Abstract
Spinal cord injury (SCI) is associated with currently irreversible consequences in several functional components of the central nervous system. Despite the severity of injury, there remains no approved treatment to restore function. However, with a growing number of preclinical studies and clinical trials, cell transplantation has gained significant potential as a treatment for SCI. Researchers have identified several cell types as potential candidates for transplantation. To optimize successful functional outcomes after transplantation, one key factor concerns generating neuronal cells with regional and subtype specificity, thus calling on the developmental transcriptome patterning of spinal cord cells. A potential source of spinal cord cells for transplantation is the generation of exogenic neuronal progenitor cells via the emerging technologies of gene editing and blastocyst complementation. This review highlights the use of cell transplantation to treat SCI in the context of relevant developmental gene expression patterns useful for producing regionally specific exogenic spinal cells via in vitro differentiation and blastocyst complementation.
Keywords
Introduction
The annual incidence of spinal cord injury (SCI) worldwide varies from 40 to 80 cases per million, with up to 90% of these cases the result of a traumatic cause 1 . In the United States alone, there are roughly 54 cases per 1 million, or about 18,000 new cases each year, with a higher prevalence in men 2 . In addition to the physical limitations that come with SCI, the associated socioeconomic burden can be crippling. Patients with SCI have estimated lifetime direct costs ranging from US$1.2 million to US$5 million, which does not include indirect costs, such as lost wages 2 . However, this cost does vary tremendously depending on life expectancy and mortality rate. Persons with SCI have an estimated two to five times greater risk of mortality than the general population, with the first year after injury holding the greatest risk 3 . There are no existing cures for individuals with SCI, and while patients with incomplete injuries may experience limited improvement, full recovery after SCI is rare4,5. The lack of available therapies along with the tremendous burden SCI has physically, economically, and emotionally highlights the need for efficacious and safe treatments for management of traumatic injury. Current experimental approaches for treatment have made use of neuron transplantation with the goal of restoring nervous system function after SCI. This review will summarize recent advances in the field of regenerative medicine on the generation and the potential use of exogenic neuronal cells to treat SCI. In addition, we will highlight the gene networks and expression patterns relevant to exogenic cell development for optimization of successful functional outcomes as a promising direction for future transplantation studies.
SCI Pathophysiology
SCI can present with a myriad of symptoms depending on the severity of the injury and its location along the spinal cord. Symptoms include partial or complete loss of motor and sensory function below the injury site, and can also affect regulatory systems that control bladder and bowel function, breathing, heart rate, and blood pressure 1 . It is important to note that no two human cases are identical as each affected person presents with varied symptoms and potential for recovery. The initial traumatic event, or primary injury, is followed by several biological events termed the secondary injury. There are, however, three pathological stages that define the secondary injury in SCI: the acute phase (48 h), subacute phase (2–14 days), and chronic phase (more than 6 months)4–7.
During the acute phase, disruption and swelling of the spinal cord occurs at the level of injury due to edema and intraspinal hemorrhage 6 . This disruption exposes the spinal cord to an influx of inflammatory cells and cytokines that cause necrotic cell death7,8. Proinflammatory cytokines and peripheral inflammatory cells are recruited to the site of injury, and phagocytes rapidly remove debris. This process generates cytotoxic byproducts, which add to cell injury9,10. The subacute stage of injury is primarily characterized by traumatic demyelination and further vascular compromise of the spinal cord 6 . Additional biochemical events, such as an accumulation of glutamate in extracellular compartments, contribute to excitotoxicity of surrounding neurons11,12. At this point, many axons affected by the injury will begin to degenerate. Macrophages can also be seen actively ingesting fragments of myelin, and myelomalacia, or softening of the spinal cord, is typical4,6. During the chronic phase, most of the necrotic tissue is eliminated and ingested by macrophages. The resulting cavities are filled with extracellular fluid, residual macrophages, and connective tissue, hindering cell regeneration, migration, and axon regrowth13,14. SCI is also characterized by Wallerian degeneration, a process that involves the anterograde degeneration and pruning of axons and their myelin sheaths, beginning in the subacute phase and persistent throughout the chronic phase. In addition, an astroglial scar at the site of injury develops from proliferative astrocytes that form tightly interwoven glial fibers around the injury site6,15.
The role of the glial scar is controversial, as initially it helps to reestablish the blood brain barrier and limit lesion size; however, later it appears to be detrimental to functional recovery by creating a barrier that prevents extensive axonal regrowth. It is also unclear the extent to which these degenerative processes have an endpoint or if they continue indefinitely, possibly due to chronic inflammation or autoimmunity. An understanding of the mechanisms of these pathophysiological events is essential, as potential therapies for SCI may target one or more of these processes. Furthermore, with the distinct progression of injury over time, it is crucial to optimize proposed therapies to the stage of SCI they will be administered.
Currently, there is no recognized treatment for the recovery of functional deficits after SCI. The current standard of care following SCI consists of a variety of management strategies, including intensive care unit admission for careful blood pressure control, optional use of corticosteroids, and early surgical intervention for decompression. Ultimately, many physicians emphasize the importance of early intervention. It has been suggested that if proper treatment is applied during the window immediately following the primary injury, the damage caused by secondary injury can be limited, thus improving functional outcomes4,9,15. Although current clinical treatments are limited to those listed above, researchers have worked for decades to develop new and promising therapies. Most of these are considered “neuroprotective” and target acute and subacute injuries; very few experimental approaches have targeted chronic SCI. However, considering that the pathophysiology of chronic SCI is relatively static, re-establishing lost connections in the chronically injured spinal cord is a mechanistically plausible target16,17. For both acute and chronic SCIs, a common strategy involves targeting regeneration of cells to restore lost connectivity, such as with cell transplantation, which is the focus of the remainder of this review. Cell replacement therapy, also referred to as cell transplantation, involves the transplantation of neural progenitor cells (NPCs), among other cell types, into the site of injury to restore function and connectivity. Numerous studies have demonstrated the safety and efficacy of this approach in animal models as well as its ability to support regeneration and functional recovery in early stage clinical trials. For the purposes of this review, we will be focusing on transplantation of NPCs, which are necessary for true neural cell replacement in the central nervous system (CNS). While there are reports of transdifferentiation from non-neural cell types to NPCs, this remains controversial, and thus will not be discussed further here.
Sources of Cells for Transplantation
Stem cells provide the ideal building blocks for cell replacement therapies because of their ability to self-renew and generate numerous cell types. Spinal neurons may be obtained from numerous stem cell sources, including fetal stem cells (FSCs), embryonic stem cells (ESCs), induced pluripotent stem cells (iPSCs), and mesenchymal stem cells (MSCs). Several methods have been developed to generate spinal neurons in vitro from their corresponding stem cell progenitors. With advances in technology and methodologies allowing for a wide breadth of cell types to be produced, transplanting spinal progenitor cells as a therapeutic approach has risen in prominence, especially with respect to treating SCI.
Preclinical and clinical studies have utilized stem cells from multiple sources for SCI, all demonstrating varying degrees of success, with primary differences being in degree of pluripotency and methods of acquisition. ESCs are pluripotent stem cells originating from the inner cell mass of the mammalian blastocyst that can differentiate into cells from each of the three germ layers 18 . FSCs are multipotent stem cells originating from the fetus that develop into progenitor cells restricted to a single germ layer. Human iPSCs (hiPSCs) are generated by cellular reprogramming of adult somatic cells via transcription factors to reset cells to a pluripotent state 18 . While first accomplished through the use of viral transduction, which carries the risk of oncogene integration and tumor formation, several non-genome incorporating or nonviral delivery methods have since been developed that may be ideal for clinical application19,20. Because hiPSCs are derived from the adult system and can be used autologously, this minimizes the need for immune suppression and greatly expands the pool of eligible recipients. MSCs are multipotent non-neural stem cells that may be obtained from adipose, bone marrow, and umbilical cord tissue 21 . Currently, MSCs are the most commonly studied for cell transplantation and have many ongoing clinical trials, likely because of their easy accessibility and capacity to be transplanted autologously. While predominantly used to produce mesoderm-derived tissues, such as bone, cartilage, and fat cells, there are also controversial reports of neural differentiation that will be highlighted below. Each of these sources has been used to generate neural or neural-like cells, making them prime candidates for use in spinal progenitor cell transplantation studies.
