Abstract
No radical treatment is available for the regeneration of dysfunction and defects in airway epithelia. Artificial tracheae made of polypropylene and collagen sponge were used in clinical studies to reconstitute tracheae after resection. For early epithelialization of the luminal surface of the artificial trachea, a model was established, that is, an artificial trachea covered with human-induced pluripotent stem cell-derived airway epithelial cells (hiPSC-AECs) was transplanted into a tracheal defect in an immunodeficient rat. Unlike the cell types of hiPSC-derived cells that are currently used in clinical studies, AECs maintain tissues by proliferation and differentiation of basal cells into various cell types that constitute AECs constantly. Therefore, post-transplantation, the proportion of each cell type, such as ciliated and goblet cells, may change; however, no studies have examined this possibility. In this study, using our hiPSC-AEC-transplanted rat model, we investigated changes in the proportion of each cell type in hiPSC-AECs pre-transplantation and post-transplantation. As a result, the proportion of each cell type changed post-transplantation. The proportion of ciliated, basal, and club cells increased, and the proportion of goblet cells decreased post-transplantation. In addition, the proportion of each cell type in engrafted hiPSC-AECs is more similar to the proportion of each cell type in normal proximal airway tissue than the proportion of each cell type pre-transplantation. The results of this study are useful for the development of therapeutic techniques using hiPSC-AEC transplantation.
Introduction
The airway is an important pathway for respiration, and airway epithelia play a role in the defense against infection through the mucus secreted by secretory cells and the ciliary motility of ciliated cells. The dysfunction of the airway epithelia in hereditary diseases, such as primary ciliary dyskinesia and cystic fibrosis, leads to infection, inflammation, and mucus plugging the airways 1 . Extensive defects of the airway epithelia associated with injury or surgery can also cause similar problems 2 . Therefore, methods for the regeneration of airway epithelial function must be developed.
We developed an artificial trachea made of polypropylene mesh and collagen sponge and used them in clinical studies for reconstruction after tracheal resection 3 . In the presence of extensive defects, the epithelialization of the tracheal lumen took 2 months, increasing the risk of infection 3 . Therefore, a model was established by transplanting the artificial trachea covered with human-induced pluripotent stem cell-derived airway epithelial cells (hiPSC-AECs)4,5 into tracheal defect in immunodeficient rats with the aim of early epithelialization 6 . In this study, hiPSC-AECs survived in the epithelia of the luminal surface at the transplanted area 2 weeks post-transplantation 6 . These results suggest that hiPSC-AEC transplantation promotes the epithelialization of the luminal surface of the tracheae.
Clinical transplantation research uses several types of hiPSC-derived cells, including cardiomyocytes, retinal pigment epithelia (RPE), dopaminergic (DA) progenitor cells, and corneal epithelial cells 7 . These cells are either terminally differentiated or have a defined differentiation direction. By contrast, in the airway epithelia, basal cells, which are tissue stem cells, maintain the tissues by proliferation and differentiation into several cell types that constitute the airway epithelia, such as ciliated, goblet, and club cells 8 . Therefore, the proportion of each cell type in the airway epithelia could be different post-transplantation. In clinical studies, the proportion of cells that are positive for markers defining each cell type has been used to ensure the quality of cells for transplantation. In hiPSC-derived RPE transplantation for macular degeneration, the proportion of BEST1/PAX6 double-positive cells in the hiPSC-derived RPE sheet must be > 95% 9 , and in hiPSC-derived DA progenitor cell transplantation, the proportion of FOXA2B/TUJ1 double-positive cells that are defined as DA progenitor cells in total cells must be > 80% 10 . However, the transplantation of the tissue containing tissue stem cells that have proliferation and differentiation potency post-transplantation, such as hiPSC-AECs, has not yet been performed in a clinical study, and no studies have reported changes in the proportion of each cell type that constitutes the tissue pre-transplantation and post-transplantation. If the proportion of each cell type that constitutes the AECs pre-transplantation is the same as the proportion of each cell type that constitutes the AECs post-transplantation, the proportion of some cell types pre-transplantation could be used as a criterion to select the cells for transplantation. However, if the proportion of each cell type changes post-transplantation, other criteria, such as marker gene expressing in basal cells that decide the differentiation directions or supplemental factors that direct the cells to differentiate into specific cell types post-transplantation are necessary.
Therefore, in this study, we investigated whether the proportion of each cell type in the hiPSC-AECs changed post-transplantation, using our previously established transplantation model in which hiPSC-AECs are transplanted into the tracheal defects of immunodeficient rats.
Materials and Methods
Culture and Induction of Airway Epithelial Cells From hiPSCs
In this study, hiPSC lines 201B7 11 and 253G1 12 were obtained from RIKEN BioResource Research Center. The 201B7 cell line was induced using four reprogramming factors (Oct3/4, Sox2, Klf4, and c-Myc), whereas the 253G1 cell line was induced by three factors without c-Myc. These undifferentiated hiPSCs were maintained in Essential 8 Medium (Thermo Fisher Scientific, Waltham, MA, USA) on culture plates coated with Geltrex (Thermo Fisher Scientific).
The induction of AECs from hiPSCs was performed according to previously described methods4,5. Undifferentiated hiPSCs were seeded on Geltrex-coated plates in the medium containing RPMI1640 (Nacalai Tesque, Kyoto, Japan), 1×B27 supplement (Thermo Fisher Scientific), 50 U/ml penicillin/streptomycin, 100 ng/ml human activin A (R&D System, Minneapolis, MN, USA), 1 μM CHIR99021 (Axon Medchem, Groningen, the Netherlands), and 10 μM Y-27632 (days 0–1), 0.25 mM (day 1) or 0.125 mM (days 2–4) sodium butyrate (FUJIFILM Wako Pure Chemical Corporation, Osaka, Japan). From days 6 to 28, the basal medium containing Dulbecco’s Modified Eagle Medium (DMEM)/F-12 with GlutaMAX (Thermo Fisher Scientific), 1×B27 supplement, 50 U/ml penicillin/streptomycin, 0.05 mg/ml
Preparation of the Artificial Trachea
Artificial tracheae were prepared with the same materials that were used in clinical treatments. Collagen powder (Nippon Meat Packers, Inc., Osaka, Japan) was dissolved in water to a concentration of 6 mg/ml at pH 7. Then, the collagen solution was centrifuged, and the pellet was frozen at −80°C and lyophilized for 10 days. Small pieces of dried collagen were dissolved in water to a concentration of 106.4 mg/ml at pH 3. A polypropylene mesh coated with the collagen solution was placed on a mold with collagen solution. An equal volume of the collagen solution was poured on it and frozen at −80°C overnight. After 1 week of lyophilization, collagen including the polypropylene mesh was cross-linked by heating. This collagen including the polypropylene mesh was used as an artificial trachea.
Animals
Male F344/NJcl-rnu/rnu nude rats were used for the transplantation experiments, which were purchased from CLEA Japan (Osaka, Japan). The transplant recipients were 9–10 weeks old and weighed 160–200 g. The animal experimental protocol for this study was approved by the Animal Experimentation Committee of Kyoto University (Med Kyo 23113). All animals received humane care in compliance with the Guidelines for Animal Experiments of Kyoto University.
Transplantation
This study used a total of 12 nude rats. The transplantation experiments were performed under general anesthesia, with an intraperitoneal injection of a mixture of midazolam (2 mg/kg), butorphanol (2.5 mg/kg), and medetomidine (0.15 mg/kg). The trachea was exposed by a midline neck skin incision, and bilateral sternohyoid and sternothyroid muscles were separated. A square tracheal defect was created by incision with a scalpel. The defect size was the size of two tracheal rings approximately 2.5 mm in length and 2 mm in width. An artificial trachea covered with a cell sheet was put over the tracheal defect with the cells facing the tracheal lumen. At this time, a portion of the cell sheets was cut off and used as pre-transplantation samples. Cell sheets containing AECs derived from the 201B7 or 253G1 cell line were transplanted into six nude rats (B7-1–B7-6, G1-1–G1-6). All rats were euthanized with carbon dioxide 2 weeks post-transplantation, and tracheae-containing grafts were harvested and used as post-transplantation samples (Fig. 1).
