Abstract
The management of granulocytopenia-associated infections is challenging, and a high mortality rate is associated with traditional supportive therapies. Neutrophils—the primary defenders of the human immune system—have potent bactericidal capabilities. Here, we investigated the dynamic in vivo distribution of neutrophil transfusion and their impact on the treatment outcome of severe granulocytopenic infections. We transfused 89Zr-labeled neutrophils in the C57BL/6 mice and observed the dynamic neutrophil distribution in mice for 24 h using the micro-positron emission tomography (Micro-PET) technique. The labeled neutrophils were predominantly retained in the lungs and spleen up to 4 h after injection and then redistributed to other organs, such as the spleen, liver, and bone marrow. Neutrophil transfusion did not elicit marked inflammatory responses or organ damage in healthy host mice. Notably, allogeneic neutrophils showed rapid chemotaxis to the infected area of the host within 1 h. Tail vein infusion of approximately 107 neutrophils substantially bolstered host immunity, ameliorated the inflammatory state, and increased survival rates in neutrophil-depleted and infected mice. Overall, massive allogeneic neutrophil transfusion had a therapeutic effect in severe infections and can have extensive applications in the future.
Introduction
Neutrophils—the predominant white blood cells in the circulation of healthy adults—are among the first responders of the innate immune system to infections. However, granulocytopenia is an inevitable side effect of cancer chemotherapy and a common but serious complication of aplastic anemia 1 . The risk of severe infections markedly increases when peripheral blood counts fall below 0.5 × 109/l 2 , and life-threatening bacterial and fungal infections in granulocytopenic patients result in organ damage and high mortality rates 3 . However, the treatment of neutropenia is a clinically challenging issue 4 . Although the outcomes of supportive granulocytopenia treatment have significantly improved with the use of granulocyte colony-stimulating factor (G-CSF) and antibiotics5,6, neutropenia remains a leading cause of morbidity and mortality in patients with hematological malignancies with mortality rates reaching up to 10%7–10. Moreover, impaired neutrophil function may increase the risk of infections even with normal neutrophil counts 11 . Therefore, novel therapeutic strategies are essential to address these challenges and improve outcomes in granulocytopenia-associated infections.
The efficacy of granulocyte transfusion therapy and higher transplant doses are directly correlated, and a minimum of 1010 granulocytes infusion are required in adults12–14. The use of drugs, such as G-CSF and corticosteroids has increased the focus on granulocyte infusion therapy. These drugs are effective in stimulating granulocytosis in healthy donors and facilitate the collection of a large number of clinically useful neutrophils5,15. Nevertheless, a limited laboratory evidence is available regarding the functional integrity of granulocytes produced through G-CSF stimulation8,16, and the exact efficacy of healthy donor-derived granulocytes in preventing and treating severe life-threatening infections remains uncertain7,17,18. Therefore, comprehensive research is required on granulocyte distribution in the host, the safety of granulocyte transplants, and the efficacy of post-transfusion therapy. Here, we conducted animal experiments to validate the intact mature neutrophil function of granulocytes derived from the bone marrow of the donor mice. In addition, we ascertained the safety of recipients after granulocyte transfusion and demonstrated a significant enhancement in the survival rate of granulocytopenic and infected mice after the therapy.
Materials and Methods
Ethics and Mouse Model
This study was approved by the Medical Ethical Committee of Suzhou Municipal Hospital (animal ethical approval No. KL901390). All experiments followed approved guidelines. Male wild-type BALB/c (C) mice aged 8–10 weeks were utilized. They were housed in a pathogen-free environment with ad libitum access to food and water. To establish the murine infection model, neutrophils were depleted by intraperitoneal injection of InVivoMAb anti-mouse Ly6G/Ly6C (Gr-1) (Bioxcell, USA) 24 h prior. Fecal samples from the same group of mice were collected and mixed with sterile saline to create a 3% fecal slurry. After centrifugation at 800 × g for 3 min, the supernatant was collected and administered intraperitoneally at 1.5 ml/100 g. The cecal ligation and puncture (CLP) model was established following the standard guidelines provided by Daniel Rittirsch et al. The cecal end was ligated by approximately 0.5 cm and punctured with a 21 G needle, followed by closure. Subcutaneous administration of 500 μl pre-warmed sterile saline was given for fluid resuscitation after suturing.
Mouse Bone Marrow Neutrophils Isolation
Isolation of mouse bone marrow neutrophils, briefly, isolate femurs and tibias from mice, flushing with Hank’s balanced salt solution (HBSS; calcium and magnesium-free, Gibco, USA) containing 2 mM ethylenediaminetetraacetic acid (EDTA; Thermo, USA) and 10% FBS (Gibco, USA), passing through a 70 μm cell strainer for filtration and debris removal, and then washing to remove impurities. The collected bone marrow cells are pelleted at 4°C, 500 × g for 7 min. Red blood cells are lysed in distilled water for 15 s. Neutrophils are enriched using the EasySepTM Mouse Neutrophil Enrichment Kit (STEMCELL, USA).
Under Agarose Neutrophil Chemotaxis Model
The under agarose neutrophil chemotaxis model, briefly, a 1.2% agarose solution was boiled and mixed 1:3 with a heated medium containing 50% HBSS with Ca2+ and Mg2+ and 50% RPMI (Roswell Park Memorial Institute) 1640 medium (20% heat inactivated fetal bovine serum [FBS]). Add 2.7 ml of the above solution into a 35 mm culture dish and cooled gently. After the agarose is cooled and solidified, three wells with a diameter of 3.5 mm and a spacing of 2.8 mm are cut into a straight line in the gel. The mesopores are filled with 10 μl chemoattractants (Trp–Lys–Tyr–Met–Val–Met–NH2, WKYMVm, 0.1 μM) (Abcam, UK), and the side wells are filled with neutrophils (10 μl, 107/ml). The gel was incubated in a 37°C/5% CO2 incubator for 2 h. Observe the chemotaxis distance with a microscope.
Flow Cytometry
To assess the post-activation surface molecule expression of neutrophils, cells were co-incubated on ice with 10 nM phorbol myristate acetate (PMA; Solarbio, China) and fluorescent antibodies for 30 min. For intracellular reactive oxygen species (ROS) detection in neutrophils, 10 μM 2′,7′-dichlorodihydrofluorescein diacetate (H2DCFDA; Solarbio, China) was utilized. CD63 (BD, USA) was employed to assess neutrophil degranulation, while SYTOX (Thermo, USA) was used to detect neutrophil extracellular trap (NETs) and nuclear morphology. Prior to evaluating the expression of the high-affinity neutrophil adhesion molecules CD11b (BD, USA) and ICAM1 (BD, USA), neutrophils at concentrations of 3 × 107/ml and 5 × 106/ml were co-cultured with 2 × 105 mouse pulmonary arterial endothelial cells at 37°C/5% CO2 for 2 h. Samples were analyzed using a FACSCanto II flow cytometer (BD Biosciences, USA) and data were processed with FlowJo V10.
