three-gene combination resulted in higher cardiomyocyte proliferation compared to single or double treatments. Further optimization of the cocktail’s concentration and ratio, along with careful evaluation of its clinical application, is essential. Consideration of time points, treatment periods, and potential long-term effects is crucial for future clinical implementation. It is noteworthy that the FoxM1 and Id1 genes can utilize the modRNA strategy to produce these target proteins, whereas the inhibition of Jnk3 expression could be achieved using a Jnk3 inhibitor, shRNA, or siRNA.
Li et al 91 . indicated that gp130, a critical regulator of cardiomyocyte proliferation and heart regeneration, plays a pivotal role in addressing the challenges posed by the inability of cardiomyocytes to renew after cardiac injury. The gene’s significance is highlighted by its potential therapeutic application in treating cardiovascular diseases, particularly MI. The study involved various genetic mouse lines, including global oncostatin M (OSM) knockout, monocyte-/macrophage-specific OSM deletion, and cardiomyocyte-specific lines with OSM receptor and gp130 deletion. The results demonstrated that OSM acts as a key upstream regulator of cardiomyocyte proliferation during heart regeneration. Conditional knockout of OSM in macrophages led to the suspension of cardiomyocyte proliferation and heart regeneration in mice after cardiac injury. Cardiomyocyte-specific deficiency of the OSM receptor heterodimers, OSM receptor, and gp130 individually reduced cardiomyocyte proliferation and hindered heart regeneration. However, the conditional activation of gp130 in cardiomyocytes promoted cardiomyocyte proliferation and heart regeneration in adult mice. Src-mediated gp130-triggered cardiomyocyte proliferation was identified, with the activation of Yap through Src playing a crucial role in this process independently of the Hippo pathway. Further experiments involving cardiomyocyte-specific conditional activation of gp130, achieved through Myh6-gp130ACT mice or gene therapy with AAV encoding constitutively activated gp130 demonstrated its effectiveness in promoting cardiomyocyte proliferation and heart regeneration in adult mice after MI. Myh6-gp130ACT mice were generated using a Cre-dependent mouse with constitutively activated gp130. This was achieved by inserting a cassette into the Rosa26 locus, which contained the coding sequence from Il6st (the gene encoding gp130) with a deletion spanning residues S187 to Y190. This deletion led to the constitutive activation of gp130, resulting in the generation of the gp130ACTfl/fl mouse. In adult mice, the activation of gp130, either through genetic manipulation or AAV9 vectors, increased proliferative cardiomyocytes, reduced the size of the infarcted area, and improved cardiac function. This suggests that gp130, through the gp130-Src-Yap pathway, may serve as a potential therapeutic target for enhancing heart regeneration after cardiac injury. The study highlights the clinical prospects of gp130 as a biological therapy to improve cardiac repair and function in the context of MI.
Ma et al 92 . showed that melatonin plays a crucial role in promoting cardiomyocyte proliferation and heart repair through the Yap/Ctnnd1 signaling pathway. In an adult MI mouse model, melatonin administration significantly improved cardiac functions and enhanced cardiomyocyte proliferation, as evidenced by improved ejection fraction and fractional shortening. Immunofluorescence staining of pH3- and Aurora B positive cardiomyocytes demonstrated increased expression of proliferation-related genes indicating enhanced cardiac myocyte mitosis and cytokinesis activity. Melatonin treatment also attenuated the increase in cardiomyocyte cross-sectional area induced by MI, further supporting its role in improving cardiac function and regeneration. Specifically, melatonin treatment suppressed miR-143-3p levels, leading to enhanced cardiomyocyte proliferation. This effect was reversed when miR-143-3p was overexpressed. Further investigation identified Yap and Ctnnd1 as target genes of miR-143-3p. Inhibition of miR-143-3p increased the protein expression of Yap and Ctnnd1, and melatonin treatment further enhanced their levels. Silencing Yap and Ctnnd1 attenuated melatonin-induced cardiomyocyte cell cycle re-entry. The study revealed that melatonin’s impact on cardiomyocyte proliferation and cardiac regeneration was dependent on melatonin receptors, particularly melatonin receptor 2. Therefore, the underlying mechanism of melatonin’s cardioprotective effects involves the upregulation of Yap and Ctnnd1 through the inhibition of miR-143-3p, facilitated by the activation of melatonin receptors. Given its pro-proliferative abilities and minimal side effects, melatonin may emerge as a promising therapeutic strategy for protecting and regenerating the cardiovascular system.
In the realm of potential applications, the use of modRNA methods emerges as a plausible avenue for harnessing the therapeutic potential of these identified genes. One of the advantages of the modRNA strategy is its ability to deliver protein expression either intracellularly or on the cell membrane, such as the expression of gp130, which is one of the subunits of the OSM receptor and is located on the cell membrane. The ability to modulate gene expression through modified RNA interventions may offer a precise and targeted approach for treating heart injury. While challenges and additional studies lie ahead, the insights gained from these investigations pave the way for innovative therapeutic strategies aimed at cardiac regeneration and functional recovery.