Advantages and limitations associated with each stem cell source must be considered when developing systems for their practical application. While ethical and immunological issues hamper the use of ESCs and FSCs in clinical settings, their application for SCI has been extensively studied in animal models 22 . To date, the most well-established protocols describing the generation of spinal neurons utilize cells derived from ESCs, providing a strong foundation for continued use 23 . While region-specific FSC-derived and fetal tissue-derived NPCs have been demonstrated to integrate well with host tissue, limitations include issues of cellular heterogeneity with non-neural cells within the grafts, limited proliferation capability, immune rejection, and the need for fetal tissue 24 . A major draw of the use of hiPSCs is their capacity to be transplanted autologously, although the current time requirements necessary to obtain donor samples, reprogram, differentiate, and perform quality control are prohibitive for acute and most subacute cases. Improved protocols for rapid induction and differentiation will be key for advancing application of autologous techniques 25 . Matched samples from hiPSCs banks may provide a middle ground, providing a cheaper, off-the-shelf product, which would still require immune suppression, but with less likelihood of rejection. Finally, MSCs, though widely used, have shown conflicting evidence regarding their neural differentiation capacity in vivo, which would significantly affect their translational capability, especially when utilized in a chronic setting. Many researchers argue their effect is limited to neurotrophic and immunomodulatory support26,27. An in-depth discussion of the history of MSC transplantation for SCI is beyond the scope of this review, therefore, we direct readers to Liau et al 21 , Rafiei et al 28 , and Ruzicka et al 29 for further study of their preclinical and clinical applications. The next section will explore the current state of preclinical and clinical applications of stem cell-derived NPCs for cell transplantation in SCI.
Cell Replacement Therapy for SCI
Significant advances in the field of cell replacement therapy in the past 10 years have highlighted both the promise along with the future directions of this approach that may promote further functional benefit as a treatment after injury. The most commonly studied cell types for transplantation include NPCs, MSCs, oligodendrocyte progenitor cells (OPCs), and Schwann cells (SCs)30–32. In general, these candidate cells can be subdivided into neuronal and non-neuronal types. While non-neuronal cells may provide some functional benefit, neuronal cells, such as NPCs, are advantageous by their additional ability to generate new neurons, astrocytes, and oligodendrocytes capable of replacing damaged or lost cells and may create a bridge across the injury site 33 . The creation of neural relays between grafted cells and host neurons could be a vital component of restoration after injury. While current cell transplantation research provides promising results for injury improvement, a better understanding of the underlying mechanisms is needed. In this review, we focus on the transplantation of NPCs because of their potential for cell replacement, which is likely more applicable than other cell therapies to chronic SCI, an unmet public health need.
Numerous preclinical experiments have transplanted cells following SCI in a range of animal models, including mice, rats, minipigs, and nonhuman primates. These preclinical studies provide invaluable insight toward understanding the mechanism and therapeutic potential of cell replacement therapy, contributing to advancements in previous and ongoing clinical trials 34 . Here, we briefly discuss functional outcomes and status of both preclinical and clinical studies using NPCs derived from fetal tissue, FSCs, ESCs, and iPSCs.
Preclinical NPC Transplantation Studies in Animal Models of SCI
Cell transplantation approaches in animal models of SCI have focused on the safety, efficacy, and extent of functional recovery in response to ESC-derived, FSC-derived, iPSC-derived, and fetal tissue-derived NPCs. These studies have demonstrated several mechanisms that promote nervous system recovery, including remyelination35–44, axonal regeneration36–38,44–52, tissue and nerve sparing35–37,45, angiogenesis36,37, and the release of neurotrophic factors38,39 (Table 1). Significantly, many of these studies highlight the successful integration of transplanted exogenic neuronal cells as observed by cell survival, migration within host tissue, and synapse formation with host cells40–43,48–52,56,59–62. These mechanisms may prove significant to restoring the cellular microenvironment at the site of injury by creating a growth permissive environment for axonal restoration and neural relay formation, ultimately promoting functional recovery. In fact, several of these studies highlight the recovery of motor function35,37–41,43–45,47,50–52,61 and reducing chronic neuropathic pain 63 in animal models of SCI. While motor and sensory recovery in animals with SCI was not a ubiquitously reported outcome, the studies that did report treatment-based functional recovery have emphasized the therapeutic benefits of cell replacement.
Common Mechanisms of Recovery After Cell Transplantation for SCI and the Associated Graft Cell Type.

For neuroprotection, references were added based on evidence of tissue sparing, improved trophic support, decreased glial scarring, or less demyelination. It is important to note that neuroprotection can also contribute to the other mechanisms of recovery as well. For axon regeneration, references were added based on evidence of regrowth of severed or damaged host axons. For relay formation, references were added based on evidence of host to graft or graft to host synapse formation. For remyelination, references were added based on evidence that transplanted cells differentiated into oligodendrocytes and myelinated host axons. For angiogenesis, references were added based on evidence of new blood vessel formation. For immunomodulation, references were included based on evidence of altered immunological response, such as decreased microglial/macrophagic activity.
In addition to the successes of NPC transplantation in animal models of SCI, other factors also assist in enhancing functional outcomes. Co-transplantation of NPCs with other cells, such as macrophages 64 , SCs 65 , OPCs 66 , or factors, such as Chondroitinase ABC 41 , has been shown to enhance cell integration and functional recovery. Designing regionally relevant neural cells for transplantation may promote further recovery, as regional specificity of ESC-derived neural cells has been shown to be a significant factor in creating a neural relay circuit for functional recovery 54 . This was similarly observed with the transplantation of regionally specific iPSC-derived NPCs into the spinal cord 57 . As such, highlighting the role of the regionally relevant microenvironment and cell populations should be a significant point of interest for transplantation-mediated SCI treatment approaches. The time, location, and manner in which cells are transplanted can also play a major role in their survival, integration, and ultimate efficacy. While the direct injection of cells at the site of injury has historically been the most common route of administration, this results in relatively poor survival and graft integration 67 . The hostile microenvironment of the acute injury site may contribute to this, with subacute transplantation and the addition of grafts rostral and caudal to the injury improving these outcomes68,69. The use of engineered biomaterials, including scaffolds, hydrogels, and nanoparticles also provides structural and nutrient support that enhances functional recovery after transplantation70–72. Though these considerations are widespread within preclinical research, their adoption in clinical studies may be pivotal for translation.
Clinical Cell Transplantation Studies in Humans for SCI
Globally, a large number of clinical trials involving cell transplantation have been conducted at all stages of injury in humans 33 . Several clinical trials have studied FSC-derived NPCs for transplantation after SCI. A phase I/II clinical trial conducted by Yonsei University Health System (KCT0000879) to evaluate the safety and efficacy of fetal telencephalon-derived NPCs in patients with cervical, motor complete SCI determined the treatment well-tolerated, safe, and neurologically beneficial, with about 25% of patients demonstrating improvement in the Abbreviated Injury Scale 73 . StemCell Inc. also tested human FSCs for feasibility and safety after transplantation in a dose-escalation study, but the study was terminated early due to inability to reach sponsor-established benchmarks (NCT02163876, NCT01725880)74–77. Finally, Neuralstem Inc. sponsored a phase I clinical trial (NCT01772810) to evaluate the safety and efficacy of fetal spinal cord-derived NPCs in 4 chronic SCI patients 78 . Notably, this is the only spinal cord-derived human cell line to have received Food and Drug Administration (FDA) approval for transplantation in humans with SCI. Neurological assessments and electromyography revealed improvement in sensory and motor scores and increased muscle activity, suggesting renewed connectivity. However, this phase I study lacked a control group, and thus lacks the statistics required to determine the extent of treatment-based functional recovery 79 . Phase I/II trials transplanting this cell line in amyotrophic lateral sclerosis (ALS) patients have also been completed, demonstrating safety but unclear functional effects (NCT01348451, NCT01730716) 80 .
The only clinical trial involving transplantation of ESC-derived NPCs in humans is currently recruiting for a phase I/II trial for subacute SCI. S. Biomedics Co. intends to recruit five patients with C4–C7 complete SCI diagnosed as a grade A injury on the American Spinal Injury Association Impairment Scale. This trial (NCT04812431) will assess the efficacy and safety of polysialic acid neural cell adhesion molecule positive (PSA-NCAM+) NPCs derived from a human ESC line 81 . While this is the first study to transplant ESC-derived neuronal cells, the SCiSTAR trial, a clinical trial started by Geron Corporation and continued by Lineage Therapeutics, involved the transplantation of ESC-derived OPCs82,83. Nearly, all patients (21 of 22) displayed at least one level of motor improvement, and seven patients showed two levels of motor improvement, which was sustained for at least 1 year post-transplantation 84 . A follow-up of five patients at 10 years post-transplantation suggests that the treatment is well-tolerated, with no evidence of neurological deterioration or tumor formation at this time point 85 . Lineage Therapeutics most recently announced its intent to initiate a new clinical trial with this cell line in both subacute and chronic SCI patients pending FDA clearance 86 .
Despite the recent developments in generating and understanding iPSC-derived NPCs in vitro and their mechanistic effects in vivo over the past 20 years, its clinical translation to human SCI has yet to be fully explored. While hiPSC-based treatments for macular degeneration and Parkinson’s disease have been documented, no clinical trial using hiPSCs for SCI has been conducted87,88. In fact, Japan is the first country to announce approval for a clinical trial using hiPSC-derived NPCs to treat subacute SCI 89 ; they intend to enroll a total of four participants and the first successful surgery was performed in December 2021 90 . The Okano group proposes to assess the safety and efficacy of hiPSC transplantation in patients with complete cervical or thoracic injury. This trial marks an incredible advancement in the field and will likely shape future clinical trials in the rest of the world.