Laser Scanning Confocal Microscopy
Phagocytosis Assay (Red E. coli, Abcam, UK) was employed to assess the phagocytic capability of bone marrow neutrophils. Neutrophils were co-cultured with red Escherichia coli at 37°C/5% CO2 for 2 h prior to nuclear staining with DAPI (4',6-diamidino-2-phenylindole; Solarbio, China). For CD11b and ICAM1 receptor staining, cells were fixed with 2% paraformaldehyde for 10 min at room temperature (RT). Permeabilization was performed using 0.01% Triton X-100 (Sigma Aldrich, USA). During CD11b (Abcam, UK) or ICAM1 (Abcam, UK) staining, cells were blocked with 5% goat serum (Solarbio, China) for 1 h. Cell nuclei were stained using DAPI. To examine the migration of allogeneic neutrophils within omental microvasculature, neutrophils were labeled ex vivo using CellTraceTM (Thermo, USA) and subsequently intravenously infused via the tail vein. A 1 × 1.5 cm skin flap was dissected from the abdominal wall after tail vein injection. Neutrophils were visualized using the LSM900 microscope (ZEISS, Germany).
Enzyme-Linked Immunosorbent Assay
EDTA-anticoagulated blood was centrifuged at 3,000 × g for 10 min at RT, and the top two thirds of the plasma was carefully removed and stored at −80°C. The experimental steps were carried out according to the reagent instructions (Abcam, UK). Gradient standard and samples at a concentration of 100 µl were added to the 96-well plate and incubated at RT. Three wells were used for each sample. Biotinylated antibody, streptavidin–horseradish peroxidase (HRP) reagent and 3,3′,5,5′-tetramethylbenzidine (TMB) substrate were added, respectively, to the wells following the procedures. The plate was developed at RT in the dark until there was an obvious difference in color, and then stop solution was added to each well. The absorbance of the enzyme was measured at 450 nm within 5 min, and the data were recorded. Constructing a standard curve, and the corresponding concentration value of each sample was calculated.
Neutrophil Radiolabeling
Oxine (Solarbio, China) was prepared as a 5 μg/μl solution in dimethyl sulfoxide (DMSO). 89Zr oxalate solution was adjusted to a neutral pH using HEPES (4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid) buffer and sodium carbonate, followed by a 15-min RT reaction with oxine. The prepared 89Zr-oxine was used to label neutrophils (25 μCi/106) in physiological saline at 37°C for 15 min, resulting in an average labeling efficiency of 38.9%.
Luminex Multiplex Cytokine Analysis
Following the instructions of the Mouse Cytokine 23-plex Assay kit (Bio-RAD, USA), 50 μl of beads, standards, controls, and samples were sequentially added and incubated at RT at 800 rpm for 1 h. Post-wash, 50 μl of detection antibodies were added and incubated at RT at 800 rpm for 1 h. Following another wash, 50 μl of phycoerythrin (PE)-conjugated streptavidin was added and incubated at RT at 800 rpm for 30 min. After a final wash, 100–150 μl of sheath fluid was added and incubated at RT at 800 rpm for 0.5–2 min. The Luminex X-200 system was then employed for analysis.
Peritoneal Lavage Bacterial Culturing
After CO2 anesthesia, mice were euthanized through cervical dislocation, followed by thorough spraying with 70% alcohol. Sterile scissors and forceps were used to make a small incision below the sternum, and the fur was gently peeled downward avoid piercing the peritoneum. Using a 10 ml syringe with a 19 G needle, a mixture of air and cold sterile phosphate-buffered saline (PBS) (1:1) was injected into the mouse’s peritoneal cavity. After massaging the mouse’s sides for 30 s, the PBS-containing bacteria was collected into a centrifuge tube using the syringe. The peritoneal cavity was opened with sterile scissors and forceps, and the remaining PBS was collected using a sterile pipette. The supernatant was collected after centrifugation at 1,000 × g, diluted 1:1,000, and then plated on Lysogeny broth (LB) agar plates for 12 h (Sangon Biotech, China).
Statistical Analysis
All statistical analysis and graphs were performed with the GraphPad Prism 8.0 software. The Shapiro–Wilk test was used to test the normality of continuous variables. Results were expressed as the mean ± standard deviation (SD). For group comparisons, one-way analysis of variance (ANOVA) was used for continuous variables with a normal distribution followed by Tukey’s post-hoc test or Dunn’s post-hoc test for multiple comparisons. Student’s t test and Wilcoxon matched-pairs signed rank test was applied to compare the differences between two groups. Statistical significance was set at P value < 0.05. The P values were represented as follows: NS, none-significant, ****P < 0.0001, ***P < 0.001, **P < 0.01, and *P < 0.05.
Results
Mouse Bone Marrow-Derived Donor Neutrophils Show Complete Mature Neutrophil Functionality
Approximately, 50% of the bone marrow cells in mice are neutrophils, which is a sufficient supply for transfusion studies. However, the proportion of mature neutrophils among the total neutrophils population should be ascertained, and the functional integrity of bone marrow-derived neutrophils should be verified. Therefore, we analyzed the functions of these bone marrow-derived neutrophils.
Flow cytometry with multicolor staining revealed that mature neutrophils (Ly6G+ CXCR2+) accounted for 66.7% of the total cells in the bone marrow after erythrocyte removal. This percentage was 85% in the peripheral blood (Fig. 1A). Utilizing magnetic beads for the extraction of bone marrow neutrophils enables the isolation of neutrophils with a purity of 90%. Post-purification, nuclear staining of the neutrophils with SYTOX revealed a substantial population of mature neutrophils, characterized by lobulated nuclei (Fig. 1B). The functions of bone marrow-derived and peripheral blood-derived neutrophils are not significantly different. These functions include ROS production (Fig. 1C), degranulation (Fig. 1D), phagocytosis (Fig. 1E), NETs formation (Fig. 1F), and chemotaxis (Fig. 1G). These findings highlighted the intact neutrophil functionality of bone marrow-derived mature neutrophils, making them suitable for transfusion therapy.
