Abstract
Peritoneal dialysis (PD) is a well-established renal replacement therapy commonly employed in clinical practice. While its primary application is in the treatment of kidney disease, its potential in addressing other systemic disorders, including neurological diseases, has garnered increasing interest. This study provides a comprehensive overview of the related technologies, unique advantages, and clinical applications of PD in the context of neurological disorders. By exploring the mechanism underlying PD, its application in neurological diseases, and associated complications, we addressed the feasibility and benefits of PD as an adjunct therapy for various neurological conditions. Our study aims to highlight its role in detoxification and symptom management, as well as its advantages over other universally accepted methods of renal replacement therapy. Our goal is to bring to the spotlight the therapeutic potential of PD in neurological diseases, such as stroke, stimulate further research, and broaden the scope of its application in the clinical setting.
Introduction
Peritoneal dialysis (PD) has traditionally been a cornerstone in renal replacement therapy. However, its application extends beyond renal conditions, and recent explorations into its use for neurological disorders are revealing its therapeutic potential. PD systemically detoxifies the body by serving as a filtration system through the peritoneal cavity. This review aims to highlight the therapeutic potential of PD in the context of neurological disease, with a particular focus on stroke, a leading cause of disability and mortality worldwide.
PD involves a dialysis system that includes a catheter, commonly the Tenckhoff catheter 1 , and a dialysate solution. Recent advancements have introduced alternatives to traditional glucose-based dialysates, such as buffered solutions2,3 and isodextrin-based options 4 , aiming to improve biocompatibility and reduce the risk of complications, such as peritoneal sclerosis5,6. Effective PD relies heavily on successful catheter placement, with techniques ranging from conventional surgical to minimally invasive percutaneous methods7–10. These advancements have made PD more accessible and adaptable to various clinical settings.
Compared with hemodialysis (HD), PD offers a more physiological and patient-friendly approach. It does not require vascular access or anticoagulants, making it a viable option for patients with HD-related thrombosis 11 or those who are hemodynamically unstable 12 . In addition, PD has been associated with a higher likelihood of renal function recovery 13 , a less complex procedure 14 , and a significant enhancement in the quality of life for patients. This is owing to its flexibility and independence from frequent clinical interventions 15 .
The intersection of PD and neurological disorders opens a new frontier in medical treatment. Given the high disability and mortality rates associated with neurological conditions like stroke, integrating PD as an adjunct therapy could offer a novel approach to management and rehabilitation. This review emphasizes the logic and significant role of PD in neurological disorders, aiming to expand its clinical applications and encouraging further research in this area. The potential of PD to serve not just as renal therapy but also as a means of improving outcomes in neurological diseases could revolutionize treatment protocols and significantly improve patient prognosis.
Materials and Methods
This review was conducted in strict adherence to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, as outlined on the PRISMA website (http://www.prisma-statement.org/). Our literature search strategy was carefully crafted to encompass a wide range of relevant topics. The key terms guiding this search included: “advancements in PD technology,” “comparison of PD and HD,” “clinical applications of PD,” “PD in the context of neurological disorders,” “PD’s role in stroke management,” “impact of PD on brain health,” and “complications associated with PD.”
The literature search was conducted over the months of April to May 2023. Our approach combined both free-text searches and targeted queries using specific terms related to PD and neurological diseases, as mentioned above. The primary database for this search was PubMed, a comprehensive resource for biomedical literature. We focused on studies published in English to ensure a thorough and inclusive review of the available literature.
For each identified study, we performed an initial screening based on abstracts, followed by a detailed examination of full papers relevant to our topics of interest. This methodical approach enabled us to compile a comprehensive and up-to-date collection of studies that provided a deep insight into the multifaceted roles of PD, particularly in the treatment of neurological disorders and stroke, as well as its technological progress and clinical implications. This review aims to present a critical analysis of these studies, offering a cohesive understanding of the current state of knowledge in this field.
Mechanism of PD
The exploration of PD mechanisms is pivotal in understanding its broader therapeutic potential, particularly in the context of neurological disorders. PD, traditionally a kidney replacement therapy, operates primarily by removing toxic substances and excess water from the body. While its conventional role in treating various kidney diseases, such as acute kidney injury, chronic kidney failure, and as a pre-operative or post-operative measure in kidney transplantation is well-recognized; its application extends beyond these realms (Fig. 1).
