Abstract
The objective of this study was to evaluate the feasibility, safety, and effectiveness of intravenous stem cell delivery utilizing ultrasound-targeted microbubble destruction (UTMD) in a rat model of middle cerebral artery occlusion (MCAO), while investigating the underlying mechanisms. Acute cerebral infarction (ACI) was induced surgically in adult rats to create the MCAO rat model. Intravenous injection of SonoVue microbubbles and bone marrow–derived mesenchymal stem cells (BMSC) was performed concurrently, with or without ultrasound targeting the stroke. The animals were divided into four groups: sham-operated group, ACI-MCAO rats treated with phosphate-buffered saline (ACI+PBS), rats receiving intravenous delivery of BMSC expressing green fluorescent protein (GFP-BMSC; ACI+BMSC), and rats receiving intravenous GFP-BMSC with simultaneous UTMD exposure (ACI+BMSC+UTMD). The efficacy of the treatments was assessed by evaluating the animals’ neurological function using the Longa score and examining histopathological changes such as cerebral infarct volume, cerebral edema, and cell apoptosis. A rat cytokine array was utilized to identify the potential cytokines that may be responsible for the therapeutic effect of UTMD-mediated BMSC treatment. Optimal UTMD parameters resulted in an increase in blood–brain barrier (BBB) permeability after 30 min, which returned to baseline 72 h later without causing any residual injury. UTMD application significantly increased the homing of intravenously delivered BMSC, resulting in a 2.2-fold increase in GFP-BMSC cell count on day 3 and a 2.6-fold increase on day 7 compared with intravenous delivery alone. This effect persisted for up to 6 weeks after injection. Intravenous BMSC delivery significantly reduced the volume of cerebral infarct and decreased cerebral edema, leading to a lower Longa score. Furthermore, this effect was further enhanced by UTMD. Acute cerebral infarction induced by MCAO led to elevated matrix metalloproteinase 8 (MMP8) levels in the cerebrospinal fluid, which were significantly reduced following UTMD-mediated BMSC treatment.
Keywords
Introduction
Acute ischemic stroke, or acute cerebral infarction (ACI), occurs due to intravascular thrombus formation that obstructs cerebral blood flow, resulting in considerable morbidity and mortality 1 . The 2020 report by the American Heart Association revealed that strokes occur at a rate of one per 40 seconds, affecting millions of individuals globally each year2,3. The current main treatments remain thrombectomy or thrombolysis with alteplase, urokinase, and tenecteplas 1 . Nevertheless, the utilization of these therapies is constrained by a narrow therapeutic window and carries significant risks of secondary brain hemorrhage 4 . Therefore, additional therapies with a less restricted therapeutic time-window and presenting with less risk are currently investigated.
In the last decade, stem cell therapy has emerged as a promising approach for improving neurological recovery and protecting against reperfusion damage in ischemic stroke5–7. Specifically, bone marrow–derived mesenchymal stem cells (BMSC) have shown potential due to their ability to generate differentiated progeny and secrete substances that activate the repair of damaged brain cells. Clinical studies have been conducted in this field, demonstrating the effectiveness of BMSC in improving behavioral performance in animal models of ischemic stroke8,9.
However, the blood–brain barrier (BBB) presents a challenge for the delivery of BMSC to the brain parenchyma. Both intravenous and intra-arterial administration methods have shown promise, but inefficient delivery to the injury sites due to limited BBB permeability remains an issue 10 . Enhancing the homing of BMSC and improving BBB permeability are crucial factors to consider for successful stem cell transplantation in ischemic stroke treatment.
Ultrasound-targeted microbubble destruction (UTMD) is a noninvasive technique that has shown promise in enhancing BBB permeability. Previous studies have demonstrated successful delivery of molecules, genes, or proteins across the BBB using UTMD11–13. However, the combination of cell therapy and UTMD in stroke treatment has been poorly studied 14 . In this study, we investigated the ability of UTMD to enhance BBB penetration and homing of BMSC in a rat model of middle cerebral artery occlusion (MCAO). We evaluated the impact of this combination on neurological outcomes and explored the underlying molecular mechanisms.
Materials and Methods
Cell Culture
Green fluorescence protein transferred bone marrow stromal cells of rat (GFP-BMSC) were constructed by Cyagen Bioscience Technology Co. (Suzhou, China) which could steadily express GFP. The cells were cultured in an Minimum Essential Medium Eagle - Alpha Modification (α-MEM) medium (Gibco, NY, USA) supplemented with 10% fetal bovine serum (Gibco, NY, USA) and 1% antibiotic-antimycotic solution (Sigma-Aldrich, USA) and kept at 37 °C, with 5% CO2 and 95% humidity. All experiments involving GFP-BMSC were conducted within the first 3 to 6 passages (Supplementary Fig. 1).
MCAO Model Inducing
A total of 200 adult male Sprague Dawley (SD) rats (260–280 g) were used in this study. Among them, 50 rats were allocated to the sham operation group for BBB permeability testing, while 150 rats were used to induce the middle cerebral artery occlusion (MCAO) model. Rats with a Longa score of 2 on the first postoperative day were included, and any rats that died during the experiment were excluded. A minimum of 3 to 5 rats per group were included for data analysis. The rats were housed in a specific pathogen-free (SPF) environment with controlled temperature and humidity, following a 12-h light/dark cycle. All experimental protocols were conducted in accordance with the guidelines of the National Institutes of Health Guide for the Care and Use of Laboratory Animals and approved by the Ethics Committee of Shanghai General Hospital.
The MCAO model was induced in SD rats using a previously described procedure. Rats were deeply anesthetized with isoflurane inhalation, and vital signs such as breathing and heart rate were monitored. The left common carotid artery (CCA) was temporarily ligated, and a silicone nylon filament was inserted into the left internal carotid artery until it reached the origin of the middle cerebral artery (MCA). After 90 min, reperfusion was achieved by withdrawing the filament and releasing the CCA ligature. Postoperative analgesics (meloxicam and ibuprofen) were administered for pain relief. The rats were kept under a warming lamp to maintain normal body temperature and physiological state. Sham-operated animals underwent exposure of arterial vessels without occlusion of the common carotid arteries.
Blood–Brain Barrier Permeability Detection
Blood–Brain Barrier permeability detection was performed as follows 15 : the fur on the head of 12 healthy SD rats was removed using depilatory cream. After anesthesia, 3% Evans blue dye (EBD, 1 mL/Kg) was injected into the tail vein and circulated for 5 min in vivo. SonoVuemicrobubbles (Bracco, Italy), approved for clinical use, were bolus-injected via the tail vein at a dose of 0.1 mL/kg, simultaneously followed by acoustic stimulation. The ultrasound apparatus Topteam161 (Physioson-Basic, Elektromedizin, AG, Germany) was used for acoustic exposure. Usually at the low acoustic frequency (1 MHz) condition, the acoustic intensity and exposure duration were two main crucial factors influencing acoustic bioeffects, therefore different intensity (1.5, 1.75, and 2.0 W/cm2) and duration (1, 5, and 10 min) were assessed and chose the optimal combination for the following study. The transducer in diameter of 2 cm was placed against the left parietal bone for 300 seconds. Thirty minutes after the end of stimulation, the animals were perfused with 4% paraformaldehyde and brains were extracted. EBD staining of the brain was visually observed. 10-μm-thick brain sections were obtained by vibratome sections and observed under fluorescence microscopy (Olympus, USA) at 30 min, 24 h, and 72 h’ time point. The excitation wavelength of EBD was selected at 470 and 540 nm with an emission wavelength at 680 nm.