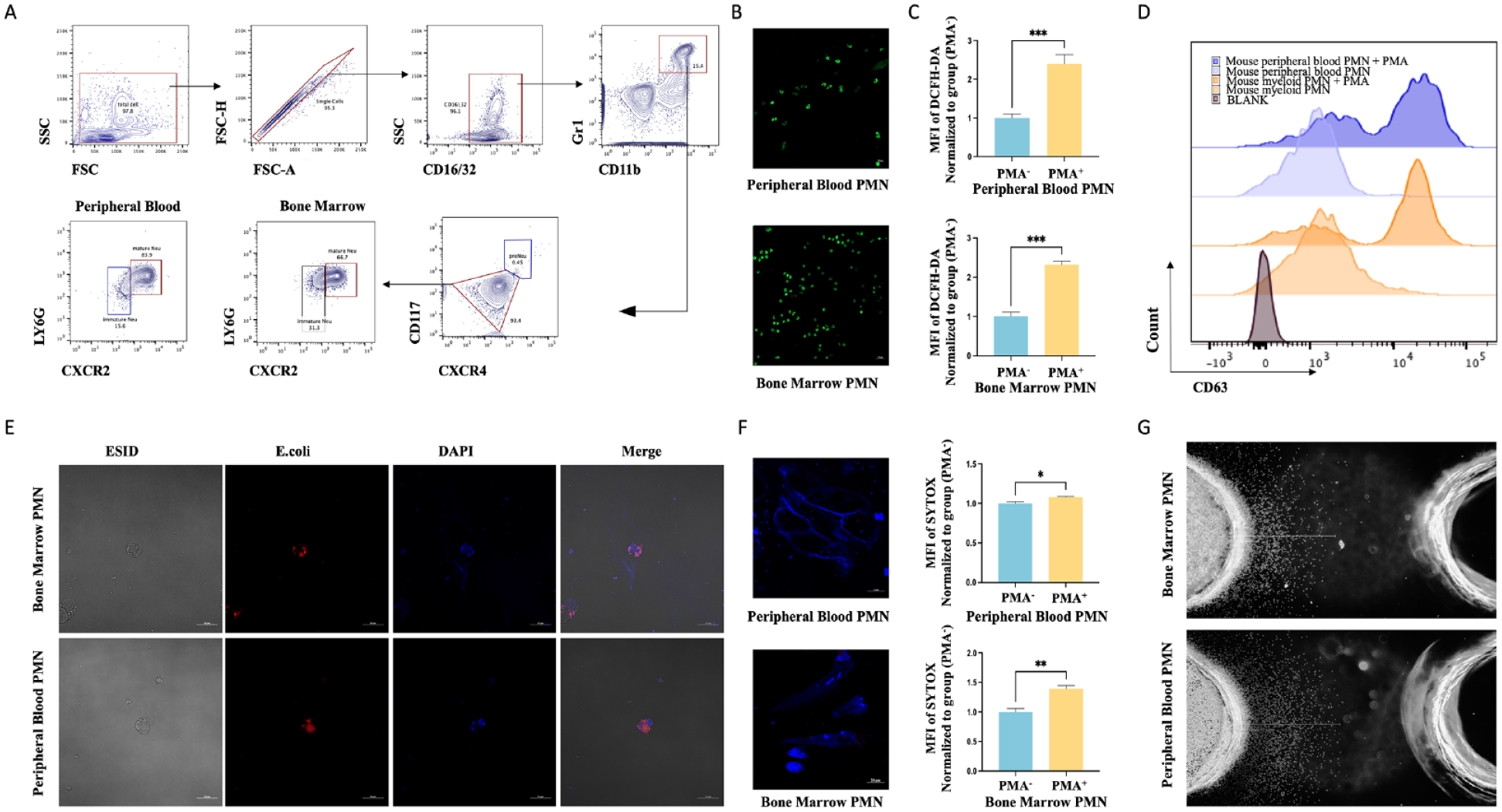
Mouse bone marrow-derived donor neutrophils show complete mature neutrophil functionality. (A) Flow polychrome staining analysis of bone marrow and peripheral blood cells after erythrocyte removal to calculate the proportion of mature neutrophils expressing CXCR2 and LY6G. (B) Neutrophils derive from bone marrow and peripheral blood are purified using the magnetic bead method, and their karyotypes are observed using confocal microscopy after SYTOX staining. (C) Flow cytometry with DCFH-DA staining results show ROS levels after neutrophil stimulation with PMA at 37°C for 30 min. (D) Flow cytometry was used to detect CD63 expression in neutrophils following PMA stimulation (and no stimulation). (E) Confocal microscopy images show phagocytosis after the co-incubation of neutrophils and fluorescent E. coli analogues at 37°C for 2 h. (F) Neutrophils were stimulated with PMA at 37°C for 4 h. The fluorescent expression was detected using flow cytometry after SYTOX staining, and the morphology of neutrophil extracellular traps was visualized using confocal microscopy. (G) Chemotaxis ability of neutrophils after incubating in the agarose chemotaxis model at 37°C for 4 h. FSC: Forward Scattering; SSC: Side Scattering; PMA: phorbol myristate acetate; PMN: polymorphonuclear neutrophil; ROS: reactive oxygen species.
Absence of Apparent Tissue or Organ Damage in Host Mice After Allogeneic Neutrophil Transfusion
The concentrations of neutrophil activation-related proteins (Fig. 2A), and liver and kidney function proteins were not significantly different between the transfusion and control groups (Fig. 2B). There was no significant increase in mortality observed in healthy mice following neutrophil transfusion (Fig. 2C). In addition, tissue histopathological analysis indicated the absence of tissue interstitial edema, cell necrosis, or structural disruption in the vital organs (Fig. 2D). We collected plasma samples within 72 h post-infusion and measured inflammatory factors to evaluate the potential adverse effects of neutrophil infusion in healthy mice. The concentrations of pro-inflammatory factors, inhibitory inflammatory factors, and specific chemokines were similar to their normal concentrations (Fig. 2E). Moreover, no apparent congestion, edema, or color changes linked to inflammatory cell infiltration were observed in the crucial organs, such as the heart, liver, spleen, lungs, and kidneys (Fig. 2F). Therefore, allogeneic neutrophil transfusion was safe in this study because it did not cause any functional damage to the tissues and organs in the host mice.