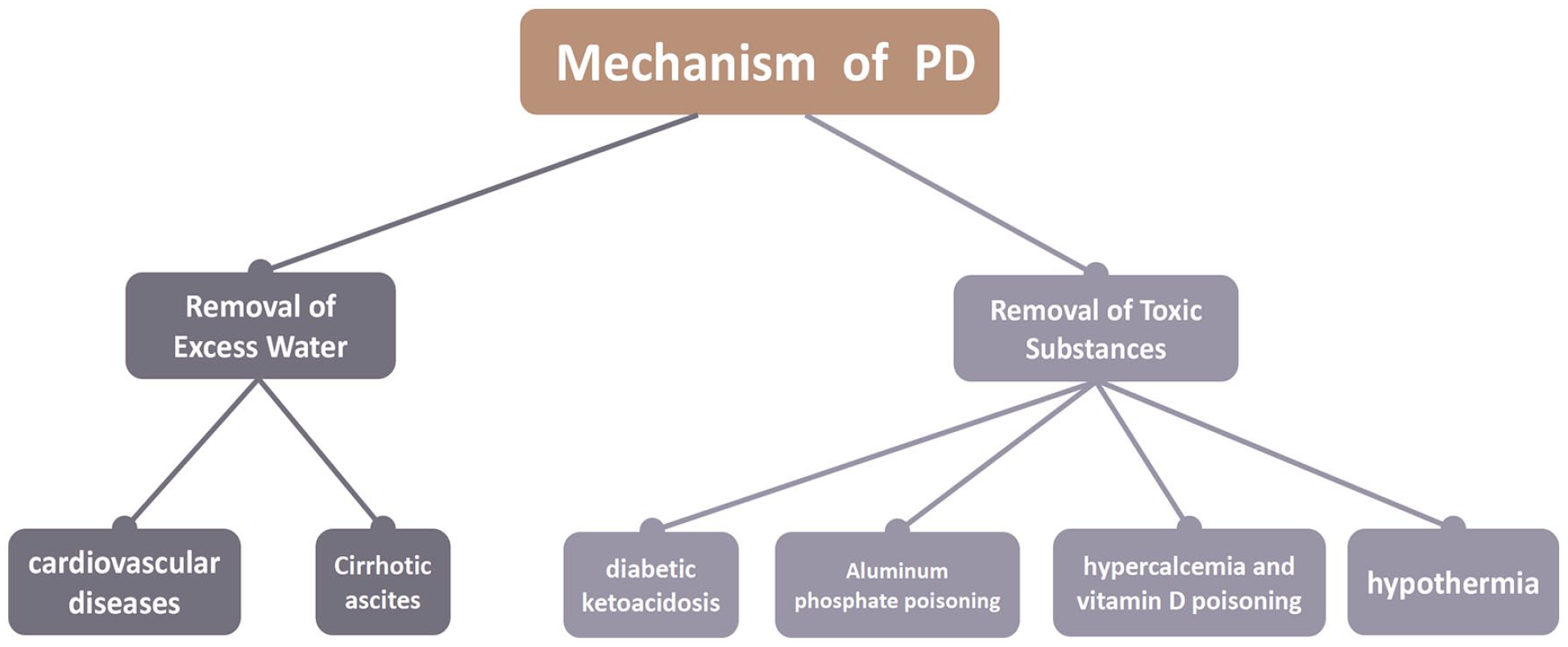
Removal of excess water in cardiovascular diseases and cirrhotic ascites, and the removal of toxic substances in aluminum phosphate poisoning, hypercalcemia, vitamin D poisoning, and hypothermia are the two primary mechanisms in which peritoneal dialysis (PD) exerts its therapeutic effect.
Understanding the mechanisms of PD is crucial for several reasons. First, it enables us to comprehend how PD can be beneficial in the treatment of neurological disorders. The process of detoxification, fundamental to PD, plays a significant role in managing these disorders. By removing toxic substances that could potentially have detrimental effects on neurological functions, PD may offer a unique approach to treating or managing neurological conditions. In addition, the mechanism of fluid removal in PD could have implications for neurological diseases where fluid balance is a concern. Conditions like cerebral edema or increased intracranial pressure, often associated with neurological disorders, such as stroke, could potentially be managed through PD’s fluid regulation capabilities.