Neurological Function Evaluation
After MCAO induced, the neurological function of all animals was evaluated daily with the Longa score which consists in a 5-point scale 16 : (0) no neurologic deficit, (1) failure to extend left forepaw fully, (2) circling to the left, (3) falling to the left, and (4) unable to walk spontaneously with reduced level of consciousness.
BMSC Engraftment and UTMD Treatment
The rats were randomly assigned to either the sham-operated group or the MCAO model group. The sham-operated rats underwent surgical exposure of the arterial vessels without occluding the common carotid arteries. All rats in the sham-operated group were assigned a Longa score of 0. Twenty-four hours after performing the MCAO model, rats with a Longa Score of 2 were subsequently randomized into the following groups: ACI+PBS group, ACI+BMSC group, and ACI+BMSC+UTMD group. Phosphate-buffered saline (PBS) of 1 mL was injected intravenously via the tail vein in ACI+PBS group 24 h after MCAO induced. A total of 5 × 106 BMSC in 1 mL PBS were injected intravenously via the tail vein in ACI+BMSC group. In the ACI+BMSC+UTMD group, intravenous injection of SonoVue microbubbles (Bracco, Milan, Italy) at a dose of 0.1 mL/kg was performed via the tail vein, followed by acoustic stimulation. After the stimulation, 5 × 106 BMSC cells were injected intravenously.
Histopathological Analysis
Brain water content (BWC) was measured to assess cerebral edema. After MCAO, brains were extracted, and the pons and olfactory bulb were removed. The residual brain was weighed to obtain its wet weight, and then dried at 95°C for 24 h to determine its dry weight. Brain water content was calculated using the formula: (wet weight − dry weight) / wet weight × 100%.
Brain infarct volume was measured using the 2,3,5-triphenyl tetrazolium chloride (TTC) methods. Five rats from each group received TTC staining 7 days after treatment. Brains were extracted and 2-mm coronal sections were obtained using vibratome section. Four sections in each brain were obtained and put in 0.5% TTC PBS solution for 10 min at 37°C without light exposure and then fixed in 4% paraform PBS. Under this preparation, normal brain tissue was stained in red and infarction area appeared white. Images of the fixed slices were obtained using an Epson V500 scanner (Epson, China) and BI2000 medical image analysis system (PVA, USA) was used to quantify infarction. The possible interference of brain edema with infarct volume was corrected by standard methods (whole contralateral hemisphere volume minus nonischemic ipsilateral hemisphere volume) and the infracted volume was expressed as a percentage of the whole contralateral hemisphere 17 .
The pathological changes of infarct brain were assessed by hematoxylin and eosin (HE) staining. Paraffin-embedded brain sections (5 μm) were subjected to HE staining according to a standard protocol 18 and observed under a light microscope (Olympus, USA).
The extent of apoptosis was analyzed by TUNEL assay using an In Situ Cell Death Detection Kit (Beyotime, China). Paraffin-embedded tissue sections were incubated with TUNEL reaction medium, counterstained for nuclei with DAPI (Sigma-Aldrich, USA), and observed under a fluorescence microscope(Olympus, USA).
Immunofluorescent (IF) Staining of Neovascularization on Brain Specimens
At 7 days after stroke, MCAO rats in each group were perfused with ice-cold PBS followed by 4% phosphate-buffered paraformaldehyde. IF was performed as described previously 19 . The primary antibodies were: Mouse anti-CD31 (GB12063, Servicebio, China), rat anti-von-Willebrand-Factor(anti-vWF) (GB11020, Servicebio, China), The secondary antibodies used were: Cy3-labeled goat anti-mouse IgG (GB21301, Servicebio, China), FITC-labeled goat anti-rabbit IgG (GB22303l, Servicebio, China).
For quantification, three randomly selected high-power fields (HPFs; 400× or 200× for IF study) were analyzed in each section. The mean number of positively-stained cells per HPF for each animal was calculated.
Cerebrospinal Fluid Collection and Cytokine Analysis
Animals were anesthetized 7 days after MCAO and were placed with their head mounted in astereotactic device. The skin was incised in the occipital part of the midline and occipito-cervical muscles were dissected. The foramen magnum and its cisterna were exposed and CSF was extracted through puncture with a 100-μL microinjector. Approximately 100 μL volume of CSF was extracted from each rat and mixed with 1 μL of phenylmethanesulfonyl fluoride (Sigma-Aldrich, USA) and then quickly transferred to −80°C for storage. Samples were removed from −80°C, thawed on ice and briefly centrifuged. CSF from all the distinct groups (Control group, ACI+PBS, ACI+BMSC and ACI+BMSC+UTMD, n = 3 from two different experiments for each group) was compared with each other using the 34-cytokine preconfigured sandwich ELISA Rat Cytokine Array G2 (RayBiotech, Norcross, GA, USA). To reduce batch variability, 100 μL of each CSF sample occupied a single array so that only one chip was used for the experiment. CSF samples were not diluted. The chip was read using a GenePix 4000B Microarray Scanner (Molecular Devices, Sunnyvale, CA, USA). Specific protocol details can be found at the website of RayBiotech Inc. (http://www.raybiotech.com).
Statistical Analysis
Data were expressed as mean ± SD and analyzed using nonparametric tests (Mann–Whitney test) when comparing two groups. Kruskal–Wallis test was used to compare variance between three or more groups. SPSS software (Version 20.0, SPSS Inc., USA) was used for all analyses. A P-value lower than 0.05(*) was considered as statistically significant.
Results
Assessing BBB Permeability Increase and Disruption by UTMD
A total of 200 rats were included in the study: 50 for the sham operation group and BBB permeability testing, while 150 were used to establish the MCAO model. Our experiment was conducted in batches, with each batch representing an experimental time point. For each time point, we ensured that each of the 4 groups consisted of either 5 or 3 rats. A scheme drawing illustrating the entire approach has been provided (Supplementary Fig. 2).
In the absence of UTMD exposure, or ultrasound without SonoVue microbubbles there was no noticeable leakage of Evans Blue Dye (EBD) observed in the rat brain 10 min after EBD injection (Supplementary Fig. 3). When using fixed conditions of low frequency (1 MHz), 1:3 duty cycle, and 0.1 mL/kg microbubbles, higher acoustic intensity and longer exposure duration resulted in a progressively increasing leakage of EBD, as depicted in Fig. 1A, B. The EBD staining was subsequently reassessed 72 h after acoustic stimulation (Fig. 1C), during which only the combination group exposed to 1.75 W/cm2 and 5 min exhibited no visible staining, indicating the closure of the BBB. Consequently, the combination of 1.75 W/cm2 and 5 min was chosen for additional experiments involving UTMD.