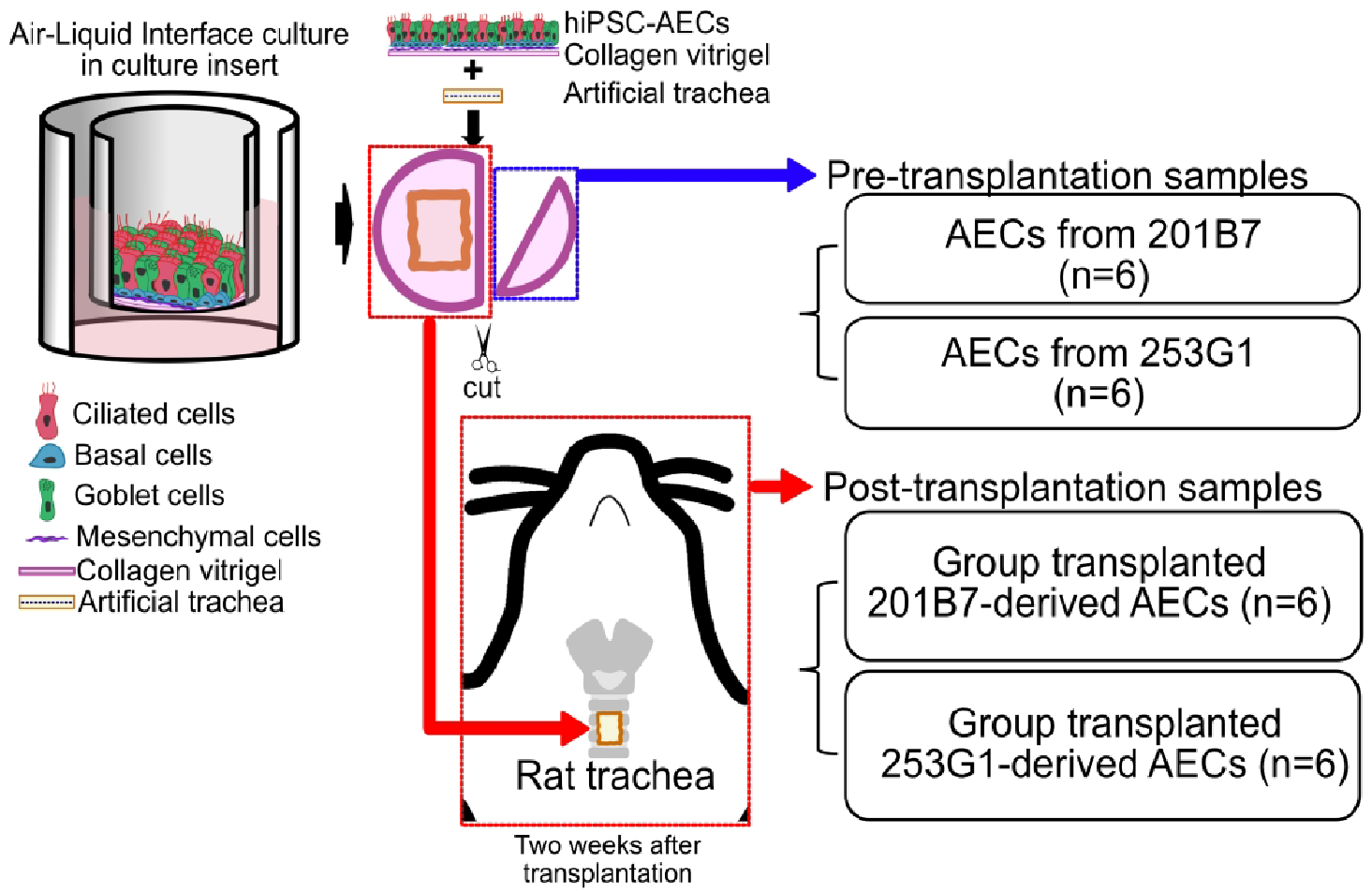
Scheme for the transplantation of hiPSC-AECs into nude rat tracheae and preparation of pre-transplantation and post-transplantation samples. An edge of the cell sheet containing hiPSC-AECs induced from 201B7 or 253G1 was cut for use as the pre-transplantation sample. Then, an artificial trachea was covered with a cell sheet and transplanted into a tracheal defect of a nude rat. Two weeks post-transplantation, rat tracheae including the grafts were collected as post-transplantation samples. Six samples in each group were prepared for subsequent studies. hiPSC-AECs: human-induced pluripotent stem cell-derived airway epithelial cells.
Immunofluorescent Staining
Pre-transplantation cell sheets and post-transplantation tracheae with the sternohyoid and sternothyroid muscles were immersed in 4% paraformaldehyde (PFA) for 24 h. Subsequently, they were immersed in 10%, 20%, and 30% sucrose for 24 h each and embedded in an optimal cutting temperature compound (Sakura Finetek Japan, Tokyo, Japan). The embedded samples were sliced at a thickness of 10 µm. Immunohistochemical analysis of 10–20 sections at 100-µm intervals of specimens from each rat was performed. For human nuclear antigen (HNA), Acetylated α-tubulin (Ac-Tub), MUCIN 5AC (MUC5AC), Vimentin, and Cytokeratin5 (KRT5), the sections were permeabilized with 0.2% Triton-X100/PBS for 5 min, treated with 1% bovine serum albumin/phosphate-buffered saline (BSA/PBS) for 10 min (for KRT5, 5% BSA/PBS for 30 min), and incubated with primary antibodies overnight at 4°C. After washing with PBS, sections were treated with Alexa Flour-conjugated secondary antibodies (Thermo Fisher Scientific), phalloidin (Santa Cruz Biotechnology, Dallas, TX, USA), and 4′,6-diamidino-2-phenylindole (DAPI) for 1 h at room temperature. For secretoglobin family 1A member 1 (SCGB1A1), sections were permeabilized with 0.2% Triton-X100 in PBS for 15 min and treated with 5% donkey serum in 1% BSA for 30 min at room temperature. After treatment of secondary antibodies, sections were washed with PBS and mounted using Fluoromount-G® Anti-Fade. Table 1 shows information on antibodies. All images were observed and analyzed using BZ-9000 fluorescence microscope and BZ-II Analyzer software (Keyence).
Antibodies Used in This Study.

SCGB1A1: secretoglobin family 1A member 1; MUC5AC: MUCIN 5AC.