These clinical trials demonstrate the potential of using NPCs to treat SCI in humans. However, while improvement in motor function was reported by some trials, others have not been as successful or require further study to determine a conclusive outcome. Ultimately, no clinical trial has reached FDA approval for SCI treatment. As already mentioned, improvements in graft integration and functional recovery when cells are tailored to regionally relevant identities have been observed in multiple preclinical studies54,57, and this may be key to drive translation within the field of cell replacement therapies.
Differentiation and Regional Specification of Spinal Neurons
Pluripotent stem cells require ex vivo differentiation to attain a neural lineage. Advancements in cell culture methodology, specifically in the field of cell transplantation over the last decade, have improved long-term survival, consistency, and replicability of differentiation protocols. This has included the implementation of defined reagents, feeder-free culture systems, and Good Cell and Tissue Culture Practice (GCCP) to guide the field closer to translation91–93. While notable progress has been made, the inherent variability of cell culture and the vast assortment of utilized methodologies remain one of the biggest hurdles to achieving reproducibility.
Considerations for Growth Factor-Directed Differentiation
The culturing of stem cells in a dish itself possesses a stochasticity that contributes to inconsistent differentiation in even carefully controlled environments 94 . Variable differentiation potential may be attributed to the process of reprogramming as well as the introduction of both genetic and epigenetic changes during culture, which are currently poorly understood95–97. Additional sources of variation include the mode of culture, cell line differences, and a vast repository of matrices, small molecules, and techniques, many of which may be proprietary or susceptible to batch variability. Thus, a successful differentiation paradigm must both carefully select the conditions to produce the desired cell type and incorporate quality control checkpoints to make note of fluctuations in outcomes when all else is the same. Considerations for directed neuronal differentiation are discussed below, and a more detailed discussion of replicability and experimental design in cell culture experiments is provided by Chan and Teo 94 .
Numerous methods for both 2D and 3D culture have been established. Culture in 2D involves the plating of adherent monolayers and is the traditional means of cell culture and differentiation. Advantages of this strategy include providing homogenous nutrient access to plated cells and enabling uniform proliferation. While single monolayers have historically been most common, micropatterning and sandwich culture are increasingly used to introduce more complex interactions to the 2D environment 98 . However, cell fate following 2D differentiation often does not correspond to differentiation in vivo, with cell type composition varying significantly following transplantation 99 . Culture in 3D provides a more complex environment that allows for greater emergence of signaling cascades, migration, and motility patterns 100 . A growing number of groups have developed robust strategies for the generation of spheroids and organoids, which can be maintained in scaffolds, sheets, hydrogels, and within microfluidic devices 98 . While these approaches produce heterogeneous populations of cells that more accurately reflect the in vivo microenvironment, they also tend to be more costly and require advanced culturing techniques that can be difficult to establish. This intricacy also leads to increased variability in outcomes that has made large-scale reproducibility difficult. Differentiation in both 2D and 3D have yielded important insights on relevant developmental pathways and the injury process, and care should be taken to determine which system will be most suitable based on the needs of a given experiment.
The use of various coating materials to provide an adhesive matrix for stem cells to grow on has been shown to influence both the efficiency of differentiation and the behavior of the terminally differentiated cells. These are generally intended to mimic the extracellular matrix (ECM) of native cells, which is known to play an important role in both the injured and diseased CNS
101
. Collagens, laminins, fibronectins, and poly-amino acids are among the most commonly used coating matrices. Laminin, a major component of ECM, has the greatest effect on NPC differentiation, expansion, and neurite outgrowth, and is often considered an essential ingredient in the coating process for both 2D and 3D applications
102
. For this reason, Matrigel is widely used for neuronal differentiation, however, its clinical relevance is impacted by its xenogeneic nature. Poly-amino acids, such as poly-
In addition to variations in the mode of cell culture, the means of differentiation may also vary substantially. Differentiation protocols themselves range from production of early progenitors to more fully developed postmitotic neurons. For this purpose, a variety of growth factors and neurotrophins have been documented to improve differentiation and transplantation outcomes 104 . Differentiation of hiPSCs commonly begins with a neural induction stage, which has been reported both using small molecules for dual SMAD inhibition 105 and morphogen-free methods 92 . Following induction, cells may be maintained in an early neuroepithelial or neuromesodermal stage via application of fibroblast growth factors (FGFs) prior to subsequent differentiation into discrete lineages 106 . Most protocols also use retinoic acid (RA) to promote neuralization and caudalization to a spinal identity. Subsequent regional specification to mature spinal interneuron subtypes involves both rostrocaudal and dorsoventral patterning, and is discussed in more detail below.
Considerations for Regional Specification of Spinal Neural Cell Types
There is profound cellular diversity within the spinal cord, and a growing body of research has revealed the importance of specific regional patterning of exogenous cells to the level and functional domain of the injury site prior to transplantation 107 . To date, most applications of cell replacement therapy utilize cells that have been differentiated to a broad neural fate without consideration for the level of the neuraxis or the necessary sensorimotor components. This has contributed to low graft survival and failure of transplanted cells to integrate with host tissue, posing a major challenge in the application of cell replacement therapies. However, recent work has shown that NPC transplantation enables robust corticospinal regeneration and functional recovery only when the cells are patterned toward a caudalized fate 54 . Furthermore, transplanted NPCs preferentially form connections with similar phenotypic regions55,58. Thus, for the creation of clinically relevant models and treatments, it is of the utmost importance that scientists recapitulate appropriate characteristics at the area of injury. This degree of specialization will allow for targeted therapies that can focus on individual outcomes, including improvements in specific locomotor circuits, sensation, amelioration of pain, remyelination, and more.
Several protocols have been developed to generate regional subtypes of spinal neurons and interneurons in vitro, allowing for a wide range of positionally specified cells to be transplanted for SCI. While spinal caudalization of NPCs is increasingly common, tailoring of cells to a specific spinal level has been rare and requires precise coordination of FGFs and wingless-related integration (Wnt) agonists and antagonists 108 . Great attention has been given to the use of motor neurons and ventral spinal interneurons for repair after injury, though the production and transplantation of dorsally specified interneurons remains an unmet need for meaningful sensory recovery. Ventral populations are widely attained by application of RA and sonic hedgehog (Shh) agonists. Derivation of V2a 58 and V3 109 interneuron populations is most commonly reported. Transplantation of ventral and motor neurons is conducted with the intention of restoring locomotion, autonomic function, and related motor activities depending on the level of injury—and many studies report success 110 . Dorsal specification requires application of bone morphogenetic proteins (BMPs) and only three protocols to date report generating these populations111–113. Considering as many as 80% of SCI patients experience chronic pain and sensory impairment, the lack of research and attention on the development of these populations neglects the devastating impact of sensory dysfunction in traumatic SCI 114 . As a result, studies moving forward should consider rostrocaudal and dorsoventral neural identity as a significant factor with transplantation. These approaches rely on an understanding of the developmental neurobiology of the spinal cord and spinal neurons to identify relevant genes and signaling pathways for producing enriched and regionally specific cell populations.
Developmental Neurobiology of the Spinal Cord
The formation of the spinal cord during development is a process that initiates during gastrulation with the formation of the primitive streak and primitive node115–118. Cells migrating through and from the primitive node will form the trilaminar embryonic disk composed of three germinal layers: endoderm, mesoderm, and ectoderm118,119. The notochord, formed in the axial mesoderm, secretes growth factors to induce the creation of the neural plate through neuroectoderm, thus initiating primary neurulation117,119. The neural plate then differentiates to create a neural groove surrounded by neural folds. These neural folds will fuse to form the neural tube, the precursor to both components of the CNS—the brain and the spinal cord 116 (Fig. 1).