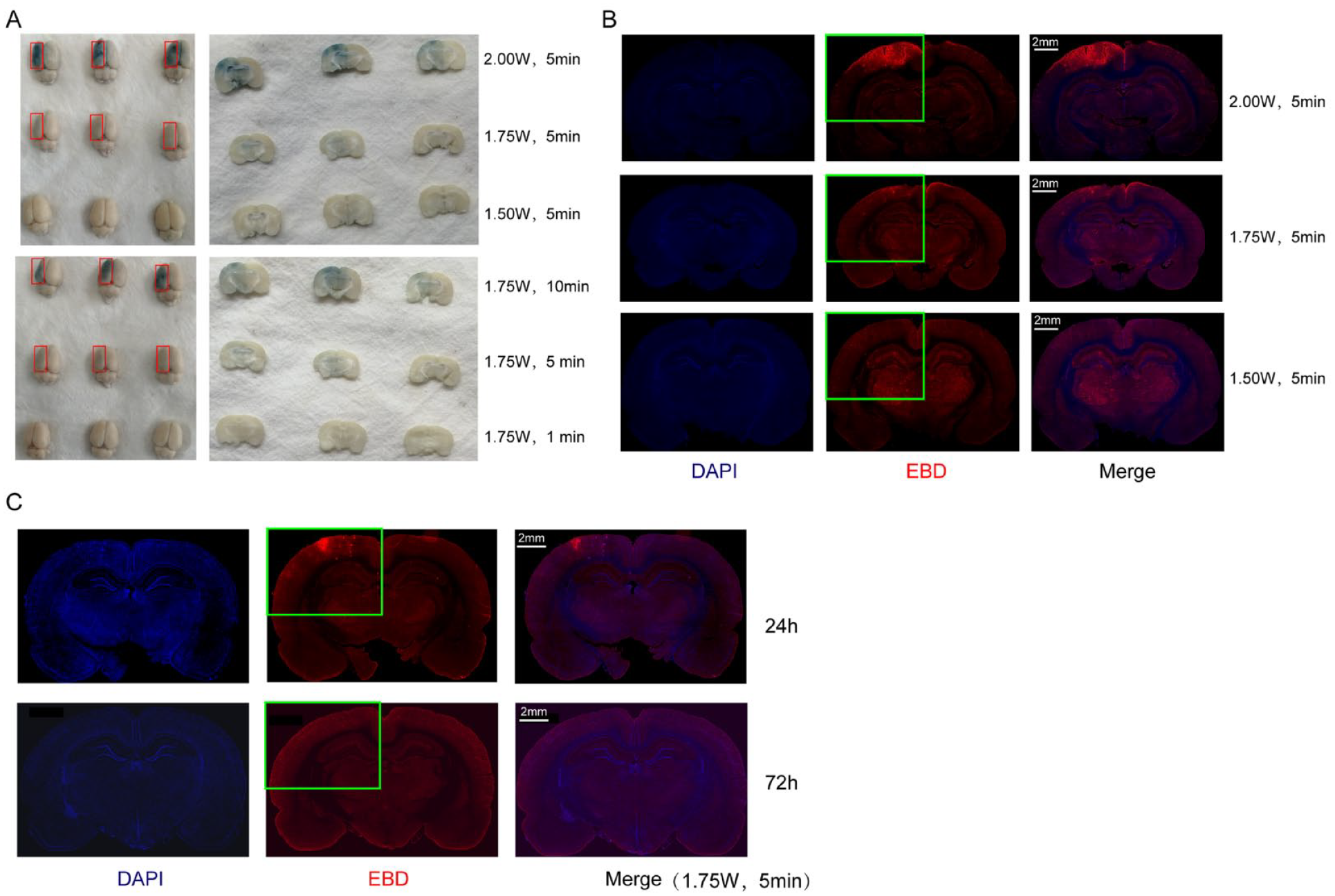
Assessing blood–brain barrier permeability increase and disruption by UTMD. (A) Under fixed conditions of low frequency 1 MHz, 1:3 duty cycle and 0.1 mL/Kg SonoVue microbubble, the blue staining of EBD on the surface and coronal sections of the left cerebral hemisphere at different ultrasound intensity (1.5, 1.75, and 2 W/cm2) and exposure duration (1, 5, and 10 min) were visually observed 30 min after UTMD treatment. (B) Correspondingly to Fig. 1A, the red fluorescence staining of EBD on coronal brain section at different intensity were observed. (C) The red fluorescence staining of EBD on coronal brain section were assessed separately at 24 and 72 h under optimal UTMD parameters’ treatment (1.75 W/cm2, 5 min, 1 MHz, 1:3 duty cycle and 0.1 mL/Kg). Green box represents the EBD staining (red, n = 650 nm). Nuclei are 4′,6-diamidino-2-phenylindole (DAPI)-stained (blue). Scale bar, 2 cm. UTMD: ultrasound-targeted microbubble destruction; EBD: Evans blue dye.
UTMD Increased Directional Homing of BMSC in MCAO Rats
SD rats were subjected to left MCAO and neurological outcome was then assessed by Longa Score 24 h after surgery. The MCAO rats exhibited impaired motor function characterized by a propensity to shift body weight toward the left, signifying impairment of the right-sided limbs resulting from a stroke in the left motor region (Fig. 2A). In order to investigate the long-term impacts of BMSC transplantation in MCAO rats, only animals with a Longa score of 2 were chosen for further experimental analysis (Fig. 2B).
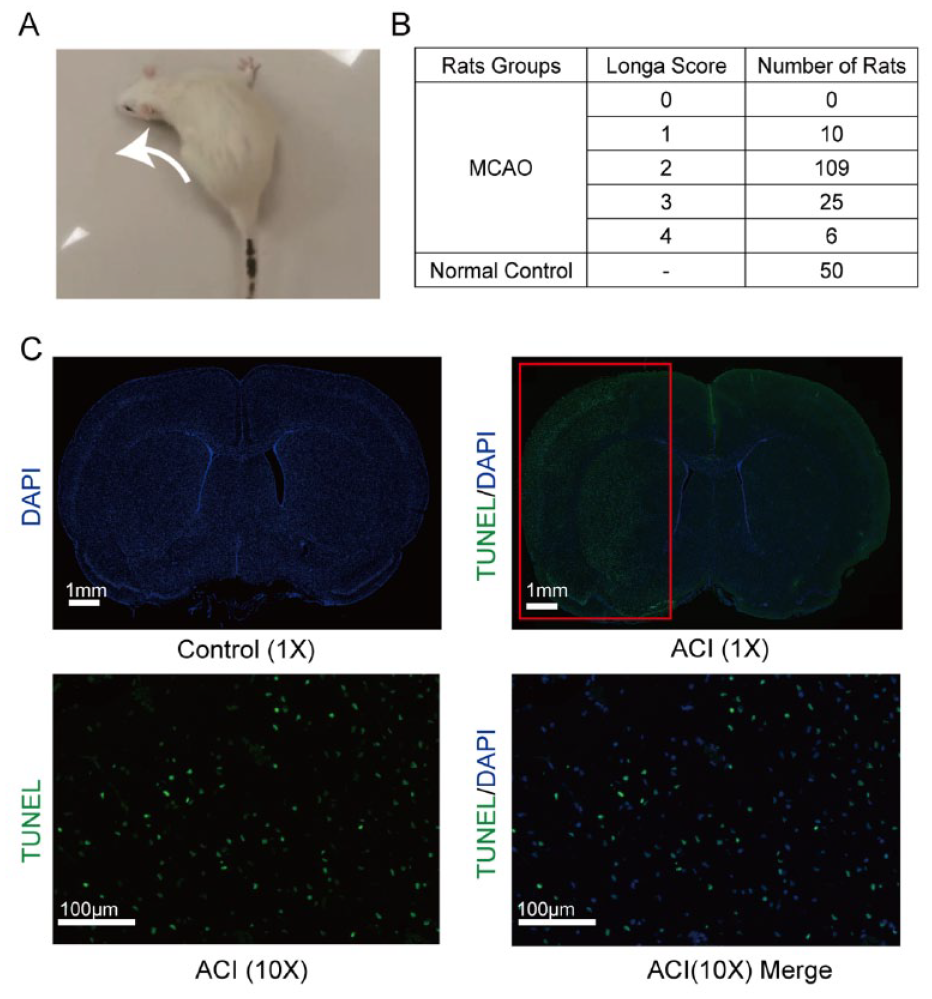
Inducing and evaluation of acute MCAO rat model. (A) Evaluation of the MCAO model with behavioral changes of rotation toward the left side (white arrow). (B) The Longa score of MCAO rats and controls (n = 50) after recovery from surgery. 109 MCAO rats scored 2; 25 MCAO rats scored 3; and 6 MCAO rat scored 4 and 10 MCAO rat scored 1 (n = 150). (C) terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) staining was used to distinguish the ipsilateral side (cerebral infarction) and the contralateral side (control). TUNEL positive cells were found on the infarct region of the acute MCAO rats. Scale bars represent 1 cm (upper) and 100 μm (lower). MCAO: middle cerebral artery occlusion; ACI: acute cerebral infarction.