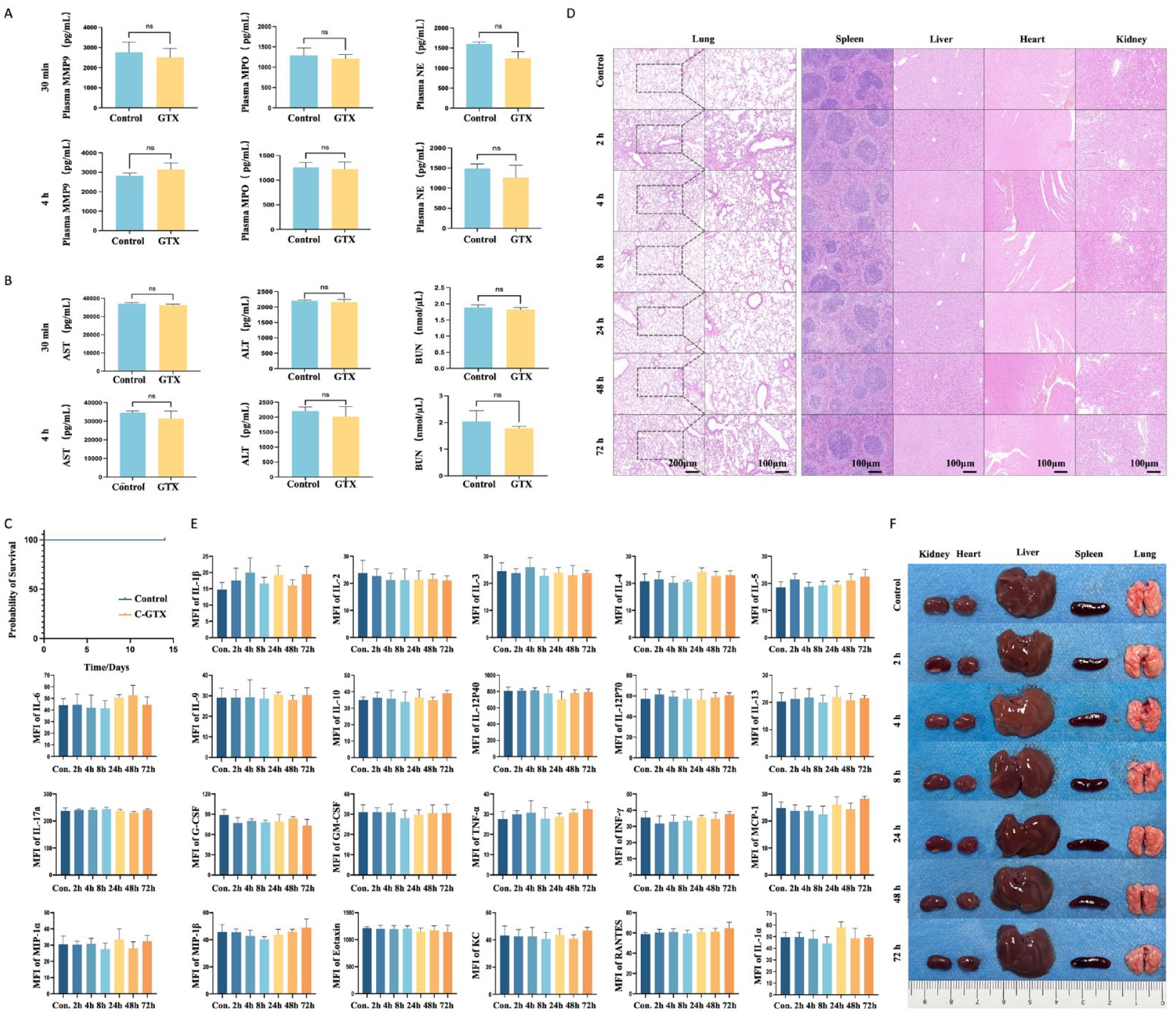
Absence of apparent tissue or organ damage in host mice after allogeneic neutrophil transfusion (A) ELISA results show plasma MPO, NE, and MMP9 concentrations in recipient mice. (P > 0.05 for all groups.) (B) ELISA results show plasma AST, ALT, and BUN concentrations in recipient mice. (P > 0.05 for all groups.) (C) Survival rate analysis after neutrophil transfusion in healthy mice. (D) HE staining results show histologic features of major organs in neutrophil allograft recipient mice. (E) Results of Luminex multifactor assay showing the concentrations of plasma inflammatory factors in recipient mice. (P > 0.05 for all groups.) (F) Images of major organs in neutrophil allograft host mice. MFI: mean fluoresence intensity; NE: Neutrophil Elastase; MPO: Myeloper-oxidase; GTX: granulocyte transfusion; ELISA: enzyme-linked immunosorbent assay; AST: aspartate aminotransferase; ALT: alanine aminotransferase; BUN: blood urea nitrogen; HE: hematoxylin and eosin.
Dynamic In Vivo Distribution of Infused Neutrophils and the Mechanisms Involved
Neutrophils were labeled with the radionuclide 89Zr, and their distribution in the host mice was monitored at various time points (Fig. 3A). The labeled neutrophils showed higher distribution density in the spleen and were transiently retained in the lungs before gradually dispersing to organs (such as the liver and bones) 4 h after transfusion (Fig. 3B, C). Confocal microscopy allowed us to observe the circulation of fluorescently labeled neutrophils in the microveins on the body surface of the host mice. We found that the number of neutrophils circulating in the peripheral veins increased gradually in the first 3 h after infusion (Fig. 3S1, Supplemental videos 1, 2) and then gradually declined at 8 h (Fig. 3S1, Supplemental video 3). After receiving neutrophil transfusion, the neutrophil absolute count in the peripheral blood of recipient mice increased at 3 h (Fig. S4A). Notably, 89Zr has a half-life of 78 h, and its radioactivity remained stable during the entire monitoring period. The retention curve of labeled neutrophils for 89Zr (Fig. 3D) and the 24-h apoptosis curve of neutrophils were identical, suggesting that 89Zr binding did not significantly affect the stability and activity of neutrophils in vitro (Fig. 3E).
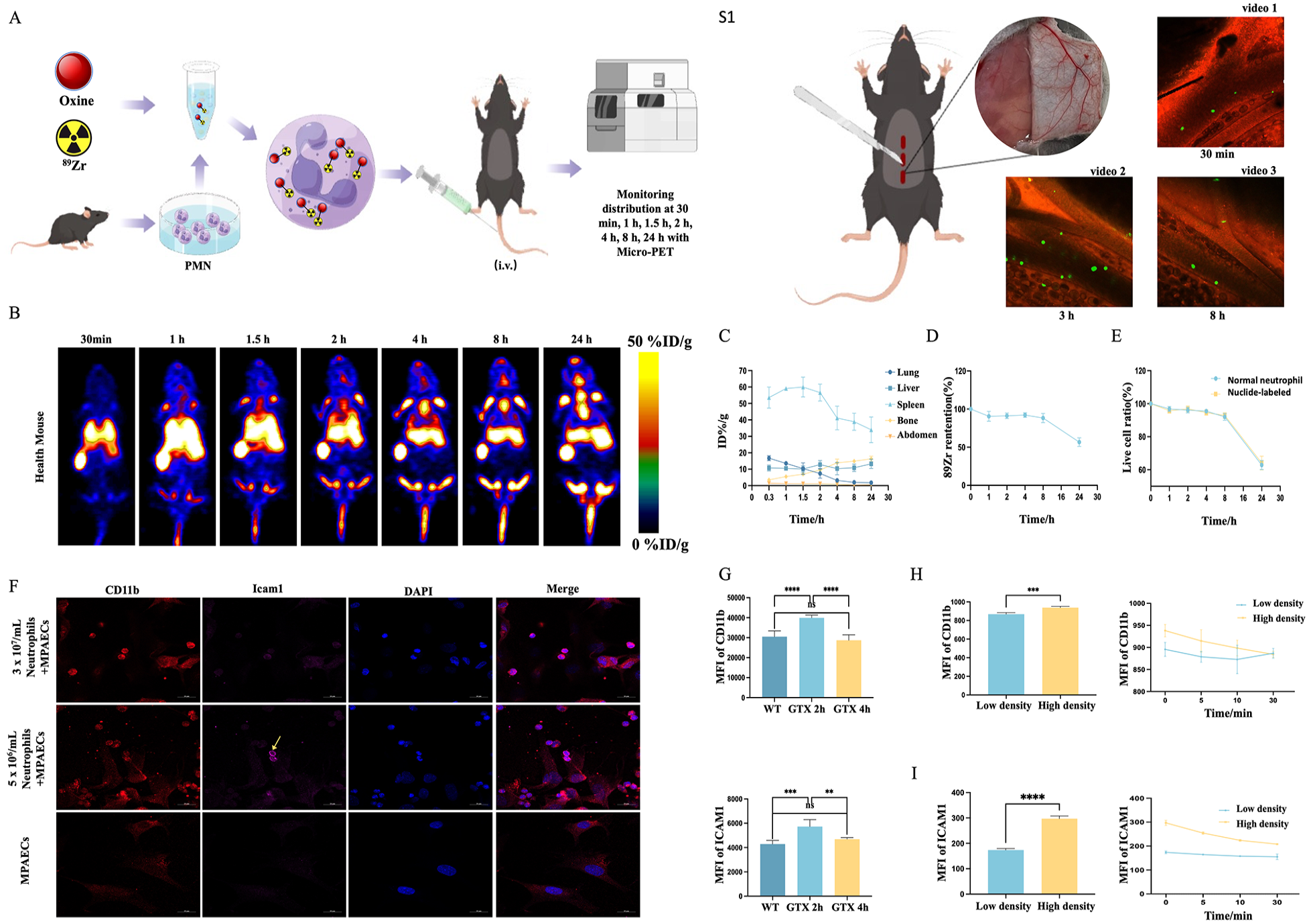
Dynamic in vivo distribution of transplanted neutrophils and the mechanisms involved. (A) Illustration of the in vivo distribution experiment of 89Zr-oxine labeled neutrophils. (B) Micro-PET monitoring of the dynamic distribution of 89Zr-labeled neutrophils in healthy mice. (C) Percentage injected dose rate per gram of tissue (%ID/g) in the abdominal region and lung, liver, spleen, and bone tissues of the host mice within 24 h. (D) Twenty-four hour of radioactivity retention rate of neutrophils after 89Zr labeling. (E) Twenty-four-hour in vitro survival rate of neutrophils after 89Zr labeling and the 24-h apoptosis rate of normal neutrophils. (F) Confocal microscopy images show the expression of ICAM1 and CD11b after co-culturing neutrophils (3 × 107/ml and 5 × 106/ml) with pulmonary arterial endothelial cells (105/mL) at 37°C for 2 h. (G) Expression of CD11b and ICAM1 on the surface of neutrophils in the lungs of host mice at 2 and 4 h after neutrophil transfusion. (H, I) Flow cytometry-based detection of ICAM1 and CD11b expression after culturing only neutrophils (3 × 107/ml and 5 × 106/ml) for 2 h. WT: wild type; %ID/g: percentage activity of injection dose per gram of tissue; MPAEC: mouse pulmonary arterial endothelial cell; PMN: polymorphnuclear neutrophil.