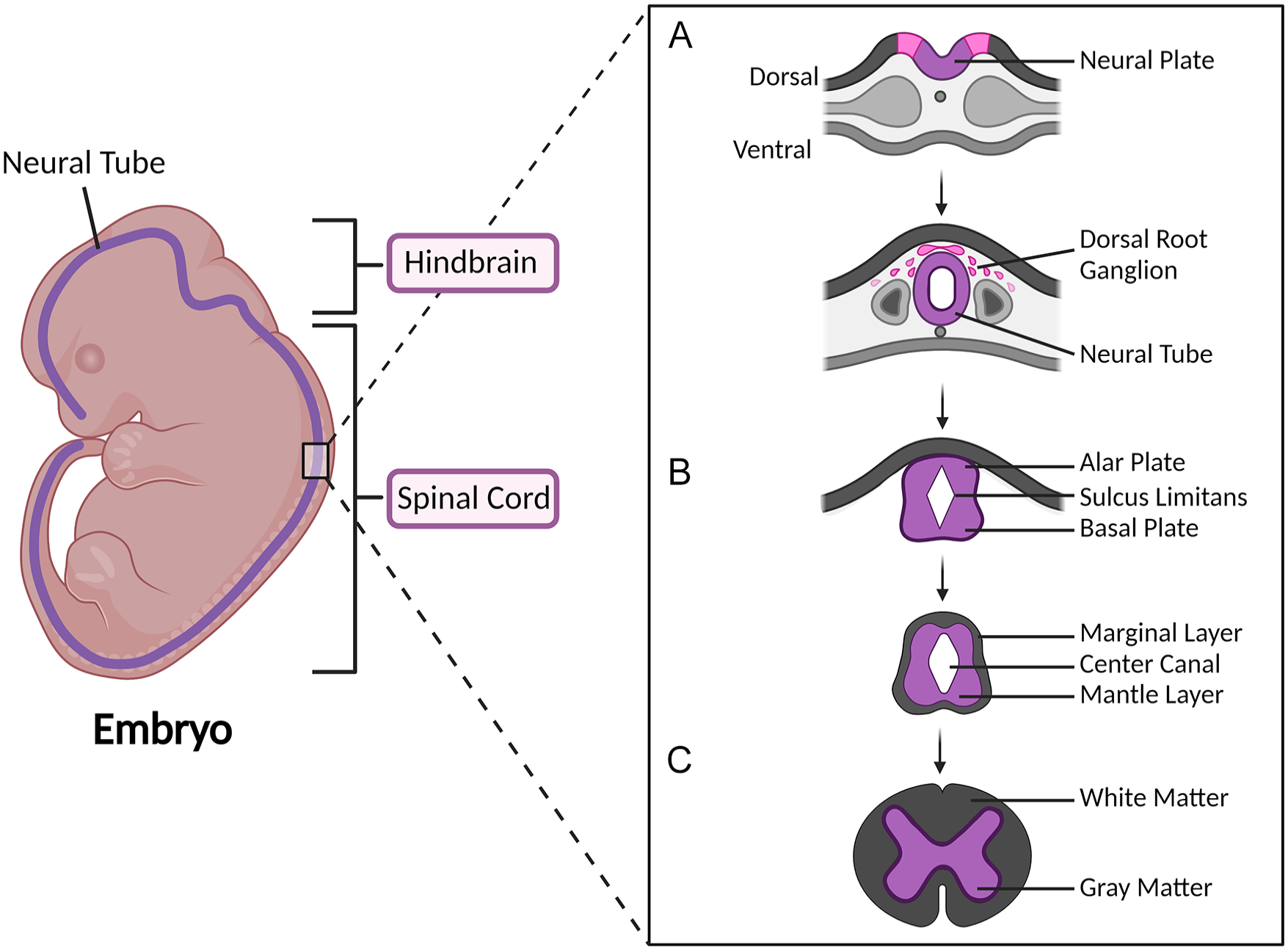
Neural tube development. Schematic illustrating the development and differentiation of the neural tube—the precursor of the hindbrain and the spinal cord. Neural tube development begins with (A) the folding of the neural plate lateral edges. The neural folds fuse to form the neural tube. Developmental progression is characterized by (B) differentiation of the neural tube, maintaining dorsoventral polarity, to form the alar and basal plates. Continued differentiation of the neuroepithelial cells results in the formation of the marginal and mantle layers, which are the precursors to (C) the white and gray matter of the spinal cord, respectively.
After neural tube closure, the neural tube maintains a dorsoventral organizational polarity, where the dorsal and ventral parts of the neural tube are characterized by the presence of the roof and floor plates, respectively120,121. Neuroepithelial cells near the neural canal differentiate and migrate laterally to form from the three central layers of the developing spinal cord: the ventricular layer, the mantle layer, and the marginal layer (Fig. 1B). Of these three layers, the marginal layer develops into the white matter of the spinal cord and the mantle layer develops into the gray matter. Further differentiation and dorsoventral polarization of the mantle layer leads to the formation of the alar and basal plates, the precursors to the dorsal and ventral horns of the spinal cord, respectively (Fig. 1C). Neuroepithelial cells of the alar plate will become sensory interneurons, whereas the cells of the basal plate will develop into the motor neurons of the developed spinal cord 122 .
In addition to dorsoventral polarity, rostrocaudal patterning is also a key factor in spinal cord development. Global rostrocaudal polarity is defined by five spinal segments: cervical, thoracic, lumbar, sacral, and coccygeal. Each spinal region is characterized by a number of extending nerve fibers from a specific dorsal root ganglion that innervate a single dermatome 123 . Local rostrocaudal patterning is specific to the organization of spinal motor neurons and interneurons. Spinal motor neuron subtype specification along the rostrocaudal axis is particularly relevant for limb-specific motor control. Spinal interneuron patterning has recently been linked to the distribution of interneuron subtypes clustered by gene expression and contributes to both sensory and motor spinal networks. The combination of both motor and interneuron regional subtypes is of particular interest considering their role in central pattern generators (CPGs) of the spinal cord124,125. Recapitulation of these developmental programs constitutes the first step in the generation of specific spinal populations.
Rostral–Caudal Patterning
During spinal cord development, the rostral and caudal identity of spinal cord cells is influenced by FGFs, RA, and growth differentiation factors (GDFs). Motor neuron identity along the rostral end of the neural tube is induced by a combination of high paraxial mesodermal RA expression and low caudal FGF8 and GDF11 expression126,127 (Fig. 2A). These signaling gradients regulate Hox gene expression throughout the developing spinal cord (Fig. 2B). Hox genes are a family of transcription factors linked to anterior-posterior cell subtype specification throughout the CNS. Hox gene expression in developing spinal motor neurons follows a rostrocaudal organization128,129. Specific Hox gene expression in turn aids in defining motor neuron columnar subtypes and regulating the motor neuron–muscle connection130,131.
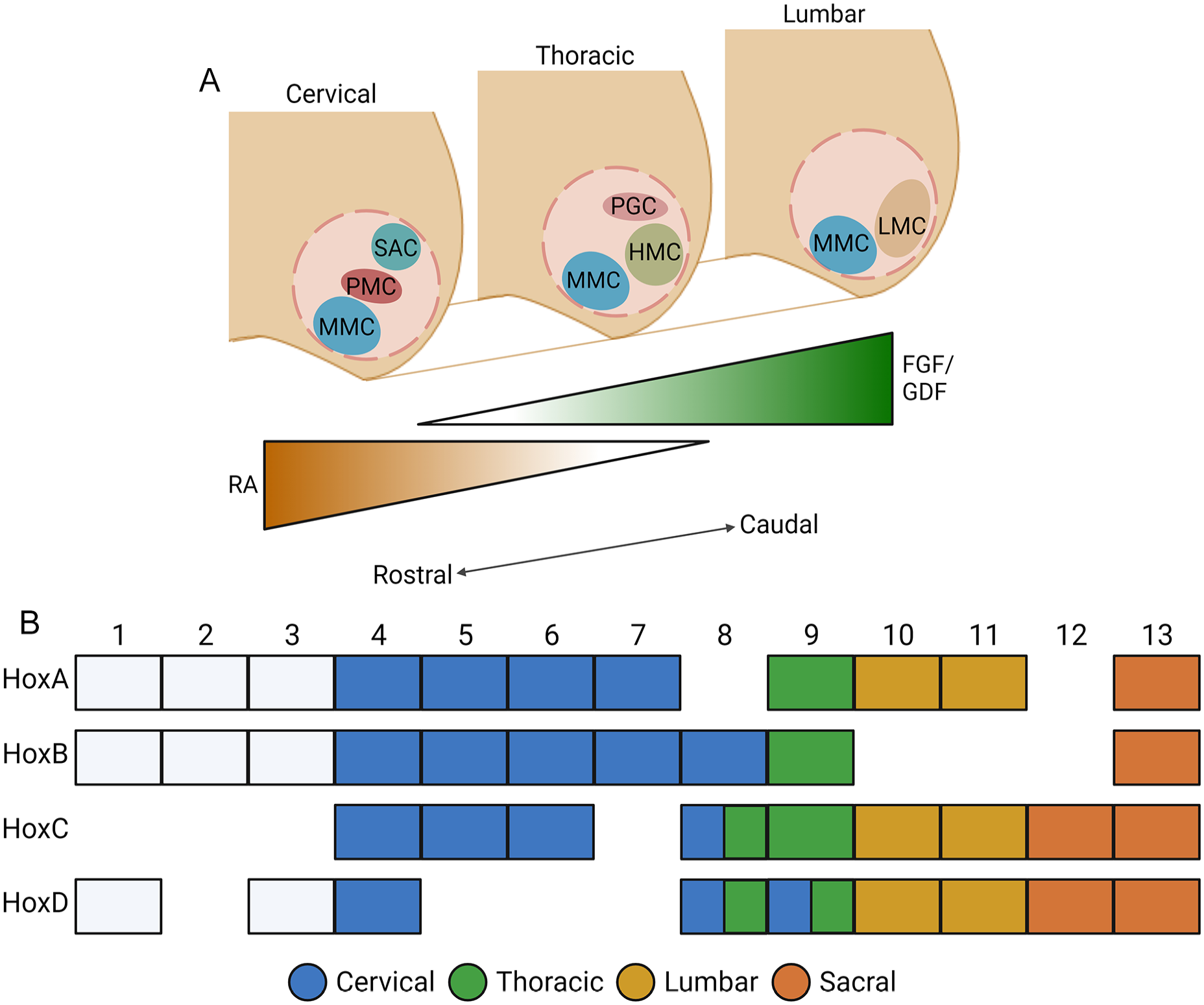
Developmental rostral–caudal patterning of the spinal cord. Schematic demonstrating the rostrocaudal patterning of transcription factors and Hox genes involved in neuronal subtype specialization. (A) Several neuronal populations are also specialized along the rostrocaudal axis, influenced by gradients of RA and FGF/GDF expression. Within the motor neuron population generated from the pMN progenitor domain, there are subpopulations of motor neurons separated into six distinct motor columns: median motor column (MMC), hypaxial motor column (HMC), preganglionic motor column (PGC), lateral motor column (LMC), phrenic motor column (PMC), and spinal accessory column (SAC). (B) Hox gene expression along the rostrocaudal axis.