Upon extraction 24 h after the lesion, the left hemisphere, where the cerebral infarction occurred, exhibited noticeable edema in comparison to the right hemisphere (control side). TUNEL staining demonstrated evident apoptosis in the left hemisphere (ipsilateral to MCAO), whereas no such apoptosis was observed in the contralateral hemisphere (Fig. 2C). Three days following GFP-BMSC engraftment in MCAO rats, the BMSC was were found within the ischemic penumbra of the left hemisphere (Fig. 3A, B). The ACI+BMSC+UTMD group exhibited a notably higher count of GFP-positive cells compared with the other groups (Fig. 3C). Furthermore, the ACI+BMSC+UTMD group displayed a 2.2-fold increase in GFP-positive BMSC on day 3 and a 2.6-fold increase on day 7 as compared with the ACI+BMSC group. GFP-positive BMSC were detectable in the lesioned hemisphere for up to 6 weeks after injection in the ACI+BMSC+UTMD group (Fig. 4).
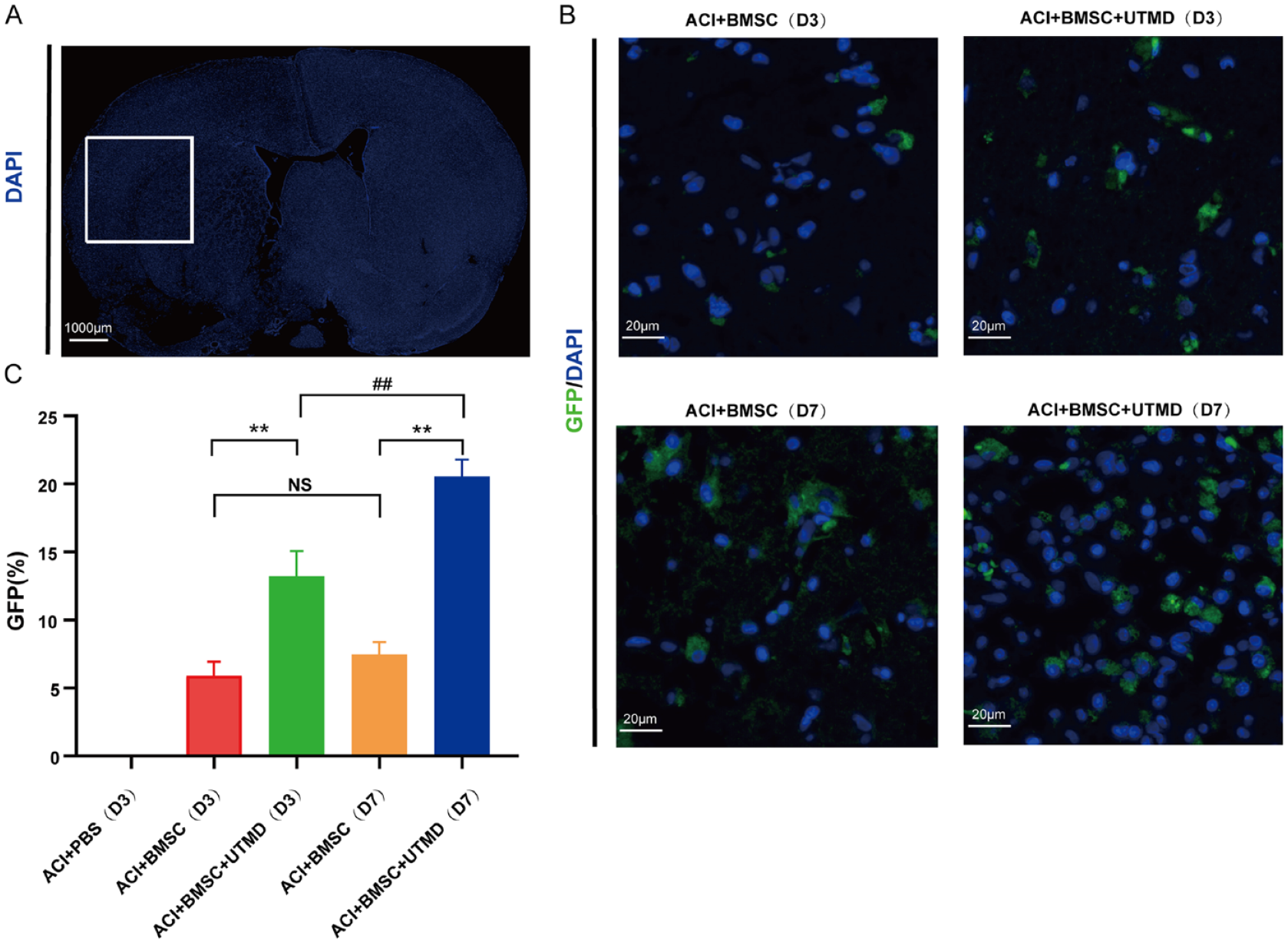
Homing of UTMD mediated BMSC in MCAO rats. (A) The ischemic penumbra (white box) on the coronal sections. 4′,6-diamidino-2-phenylindole (DAPI) indicates the DAPI staining of the nucleus in the brain. Scale bar, 1000 μm. (B) GFP-BMSC were detected in the ischemic penumbra at 3 days (D3) and 7 days (D7) after MCAO induced. (C) At 3 or 7 days after MCAO, the number of GFP-BMSC was significantly higher in ACI+BMSC+UTMD group than in ACI+BMSC group. *P < 0.05. **P < 0.01. At 7 days after MCAO, GFP-positive cells were significantly increased higher in ACI+BMSC+UTMD group. D7 vs. D3, #P < 0.05. ##P < 0.01. Values were expressed as means ±SDs. n = 5 rats per group. While there was no significant difference between D3 and D7 in ACI+BMSC group (NS, P >0.05). Green, GFP-BMSC; blue, DAPI nuclear staining. UTMD: ultrasound-targeted microbubble destruction; BMSC: bone marrow–derived mesenchymal stem cells; MCAO: middle cerebral artery occlusion; GFP: green fluorescent protein; ACI: acute cerebral infarction.
