Abstract
Recently, we and others generated induced tissue-specific stem/progenitor (iTS/iTP) cells. The advantages of iTS/iTP cells compared with induced pluripotent stem (iPS) cells are (1) easier generation, (2) efficient differentiation, and (3) no teratomas formation. In this study, we generated mouse induced pancreatic stem cells (iTS-P cells) by the plasmid vector expressing Yes-associated protein 1 (YAP). The iTS-P YAP9 cells expressed Foxa2 (endoderm marker) and Pdx1 (pancreatic marker) while the expressions of Oct3/4 and Nanog (marker of embryonic stem [ES] cells) in iTS-P YAP9 cells was significantly lower compared with those in ES cells. The iTS-P YAP9 cells efficiently differentiated into insulin-expressing cells compared with ES cells. The ability to generate autologous iTS cells may be applied to diverse applications of regenerative medicine.
Keywords
Embryonic stem (ES) cells are derived from the inner cell mass of a blastocyst and generate all cell types derived from the three germ layers. Induced pluripotent stem (iPS) cells, which are generated by reprogramming somatic cells with diverse phenotypes, closely resemble ES cells in terms of their morphology, growth characteristics, gene expression patterns, epigenetics, and differentiation potential 1 . The reprogramming is typically achieved by introducing a combination of specific transcription factors such as Octamer-binding transcription factor (Oct)3/4, Sex-determining region Y-box (Sox)2, Kruppel-like factor (Klf)4 and c-Myc (OSKM) into the somatic cells. The ES/iPS cells are capable of differentiating into various cell types in the body. Therefore, ES/iPS cells have been expected for their potential applications in regenerative medicine. However, ES/iPS cells have low efficacy to generate differentiated cells in vitro and the potential to form tumors, particularly when used in transplantation therapies. Tissue-specific stem/progenitor cells, also known as adult or somatic stem/progenitor cells, may represent an alternative source for the treatment of disease2–4. Tissue-specific stem cells, such as hematopoietic stem cells, neural stem cells, hepatic stem cells, and pancreatic stem cells, are undifferentiated cells found in specific tissues of the body and are multipotent, not pluripotent. Tissue-specific stem/progenitor cells can only differentiate into a limited number of cell types within the tissue in which they reside. However, to isolate and/or expand tissue-specific stem/progenitor cells from native tissues is not easy, because the population of tissue-specific stem/progenitor cells is very small 4 .
Recently, we have developed a method for generating induced tissue-specific/progenitor stem (iTS/iTP) cells by the repeated transfection of polycistronic plasmids expressing OSKM 5 , and OCT3/4, SOX2, KLF4, GLIS family zinc finger 1 (GLIS1) 6 , and OCT3/4, SOX2, KLF4, L-MYC, LIN28, and p53 shRNA 7 , followed by selection using tissue-specific markers. The iTS cells derived from pancreatic tissue (iTS-P cells), which express several endoderm/pancreatic markers, differentiate into insulin-expressing cells more efficiently than ES/iPS cells. More important, iTS cells do not form teratomas when subcutaneously transplanted into immunodeficient mice5–8. Therefore, iTS cells provide important advantages over ES/iPS cells. Recently, other groups reported induction of tissue-specific stem cells by the transient expression of Yes-associated protein 1 (YAP)/tafazzin (TAZ) (YAP induced stem cells: ySCs) 9 as well as induced endodermal stem/progenitor cells by small molecules 10 . These reports provide compelling evidence that the protocol for generating iTS/iTP cells will advance the field of regenerative medicine.
We transfected the YAP plasmid into pancreatic tissue obtained from 24-week-old mouse (C57BL/6) on days 1, 3, 5, and 7 (Fig. 1A). We generated 11 colonies of iTS-P cells, but no colony of iPS cells, using the YAP plasmid (Fig. 1B, C). The morphology of iTS-P cells by YAP plasmid was similar to that of iTS-P cells which we previously reported5–7 and mouse pancreatic stem cells without genetic manipulation 3 . We evaluated the self-renew of the iTS-P cells by YAP plasmid. The iTS-P YAP9 cells, which are one of 11 colonies, divided beyond passage 50 without detectable changes in growth rate (Fig. 1D).