Removal of Excess Water
PD is the preferred treatment for end-stage renal failure patients with cirrhosis and ascites. It offers a slow and continuous treatment that maintains hemodynamic stability while simultaneously removing ascites, without the need for anticoagulation, thus reducing the risk of bleeding 16 . PD not only drains ascites but also prevents its accumulation by increasing ascites static pressure 17 . Furthermore, dialysate may serve as a calorie source, which is beneficial for cirrhotic patients experiencing malnutrition and appetite loss 18 . Clinical trials have shown that concerns regarding albumin loss and a high incidence of infectious peritonitis did not materialize 19 . PD has also been used as an alternative to repeated large-capacity abdominal punctures for treating malignant refractory ascites, demonstrating its safety and effectiveness 20 . This approach can alleviate breathing difficulties and improve the quality of life for patients with malignant ascites 20 .
PD also has a therapeutic effect on cardiovascular diseases, which can cause great psychosocial stress21,22. PD can optimize the volume status in heart failure patients, allowing for customized removal of water and sodium, thus, making it an effective adjunct to individualized drug therapy23,24. Numerous clinical studies have demonstrated that PD effectively alleviated heart failure symptoms and improved cardiac function 25 . Barman et al. compared the effects of continuous ambulatory peritoneal dialysis (CAPD) and HD in end-stage renal disease patients with heart failure and found that the CAPD group experienced better improvements in cardiac function, thus, proving PD to be more advantageous for heart failure patients than HD 26 . In a systematic review, Lu et al. reported that PD significantly reduced length of stay, improved cardiac function, and had a low incidence of peritonitis in refractory congestive heart failure patients, therefore demonstrating its safety and effectiveness as a volume-reducing method 27 .
Removal of Toxic Substances
In 1967, Knight R.K. employed PD to save the life of a young woman suffering from suicidal chlorate poisoning 28 . In a case reported by Harpreet Singh, PD was used to correct diabetic ketoacidosis, as traditional bicarbonate treatment failed to adequately control blood sugar levels and improve symptoms of refractory metabolic acidosis 29 . Krystal et al. proved that PD could effectively correct vitamin D poisoning by leveraging the calcium loss during the dialysis process 30 . This case also highlights the need to consider calcium and vitamin D loss in PD patients. PD can also correct refractory metabolic acidosis caused by aluminum phosphate 31 . Maj and colleagues used warm dialysate in PD to successfully correct hypothermia in a traumatic paraplegic patient, suggesting that PD fluid can also be employed for temperature regulation 32 .
Taken together, significant emphasis has been placed on the mechanisms underlying PD application, providing new insights and potential therapeutic approaches.
PD Complications
Addressing complications associated with PD is crucial for the safety and efficacy of this therapy. While PD offers significant benefits for patients with renal and neurological disorders, its effectiveness can be severely compromised by complications, such as infections and mechanical issues. Infections, particularly at the catheter site and peritonitis, are among the most common challenges. These can significantly impact patient health, leading to increased morbidity and potentially limiting the effectiveness of PD. Effective management of these infections, typically through stringent hygiene protocols and appropriate antibiotic use, is vital to safeguard the health of patients undergoing PD. Mechanical complications, though less frequent than infections, present a substantial risk. Issues like organ perforation or dialysate leakage can have serious consequences and often necessitate swift medical intervention. These complications underscore the importance of thorough pre-operative risk assessments and skilled catheter handling by healthcare professionals.