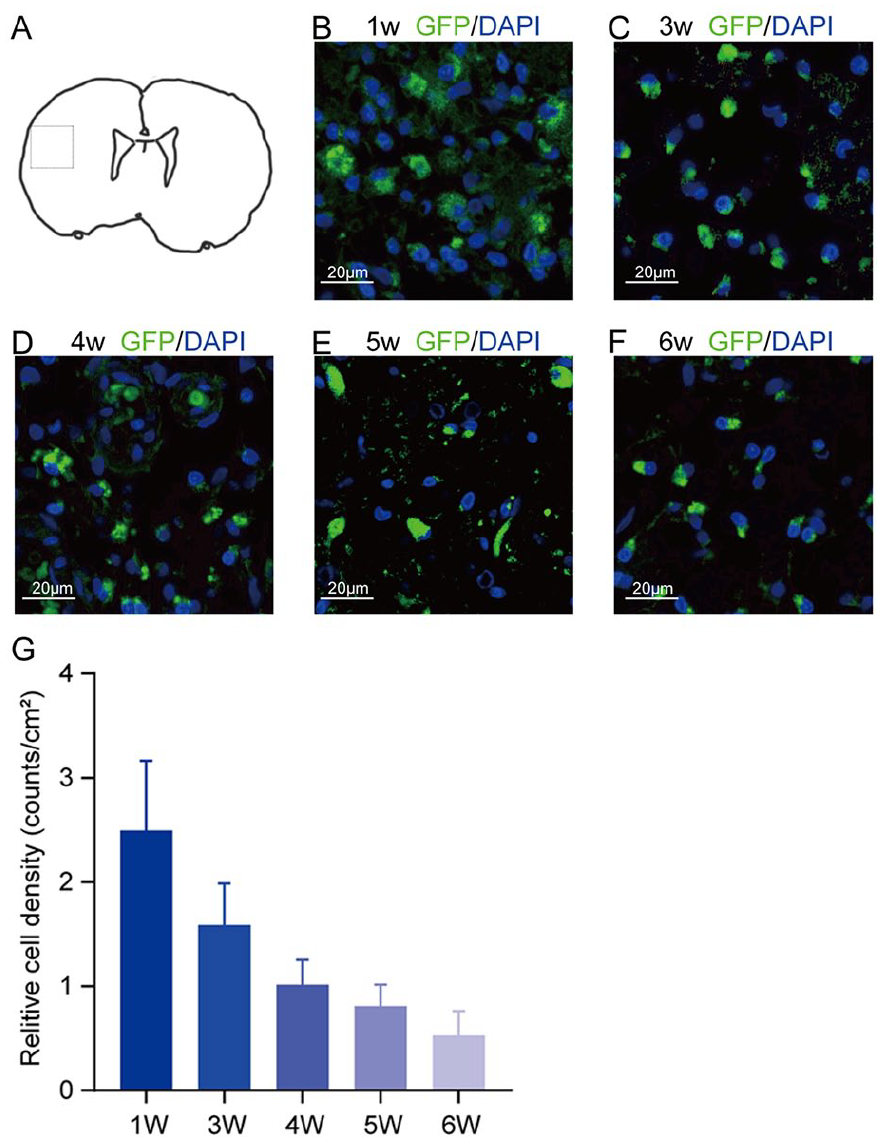
The persistent existence of GFP-positive BMSC in the lesioned hemisphere. (A) Pattern diagram of the MCAO rat brain coronal section and dashed box represented observation area. (B)–(F) represented the distribution of areas of the GFP positive cells at 1 to 6 weeks after surgery. n = 15 (3 rats were sacrificed every week). (G) Fluorescence microscopy analysis was employed to quantify GFP-labeled BMSC in the ACI area of the ACI+BMSC+UTMD group. The BMSC cell count, distinguished by GFP-positive cells, was determined in at least three distinct regions (at 10× magnification). Green, GFP-BMSC; blue, 4′,6-diamidino-2-phenylindole (DAPI) nuclear staining. Scale bar, 20 μm. GFP: green fluorescent protein; BMSC: bone marrow–derived mesenchymal stem cells; MCAO: middle cerebral artery occlusion; ACI: acute cerebral infarction; UTMD: ultrasound-targeted microbubble destruction.
UTMD Potentiates BMSC-Induced Improvement of Neurological Function and Histopathological Changes After MCAO
At 48 h post-BMSC engraftment, ACI+BMSC rats exhibited a lower Longa score in comparison to the ACI+PBS group. This disparity remained significant even after 15 days since MCAO. Importantly, the combination of UTMD with BMSC yielded additional enhancements in neurological outcomes when compared with BMSC treatment alone (Fig 5A). Furthermore, the infarct volume in the ACI+BMSC group exhibited a significant decrease when compared with the PBS group at 7 days posttreatment, and the addition of UTMD resulted in additional reduction of the infarcted area (P < 0.01) (Fig. 5C, D). Cerebral edema is recognized as a crucial contributor to the damage associated with the acute phase of stroke. Treatment with BMSC, regardless of UTMD inclusion, resulted in a noteworthy reduction in cerebral edema compared with the PBS group. However, no statistically significant difference was observed between the ACI+BMSC and ACI+BMSC+UTMD groups (Fig. 5B). Finally, ischemic changes characterized by cytoplasmic vacuoles and karyopyknosis formation were detected in the cortical layer of the left hemisphere, primarily supplied by the middle cerebral artery (Fig. 5E).
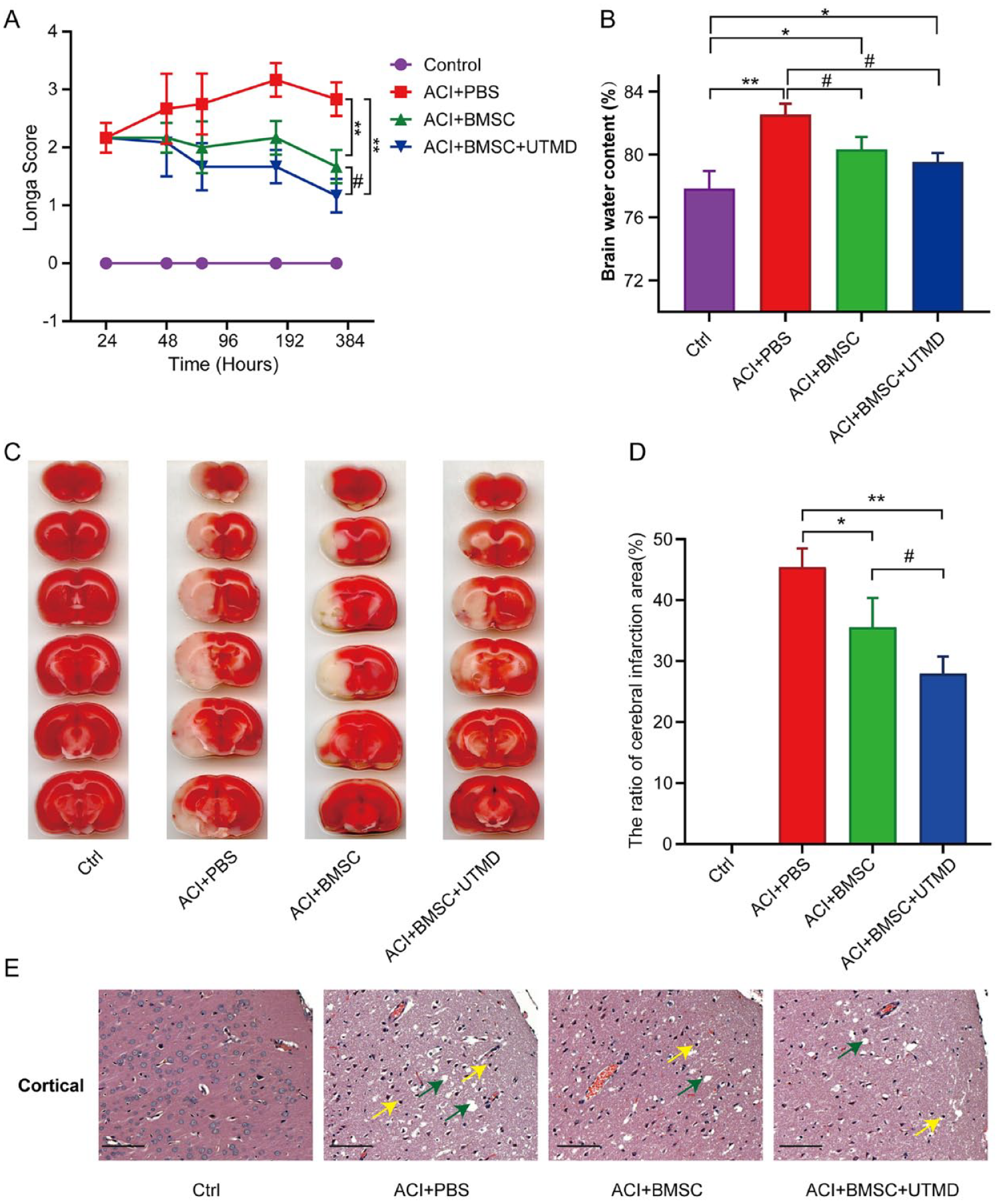
UTMD-mediated BMSC improved the neurological function and histopathological change in MCAO rat model. (A) Neurological performance as revealed by Longa score at indicated time points. Rats of ACI+BMSC+UTMD group showed significant recovery, compared with all other groups at 3, 7, and 15 days after MCAO. (B) UTMD mediated BMSC reduced cerebral edema of rats at 7 days after MCAO. The brain water content (BWC) of MCAO was significantly high compared with