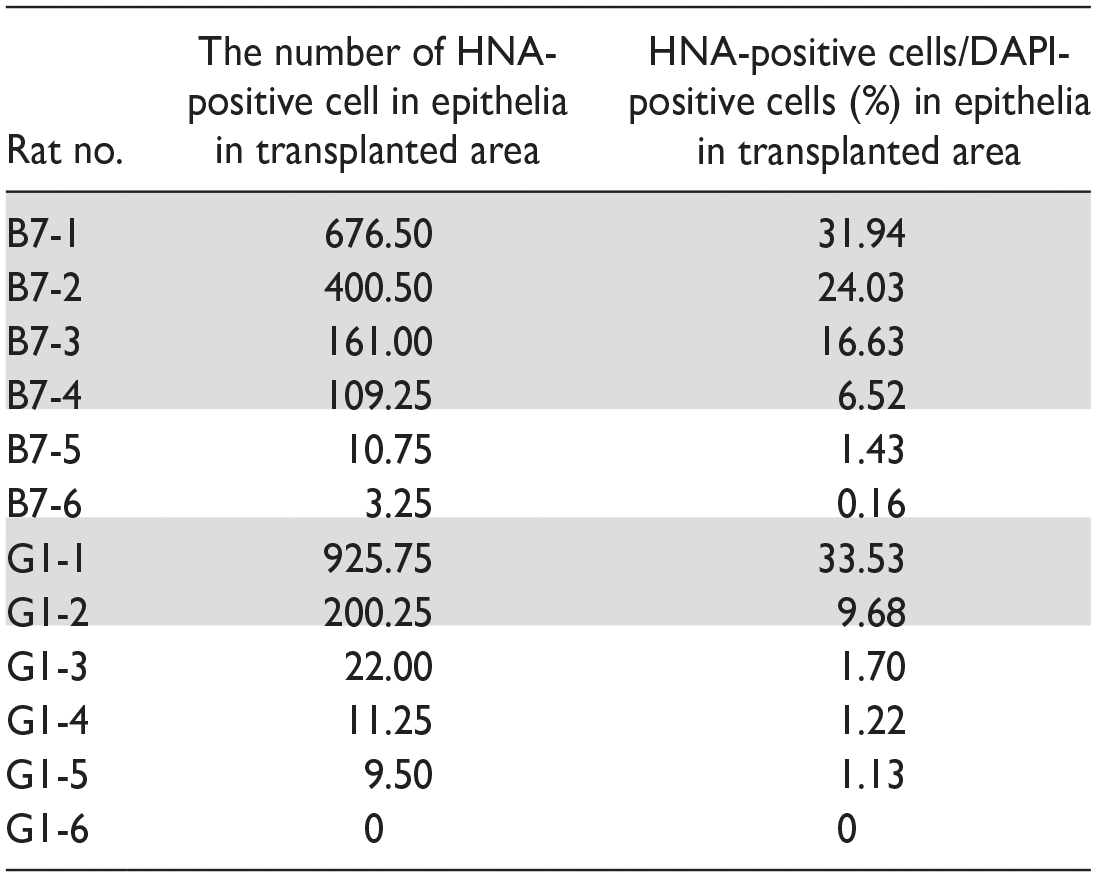
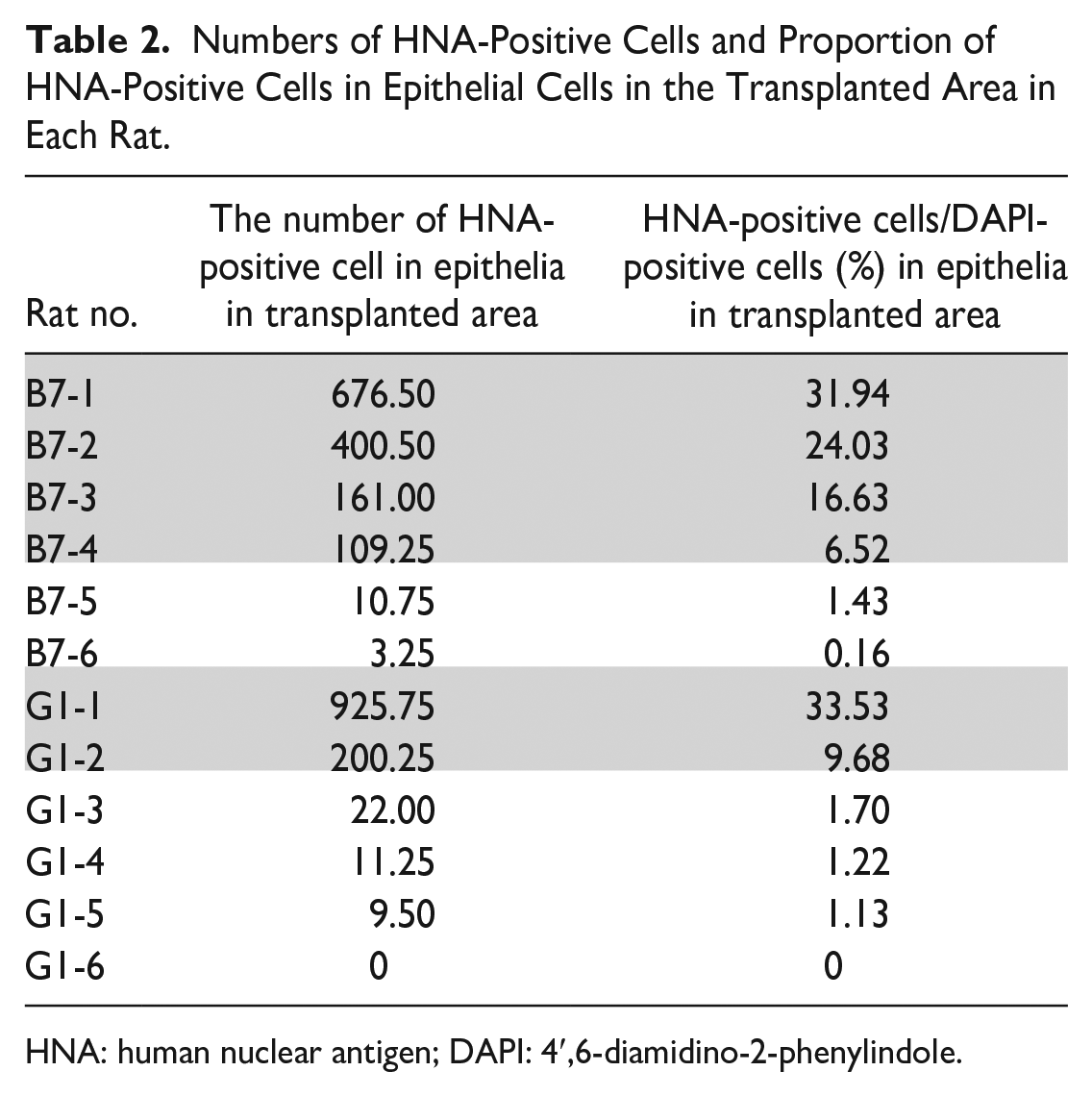
Quantification
To evaluate changes in the proportion of each cell type in hiPSC-AECs, immunofluorescent staining was performed using vertical sections of the pre-transplantation (Fig. 2A1, B1) and post-transplantation samples (Fig. 2A3, B3). Table 2 shows the number of HNA-positive cells and proportion of HNA-positive cells in total epithelial cells at the transplanted area (= survival rate) in each transplanted rat. The proportion of HNA-positive cells in total epithelial cells at the transplanted area was defined as the survival rate. Samples G1-6 were omitted following analyses due to the absence of engraftment. Ac-Tub-positive cells, KRT5-positive cells, SCGB1A1-positive cells, MUC5AC-positive cells, Vimentin-positive cells, and anti-HNA-positive cells were defined as ciliated, basal, club, goblet, mesenchymal, and hiPSC-derived cells, respectively. DAPI staining was used to define cells, and the cells of the luminal side on the phalloidin-positive basement lamina were defined as AECs13–15. The proportion of cells that express markers of each cell type and HNA was used as the proportion of each cell type in hiPSC-AECs post-transplantation. The number of surviving cells in the tracheal epithelium was counted in two areas with a width of 350 μm (side and center of the tracheostomy site). The Vimentin-positive area/1 μm of the cell sheet and the HNA-positive area/1 μm of the epithelia in the transplanted area were used to indicate the number of mesenchymal cells in the pre-transplantation (Fig. 2B1, B2) and post-transplantation (Fig. 2B3, B4) samples, respectively. Vimentin-positive or HNA-positive area in each sample was measured using ImageJ 16 .