Spinal interneurons are similarly organized depending on rostrocaudal positioning. Several studies have implicated FGFs, RA, and Wnts in controlling the Hox gene expression of ventral interneurons. V0, V1, V2, and V3 interneurons have been shown to have differential gene expression along the rostrocaudal axis of the developing spinal cord, particularly subdivided at the brachial, thoracic, and lumbar levels132,133.
The local distribution of spinal motor neurons and interneurons has been identified as a factor in the development of CPG circuits in the spinal cord 124 . Considering the extensive role of spinal CPG networks for locomotion, the developmental rostrocaudal diversification of spinal motor neurons and interneurons has gained attention with the generation and transplantation of neurons and neural stem cells for nervous system repair after SCI. With the goal of cell transplantation studies being to restore lost function and repair neural circuits, the generation of neurons with regional specificity must be taken into consideration to account for pre-established local motor neuron–interneuron interactions along the rostrocaudal axis.
Dorsal–Ventral Patterning
Differences in form and function are most pronounced between the dorsal and ventral domains of the spinal cord, with dorsal progenitors generally giving rise to domains that process sensory input and ventral progenitors giving rise to domains that process motor output (Fig. 3). Readers may refer to Zholudeva et al 107 , Sagner and Briscoe 134 , and Hernandez-Miranda et al 135 for extended discussion of the developmental origins of these spinal neurons. Generation of ventral cell types requires activation of Shh signaling pathways. Shh is secreted by the floor plate during development and gives rise to all ventral progenitor domains in a concentration dependent fashion 135 . Within the dorsal horn, a variety of spinal interneuron subtypes process sensory information from primary afferent inputs and modulate transmission to spinal motor circuits and supraspinal regions 134 . However, very little progress has been made on the derivation of dorsal and sensory populations for transplantation in SCI. Early “Class A” dorsal progenitors arise via Wnt and BMP signaling 135 . Late “Class B” dorsal progenitors arise independent of these signals, and the conditions driving their development in vitro have been more difficult to elucidate136,137. To design protocols aimed at generating spinal neuronal cells with regionally relevant identity, the emerging technology of generating exogenic cells and organs via targeted gene editing, as well as blastocyst complementation may provide additional sources of authentic neuronal progenitor cells for transplantation for repair in SCI.
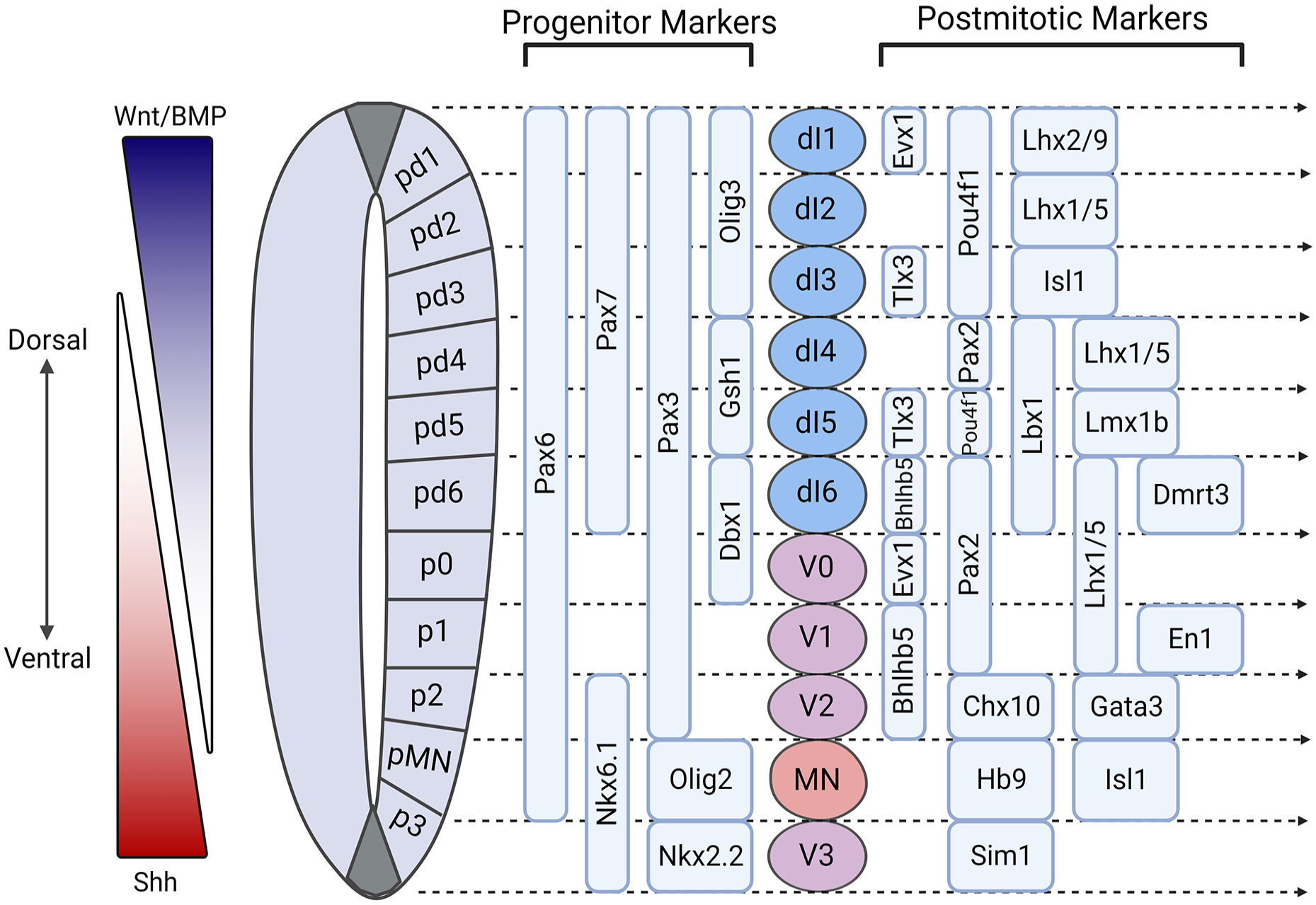
Developmental dorsal–ventral patterning of the spinal cord. Wnt and BMP secreted from the roof plate and Shh secreted from the floor plate are the main factors influencing neuronal subtype specification along the dorsoventral gradients. The progenitor domains, pd1–pd6, p0–p3, and pMN, generate dorsal interneurons (dI1–dI6), ventral interneurons (V0–V3), and motor neurons (MN), respectively. Several progenitor and postmitotic markers have been identified for particular subpopulations of dorsal and ventral interneurons along the dorsoventral axis, acting as potential gene candidates for region-specific cell differentiation protocols.
Developmental Gene Networks for Exogenic Spinal Cell Specification and Differentiation
While several protocols have been established to generate different spinal neuron populations, the regional diversity of spinal neurons has yet to be fully recapitulated in vitro. However, the few protocols that have been developed to tailor neuronal differentiation toward specific subpopulations of spinal neurons have made use of relevant developmental cues and gene networks. Here, we discuss the subtype-specific genes that could be targeted to produce spinal neurons with regional specificity (Fig. 4).