Circulating neutrophils can adhere with endothelial cells or with each other by upregulating the expression of CD11b and ICAM1. We examined the expression of CD11b and ICAM1 on the surface of lung neutrophils in the host mice at 2 and 4 h after infusion to investigate the mechanism underlying neutrophil accumulation in lungs. The expression levels of CD11b and ICAM1 were upregulated in the 2-h group, and the expression levels in the 4-h group were similar to those observed in the lung neutrophils of healthy mice (Fig. 3G).
Next, we co-cultured neutrophils (3 × 107/ml [low concentration] and 5 × 106/ml [high concentration]) and pulmonary arterial endothelial cells for 2 h to determine whether the upregulation of surface adhesion molecules resulted from interactions of neutrophils with pulmonary endothelial cells or among themselves. In the high-concentration neutrophil group, the CD11b expression was upregulated on both neutrophils and pulmonary endothelial cells, whereas high expression of ICAM1 was observed only on neutrophils (Fig. 3F). When only neutrophils were incubated for 2 h at these concentrations, both CD11b and ICAM1 expressions were upregulated on the surface of neutrophils in the high-concentration group followed by a gradual downregulation within 30 min after cell dilution (Fig. 3H, I). These findings suggest that the early aggregation of infused neutrophils in the lungs involves interactions between neutrophils and pulmonary endothelial cells, but neutrophils play a dominant role. Moreover, the downregulation of adhesion molecules after diluting neutrophil concentration may have contributed to the changes in neutrophil distribution after 4 h of infusion.
Neutrophil Transfusion Markedly Enhances Survival in Neutrophil-Depleted Mice Having CLP-Induced Sepsis
Neutrophils phagocytose bacteria and subsequently use intracellular lysozymes and peroxides to eradicate them. However, these bactericidal processes are dependent upon the intact chemotaxis of neutrophils, which enables them to migrate across the endothelium toward infected tissues. We transplanted allogeneic 89Zr-labeled neutrophils to mice having high-grade CLP-induced sepsis and monitored their distribution using micro-PET over 24 h (Fig. 4A). The allogeneic neutrophils were observed in the infected abdominal region within 1 h and their distribution in other organs was similar to that in healthy mice (Fig. 4 B, C). After receiving neutrophil transfusion, the neutrophil absolute count in the peripheral blood of recipient CLP sepsis mice increased at 3 h (Fig. S4B).
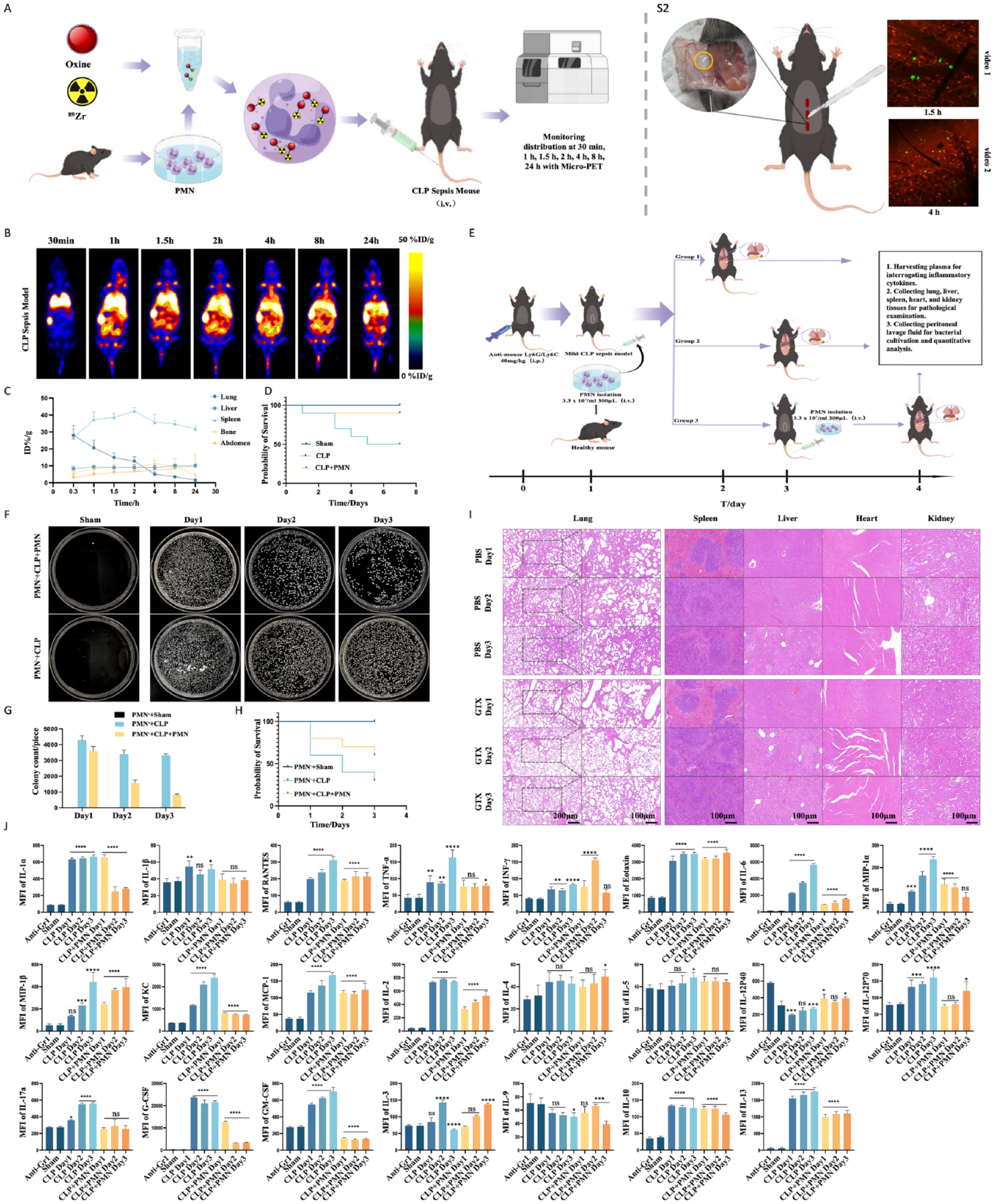
Neutrophil transfusion markedly enhances survival in neutrophil-depleted mice having cecal ligation puncture (CLP)-induced sepsis. (A) Illustration of the experiment indicating in vivo distribution of 89Zr-labeled neutrophils in the CLP-induced sepsis mouse model. (B) Micro-PET image shows the in vivo distribution of 89Zr-labeled neutrophils in the CLP-induced sepsis mouse model. (C) Percentage injected dose rate per gram of tissue (%ID/g) in the abdominal region and lung, liver, spleen, and bone of host mice within 24 h. (D) Seven-day survival rate of mice with severe CLP-induced sepsis (n = 10) after neutrophil transfusion. (E) Schematic of neutrophil transfusion for the treatment of neutrophil-depleted mice having CLP-induced sepsis. (F) Bacterial growth in the peritoneal lavage fluid in neutrophil-depleted mice with CLP-induced sepsis. (G) Bacterial colony enumeration and statistical analysis. (H) Survival rate after neutrophil transfusion in neutrophil-depleted mice with CLP-induced sepsis (treatment group = 10, non-treatment group = 20). (I) Tissue sections of vital organs of mice with CLP-induced sepsis. (J) Luminex multiplex assay results showing inflammatory factors in the plasma of neutrophil-depleted mice having CLP-induced sepsis. MCP: monocyte chemotactic protein; PET: positron emission tomography; NS: none-significant; PMN: polymorphnuclear neutrophil. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001, compare with Sham group.