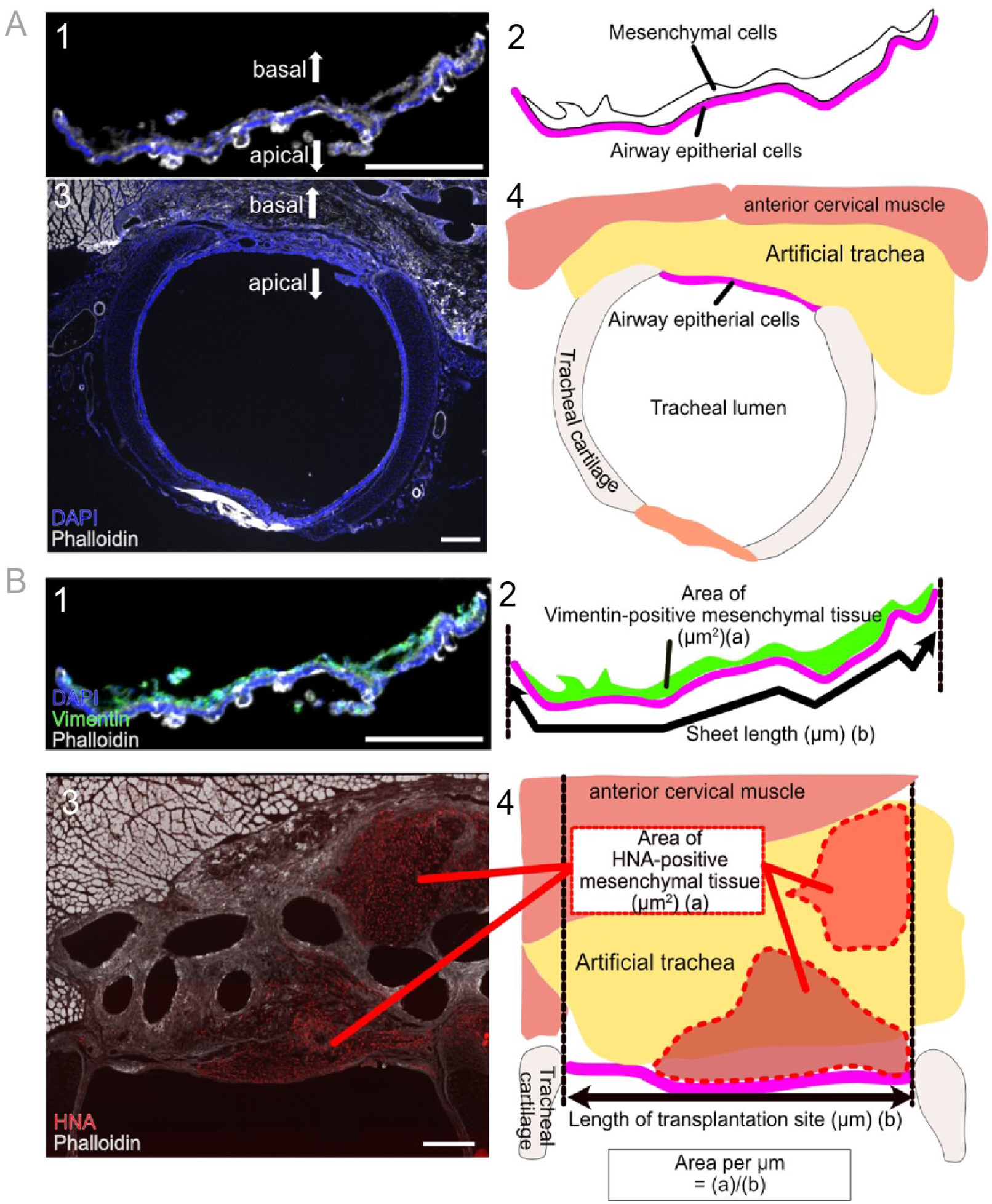
Vertical section of pre-transplantation and post-transplantation samples. (A) Vertical section of pre-transplantation and post-transplantation samples (1, 3) and the scheme for the vertical section to indicate the positional relationship of the pre-transplantation and post-transplantation samples (2, 4). (B) Vertical section of pre-transplantation and post-transplantation samples (1, 3) and the scheme for the vertical section to explain the measurement method for the mesenchymal cell area in the pre-transplantation and post-transplantation samples (2, 4). Vimentin (green) and human nuclear antigen (red) indicate mesenchymal cells in each sample (B1, 3). Phalloidin (white) and DAPI (blue) indicate the cell or tissue outline and nuclei respectively. Scale bars = 300 μm. DAPI: 4′A6-diamidino-2-phenylindole; HNA: human nuclear antigen.
Numbers of HNA-Positive Cells and Proportion of HNA-Positive Cells in Epithelial Cells in the Transplanted Area in Each Rat.
HNA: human nuclear antigen; DAPI: 4′,6-diamidino-2-phenylindole.
Statistical Analysis
Statistical analyses were performed using EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan), which is a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria) for Windows 17 . Data are expressed as median (range, minimum–maximum value). The Mann–Whitney U-test or paired-samples t test was used to compare the two groups. A significance level of 5% was adopted in all analyses.
Results
Characterization of hiPSC-AECs on Collagen Vitrigel Membrane From Two hiPSC Lines
In this study, two cell lines, namely, 201B7 and 253G1, were used. On day 56, the Ac-Tub-positive ciliated, KRT5-positive basal, and MUC5AC-positive goblet cells were found in the epithelial layer of the induced cell sheets (Fig. 3A1, A2, A4 and Supplemental Fig. S1A, B, D). On the contrary, no SCGB1A1-positive club cell was observed in all cell sheets induced from both cell lines (Fig. 3A3 and Supplemental Fig. S1C). Vimentin-positive mesenchymal cells were confirmed in the submucosal layer of the induced cell sheets, which indicate that the two hiPSC lines differentiated not only into AECs but also into mesenchymal cells (Fig. 3A5 and Supplemental Fig. S1E). These results indicate that AECs were induced from two hiPSC lines on commercially available collagen vitrigel membranes, as in previous studies.