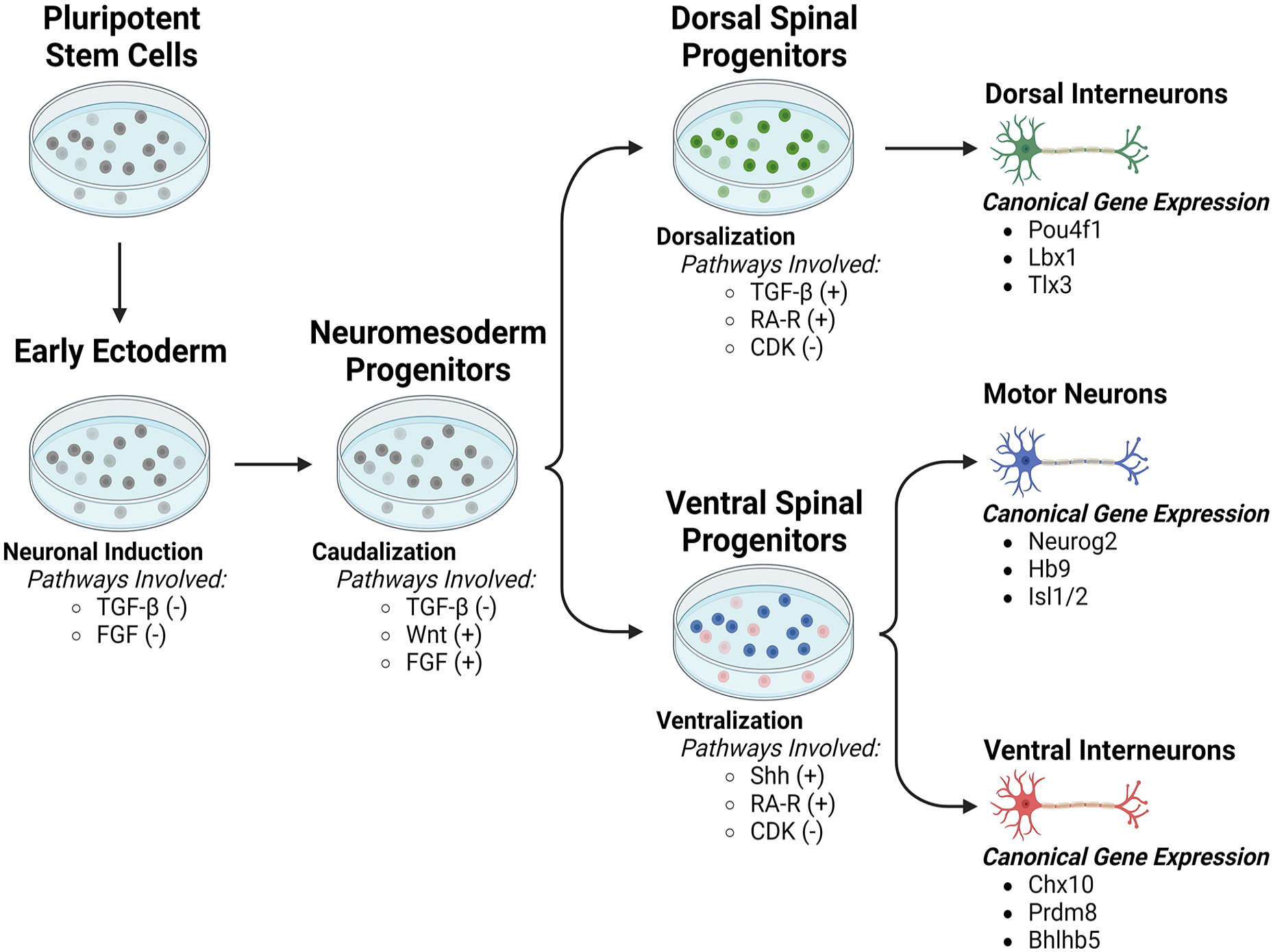
Generating neuronal progenitor subtypes for cell transplantation by temporal enhancement or suppression of key developmental pathways. Pluripotent stem cells (PSCs) must first undergo neural induction, traditionally to an early ectoderm phenotype, prior to further differentiation to specific neural types. Subsequent caudalization to a spinal identity is essential for production of dorsal, ventral, and motor domains. For generation of dorsal populations, TGF-β pathway agonists, such as BMPs, allow for maturation into dorsal interneurons upon extended culture. Particular networks of expression, such as Pouf41, Lbx1, and Tlx3, may be utilized for identification of specific interneuron subtypes. For generation of ventral motor and interneurons, sonic hedgehog (Shh) pathway agonists allow for maturation into motor neurons or ventral interneurons upon extended culture. Particular networks of expression, such as Neurog2, Hb9, and Isl1 for motor neurons and Chx10, Bhlhb5, and Prdm8 for ventral interneurons, may be utilized for identification of specific interneuron subtypes.
Motor Neurons
Motor neuron development is largely influenced by developmental gradients of RA, Shh, and FGF signaling127,138. These signaling pathways influence the generation of the pMN progenitor domain where all spinal motor neurons arise. The pMN progenitor domain is characterized by the expression of homeodomain proteins PAX6, NKX6.1, and basic helix–loop–helix (bHLH) protein OLIG2139–141 (Fig. 3). Motor neuron progenitors begin differentiation as a result of RA and OLIG2-mediated gene expression of GDE2 142 and Neurog2 143 , respectively. In particular, OLIG2 and Neurog2 promote transcription of downstream motor neuronal genes, such as homeobox gene Hb9, early in development144–146. In addition, the ISL1–LHX3 complex has been shown to be significant in specifying motor neuronal fate over an interneuron fate147,148.
Developmental motor neurons will adopt a fate dependent on the muscle it targets and its location along the spinal cord, thus relying on further differentiation cues along both the dorsoventral and rostrocaudal axes, as described above. Significantly, motor neurons follow a columnar organization, separating pools of motor neurons by their location and muscle targets. The four well-characterized motor columns are the median motor column (MMC), lateral motor column (LMC), hypaxial motor column (HMC), and the preganglionic column (PGC) 138 (Fig. 2A). MMC neurons are located all throughout the spinal cord and are characterized by the co-expression of Hb9, ISL1/2, OCT6, and LHX3/4149,150. LMC neurons are found in the brachial and lumbar levels of the spinal cord, and are characterized by the expression of ISL2, FOXP1, and RALDH2 130 . HMC neurons are located in the thoracic level of the spinal cord, and are known to express Hb9, ISL1, and ER81 130 . PGC motor neurons are localized to the thoracic and upper-lumbar segments of the spinal cord and known to express SMAD1, NOS1, ZEB2, HNF6, and FOXP1130,150,151. In addition to the MMC, LMC, HMC, and PGC, there are two additional, less-characterized motor columns—phrenic motor column (PMC) and spinal accessory column (SAC)—that warrant future research into the gene expression profiles of its neuronal populations.
Several protocols have been developed to generate motor neurons in vitro from ESCs and iPSCs. These protocols generally follow the processes of neural induction, differentiation, and maturation described above. Neural induction and differentiation are promoted by culturing the ESCs or iPSCs with BMP inhibitors, ROCK inhibitors, SMAD inhibitors, TGF-β inhibitors, Shh agonists, RA, and neurotrophic growth factors. Motor neuron differentiation is promoted by the presence of key developmental factors, such as Shh, RA, brain-derived neurotrophic factor (BDNF), and FGF. Subsequent motor neuron maturation involves several neurotrophic and growth factors91,105,152–162. While these protocols have been shown to generate motor neurons in vitro, the majority of protocols generate neurons that are often immature, uncharacterized, or limited to the upper motor neuron subtype, therefore likely not locally relevant to an injury within the spinal cord. Considering the range of motor neuron subtypes throughout the spinal cord, each expressing different combinations of genes designed for specific muscle targeting, current studies have started to focus on generating subtype-specific motor neurons in vitro.
Spinal Interneurons
Dorsal interneurons
As described above, early dorsal spinal specification is driven largely by BMP and Wnt pathway signaling via secretion of factors from the roof plate. As cellular identity advances, a plethora of regulated gene networks coordinates the onset of distinct dorsal domains. Homeobox genes, including HOX genes and PAX genes, play a critical role in broad patterning of the spinal cord along the rostrocaudal and dorsoventral axes, respectively. For more precise dorsal specification, bHLH transcription factors contribute to development of distinct progenitor layers 163 . Much of the work elucidating gene network expression in the spinal cord has been performed in chick and mouse knockout studies164,165.
Class A dorsal progenitors, which are dependent on roof plate signaling for appropriate specification, predominantly express AtOH1 and Olig3. Multiple studies have confirmed Olig3 is necessary for the specification of class A fate165–167. Class B dorsal progenitors rely more on a combination of homeobox and bHLH expression, including Ascl1, Pax7, and Ngn2. Combinatorial expression of multiple genes can be used to further classify distinct populations 168 .