Next, we infused labeled neutrophils in healthy mice and injected a chemotactic peptide into the medial side of the abdominal wall and observed the behavior of the neutrophils in the microvessels using confocal microscopy (Fig. 4S2). A slow flow of neutrophils adhered to the wall in the microvessels near the chemotactic peptide (Fig. 4S2, Supplemental video 1) after 1.5 h of infusion, and some neutrophils had already migrated across the endothelium into the tissue by 4 h (Fig. 4S2, Supplemental video 2). Notably, the transfusion of neutrophils into severely infected mice (CLP-induced sepsis) resulted in a substantial increase in survival rates in the treatment group compared with the untreated group (Fig. 4D).
Neutrophil depletion was induced by intraperitoneally injecting anti-Gr1 antibody at least 24 h before establishing the CLP-induced sepsis mouse model to assess the therapeutic efficacy of neutrophil transfusion in granulocytopenia-associated infections. Then, we simultaneously transplanted neutrophils and established the CLP model. The plasma, intraperitoneal washings, and organ samples from the lungs, spleens, livers, hearts, and kidneys were collected to assess the treatment outcomes (Fig. 4E). After 3 h of receiving neutrophil transfusion, both neutrophil-depleted mice and neutrophil-depleted CLP sepsis mice showed an increase in the neutrophil absolute count in peripheral blood (Fig. S4C, D). Bacterial counts were significantly lower in the abdominal washings from the transplanted group than in the non-transplanted group, and these counts progressively reduced with the treatment (Fig. 4F, G). Moreover, the survival rate of neutropenic CLP-infected mice was substantially improved after neutrophil transfusion (Fig. 4H).
The lung tissue sections of septic neutrophil-deficient mice showed thickened lung interstitium, collapsed alveolar atelectasis, and compromised ventilation and gas exchange functions, along with structurally disrupted liver lobules and numerous necrotic cells. In contrast, the lung tissue sections of neutrophil-transplanted host mice demonstrated substantial improvements in morphology, which gradually improved with the treatment. Moreover, the liver tissue showed no apparent necrosis in the treatment group. (Fig. 4I).
Furthermore, the plasma cytokine analysis revealed a notable increase in the secretion of pro-inflammatory cytokines, including interleukin (IL)-1α, RANTES (regulated upon activation, normal T cell expressed and secreted [also known as CCL5]), tumor necrosis factor-alpha (TNF-α), interferon-gamma (INF-γ), Eotaxin, IL-6, macrophage inflammatory protein (MIP)-1α, MIP-1β, keratinocytes (KC), and IL-2, as well as factors regulating granulocyte regeneration, such as G-CSF, granulocyte-macrophage colony-stimulating factor (GM-CSF), and IL-3, along with the secretion of the pro-respiratory mucin secretion factor IL-13, and the suppressive cytokine IL-10 following infection. Furthermore, the secretion levels in the untreated group exceeded those in the neutrophil-treated group (Fig. 4J). These cytokine findings suggest that in infections lacking neutrophils, the body secretes an increased amount of pro-inflammatory cytokines, enhancing the functionality of monocytes and macrophages, and promoting the proliferation and differentiation of B and T cells, simultaneously, factors like G-CSF drive granulocyte production, which all leading to a state of excessive inflammation. On the contrary, after receiving neutrophil therapy, the secretion of corresponding inflammatory factors relatively decreased. Neutrophil infusion therapy not only exerts bactericidal effects but also ameliorates the inflammatory status of the body. These findings suggest that allogeneic neutrophils show bactericidal functions in host mice and ameliorate the inflammatory state, thereby increasing survival.