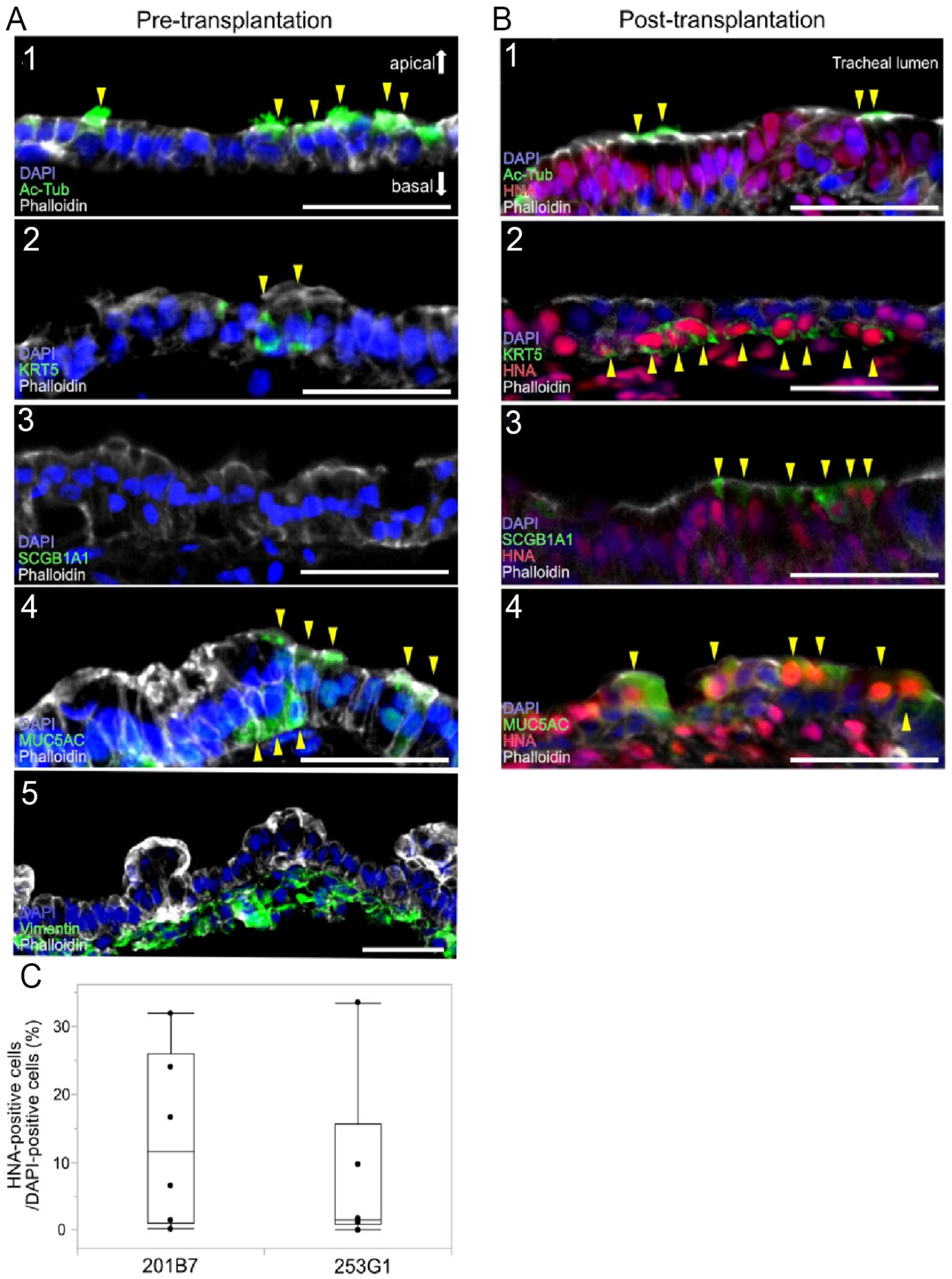
Immunofluorescence staining images of pre-transplantation and post-transplantation samples. Representative images of cross-sections of pre-transplantation (A) and post-transplantation (B) samples stained with antibodies for Ac-Tub (1), KRT5 (2), SCGB1A1 (3), MUC5AC (4), and Vimentin (5). Surviving transplanted cells were labeled with an antibody for HNA. Images of DAPI staining and phalloidin staining are also shown; scale bars = 50 μm. Yellow arrows indicate each cell type marker-positive cells. (C) Comparative analysis of the survival rates in the two cell lines. The Mann–Whitney U-test was used. Ac-Tub: Acetylated α-tubulin; KRT5: Cytokeratin5; SCGB1A1: secretoglobin family 1A member 1; MUC5AC: MUCIN 5AC; HNA: human nuclear antigen; DAPI: 4′,6-diamidino-2-phenylindole.
Confirmation of Engrafted hiPSC-AECs Transplanted Into Immunodeficient Rat Tracheae
Two weeks post-transplantation, transplanted areas were examined by immunofluorescent staining. HNA-positive hiPSC-AECs were confirmed in the luminal epithelial layer at the transplanted area of the post-transplantation samples. Furthermore, some of these HNA-positive cells express Ac-Tub, KRT5, MUC5AC, or SCGB1A1 (Fig. 3B1–B4). First, we examined the difference in the survival rates of the two cell lines. The median rates of HNA-positive cells in the total epithelial cells at the transplanted area was 11.58% (0.16%–31.94%) in 201B7-transplanted rats (n = 6) and 1.46% (0%–33.53%) in 253G1-transplanted rats (n = 6). In two cell lines, a significant difference was not found (Fig. 3C).
Examination of Changes in the Proportion of Component Cell Types in the Pre-Transplantation and Post-Transplantation Samples
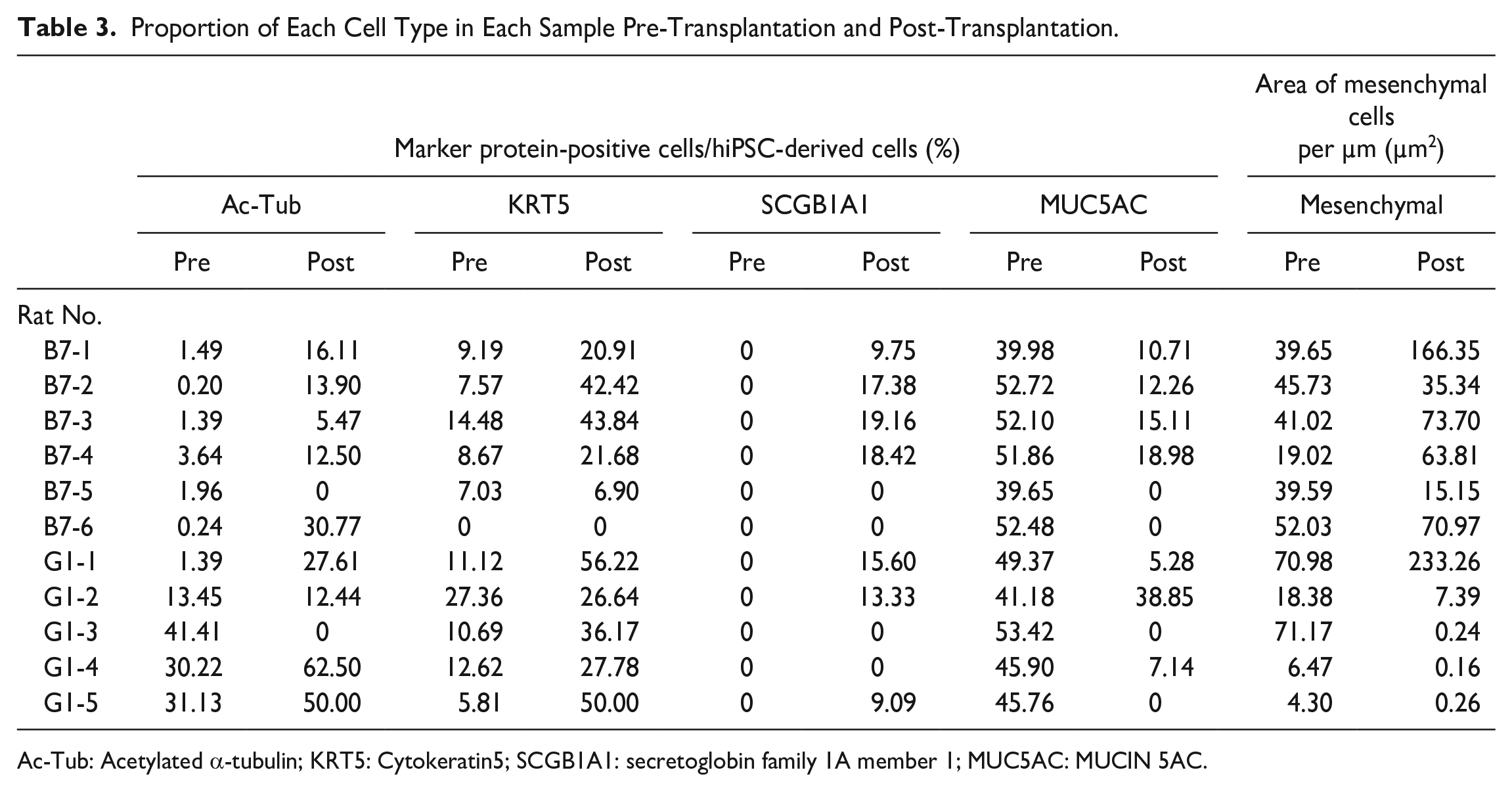
Changes in the proportion of each cell type in hiPSC-AECs pre-transplantation and post-transplantation are shown as line graphs (Fig. 4A and Supplemental Fig. S2). The proportion of each cell type in each pre-transplantation and post-transplantation sample is shown in Table 3. Club cells were not observed in the pre-transplantation samples but confirmed in the post-transplantation samples. Only the abundance of goblet cells decreased post-transplantation.