control group (*P < 0.05, **P < 0.01, compared with control group) and a significant reduction when treated with BMSC/ UTMD+BMSC (#P < 0.05, ##P < 0.01, compared with ACI+PBS group). Values were expressed as mean±SDs. n = 5 rats per group. (C) The therapeutic effect of UTMD mediated BMSC transplantation treatment on MCAO was determined by brain images of coronal brain sections of rats with 2,3,5-triphenyl tetrazolium chloride (TTC) staining. (D) The infarct areas of rats in ACI+BMSC+UTMD group significantly decreased compared with rats in other groups at 7 days after MCAO. Values were expressed as means ±SDs. n = 10 rats per group. *P < 0.05, **P < 0.01, compared with ACI+PBS group; # P < 0.05, compared with ACI+BMSC group. (E) The normal structure of the brain was demonstrated at the cortical in the ipsilateral hemisphere of the control group. cytoplasm vacuoles (green arrowheads) and karyopyknosis (yellow arrowheads) formation were demonstrated at the cortical in ACI+PBS group and BMSC/BMSC+UTMD group. Scale bar, 50 μm. UTMD: ultrasound-targeted microbubble destruction; BMSC: bone marrow–derived mesenchymal stem cells; MCAO: middle cerebral artery occlusion; ACI: acute cerebral infarction; PBS: phosphate-buffered saline.
Cerebral infarction repair is frequently accompanied by neovascularization. Fluorescence detection of CD31 and vWF was used to assess the angiogenesis of cerebral infarction. The fluorescence detection results revealed a significant increase in the density of CD31+ endothelial cells in the ACI+BMSC group compared with the ACI group, as well as a significant increase in the density of CD31+ cells in the UTMD treatment group compared with the BMSC alone group (Fig. 6A, B). The expression pattern of vWF protein correlated with that of CD31 protein in each group (Fig. 6A, C).
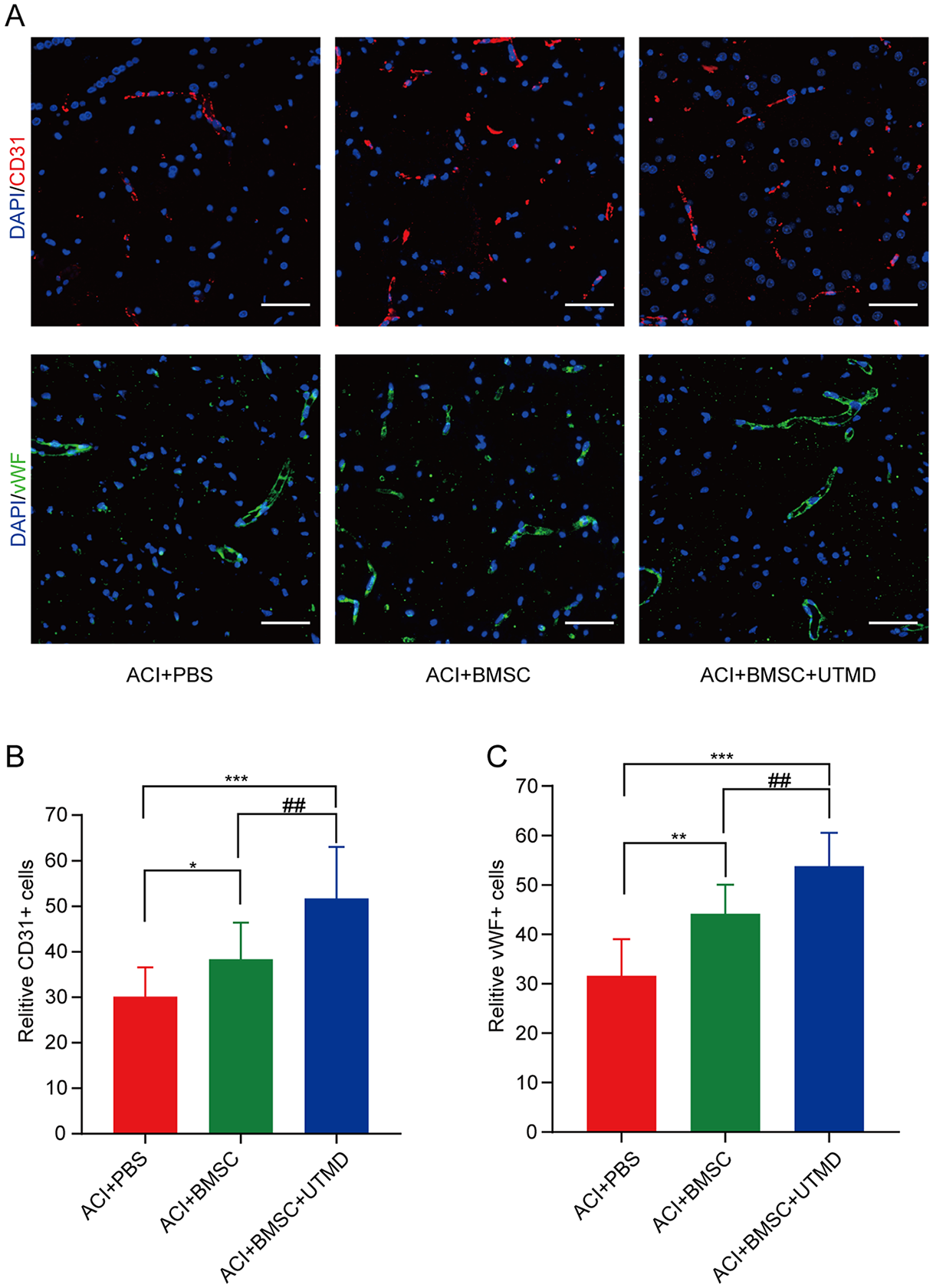
Angiogenesis biomarkers of CD31 and vWF positively stained vessels in brain infarct area by day 7 after MCAO. (A) The immunofluorescent microscopic findings (200×) of CD31+ cells (red color) and vWF+ cells in brain infarct area. (B) Statistical analysis of numbers of CD31+ cells, *P < 0.05, ***P < 0.001, compared with ACI+PBS group; ##P < 0.01, compared with ACI+BMSC group. The scale bars in right lower corner represent 50 µm. (C) Statistical analysis of numbers of vWF+ cells, **P < 0.01, ***P < 0.001, compared with ACI+PBS group; ##P < 0.01, compared with ACI+BMSC group. The scale bars in right lower corner represent 50 µm. vWF: anti-von-Willebrand-Factor; MCAO: middle cerebral artery occlusion; ACI: acute cerebral infarction; PBS: phosphate-buffered saline; BMSC: bone marrow–derived mesenchymal stem cells.