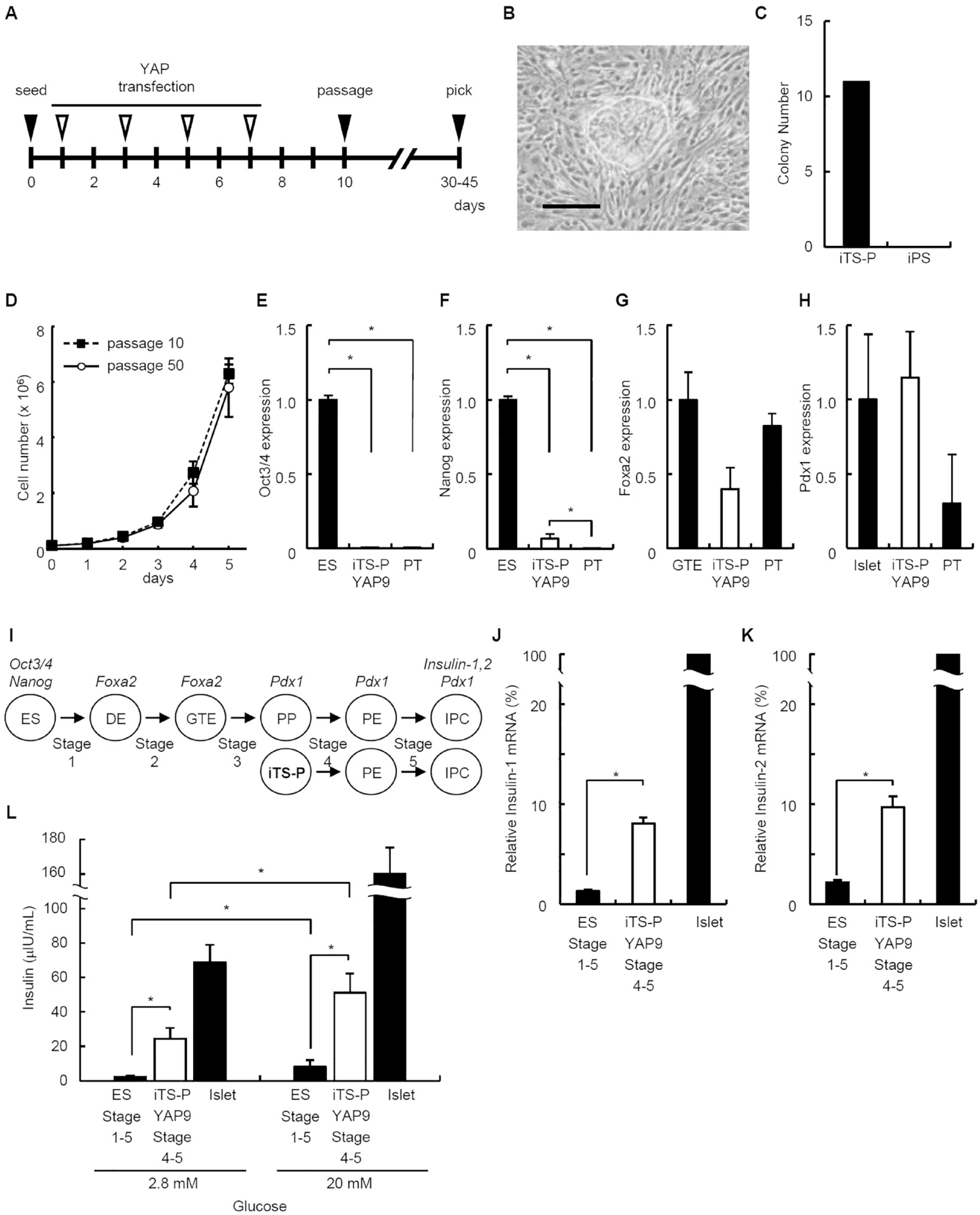
Generation of iTS-P cells using YAP plasmid. (A) Time schedules for the induction of tissue-specific stem cells transfected with the plasmids. Solid arrowheads indicate the times of cell seeding, passaging, and colony selection. Open arrowheads indicate the times of transfection. (B) Morphologies of iTS-P YAP9 cells. Scale bars = 100 µm. (C) Numbers of colonies formed by iTS-P and iPS cells. The YAP plasmid was used to transfect pancreatic tissue from 24-week old C57/BL6 mouse, and the number of colonies was counted after 30–45 days. (D) Growth rate of iTS-P YAP9 cells (passage 10 and 50). (E-H) Quantitative RT-PCR analysis of Oct3/4 (pluripotent marker) (E), Nanog (pluripotent marker) (F), Foxa2 (endodermal marker) (G), and Pdx1 (pancreatic marker) (H) in iTS-P YAP9 cells. ES cells (E, F), Gut tube endoderm (GTE) cells differentiated from ES cells (stage 1-2) (G), pancreatic islets (H), and pancreatic tissue (PT; including islet/acinar/duct cells) served as controls. The data are expressed as the gene-to-Gapdh ratio, with the ratio of the control cells arbitrarily defined as 1 (n = 5). (I) Schematic of the stepwise differentiation of ES and iTS-P cells to insulin-expressing cells. The cells of the definitive endoderm (DE) and the GTE express Foxa2; pancreatic progenitors (PP), pancreatic endocrine precursors (PE), and insulin-producing cells (IPC) express Pdx1; and IPC also express Insulin-1 and 2. (J, K) Quantitative RT-PCR analysis of Insulin-1 and 2 genes in differentiated iTS-P YAP9 cells. Pancreatic islets served as a positive control. The data are expressed as the Insulin-1 (J) or 2 (K): Gapdh ratio compared with that of islets, arbitrarily defined as 100 (n = 5). (L) Insulin release assay. Differentiated iTS-P YAP9 cells by protocol stages 4–5 protocol, derived from ES cells by protocol stages 1–5, and pancreatic islets were treated with 2.8 mM and 20 mM
We next investigated the expression of pluripotency marker genes 1 by iTS-P YAP9 cells. The iTS-P YAP9 cells expressed Oct3/4 and Nanog significantly lower than those in ES cells (from C57BL/6, ATCC, Manassas, VA, USA) (Fig. 1E, F). There are no significant differences between the expression of Oct 3/4 in iTS-P YAP9 cells and those in pancreatic tissue (Fig. 1E), while pancreatic tissue expressed Nanog significantly lower than those in iTS-P YAP9 cells (Fig. 1F). We also investigated the expression of endodermal/pancreatic markers 11 . The iTS-P YAP9 cells expressed Forkhead box protein a2 (Foxa2) (endodermal marker gene) (Fig. 1G) and the Pancreatic and duodenal homeobox factor-1 (Pdx1) (Fig. 1H), similar to the results of previous analyses of iTS-P cells and mouse pancreatic stem cells without genetic manipulation3,5.