Infections
Common infections in PD patients include catheter exit site infections and PD-associated peritonitis. For exit site and catheter infections, proper care of the exit site plays a crucial role in prevention. Guidelines for PD-associated peritonitis management recommend daily application of an antibiotic cream or ointment at the catheter exit site 33 . Gao et al. demonstrated that the topical application of mupirocin cream or ointment was effective in reducing exit site infections and peritonitis caused by Staphylococcus aureus 34 . PD-associated peritonitis is a common complication in PD patients, and a single episode of severe peritonitis often results in decreased peritoneal ultrafiltration capacity, consequently making it the most common reason for conversion to long-term HD 35 . However, with equipment and technological advancements, as well as the implementation of global guidelines for PD peritonitis prevention and management, there has been a decrease in PD-associated peritonitis33,36. Antibiotics play a key role in preventing PD-associated peritonitis. Care guidelines support the prophylactic use of antibiotics before catheter insertion, with antibiotic selection being based on local resistance profiles 33 . Catheter exit site infections are associated with peritonitis 37 . Therefore, early detection and prompt antibiotic treatment of exit site and catheter infections can effectively prevent peritonitis. In addition, using a “flush and refill” design for the separated PD system and training patients, and caregivers can also effectively reduce the incidence of peritonitis 38 .
Mechanical Complications
Mechanical complications include peritoneal organ perforation, dialysate leakage, and poor drainage caused by improper positioning, blockage, displacement, or entanglement of tubes. Although operational risks cannot be entirely eliminated, the incidence of such complications is relatively low 39 . Pre-operative identification and assessment of mechanical complication risks can effectively reduce their occurrence. The surgeon’s experience is inversely proportional to the rate of mechanical complications, making standardized procedures and individualized placement programs crucial 40 . Studies have also shown that delaying the initiation of dialysis after peritoneal catheterization can reduce catheter-related mechanical complications 41 .
Taken together, understanding, preventing, and managing these complications are essential steps in ensuring that PD remains a viable and effective treatment option. By minimizing these risks, we would improve patient outcomes, maintain the high efficacy of PD therapy, and enhance overall patient well-being. This focus on complication management is not only about preserving the integrity of the treatment but also about respecting and improving the quality of life for those relying on PD.
Application of PD in Nervous System Diseases