UTMD and BMSC Combination Treatment Reduces MMP8 Levels After MCAO
We investigated the cytokines that may contribute to the poststroke recovery associated with BMSC treatment by employing a rat cytokine antibody array (G-series). The levels of these cytokines in cerebrospinal fluid (CSF) samples were measured. Among the numerous candidates examined, the expression levels of matrix metalloproteinase 8 (MMP8) in the CSF were significantly elevated in MCAO rats compared with controls. The infusion of BMSC led to a reduction in MMP8 levels. The addition of UTMD to BMSC engraftment resulted in a further decrease in MMP8 levels, bringing them to a level comparable with that observed in nonlesioned control animals (Fig. 7). It is worth mentioning that abnormal levels of vascular endothelial growth factor (VEGF) and Fractalkine were likewise observed after MCAO, although their deviations were not as pronounced as that of MMP8. Notably, treatment with the combination of BMSC and UTMD restored the expression levels of these proteins to normalcy (Fig. 6).
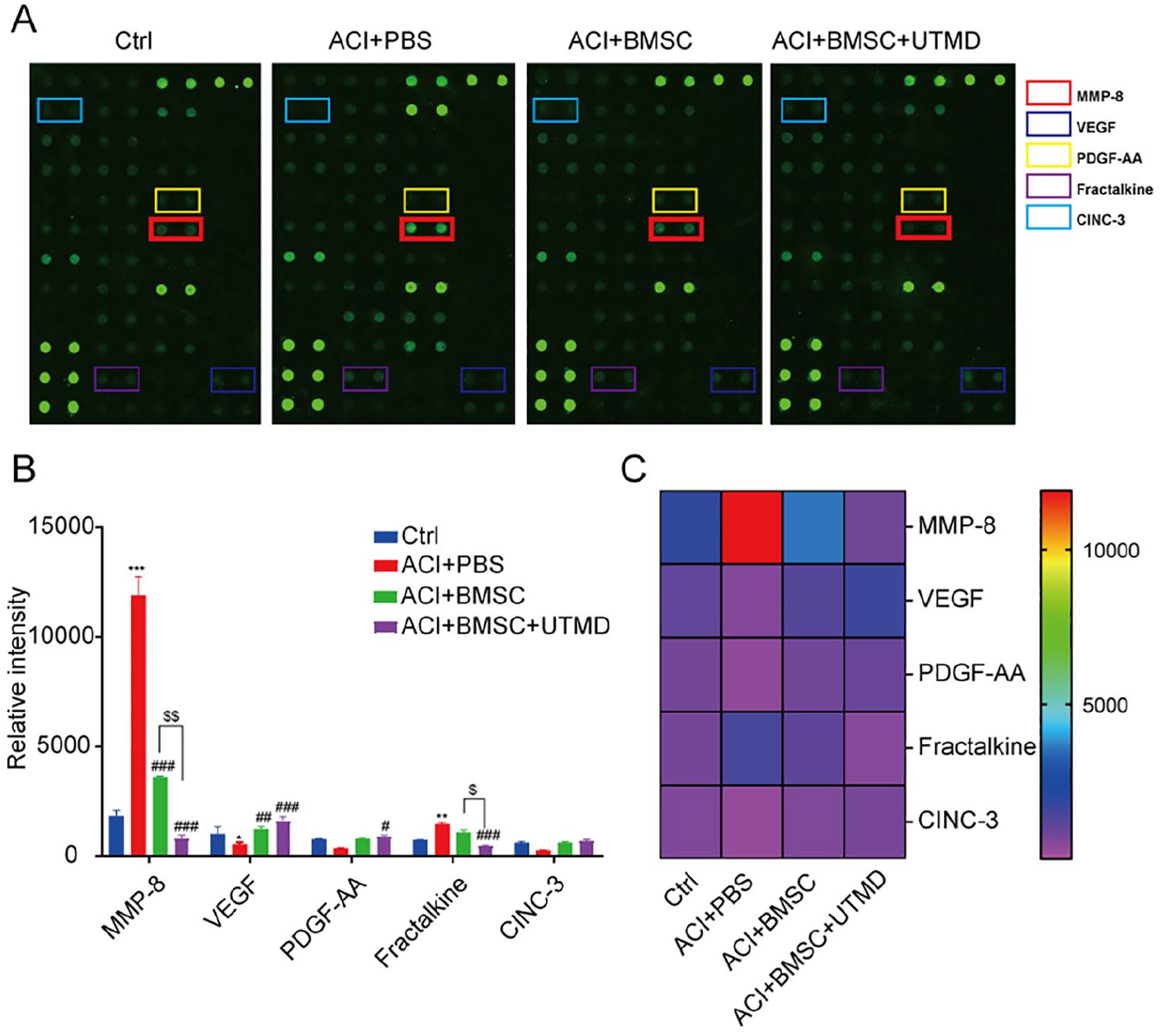
The potential candidate cytokines involved in UTMD mediated BMSC therapy in MCAO model. (A) Array format. Examples of rat cytokine antibody array (G-series) blots probed with the CSF sample in different groups. MMP8 was displayed in red box. (B, C) Bar graph and heat map represented the candidate cytokines which showed significant differences in the 4 groups. *P < 0.05, **P < 0.01, ***P < 0.001, compared with control group; # P < 0.05, ## P < 0.01, ###P < 0.001, compared with ACI+PBS group; $P < 0.05, $$P < 0.01, compared with ACI+BMSC group. UTMD: ultrasound-targeted microbubble destruction; BMSC: bone marrow–derived mesenchymal stem cells; MCAO: middle cerebral artery occlusion; MMP8: matrix metalloproteinase 8; ACI: acute cerebral infarction; PBS: phosphate-buffered saline.
Discussion
This study aimed to investigate the feasibility, safety, and effectiveness of intravenous delivery of stem cells using UTMD in rat models of ACI. The study found that UTMD can effectively and reversibly increase the permeability of the BBB, leading to increased homing of BMSC to the damaged area. This resulted in reduced histopathological alterations and improved neurological function.
Since 2009, multiple studies have reported that stem cells transplantation can effectively treat ischemic stroke20,21. Considering various factors such as feasibility and safety, intravenous administration of stem cells was chosen over arterial administration 22 . Although it is true that large cells like BMSC can be filtered in pulmonary capillaries 23 , it is important to note that a fraction of these cells (about 1%–5%) can still bypass filtration and enter the systemic circulation 24 . In our study, we utilized UTMD as a technique to transiently and reversibly modulate the BBB, facilitating enhanced entry of stem cells into the brain through the venous route and promoting their functional impact. This approach may increase the utilization of BMSC from venous transplantation.