To investigate differentiational potency of iTS-P YAP9 cells into insulin-expressing cells, we applied the stepwise differentiation protocol (Fig. 1I) 11 . We applied stages 4 and 5 of the protocol in iTS-P YAP9 cells because the characteristics of iTS-P cells which we reported previously5–7 was similar to Pdx1-expressing pancreatic progenitors (PP) (Fig. 1I). Pancreatic islets and differentiated ES cells (generated by the stepwise differentiation protocol, stages 1–5) served as controls. The levels of mRNAs encoding Insulin-1 and Insulin-2 in differentiated iTS-P YAP9 cells were significantly higher than those in differentiated ES cells (Fig. 1J, K). To determine whether the cells differentiated from iTS-P YAP9 cells were sensitive to glucose, they were treated with 2.8 mM or 20 mM glucose. The differentiated cells treated with 20mM glucose released significantly higher amounts of mouse insulin than the cells treated with 2.8 mM glucose (Fig. 1L). The cells differentiated from iTS-P YAP9 cells released approximately 6-fold higher amounts of mouse insulin than an ES-derived population at both glucose concentrations (Fig. 1L).
We previously reported that the iTS-P cells transfected with the OSKM plasmid inherit numerous components of epigenetic memory from pancreatic cells and acquire self-renewal potential. In contrast, other evidence indicates that transient ectopic expression of YAP in differentiated primary mouse cells induces conversion to a tissue-specific stem or progenitor state 9 . YAP is a key transcriptional co-activator that plays a significant role in the Hippo signaling pathway, a highly conserved signaling pathway involved in regulating cell proliferation, organ size, and tissue homeostasis. YAP functions as a transcriptional effector which reprograms nonstem cancer cells into cancer stem cells 12 . YAP translocates from the cytoplasm to the nucleus in cells located in tissues enriched in tissue stem cells, suggesting its regulation by structural and chemical signals associated with the stem cell niche 13 . Although genetic ablation of Yap and Taz from certain organs of adult mice shows that they are dispensable for normal tissue homeostasis, YAP/TAZ are essential for organ regrowth after tissue damage14,15. These reports suggest that YAP and TAZ are inactive in normal adult tissues but are called into action to generate new stem cells.
When differentiated iTS-P YAP9 cells were transplanted into syngeneic mice with streptozotocin-induced diabetes, the mice did not become normoglycemic. The amounts of insulin secreted by differentiated iTS-P YAP9 cells was quite low compared with that of pancreatic islets (Fig. 1L), suggesting that differentiated iTS-P YAP9 cells were not fully matured, although the amount of insulin secreted by iTS-P YAP9 cells was higher than that of differentiated ES cells. Establishing an efficient, reproducible protocol for generating insulin-expressing cells is critically important for clinical applications.
In summary, we generated iTS-P cells through expression of YAP. Although YAP plasmid does not induce iPS cells, we cannot conclude that generation of iTS-P cells using the YAP plasmid is safer than using OSKM plasmid, because activation of YAP can reprogram nonstem cancer cells into cancer stem cells 12 . The mechanisms of reprogramming by OSKM and YAP may differ. We will compare iTS-P cells by OSKM or YAP plasmid in future study.
Footnotes
Ethical Approval
The review committee of University of the Ryukyus approved the studies using mice (A2017063, A2017145, A2019040).
Statement of Human and Animal Rights
This study does not contain human subjects.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported, in part, by JSPS KAKENHI Grant Numbers 23H029733 and 22K08759, and the Okinawa Science and Technology Innovation System Construction Project.