Allogeneic Neutrophil Transfusion Improves the Survival Rate of Neutrophil-Depleted Mice With Multi-Organ Dysfunction Caused by Abdominal Sepsis
We used a mouse model that enabled us to control infection severity—the intraperitoneal auto-fecal infection leading to multiple organ dysfunction model—to investigate the efficacy of neutrophil allografts in treating long-term granulocytopenic infections. Neutrophils were depleted by intraperitoneally injecting anti-Gr1 antibody 24 h before modeling. The mouse model was established by intraperitoneally injecting 3% supernatant of fresh fecal homogenate, and systemic symptoms of infection, such as listlessness, disheveled hair, and increased ocular secretion, were observed after 12 h. The model mice were divided into three groups, and neutrophils were transplanted at 1-day interval with mice in the third group receiving three treatments. As the anti-Gr1 antibody maintained the neutrophil-deficient state for approximately 5 days (Fig. 5, S3), the same dose of anti-Gr1 antibody was administered again to mice in all groups on the third day of the experiment for preventing drug interactions and ensure sustained therapeutic efficacy. The therapeutic effect was assessed by measuring plasma inflammatory factors, determining the bacterial count in the abdominal flushing fluid, and observing histopathologic changes in the vital organs of the mice (Fig. 5A). We observed a significant increase in the survival rate of the mice in the treatment group (Fig. 5B). The bacterial count was significantly lower in the abdominal rinse fluid of the transplanted group than that of the non-transplanted group, and the count gradually decreased with the progression of treatment (Fig. 5C, D). After 3 h of neutrophil transfusion, neutrophil-depleted intra-abdominal infection (IAI) mice exhibited an increase in the neutrophil absolute count in peripheral blood (Fig. S4E). Histopathologic analysis showed changes, such as interstitial thickening, alveolar collapse, and fusion in the lungs of infected mice, and the normal state was gradually restored in the interstitium and alveoli after neutrophil transfusion. As the treatment progressed, the spleen parenchyma of the treated group showed more neutrophil infiltration compared with other groups. Hepatic pathologic staining showed structural destruction of the hepatic liver lobule with a large number of liquefied and necrotic cells in the non-treated group on the seventh day (Fig. 5E). G-CSF was progressively produced, pro-inflammatory factors were secreted in large quantities, and helper T cells secreted numerous chemokines for activating monocytes and macrophages in the non-transplanted group. In the transplanted group, the overall level of inflammatory factors was relatively low, mainly consisting of inhibitory inflammatory factors and some monocyte and macrophage chemokines. The level of G-CSF gradually decreased with the progression of treatment (Fig. 5F). Therefore, transfusion of a large number of neutrophils ameliorated the infection, restored the regulation of normal inflammatory cells, enhanced the overall immune response to infections, and decreased the high mortality because of infections caused by neutrophil deficiency.
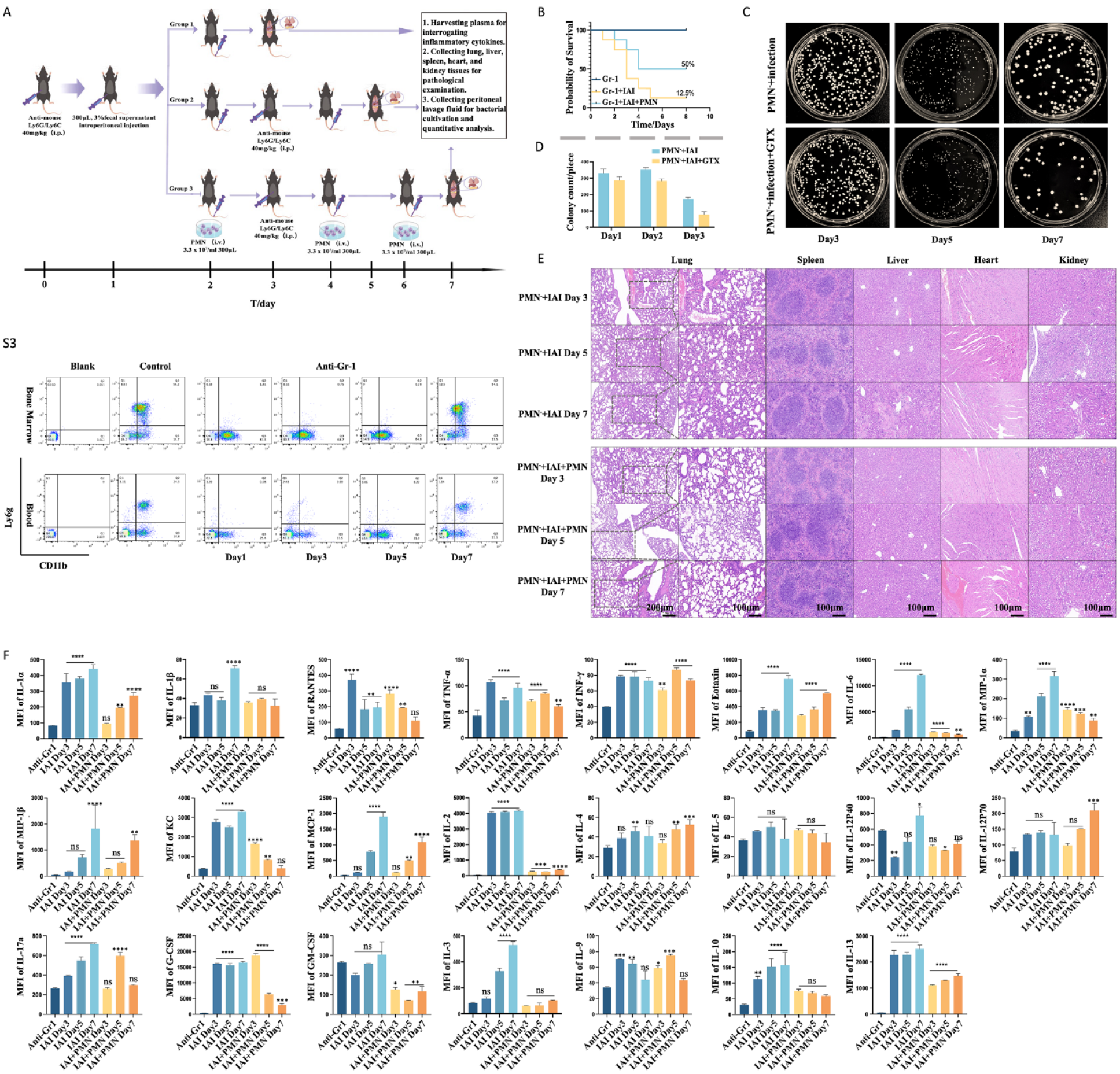
Allogeneic neutrophil transfusion improves the survival rate of neutrophil-depleted mice with multi-organ dysfunction caused by abdominal sepsis (A) Neutrophil transfusion strategy for treating granulocytopenic abdominal infection with multiple organ dysfunction in mice. (B) Seven-day survival rate of severe granulocytopenic abdominal infection with multiple organ dysfunction mice treated with neutrophil transfusion (n = 10). (C and D) Bacterial counts and quantitative analysis in abdominal lavage fluid collected from mice with granulocytopenic abdominal infection and multiple organ dysfunction. (E) Histopathologic examination of lungs, spleens, livers, hearts, and kidneys in mice with multiple organ dysfunction resulting from abdominal infection. (F) Results of Luminex multiplex assay for inflammatory cytokines in the plasma of mice with granulocytopenic abdominal infection and multiple organ dysfunction. GTX: granulocyte transfusion; IAI: intra-abdominal infection; NS: none-significant; PMN: polymorphnuclear neutrophil. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001, compare with anti-Gr1 group.