Challenges
The introduction of many chemical modifications to mRNA synthesis has contributed to enhancing target binding affinity, cellular uptake, pharmacokinetics, and drug efficacy, which has resulted in significant progress in modRNA therapy. Due to the outbreak of the SARS-CoV-2 pandemic worldwide in 2019, modRNA-based vaccines were rapidly developed to combat SARS-CoV-2 infection. We are witnessing tremendous advances in the field of modRNA therapeutics, as well as the rapid translation of experimental research into human clinical trials, paving the way for cardiac regenerative medicine. However, several challenges need to be overcome before modRNA-based protein replacement can effectively evolve into clinical cardiac drugs. These are as follows: (a) minimizing off-target effects, (b) minimizing immunogenicity, and (c) improving organ-specific delivery. These remain the most challenging issues before this exciting technology can be brought into clinical practice on a large scale. Scientists are making significant efforts to avoid unexpected effects.
Off-target effects: These involve unexpected side-effects that may be harmful when modRNA strategy is conducted for heart disease. For example, sustained overexpression of VEGF-A causes the leaky formation of blood vessels via vascular hyperpermeability and hypotension via the excessive release of nitric oxide101,102. Although modRNA-induced transient expression can reduce this effect, the optimal dosages of modRNA and the number of injections need to be considered to avoid unexpected toxicity. When MI occurs, the modRNA injection timing is also very important. Understanding its expression period and levels in tissues will allow us to more accurately predict the appropriate timing of treatment, which may affect the outcome of the modRNA strategy.
Immunogenicity: Much evidence indicates that inflammatory immune cells play a critical role in the pathogenesis of cardiovascular disease 103 . Thus, modRNA-elicited immunogenicity is undesirable in the disease. It was found that modified mRNAs evaded the host’s innate immunity and allowed the effective translation of target proteins in vivo when modified nucleosides such as m5C, m5U, m6A, s2U, and ΨU were incorporated into mRNA 29 . modRNA purified via HPLC to remove contaminants was demonstrated to largely reduce effects on the production of pro-inflammatory cytokines 35 . In addition, the incorporation of m1Ψ into modRNA synthesis indicated decreased immunogenicity in mammalian cells by inhibiting endosomal TLR3 activation and downstream innate immune signaling 104 . Therefore, the choice of modified bases for modRNA transcription needs careful consideration.
Organ-specific delivery: According to the current research, direct intracardiac injection of modRNA into myocardial tissue is the most effective method for gene delivery to the heart. However, this direct intracardiac injection will cause stress and local damage to the heart tissue. If repeated injections of modRNA are required, the damage will be increased. Therefore, there is a need to develop modRNA delivery systems that are noninvasive and target cardiac trafficking. A new delivery system comprising selective organ targeting (SORT) lipid nanoparticles including internal charge adjustment (SORT molecules) can specifically be delivered into the lung, spleen, or liver after intravenous injection105,106. In addition, the systemic administration of LNPs for delivering modRNA to the damaged myocardium holds significant promise for modRNA-based cardiac therapies. For instance, employing a noninvasive intravenous injection method to introduce LNPs encapsulating modRNA into the animal’s body, it was observed that LNPs accumulated in the ischemic regions of the heart after ischemia-reperfusion injury. Compared to sham-operated animals, the tissue accumulation of LNP-modRNA increased, leading to elevated production of functional proteins in the heart, albeit at a lower expression level compared to LNP-modRNA delivery to the liver 77 . Finally, plans to develop modRNAs as therapeutic interventions must consider production costs. Given the large quantities of modRNA required to transfect an organ as large as the human heart, production costs must be reduced.
Conclusions
modRNA is a promising approach to improving heart disease because it overcomes key difficulties posed by recombinant protein therapy and traditional mRNA-based gene therapy. While there are three relevant articles discussing the application of modRNA in cardiac therapy today, the literature they have compiled is not the most up-to-date, containing about 11 to 17 references42,107,108. However, in this article, Table 2 and Fig. 1 present a compilation of approximately 25 articles on the therapeutic applications of modRNA for cardiac diseases in animal models. Among them, the most notable is a publication from 2022, which employs the technique of CD5-conjugated LNP encapsulating modRNA to directly generate FAP CAR T cells in vivo, thereby reducing cardiac fibrosis and restoring cardiac function. In addition, Table 3 and Fig. 2 have organized some genes related to cardiac cell regeneration. These studies primarily validate the characteristics of cardiomyocyte proliferation through loss-of-function, gain-of-function, and recombinant protein administration. Although these genes have not yet been applied in cardiac diseases using modRNA technology, this table can provide helpful information for scholars interested in utilizing the modRNA platform for promoting research on cardiac cell regeneration. modRNA delivery leads to transient expression of functional proteins and is, thus, an attractive tool for cellular processes that do not need long-term protein expression, such as cardiomyocyte proliferation and cardiac reprogramming. The research on modRNA has improved safety and scalability, resulting in the development of cost-effective clinical-grade materials, robust delivery methods, and lower treatment costs. modRNA technology will be an excellent therapeutic agent via which to induce cardiac regeneration and promote cardiac function in ischemic heart disease for experimental and clinical needs.