As progenitors further differentiate into a postmitotic state, gene expression undergoes a transition to six distinct interneuron populations. Class A progenitors give rise to interneuron population 1–3 (dI1–dI3) and class B progenitors give rise to interneuron populations 4–6 (dI4–dI6). In class A progenitors, AtOH1 and Olig3 expression gives way to Pou4f1 expression. Further specification can be seen in expression of Lhx9 by dI1s, FoxD3 by dI2s, and Tlx3 by dI3s. In class B progenitors, Ascl1 expression gives way to Lbx1 expression. Further specification can be seen in expression of Pax2 by dI4s, Lhx1/5 by dI5s, and co-expression of Pax2 and Bhlhb5 in dI6s. These cardinal classes have been defined by morphological, electrophysiological, and functional properties in addition to their distinct genetic profile 107 . Subtype specific gene modification has proved valuable in generation of ventral populations, as described below, and future research may explore this possibility for dorsal interneuron specification.
With the limited work that has been done generating dorsal spinal interneuron populations, targeted gene editing to drive a dorsal phenotype has yet to be attempted. Upregulation of Wnt/β-catenin signaling pathways using exogenous factors has demonstrated strong results in the generation of class A progenitors, though a similar signaling cascade that regulates class B progenitors has yet to be identified.
Ventral interneurons
Ventral interneuron positioning is largely influenced by Hox gene expression, controlled by FGFs, RA, and Wnt signaling, and much like the dorsal interneurons, gene expression transitions as progenitors differentiate into the postmitotic stage, thus resulting in the four distinct cardinal classes (V0–V3). V0 interneurons arise from the dorsal–most ventral progenitor p0, and are defined by the expression of Dbx1 169 , with postmitotic markers Evx1/2, Pax2, Lhx1/5. V1 identity arises in the p1 progenitor domain where the interneurons initially express Irx3 and Dbx2. Following maturation, these interneurons can be identified by the postmitotic marker En1. The p2 progenitor domain gives rise to V2 interneurons that express Irx3, Lhx3 and Foxn4131,132. This cardinal class of interneurons is further subdivided into the well-characterized V2a and V2b interneurons. The segregation of these two subtypes is driven by Notch-Delta signaling followed by differential gene expression. V2a interneurons can be identified by their postmitotic marker Chx10, whereas V2b interneurons express Gata3. Finally, V3 interneurons arise from the p3 progenitor domain and largely express Nkx2.2 and Nhn3 along with the postmitotic marker, Sim1 170 .
In the development of therapeutic cell transplantation models for SCI, the addition of regionally relevant cell specification procedures has enhanced recovery parameters after injury. V2a and V3 neurons have been generated in mouse and human stem cells with much success170–173. V2a interneuron induction was first achieved using an in vitro protocol for differentiating mouse ESCs into interneurons through induction with RA, purmorphamine (Shh agonist), and DAPT (N-[N-(3, 5-difluorophenacetyl)-l-alanyl]-s-phenylglycinet-butyl ester; Notch inhibitor). V2a enrichment in vitro was achieved by ectopic expression of puromycin N-acetyltransferase under the Chx10 promoter where puromycin resistance would be found in Chx10-expressing cells specifically. Considering that V2a interneurons are characterized by Chx10 expression during development, this protocol results in an interneuron population highly enriched with V2a interneurons. This differentiation protocol was also translated to hiPSCs, showing the robustness of this combination of morphogens and small molecules for developing V2a-enriched neuronal populations 172 . Similarly, the differentiation of V3 interneurons from mouse ESCs was achieved through lower concentrations of RA combined with longer exposure to a Shh agonist. The resulting population of interneurons displayed expression of V3 interneuron-specific genes Sim1 and Nkx2.2, suggesting a V3-enriched population of interneurons 171 . Although V2a and V3 interneurons have been generated in vitro, the other ventral neuron populations and subpopulations remain to be explored.
Generating Exogenic Neuronal Cells Via Blastocyst Complementation as a Source of Cells for Repair of SCI
Blastocyst Complementation
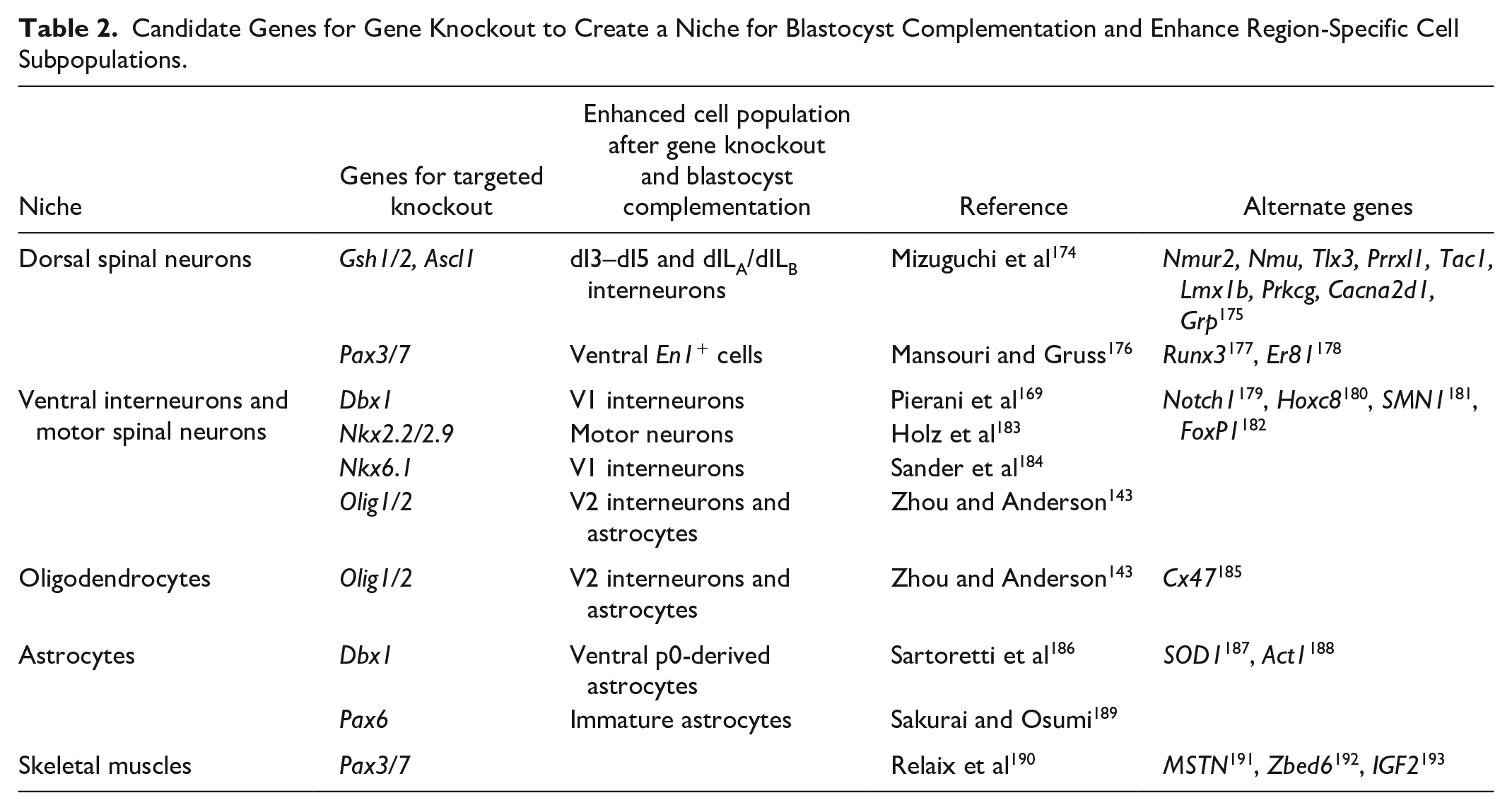
A recent novel approach in the field of regenerative medicine is the use of blastocyst complementation for the generation of exogenic organs and cells. The process of blastocyst complementation involves genetically engineering embryos from one organism to knockout specific target genes that are critical for cell specification, creating a developmental niche leading to agenesis of the cell type or organ of interest (Fig. 4, Table 2). Donor pluripotent stem cells from the same or different species are then injected into the modified blastocysts and transferred to pseudo-pregnant maternal surrogates. The healthy pluripotent stem cells injected can then occupy the empty niche and continue with normal development of the cells and organs of interest 194 . It has been suggested that any cell type or organ has the potential to create developmental niches via specific target genes 195 . This has shown to be the case with intraspecies and interspecies blastocyst complementation in recent years, with great success in generating a wide range of cells and organs, including inner ear sensory neurons, liver, forebrain, lymphocytes, kidney, pancreas, lung, thyroid, and eye196–203.
Candidate Genes for Gene Knockout to Create a Niche for Blastocyst Complementation and Enhance Region-Specific Cell Subpopulations.