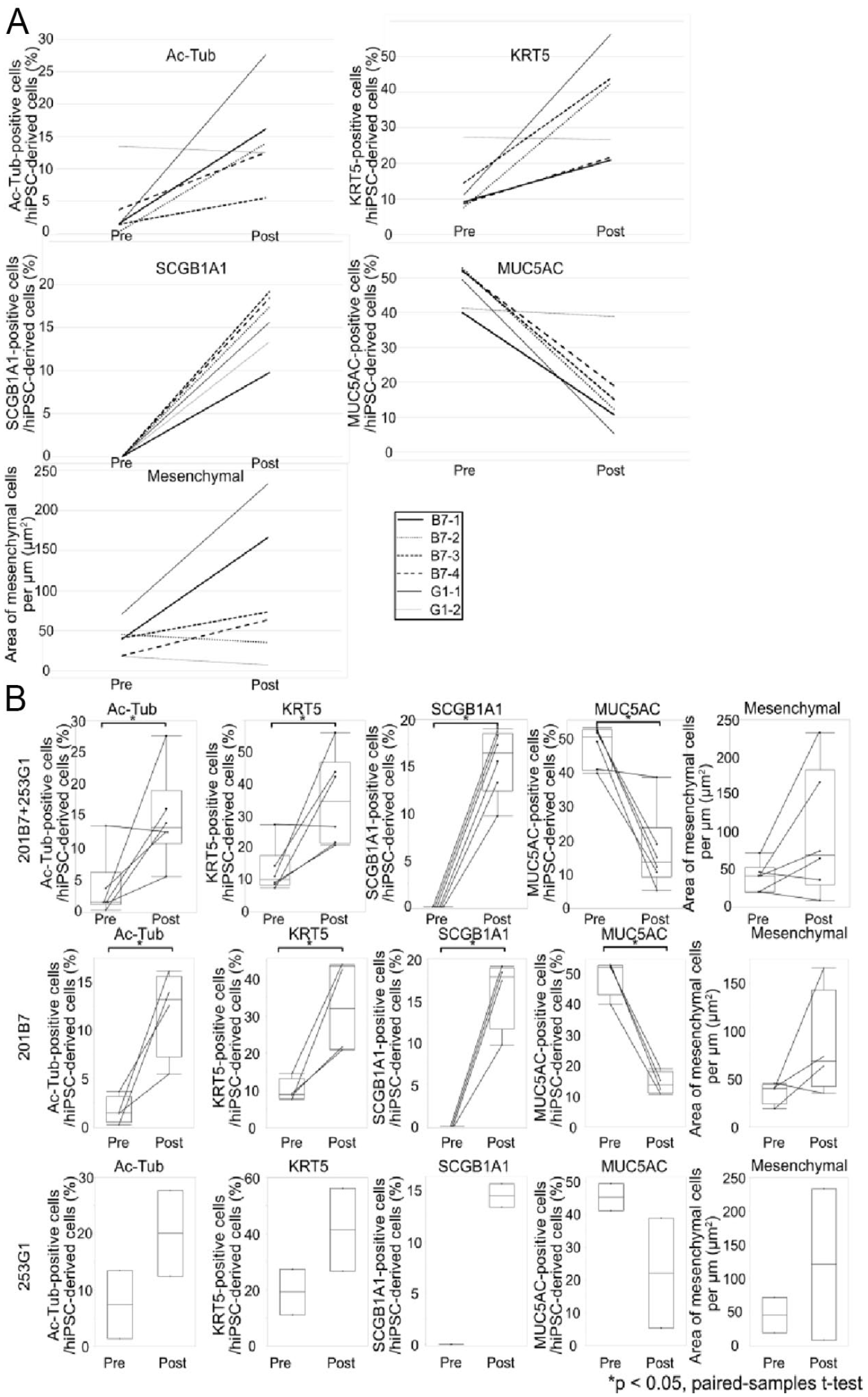
A comparison of the proportion of each cell type in hiPSC-derived cells pre-transplantation and post-transplantation using only samples showing high survival rates. (A) Line graphs show the proportion of each cell type in hiPSC-derived cells pre-transplantation and post-transplantation. (B) Comparative analyses of the proportion of each cell type in hiPSC-derived cells pre-transplantation and post-transplantation in both or each cell line. The paired-samples t test was used. (*P < 0.05). hiPSC: human-induced pluripotent stem cell; Ac-Tub: Acetylated α-tubulin; KRT5: Cytokeratin5; SCGB1A1: secretoglobin family 1A member 1; MUC5AC: MUCIN 5AC.
Proportion of Each Cell Type in Each Sample Pre-Transplantation and Post-Transplantation.
Ac-Tub: Acetylated α-tubulin; KRT5: Cytokeratin5; SCGB1A1: secretoglobin family 1A member 1; MUC5AC: MUCIN 5AC.
Furthermore, to eliminate errors caused by samples with low survival rates, only samples including over 100 hiPSC-AECs (survival rate > 6.5%, B7-1–B7-4, G1-1 and G1-2, samples highlighted in gray [Table 2]) were used for analyses (Fig. 4A). In the examination of the changes in the proportion of each cell type in hiPSC-AECs pre-transplantation and post-transplantation, the proportions of ciliated, basal, and club cells increased, the proportion of goblet cells decreased, and that of mesenchymal cells did not show a trend post-transplantation (Fig. 4A). Based on the data from two cell lines, changes in the proportion of each cell type in hiPSC-AECs were analyzed statistically (Fig. 4B). The median rates of ciliated, basal, club, and goblet cells in hiPSC-AECs pre-transplantation and post-transplantation were 1.44% (0.20%–13.45%) vs 13.20% (5.47%–27.61%) (P < 0.05 for all data), 10.16% (7.57%–27.36%) vs 34.53% (20.91%–56.22%), 0% (0%–0%) vs 16.49% (9.75%–19.16%), 50.61% (39.98%–52.72%) vs 13.69% (5.28%–38.85%). The median rates of mesenchymal cells in hiPSC-AECs pre–post-transplantation were 40.33 µm2 (18.38–70.98 µm2) vs 68.76 μm2 (7.39–233.26 µm2) (P = 0.11). In the data from the 201B7 lines, changes in the proportion of each cell type in hiPSC-AECs were also analyzed (Fig. 4B). The median rates of ciliated, basal, club, and goblet cells in hiPSC-AECs pre-transplantation and post-transplantation were 1.44% (0.20%–3.64%) vs 13.20% (5.47%–16.11%) (P < 0.05 for all data), 8.93% (7.57%–14.48%) vs 32.05% (20.91%–43.84%), 0% (0%–0%) vs 17.90% (9.75%–19.16%), and 51.98% (39.98%–52.72%) vs 13.69% (10.71%–18.98%). The median rates of mesenchymal cells in hiPSC-AECs pre-transplantation and post-transplantation were 40.33 µm2 (19.02–45.73 µm2) vs 68.76 μm2 (35.34–166.35 µm2) (P = 0.19). Although statistical analysis was not available, the 253G1 line showed a similar trend (Fig. 4B). The proportion of ciliated, basal, and club cells increased, the proportion of goblet cells decreased, and the proportion of mesenchymal cells showed no trends.