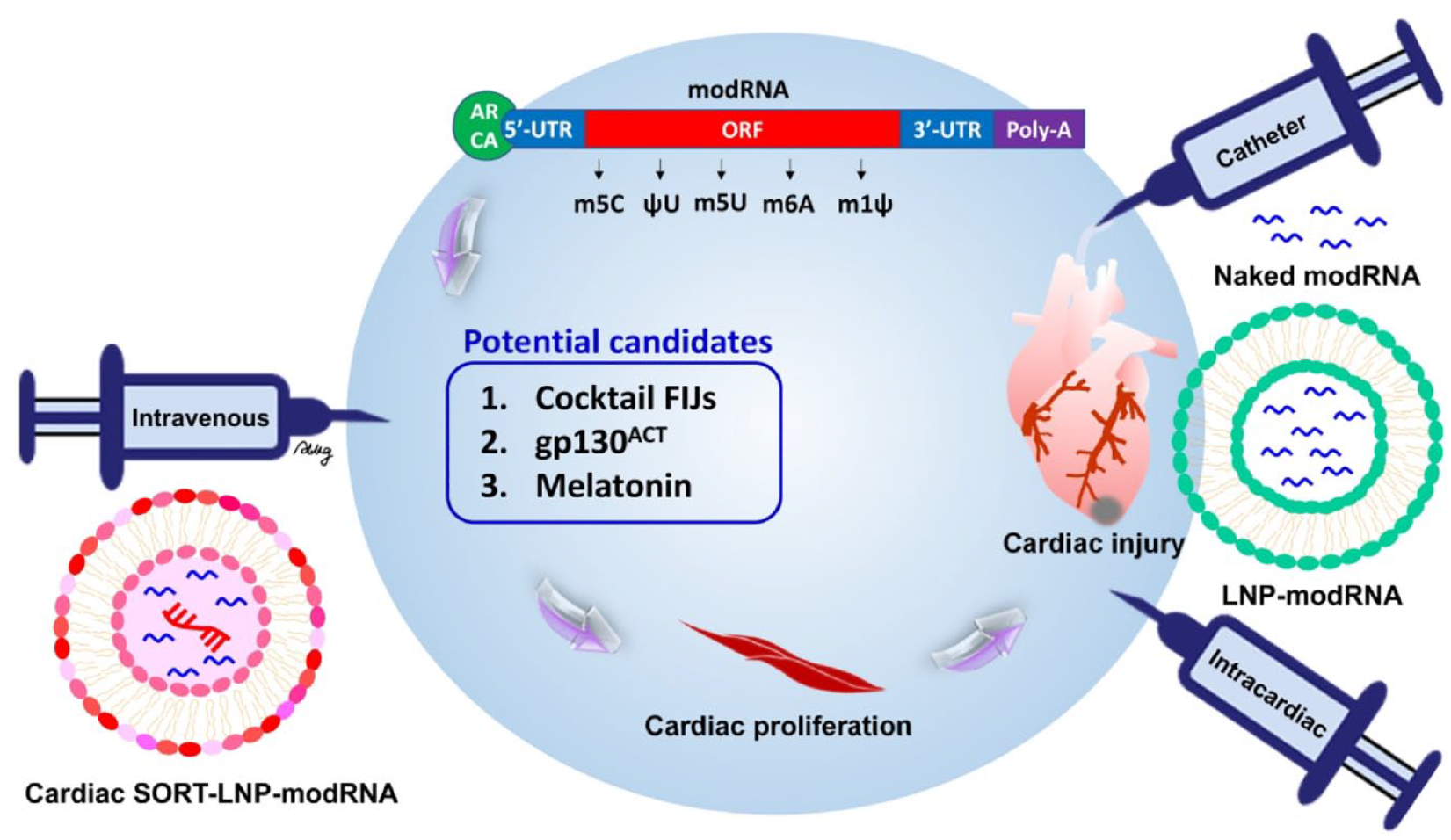
The prevention of heart disease involves utilizing the modRNA platform to target potential genes associated with cardiac proliferation, as outlined in Table 3.
Footnotes
Author Contributions
A.Y.L.W. conceptualized, designed, and coordinated the writing of the whole manuscript. Y.C.C. K.H.C. and organized the three tables. C.Y.Y.L. revised the manuscript. All authors contributed to critically revising and approving the final version of this manuscript.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by grants from the Ministry of Science and Technology, Taiwan (grant nos. MOST 109-2314-B-182A-056-MY3 and MOST 110-2314-B-182A-137-MY3), the National Science and Technology Council, Taiwan (grant no. NSTC 112-2314-B-182A-045-MY3), and the Chang Gung Medical Foundation, Chang Gung Memorial Hospital, Taiwan (grant nos. CMRPG3M0281, CMRPG3M0282, and CMRPG3M0283).