PD is a renal replacement therapy with unique advantages in the treatment of various nervous system diseases (Fig. 2). In stroke treatment, PD has been shown to effectively reduce plasma glutamate levels, providing a minimally invasive and hemodynamically stable alternative to HD. In mitochondrial neurogastrointestinal encephalopathy (MNGIE), PD can rapidly improve clinical symptoms and serve as a bridge treatment before receiving bone marrow transplantation (BMT). In the case of valacyclovir neurotoxicity, incremental PD has shown promise in alleviating symptoms. PD has also been applied in treating hepatic encephalopathy and hyperammonemic encephalopathy caused by valproate (VPA). Though the safety and effectiveness of PD in these contexts require further validation, its minimally invasive nature, hemodynamic stability, and continuous filtration capabilities make it an appealing option for treating a variety of nervous system diseases.
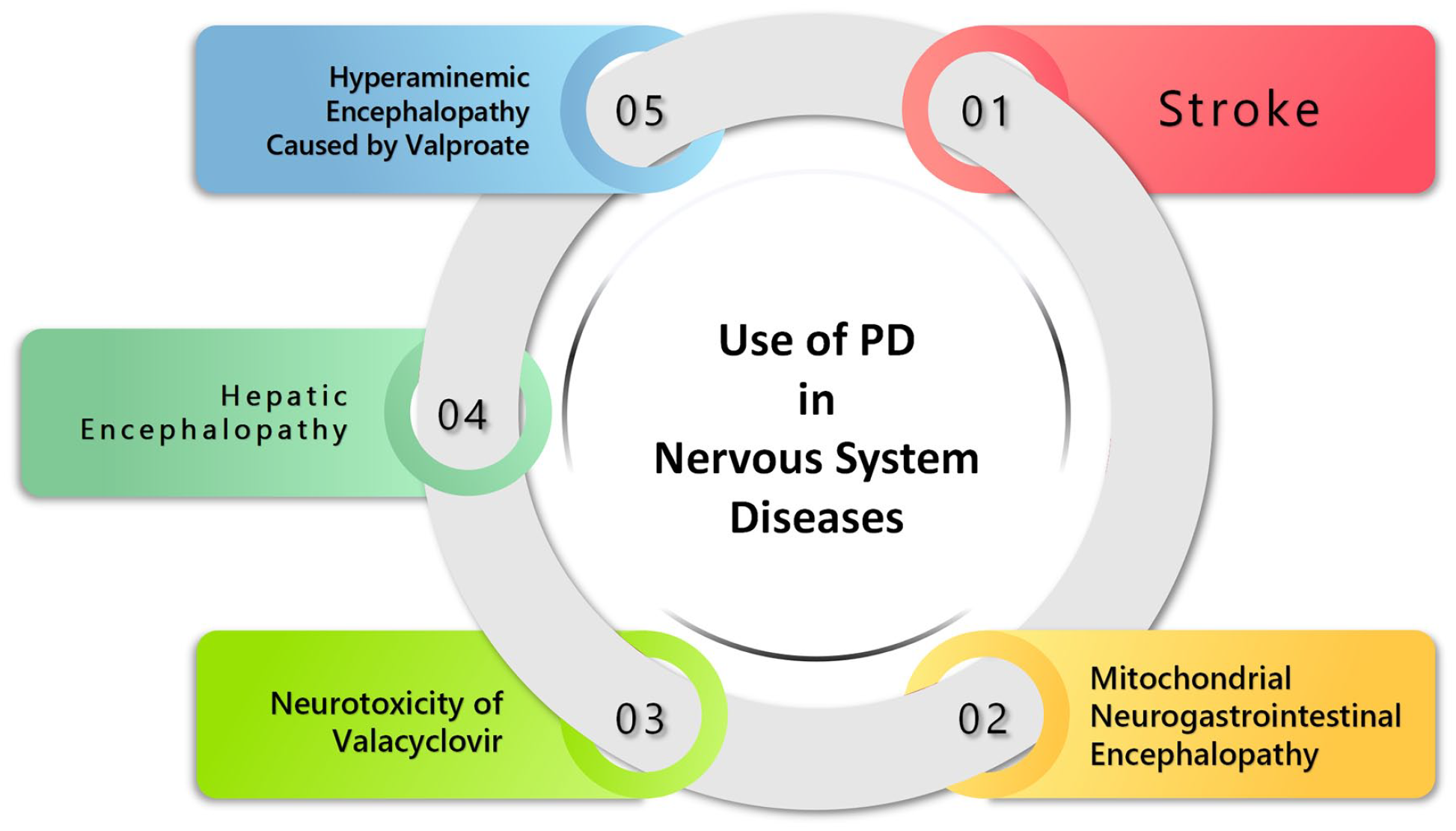
Peritoneal dialysis (PD) has been implemented in a variety of neurological diseases and it has shown clinical benefits in stroke management, MNGIE, and valproate-induced hyperammonemic encephalopathy. Further studies should expand on the safety and optimal usage of PD in valacyclovir-induced neurotoxicity and hepatic encephalopathy.
Stroke
Excitatory amino acids (EAAs), particularly glutamate, have been implicated in the pathogenesis of ischemic neuronopathy. During ischemia, excessive glutamate release from neurons and astrocytes leads to cellular calcium overload, primarily through its action on calcium-permeable N-methyl-
Previous research has demonstrated that intravenous administration of glutamate scavengers, such as pyruvate and oxaloacetic acid (OxAc), can decrease blood glutamate levels and enhance brain–blood glutamate outflow, leading to reduced cerebrospinal fluid (CSF) glutamate levels46–49. Although some human clinical trials have investigated NMDA receptor antagonists, they were discontinued prematurely due to adverse events and even fatalities 50 . As a result, non-pharmacological or extracorporeal approaches for lowering glutamate levels have become increasingly appealing.
Rogachev and colleagues assessed glutamate levels during HD in patients with stage V chronic kidney disease, showing that HD effectively reduced glutamate levels in patients with end-stage renal failure 51 . However, as previously mentioned, HD faces challenges in clinical application, including heparin anticoagulation and hemodynamic instability, among others. These limitations are particularly relevant for patients with hemodynamic instability, therefore restricting the use of HD to some extent. PD has been shown by Rogachev and colleagues to effectively reduce plasma glutamate levels 52 . Building on this finding, a study using a rat model of permanent middle cerebral artery occlusion (pMCAO) found that reducing plasma glutamate levels with PD led to a decrease in cerebral infarction volume 53 . Blood oxygen level-dependent (BOLD) functional magnetic resonance imaging (fMRI) demonstrated that PD could partially prevent functional deficits resulting from ischemic injury54,55.