Discussion
Neutrophils—crucial for combating infections—are an essential component of the innate immune system. A decrease in neutrophil count markedly elevates the risk of severe infections in patients19–21. Despite advancements in antibiotic therapy and supporting treatments, the management of infections in patients with granulocytopenia remains challenging. Prolonged neutrophil deficiency leads to refractory fungal infections and higher patient mortality22,23. Even in cases where neutrophils are present in sufficient numbers, their impaired function contributes to increased infection risk. Therefore, researchers have explored strategies to promote neutrophil generation and engraftment, such as using G-CSF to stimulate neutrophil production 5 . Although G-CSF treatment has facilitated the procurement of abundant neutrophils from a single donor, laboratory evidence is inadequate to confirm the functional integrity of such neutrophils 8 . In addition, clinical studies on granulocyte transfusion therapy are inconclusive 24 . Therefore, further research is essential to establish the effectiveness of neutrophil transfusion therapy for future applications and provide reliable guidance to clinicians for improving treatment outcomes in infected patients.
The efficacy of granulocyte transfusion therapy is positively correlated with the infusion dose. Engelfriet et al. found that the optimal infusion dose of granulocyte infusion therapy in adults was at least 1010 polymorphonuclear leukocytes/m2 12 ,25. This lower limit represents approximately half of the total peripheral blood neutrophil count in adults. Therefore, maintaining sufficient circulating neutrophils during granulocyte transfusion is essential for treating granulocytopenia-associated infections. Nevertheless, when assessing neutrophil counts in the peripheral blood of recipient mice post-transfusion, we observed an increase in absolute neutrophil count. However, this increase was significantly lower than the number of transplanted cells, and in a few mice, neutrophil counts remained unchanged. This phenomenon may be associated with the potential presence of allogeneic neutrophils that could be lingering in the marginal pool 26 . We used 7-week-old healthy C57BL/6 mice having a total peripheral blood neutrophil count ranging from approximately 1.8 × 106–6.6 × 106 27 . We compared purified bone marrow neutrophils with mature neutrophils from peripheral blood and found that bone marrow neutrophils also possess functional integrity and can be effectively used for transfusion therapy.
Exogenous neutrophils are allogeneic cells that may elicit an immune rejection response in the host. This reaction could be linked to the human leukocyte antigen (HLA) antigens present on the neutrophil surface, as well as the small number of erythrocytes and monocytes that accompany the graft28–33. Some authors have indicated possible symptoms of fever and impaired lung function in patients following neutrophil transfusion 34 , this symptom may resemble the occurrence of a cytokine storm observed during chimeric antigen receptor (CAR)-T cell therapy35,36, underscoring the importance of addressing immune rejection. Therefore, we used magnetic bead sorting to purify neutrophils (neutrophil purity approximately is 90%) and eliminate erythrocytes and other heterocytes, thereby minimizing the possibility of immune rejection. Furthermore, we selected C57BL/6 inbred mice as recipients to minimize immune rejection during neutrophil transfusion. We evaluated tissue and organ damage in immunocompetent host mice after transfusion and observed a minimal immune rejection with our transfusion approach because of the use of purified neutrophils and suitable recipient mice. Overall, we found that the use of highly purified neutrophils and careful control of immunogenicity between donors and recipients enhanced the immunological safety of the transfusion process.
Next, we investigated the distribution of transplanted neutrophils in mice to comprehend their proper functioning. We observed a notable accumulation of exogenous neutrophils in the host lungs during the early transfusion phase. We believe that this phenomenon was linked to the transiently elevated expression of ICAM1 in neutrophils and that of CD11b in both neutrophils and endothelial cells at a high cell density. Interestingly, transplanted neutrophils showed a rapid response to infection, independent of the lung stasis phenomenon, in a severe CLP-induced sepsis mouse model. This finding validated the use of neutrophils for treating granulocyte deficiency-associated infections. Overall, our neutrophil transfusion treatment had a high immune safety profile, and the transplanted neutrophils were functional in response to infection.
Ly6G is differentially expressed during the development of myeloid lineage cells, such as monocytes, macrophages, granulocytes, and neutrophils 37 . While monocytes transiently express Ly6G during development, only mature granulocytes and peripheral neutrophils retain Ly6G expression 38 , making it a valuable surface marker for identifying these cell populations. The RB6-8C5 monoclonal antibody is strongly reactive to mouse Ly6G 39 . Therefore, it is possible to effectively deplete the original neutrophils in the host mice without destroying other immune cells using the anti-Gr1 treatment. Here, the use of anti-Gr1 antibodies against the Ly6G surface marker allowed us to establish a robust model for investigating neutrophil transfusion therapy. The neutrophil-depleted mice showed high mortality even with mild infections. However, neutrophil transfusion treatment improved the survival rates in addition to reducing bacterial load, leading to effective bacterial clearance. Moreover, the damaged organ tissues showed gradual recovery. Overall, neutrophil transfusion was effective in treating granulocytopenic infections, and our results provided a baseline for the future development and optimization of neutrophil transfusion strategies.
In conclusion, neutrophil transfusion therapy is a novel therapeutic approach for the management of granulocytopenic infections; however, specific challenges persist. Further research is required to assess the effectiveness of this therapy in human patients and explore the pertinent issues about its clinical application. The current clinical data are insufficient to draw definitive conclusions regarding the efficacy of the treatment; the data merely indicate that extensive neutrophil transplants yield benefits for patients. Therefore, it is imperative to establish the effectiveness of neutrophil transfusion therapy through rigorous validation and provide a theoretical foundation for its future use in clinical practice to improve patient outcomes. Continuous research efforts will enable us to refine the techniques and methods of neutrophil transfusion, thereby enhancing therapeutic efficacy and minimizing potential immune rejection. Our future research will concentrate on refining neutrophil acquisition and storage techniques, optimizing transfusion dosage and timing, enhancing the safety of neutrophil transfusion, and conducting further clinical trials to substantiate its value in clinical practice. We firmly believe that neutrophil transfusion therapy holds the promise of broader applications in the near future.
Supplemental Material
sj-docx-1-cll-10.1177_09636897241228031 – Supplemental material for Investigation of Allogeneic Neutrophil Transfusion in Improving Survival Rates of Severe Infection Mice
Supplemental material, sj-docx-1-cll-10.1177_09636897241228031 for Investigation of Allogeneic Neutrophil Transfusion in Improving Survival Rates of Severe Infection Mice by Linbin Li, Yunxi Yang, Zaiwen Guo, Xi Gao, Lu Liu, Jiamin Huang and Bingwei Sun in Cell Transplantation
Footnotes
Author Contributions
LL and BS designed the study and wrote the paper. LL, YY, ZG, XG, LL, and JH performed the experiments. ZG and BS performed the statistical analysis. All authors read and approved the final manuscript.
Availability of Data and Materials
Data are available from the authors upon reasonable request.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China, No. 82072217, 81772135, and U21A20370.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