Relation of the Proportion of Each Cell Type in Engrafted hiPSC-AECs and the Efficiency of Engraftment of hiPSC-AECs
To examine whether the proportion of each cell type in engrafted hiPSC-AECs related with the efficiency of engraftment of hiPSC-AECs, all sample data were shown in scatter plots (Fig. 5). In samples with low survival rates, including less than 100 hiPSC-AECs, some Ac-Tub, or KRT5, high-positive rate samples were observed. In the graph of Ac-Tub or KRT5, 60% of the samples (3/5) show higher than median percentage of Ac-Tub-positive or KRT5-positive cells. In the graph of SCGB1A1 or MUC5AC, 80% of the samples (4/5) contained no SCGB1A1-positive or MUC5AC-positive cells. All samples containing more than 100 hiPSC-AECs contained all of Ac-Tub-positive, KRT5-positive, SCGB1A1-positive, and MUC5AC-positive cells.
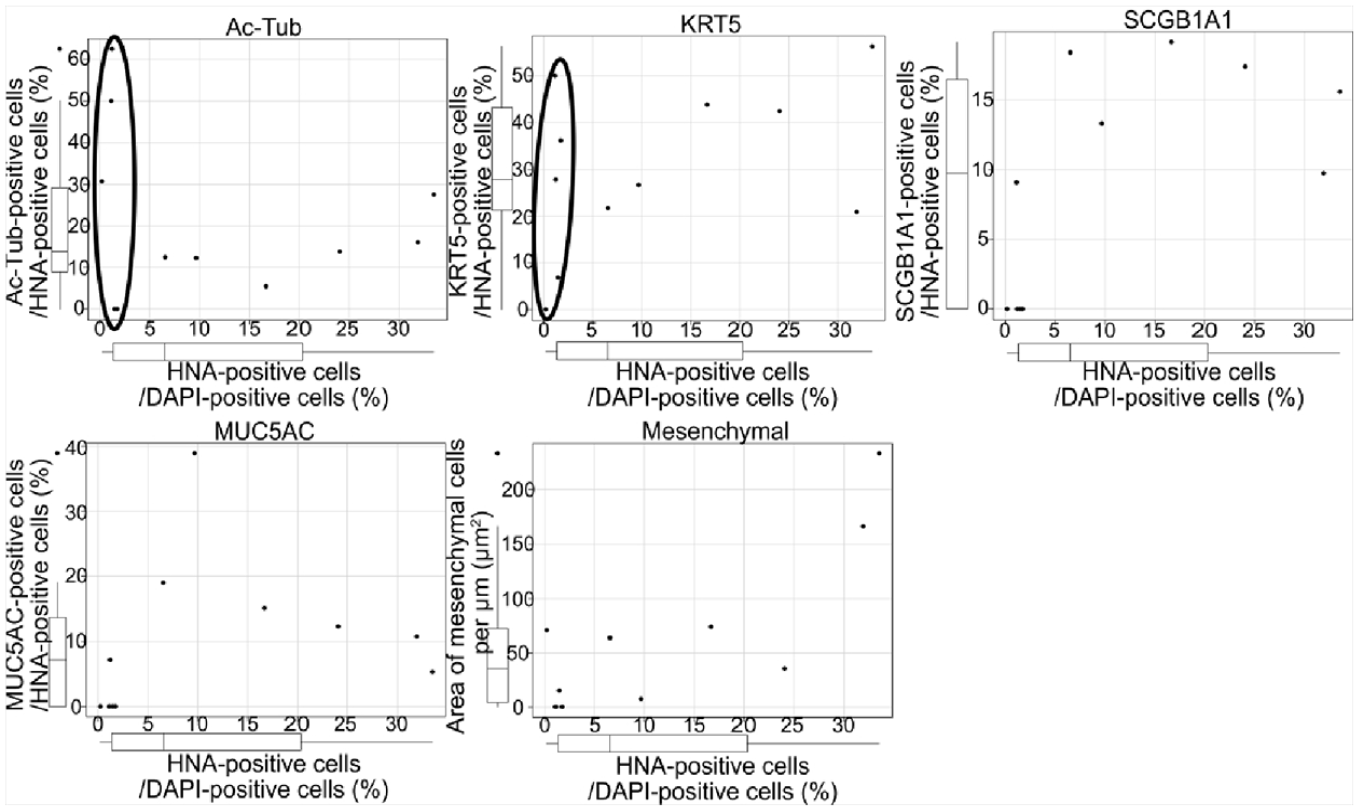
Scatter plots of the relation of the proportion of each cell type in HNA-positive cells with survival rate post-transplantation. Black circles indicate samples with low survival rates including less than 100 hiPSC-AECs in the plots for the examination of Ac-Tub and KRT5. HNA: human nuclear antigen; hiPSC-AECs: human-induced pluripotent stem cell-derived airway epithelial cells; Ac-Tub: Acetylated α-tubulin; KRT5: Cytokeratin5; DAPI: 4′,6-diamidino-2-phenylindole; MUC5AC: MUCIN 5AC.
Discussion
In this study, we compared the proportion of each cell type in hiPSC-AECs pre-transplantation and post-transplantation. We observed statistically significant differences in the proportion of four cell types that were positive for Ac-Tub, KRT5, SCGB1A1, and MUC5AC (Fig. 4B), indicating that the proportion of cell types constituting the hiPSC-AECs changes during pre-transplantation and post-transplantation. Therefore, it is difficult to consider the proportion of each cell type pre-transplantation as the proportion of each cell type post-transplantation. Thus, it is necessary to establish different criteria for selecting the cell sheet for transplantation or to develop the method for differentiation into a specific cell type. In addition, the proportion of Ac-Tub-positive, KRT5-positive, and SCGB1A1-positive cells tended to increase, whereas the proportion of MUC5AC-positive cells tended to decrease post-transplantation (Fig. 4B). Further studies with larger sample size will enable us to predict the proportion of each cell type post-transplantation from the proportion of each cell type pre-transplantation if the trend of these changes and the rate of change are always constant. The proportions of ciliated, basal, club, and goblet cells in hiPSC-AECs pre-transplantation were 1.44% (0.20%–13.45%), 10.16% (7.57%–27.36%), 0% (0%–0%), and 50.61% (39.98%–52.72%), respectively. On the contrary, the proportions of ciliated, basal, club, and goblet cells in engrafted hiPSC-AECs were 13.20% (5.47–27.61%), 34.53% (20.91–56.22%), 16.49% (9.75%–19.16%), and 13.69% (5.28%–38.85%), respectively. The proportions of ciliated, basal, and secretory cells (including club and goblet cells) in biopsy samples of human tracheal epithelia were 4.08% (0%–13.06%), 60.86% (17.69%–74.86%), and 4.19% (0%–12.32%), respectively 18 . The proportion of ciliated, basal, and club cells increased, whereas the proportion of goblet cells decreased, resulting in the proportion of each cell type in hiPSC-AECs closer to the proportion of each cell type in the biopsy tissue of human proximal AECs 18 . These results suggest that basal cells may show a differentiation direction post-transplantation different from pre-transplantation and that the transplanted hiPSC-AECs are subject to certain regulations, such as cell–cell interaction and/or paracrine by the recipient tissue 19 . The overgrowth of goblet cells causes impaired ejection function and leads to impaired airway defense, tumorigenesis of the transplanted tissue, tracheal obstruction, asthma, and other respiratory diseases 1 ; therefore, extreme changes in the proportion of each cell type post-transplantation could have risk. However, the direction of differentiation of hiPSC-AECs could be adjusted to the proportion of each cell type in the recipient’s tissue, and expectedly, the abnormal proliferation of specific cell types will be suppressed.