While the clinical feasibility and safety of this treatment method require further exploration, it offers a new approach for stroke treatment. PD has unique advantages in stroke treatment. It is minimally invasive, does not require vascular access, utilizes simple equipment, can be performed at the bedside, and only takes about 20 min to set up, therefore ensuring early treatment for acute stroke patients. Compared with HD, PD offers greater hemodynamic stability and has less impact on blood flow dynamics and blood oxygen saturation in brain tissue56–58, thus making it more suitable for critically ill patients with hemodynamic disorders. In addition, since heparin anticoagulation is not required, PD can be used for both ischemic and hemorrhagic stroke patients.
Mitochondrial Neuro-Gastrointestinal Encephalopathy
MNGIE is a rare condition characterized by the accumulation of thymidine (dThd) and deoxyuridine (dUrd) due to thymidine phosphorylase (TP) deficiency. Major clinical manifestations include gastrointestinal disorders, ophthalmoplegia, and peripheral neuropathy 59 . The prognosis for MNGIE patients is typically poor, with high mortality rates. Common treatment approaches include platelet transfusion, allogeneic BMT, and dialysis. Platelet infusion and HD can only temporarily reduce dThd and dUrd levels. Although BMT offers a long-lasting therapeutic effect, the risk of transplant rejection compromises its safety. Yavuz et al. first reported the use of PD in MNGIE, which led to an improvement in gastrointestinal symptoms 60 . In another case reported by Claudia et al., a patient underwent 15 months of CAPD before BMT, which resulted in improved gastrointestinal and neurological symptoms, ultimately allowing the patient to undergo BMT 61 . In another case report, a young male patient experienced significant relief and weight gain following PD treatment62,63.
Compared with other treatment methods, PD can rapidly improve clinical symptoms, and its simple operation makes it suitable for symptomatic support management during acute attacks. CAPD can alleviate symptoms by continuously and slowly removing dThd and dUrd, therefore ensuring patients’ quality of life. It can serve as a bridge treatment by improving a patient’s clinical condition before undergoing BMT. While no treatment has been reported to effectively extend the life span of MNGIE patients, the symptom improvement and enhanced quality of life associated with PD suggest that it may hold greater potential for MNGIE treatment than current options.
Neurotoxicity of Valacyclovir (Valtrex)
Valacyclovir is used to treat herpes virus infections, including herpes labialis (cold sores), herpes zoster (shingles), and herpes simplex (genital herpes) in adults, as well as chickenpox and cold sores in children. About 90% of valacyclovir is eliminated by the kidneys through urine. Studies have shown that in CAPD patients, its elimination half-life extends from 2.5 to 3.3 to 14 to 20 h 63 , which exacerbates neurotoxicity, consequently leading to a series of neurological symptoms. Continuing CAPD without increasing the dialysis dose can fully resolve nerve toxicity symptoms within 7 days 64 . In Chrysoula’s case, incremental dialysis took 3 days to alleviate symptoms 65 . Bhanu improved symptoms within 24 h by increasing supportive treatment alongside incremental PD treatment and discharged the patient after 3 days 66 .
Although many studies have suggested that incremental PD can be used as a treatment for removing toxins and fluid, the limited efficacy of PD over HD must be noticed. While PD is sometimes not fast enough, HD is required. HD has long been considered a rapid and effective method for alleviating the neurotoxic symptoms of valacyclovir, as it significantly lowers serum drug levels 67 . Earlier studies indicated that approximately 60% of acyclovir was cleared after 4 h of HD in patients undergoing this procedure. Valacyclovir, as a precursor of acyclovir, can be rapidly eliminated by HD 64 . However, for CAPD patients, PD does not require establishing new dialysis access and can be initiated urgently, which may be more suitable for improving neurological symptoms during acute attacks. Although HD remains the first choice when valacyclovir causes severe neurotoxic effects, the general condition of patients and medical history should be considered before deciding on a dialysis plan. For patients with stable overall health, PD may be chosen, while patients with severe neurological or psychiatric symptoms and poor overall condition may require HD to rapidly alleviate symptoms 52 . Due to the limited number of cases, the effectiveness of incremental PD requires further validation.