Additional knowledge is necessary to apply such technology to humans as there are still a number of barriers and challenges to overcome with this approach. A major concern involves the ethical debate surrounding human-animal chimeras, and the risk of humanization. However, a review of 150 peer-reviewed transplantation studies involving relatively high degree of neurological chimerism, found no evidence to suggest humanization of human-animal chimeras 204 . In addition, a survey measuring public readiness of human-animal chimeric research in the United States found that more than half of the population was accepting of blastocyst complementation research, making this approach all the more a viable option for future therapeutics 205 . There are also technical barriers, such as off-target chimerism within the brain and reproductive systems of human cells in host animals204,205. Although there is no current research on the treatment of SCI with blastocyst complementation, this approach provides a promising future. Blastocyst complementation avoids the challenges associated with many other approaches, including the need for immunosuppression and risk of chemotherapy-associated toxicity (Fig. 5). While not yet attempted within the context of SCI, this novel approach, in theory, can be used to create human-animal chimeras to generate authentic exogenic spinal neurons for transplantation to restore function and connectivity following SCIs.
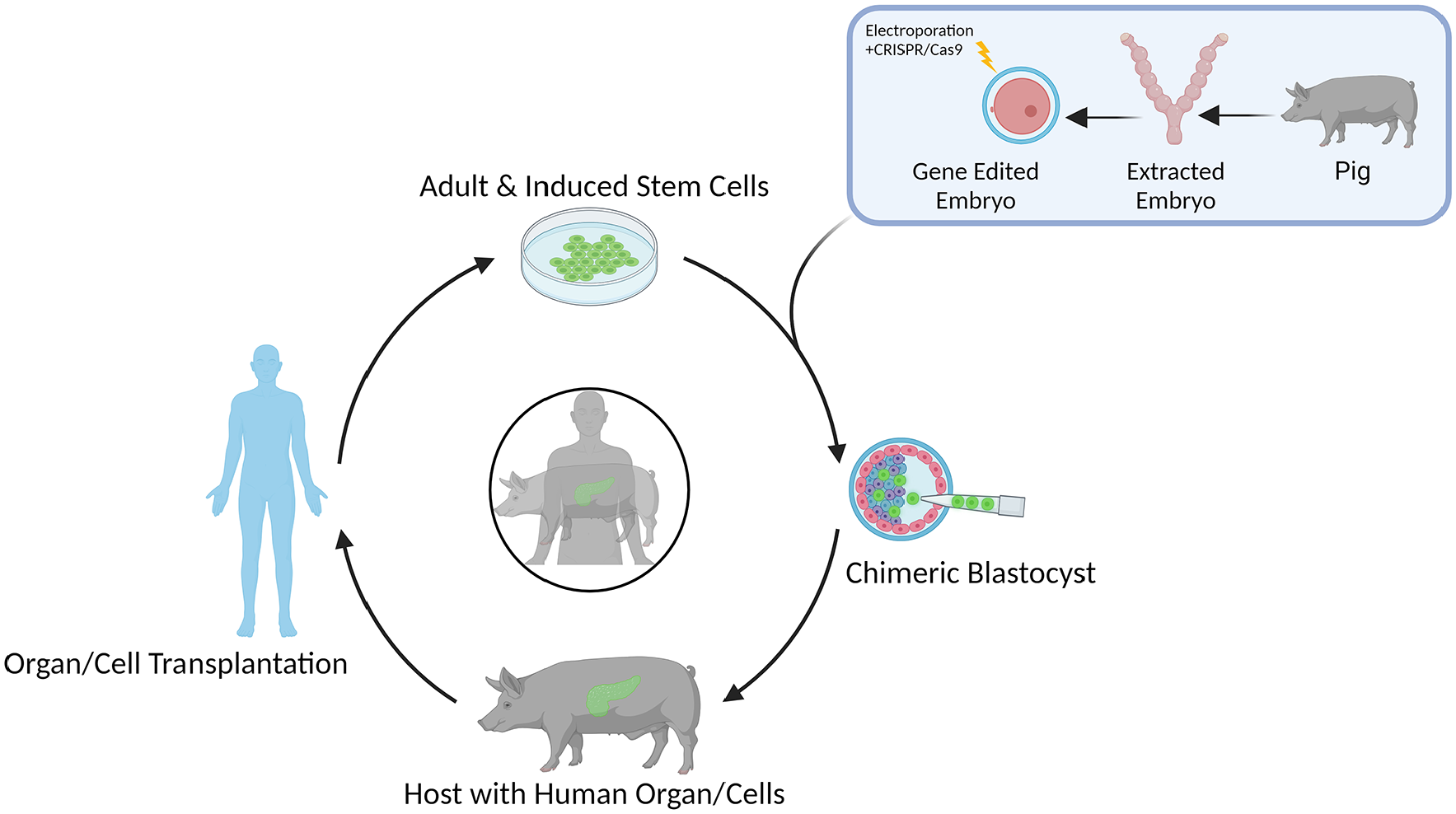
Schematic of blastocyst complementation. Human pluripotent stem cells in vitro are injected into genetically engineered porcine blastocysts, which are then transferred to pseudo-pregnant dams. The chimeric blastocysts are left to develop to a fetal stage in which human neuronal stem/progenitor cells can be harvested or they can be left to mature into human organs to be harvested and processed for transplantation into patients.
Gene Editing for Blastocyst Complementation
A key step in generating exogenic cells via blastocyst complementation is the need to knockout specific genes in developing embryos to create a niche for injected wild-type stem cells to occupy and differentiate into the desired cell phenotype. Advances in genome editing with synthetic nucleases, such as transcription activator-like effector nucleases (TALEN), and CRISPR/Cas systems, are now being used to modify stem cell-derived populations for the production of relevant disease lines206,207. These approaches are now being utilized to knockout genes with developing embryos as hosts for receiving injections of pluripotent stem cells to generate intraspecies and interspecies chimeras enabling the growth of specific organs and cells 195 . The selection of appropriate genes for targeted knockout to create niches for producing chimeric animals for generating exogenic organs and cells is dependent on the understanding of the genes required for the developmental biology of the targeted organs and cells; see Table 2 for a comprehensive list of relevant spinal neuronal populations and corresponding target genes to knockout for generation of a developmental niche. It is important to consider, however, the potential limitation that presents with knocking out cardinal classes is the occurrence of divergence within the classes. For example, Hayashi et al 208 demonstrate the divergence of Chx10 expression in V2a interneurons along the rostrocaudal axis that ultimately generates multiple subtypes of V2a interneurons. This divergence can be limiting for many current approaches in the regenerative medicine field. However, with blastocyst complementation, this challenge is less limiting since knocking out the cardinal class would ideally create the developmental niche that donor cells would take up completely. The donor cells would then grow and diverge normally during the developmental process, thus generating authentic exogenic neuronal populations that could be separately identified and used for region-specific cell transplantation within the spinal cord.
Ultimately, the goal is to identify the appropriate genes responsible for the cell or organ of interest, knock them out to create a developmental niche for the transplant of donor cells to grow said cell or organ of interest. Within the context of SCI, blastocyst complementation may be considered in the future for generating human-animal chimeras for a variety of spinal neuron populations.
Conclusion
SCI remains a significant area of study for treatment development. With the variety of therapeutic approaches available and studied to address SCI neuropathologies, progenitor cell transplantation has been at the forefront of several preclinical and clinical studies. The transplantation of progenitor cells has found various degrees of success in animal models of SCI and in human clinical trials. One key consideration with the generation of exogenic neuronal cells to optimize for success is the regional specification of cells for transplantation. Considering the importance of regional and subtype specificity during the development of neuronal cells of the spinal cord, similar considerations being made for cell transplantation may enhance therapeutic benefit. In fact, the adoption of regional specification in differentiation protocols has already improved transplantation outcomes and will continue to do so as techniques are refined and replicated. However, while distinct developmental pathways contribute to the production of these varied spinal cell types, their functional classification within either sensory or motor domains is an oversimplification. In truth, the spinal cord is a complex, interconnected system, and one domain cannot operate without the input from the other. Future work must consider application of both cell types in tandem to address the multifaceted nature of injury and recovery. Furthermore, the advent of blastocyst complementation to generate exogenic spinal neurons may provide regionally specific cells needed for appropriate neural connectivity and more robust functional recovery.
Footnotes
Author Contributions
Initial planning and drafting of this manuscript were completed by Dr Walter Low, Dr Ann Parr, Alex R., Anne H.-S., Madison W., and Zainab K. Significant writing, restructuring, editing, and reviewing were completed by Alex R., Anne H.-S., and Madison W. Figures were drafted and edited by Alex R., Anne H.-S., Madison W., and Jeffrey B. Final reviews and overviews were conducted by Dr Ann Parr and Dr Walter Low.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was supported by NIH grants R01 NS119297 and R01 AI173804, funds from the State of Minnesota Office of Higher Education Grant Program on Spinal Cord Injury and Traumatic Brain Injury Research, and the Suzanne M. Schwarz Fund.