Studies have demonstrated that BMSC transplantation improves neurological functions in animals with MCAO, indicating its potential for poststroke treatment8–10. The therapeutic mechanism of MSC in ischemic stroke is that MSCs secrete substances that activate the repair of damaged brain cells instead of replacing them. These substances create a favorable environment in the brain, promoting the migration and growth of cells derived from progenitor and neural stem cells. Studies have shown that MSCs have anti-apoptotic and pro-proliferative effects in ischemic regions, leading to improved behavioral performance in rats. This study provides confirmation that UTMD, when used in conjunction with BMSC treatment, leads to increased homing of BMSC to the injured brain, resulting in a reduction in brain infarct size and improved neurological function compared with BMSC treatment alone. Furthermore, at the cellular level, UTMD adjunct to BMSC treatment reduces necrosis in the brain compared with BMSC treatment alone, and increase neovascularization on injured area.
Ultrasound-targeted microbubble destruction has been shown to disrupt tight-junction protein complexes between endothelial cells, leading to an increase in the permeability of the BBB. This creates temporary openings between cells and enhances the transport of substances across the BBB through transcytosis11–13. In vivo studies using magnetic resonance imaging have confirmed the transient opening of the BBB by UTMD 25 . It is now widely agreed that permanent tissue damage can be avoided by using appropriate ultrasound settings and microbubble dosage 26 . In this study, we observed the temporary nature of BBB permeability at 72 h post-UTMD. However, we continued to observe an increase in the presence of BMSC in the brain from day 3 to day 7 following MCAO. It could be due to delayed migration of BMSC despite BBB closure or in-situ proliferation of BMSC. In addition, UTMD was found to stimulate a neuroinflammatory response in the BBB, leading to upregulation of chemokines, cytokines, and other trophic factors in animal studies. This response may provide more explanation for the increased and delayed homing of BMSC.
To investigate the therapeutic effects of UTMD-enhanced BMSC, we conducted a rat cytokine array analysis to identify potential cytokines that could contribute to neurological recovery following BMSC transplantation. BMSC migrated to the site of ischemic infarction by traversing the BBB and CSF, suggesting that the cytokines present in CSF might exert significant influence on the ischemic brain microenvironment. The cytokines present in CSF therefore may play important roles in the ischemic microenvironment of the brain. Of the multiple candidate cytokines in the CSF, only the level of MMP8 exhibited a notable increase in rats with MCAO as compared with sham-operated animals. Interestingly, MMP8 levels demonstrated significant reduction in the BMSC group and were further diminished in the BMSC+UTMD group. Earlier studies have established that matrix metalloproteases can induce disruption of the BBB by degrading its matrix proteins 27 . Notably, a specific study indicated an association between elevated serum MMP8 levels and heightened risk of small vessel stroke 28 . In addition, Han et al 29 . reported that intracerebroventricular microinjection of MMP8-specific shRNA lentivirus could diminish the magnitude of ischemia-induced brain injury. MMP8 levels have been found to be elevated in patients with ischemic stroke and are closely associated with both BBB leakage and inflammatory responses30–32. Furthermore, BMSC transplantation has the potential to downregulate MMP8 levels, possibly by influencing the differentiation of BMSC into multiple lineages33,34. Our findings align with these previous observations, underscoring the need for additional investigations into the connections between MMP8 and the therapeutic applications of BMSC and UTMD-enhanced BMSC.
Conclusion
This study demonstrates that UTMD can safely and effectively enhance the homing of intravenously administered BMSC to the injured brain by transiently augmenting BBB permeability. As a result, there was an enhanced survival of transplanted BMSC, reduced size of the ischemic lesion, and improved neurological outcomes in an ACI model. These effects may be attributed to the potential reduction in MMP8 levels within the CSF.
Supplemental Material
sj-docx-1-cll-10.1177_09636897231223293 – Supplemental material for Ultrasound-Targeted Microbubble Destruction Increases BBB Permeability and Promotes Stem Cell–Induced Regeneration of Stroke by Downregulating MMP8
Supplemental material, sj-docx-1-cll-10.1177_09636897231223293 for Ultrasound-Targeted Microbubble Destruction Increases BBB Permeability and Promotes Stem Cell–Induced Regeneration of Stroke by Downregulating MMP8 by Yun Bai, Yichao Du, Yin Yang, Thomas Wälchli, Paul E Constanthin and Fan Li in Cell Transplantation
Supplemental Material
sj-tif-2-cll-10.1177_09636897231223293 – Supplemental material for Ultrasound-Targeted Microbubble Destruction Increases BBB Permeability and Promotes Stem Cell–Induced Regeneration of Stroke by Downregulating MMP8
Supplemental material, sj-tif-2-cll-10.1177_09636897231223293 for Ultrasound-Targeted Microbubble Destruction Increases BBB Permeability and Promotes Stem Cell–Induced Regeneration of Stroke by Downregulating MMP8 by Yun Bai, Yichao Du, Yin Yang, Thomas Wälchli, Paul E Constanthin and Fan Li in Cell Transplantation
Supplemental Material
sj-tif-3-cll-10.1177_09636897231223293 – Supplemental material for Ultrasound-Targeted Microbubble Destruction Increases BBB Permeability and Promotes Stem Cell–Induced Regeneration of Stroke by Downregulating MMP8
Supplemental material, sj-tif-3-cll-10.1177_09636897231223293 for Ultrasound-Targeted Microbubble Destruction Increases BBB Permeability and Promotes Stem Cell–Induced Regeneration of Stroke by Downregulating MMP8 by Yun Bai, Yichao Du, Yin Yang, Thomas Wälchli, Paul E Constanthin and Fan Li in Cell Transplantation
Supplemental Material
sj-tif-4-cll-10.1177_09636897231223293 – Supplemental material for Ultrasound-Targeted Microbubble Destruction Increases BBB Permeability and Promotes Stem Cell–Induced Regeneration of Stroke by Downregulating MMP8
Supplemental material, sj-tif-4-cll-10.1177_09636897231223293 for Ultrasound-Targeted Microbubble Destruction Increases BBB Permeability and Promotes Stem Cell–Induced Regeneration of Stroke by Downregulating MMP8 by Yun Bai, Yichao Du, Yin Yang, Thomas Wälchli, Paul E Constanthin and Fan Li in Cell Transplantation
Footnotes
Acknowledgements
We are grateful to Professor Lianfang Du for the theoretical guidance with this study.
Author Contributions
FL conceived and designed the experiments. YB and YD performed the experiments. YB and YY performed the data analysis, statistical analysis, and finished the manuscript writing. TW and PEC provide technical support.
Availability of Data and Material
All data generated or analyzed during this study are included in the main text and the supplementary information files.
Ethical Approval
The Animal ethical committee of Shanghai General Hospital (SYXK[沪] 2019-0028) approved the study.
Statement of Human and Animal Rights
All experimental protocols were conducted in accordance with the guidelines of the National Institutes of Health Guide for the Care and Use of Laboratory Animals and were approved by the Ethics Committee of Shanghai General Hospital.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by following foundations: National Natural Science Foundation of China (Project No. 82272012), Natural Science Foundation of Shanghai (Project No. 20ZR1444600) and Special fund for science and technology innovation of Shanghai Jiao Tong University (Project No. YG2019ZDA27)
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