Because no club cells were observed in hiPSC-AECs pre-transplantation (Fig. 3A3), the proportion of club cells was considered to increase by differentiation from basal cells post-transplantation 20 . Differentiation into club cells from hiPSC-AECs post-transplantation may have been affected by the surrounding environment. SCGB1A1-producing club cells have anti-immune and anti-inflammatory effects due to the suppression of neutrophils, monocyte-mediated chemotaxis and phagocytosis, and suppression of interferon-γ, tumor necrosis factor-alpha, and interleukin (IL)-1 production21–23. In clinical studies, low circulating SCGB1A1 levels were not only found in asthma 24 ; however, they may be an independent actor to predict deficits in lung function and growth in childhood as well as for accelerated lung function decline and incident chronic obstructive pulmonary disease (COPD) in adult life 25 . Therefore, increasing the number of club cells may prevent these diseases.
Mesenchymal cells are already present prior to transplantation as shown in Fig. 3A5 and Supplemental Fig S1E. Mesenchymal cells are non-epithelial cells that are not objective cells for induction and must be removed to avoid tumorigenesis. After ventralized anterior foregut endoderm cell induction, NKX2.1-positive cells were purified by MACS using anti-CPM antibody5,26,27. Given that the efficiency of purification by MACS is not 100% 28 , non-objective cells remain in the cell sheet29,30. A method of removing undifferentiated hiPSCs was reported 31 , and the purity of transplanted cells must be increased using these methods. After the removal of residual hiPSC-derived mesenchymal cells in this manner, artificial tracheae-containing validated mesenchymal stem cells (MSCs) can be used as needed.
The relations of the proportion of hiPSC-AECs in the total epithelia in the transplanted area and the proportion of each cell type in engrafted hiPSC-AECs were examined. In samples with low survival rates of hiPSC-AECs, the proportions of Ac-Tub-positive and KRT5-positive cells in engrafted hiPSC-AECs largely varied among samples (Fig. 5, black circles). Because ciliated and basal cells were also observed in samples with low survival rates of hiPSC-AECs, these cells have a high ability to survive post-transplantation. As hiPSC-AECs transplanted with a high proportion of ciliated cells show high survival rates post-transplantation 32 , the area containing ciliated cells could preferentially remain in transplanted AECs. Basal cells have high resistance to drug-related injury compared with other cell types 33 . In primary culture, basal cells adhere to plastic dishes, proliferate, and differentiate into other cell types that constitute the airway epithelia 34 . These reports support the ability of basal cells to survive in the recipient tissue. The survival rate may be improved by increasing the proportion of ciliated and basal cells in hiPSC-AECs pre-transplantation.
In this study, we evaluated hiPSC-AECs pre-transplantation and post-transplantation by histological analyses, confirmed the change in the proportion of each cell type that constitutes the AECs pre-transplantation and post-transplantation, and obtained essential information for the realization of hiPSC-AEC transplantation in the future. In this study, we used the same markers used to identify cell types as in the work of Konishi et al 5 , who reported the induction method of hiPSC-AEC, and defined the cell types as ciliated, goblet, club, and basal cells. However, previous studies have reported that many hiPSC-derived cells show a degree of differentiation equivalent to that of the fetal stage in vitro35,36. In addition, as the airway epithelium differentiates from basal cells to ciliated cells or goblet cells via club cells, some cells express multiple differentiation markers; thus, more detailed studies using multiple markers are required in the future. Furthermore, this study performed only tissue evaluation. The mechanisms, such as intercellular signaling, have been unclear and must be elucidated in the future. In addition, since this study was performed 2 weeks post-transplantation and used only two hiPSC lines, further studies for long-term transplantation of AECs induced from various hiPSC lines using various induction methods and scaffolds are necessary.
In conclusion, we first demonstrated that the proportion of each cell type in hiPSC-AECs changes pre-transplantation and post-transplantation, and confirmed that the proportion of each cell type in hiPSCs post-transplantation is closer to the proportion of each cell type in human proximal AECs. This study provides important information for the development of therapeutic methods using hiPSC-AEC transplantation.
Supplemental Material
sj-docx-1-cll-10.1177_09636897241228026 – Supplemental material for Changes in the Proportion of Each Cell Type After hiPSC-Derived Airway Epithelia Transplantation
Supplemental material, sj-docx-1-cll-10.1177_09636897241228026 for Changes in the Proportion of Each Cell Type After hiPSC-Derived Airway Epithelia Transplantation by Masayuki Kitano, Yasuyuki Hayashi, Hiroe Ohnishi, Hideaki Okuyama, Masayoshi Yoshimatsu, Keisuke Mizuno, Fumihiko Kuwata, Takeshi Tada, Yo Kishimoto, Satoshi Morita and Koichi Omori in Cell Transplantation
Supplemental Material
sj-tiff-2-cll-10.1177_09636897241228026 – Supplemental material for Changes in the Proportion of Each Cell Type After hiPSC-Derived Airway Epithelia Transplantation
Supplemental material, sj-tiff-2-cll-10.1177_09636897241228026 for Changes in the Proportion of Each Cell Type After hiPSC-Derived Airway Epithelia Transplantation by Masayuki Kitano, Yasuyuki Hayashi, Hiroe Ohnishi, Hideaki Okuyama, Masayoshi Yoshimatsu, Keisuke Mizuno, Fumihiko Kuwata, Takeshi Tada, Yo Kishimoto, Satoshi Morita and Koichi Omori in Cell Transplantation
Supplemental Material
sj-tiff-3-cll-10.1177_09636897241228026 – Supplemental material for Changes in the Proportion of Each Cell Type After hiPSC-Derived Airway Epithelia Transplantation
Supplemental material, sj-tiff-3-cll-10.1177_09636897241228026 for Changes in the Proportion of Each Cell Type After hiPSC-Derived Airway Epithelia Transplantation by Masayuki Kitano, Yasuyuki Hayashi, Hiroe Ohnishi, Hideaki Okuyama, Masayoshi Yoshimatsu, Keisuke Mizuno, Fumihiko Kuwata, Takeshi Tada, Yo Kishimoto, Satoshi Morita and Koichi Omori in Cell Transplantation
Footnotes
Acknowledgements
The authors thank all the members of our laboratory (Department of Otolaryngology, Head and Neck Surgery, Faculty of Medicine, Kyoto University) for the helpful discussions.
Author Contributions
Masayuki Kitano: Data collection and assembly, data analysis and interpretation, manuscript writing, and final manuscript approval. Yasuyuki Hayashi: Data collection and assembly, data analysis and interpretation, manuscript writing, and final manuscript approval. Hiroe Ohnishi: Conception and design, data collection and assembly, data analysis and interpretation, manuscript writing, and final manuscript approval. Hideaki Okuyama: Data collection and assembly, data analysis and interpretation, and final manuscript approval. Masayoshi Yoshimatsu: Data collection and assembly, and final manuscript approval. Keisuke Mizuno: Data collection and assembly, and final manuscript approval. Fumihiko Kuwata: Data analysis and interpretation, and final manuscript approval. Takeshi Tada: Data analysis and interpretation, and final manuscript approval. Yo Kishimoto: Conception and design, administrative support and final manuscript approval. Satoshi Morita: Conception and design, data analysis, and final manuscript approval. Koichi Omori: Conception and design, administrative support, and final manuscript approval.
Availability of Data and Materials
The materials used in the current study are available from the research institutes or companies described in this article.
Ethical Approval
This study was approved by the Kyoto University Graduate School and Faculty of Medicine, Ethics Committee (Med Kyo 23113).
Statement of Human and Animal Rights
The animal experimental protocol for this study was approved by the Animal Experimentation Committee of Kyoto University. All animal experiments were performed following the “Guidelines for Animal Experiments of Kyoto University.” And the experiment using human cell line was approved by the Kyoto University Graduate School and Faculty of Medicine, Ethics Committee.
Statement of Informed Consent
Informed consent for patient information to be published in this article was not obtained, because we used only hiPSCs, which had already been established and widely distributed by cell banks.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI (22K09666) and the alumni Otolaryngology Fund from the Department of Otolaryngology—Head and Neck Surgery, Graduate School of Medicine, Kyoto University.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.