Hepatic Encephalopathy
Both PD and HD are considered useful in treating hepatic encephalopathy by removing ammonia from the blood. Pipili et al. reported a case in which a patient improved clinically and they regained basic self-care ability after 10 months of CAPD treatment 68 . However, Suleyman’s patient needed to switch from PD to HD to achieve the desired effect 69 . In other reported cases, treatment outcomes have been inconsistent, with some patients requiring both methods simultaneously 70 .
Hemodynamic stability, the absence of anticoagulation, and improved quality of life are regarded as the main advantages of PD in treating hepatic encephalopathy. Additionally, the continuous clearance provided by PD helps maintain stable blood ammonia levels, thereby aiding in the prevention of recurrent hepatic encephalopathy and effectively treating ascites in cirrhosis simultaneously 17 . Previous case reports suggested that extracorporeal removal of blood ammonia through dialysis had a therapeutic effect on patients with hepatic encephalopathy. Although PD has unique advantages, the choice of dialysis modality should be tailored to each patient’s individual circumstances.
Hyperammonemic Encephalopathy Caused by VPA
Sodium VPA is used to treat epilepsy, bipolar disorder, and is occasionally used to prevent migraine headaches. The mechanism of PD for removing blood ammonia can also be applied to treat hyperammonemic encephalopathy caused by VPA. Although the mechanism of hyperammonemia differs from hepatic encephalopathy. VPA can affect liver and kidney metabolism, resulting in decreased ammonia clearance. Studies have shown that 20.4% of general hospitalized adults receiving VPA develop hyperammonemia 71 . In some cases, hyperammonemia led to severe hyperammonemic encephalopathy 72 .
Multiple case reports have demonstrated that HD is effective in rapidly normalizing ammonia levels and reversing encephalopathy symptoms73–75, with VPA and ammonia levels quickly returning to normal, thus indicating that dialysis is a life-saving intervention. PD is a preferable option for hemodynamically unstable patients. Amandeep et al. reported a successful case of PD in treating VPA-induced hyperammonemic encephalopathy 76 , demonstrating that PD can effectively and rapidly clear blood ammonia without causing other complications. Although most VPA-induced hyperammonemia cases resolve spontaneously after drug discontinuation, PD should be the preferred life-saving treatment for severe refractory cases, especially those with rapid clinical deterioration that does not respond to discontinuation of VPA and supportive measures.
Perspectives and Prospective
As a widely accepted renal replacement therapy, PD offers several advantages over other methods, such as renal function preservation, simpler operation, and stable hemodynamics. With advancements in related technologies and equipment, PD has increasingly gained clinical acceptance. In addition to renal replacement, PD serves as an adjunct therapy for various systemic diseases, primarily for removing toxic substances and excess water. Numerous animal and clinical studies have demonstrated that this detoxifying effect can also play a role in treating neurological disorders because of its safety and effectiveness. In the context of neurological diseases, the unique advantages of PD make it particularly suitable for stroke treatment. Therefore potential therapeutic impact of PD is worth further exploration.
Ongoing research should focus on understanding the underlying mechanisms of PD’s neuroprotective effects and optimizing PD protocols to maximize its benefits for patients with neurological disorders. Moreover, investigating the long-term outcomes of PD in neurological conditions and comparing its effectiveness with other treatment modalities will provide valuable insights. As our understanding of PD’s potential in treating neurological disorders strengthens, it is likely that PD will play an increasingly significant role in managing these conditions and improving patient quality of life.
Footnotes
Acknowledgements
The authors would like to thanks to Dr Sichao Guo for her help in the content and structure of the article.
Author Contributions
Muyuan Cheng and Yuchuan Ding are responsible for the acquisition, analysis and interpretation of the data, and drafting of the manuscript. Enoch Kim, Xiaokun Geng contributed to the critical revision of the manuscript.
Availability of Data and Material
Not applicable.
Ethical Approval
Not applicable.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
Not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by the Capital Clinical Characteristic Application Research Project of Beijing Science and Technology Commission (Z221100007422085), the Youth Plan of Beijing Luhe Hospital (LHYY2023-JC101, LHYY2023-JC102), and the Beijing Tongzhou District Financial Fund (2023).
