Abstract
Low back pain (LBP) is a leading cause of long-term disability globally. Intervertebral disk degeneration (IVDD) is mainly responsible for discogenic pain in LBP-affected young patients. There is no effective therapy to reverse disease severity and IVDD progression. This study investigates the effect of human peripheral blood-derived mononuclear cells (PBMCs) on pain relief and life quality improvement in IVDD patients. The enriched monocytes of the PBMCs could differentiate into CD14 and CD206 double-positive M2 macrophages in vitro. Preclinical evidence in rats showed that the transplanted PBMCs exhibited anti-inflammatory and moderate tissue-repair effects on controlling IVDD progress in the rat model. The PBMCs significantly steered the aggrecan and type II collagen expressions and attenuated the pro-inflammatory cytokines in the affected disk. Based on the animal results, 36 patients with chronic low back pain (CLBP) were included in clinical trials. The control group was conservative care only, and the experimental group was platelet-rich plasma (PRP) and PBMCs intradiscal injections. We first confirmed the single lumbar disk causing the discogenic pain by provocative discography or magnetic resonance imaging (MRI). Discogenic LBP participants received one intradiscal injection of autologous PBMCs and followed for 6 months. Our clinical trial showed that patients’ LBP and disability were significantly ameliorated after the PBMCs transplantation rather than PRP. These preclinical and pilot clinical studies indicate that intradiscal injection of the enriched PBMCs might be a feasible and potential cell therapy to control pain and disability in IVDD patients.
Keywords
Introduction
Low back pain (LBP) is the most common cause of disability and brings enormous social impact globally 1 . In the affected young patients, LBP is mainly caused by gradual and irreversible damage to the intervertebral disk (IVD) 1 . Non-surgical analgesics and rehabilitation treatments for intervertebral disk degeneration (IVDD) show limited effects on preventing disease progression, and the disease status is prone to become chronic low back pain (CLBP) 2 . Few practical solutions are available to manage the symptoms of IVDD patients before receiving surgical disk replacement.
Intradiscal transplantation of mesenchymal stem cells (MSCs) shows promising long-term effects on controlling discogenic pain in CLBP patients3,4. Both autologous and allogenic MSCs are highly tissue comparable and have a high potential for anti-inflammation and tissue regeneration. Co-injection of MSCs and hyaluronic acid (HA) showed significant pain relief effects in the CLBP population within 3 years of follow-up 5 . However, the MRI examination did not exhibit tissue repair in the treated disk 5 . Whether the MSC administration provides effective opioid-sparing therapy for IVDD patients is still under investigation. Besides the MSCs, biomaterials and other cells for disk regeneration have been evaluated, such as HA alone, platelet-rich plasma (PRP), bone marrow aspirate concentrate (BMAC), and allogenic chondrocytes6,7. However, their efficacy is still inconclusive due to lacking qualified patient numbers, heterogeneous recruited populations, and single-arm experimental design.
Isolated autologous or allogenic MSCs require the cell expansion for clinical application. In contrast, BMAC harvest is a surgical approach to provide a viable and sufficient autologous cell source with minimal cellular manipulation. Some trials demonstrated that engrafting BMAC into the affected disk may moderately attenuate the pain score and improve the life quality8–10. It is postulated that CD34+ hematopoietic stem cells (HSCs) and MSCs in the BMAC may contribute to the anti-inflammation in IVDD patients11–13.
In addition to the stem cells, monocytes in the BMAC can differentiate into anti-inflammatory M2 macrophages after the migration into the destructive tissues14,15. Interestingly, previous studies illustrated that transplanting blood-derived M2 cells were safe and beneficial for neurological outcomes in stroke patients 16 and pain relief in osteoarthritis patients 17 , respectively. These preliminary results indicate that monocyte/M2 macrophages might control inflammatory diseases as in vitro amplified MSCs18,19.
Here, we examine the therapeutic efficacy of monocyte-enriched peripheral blood-derived mononuclear cells (PBMCs) for rescuing pain and disability in CLBP patients. We first conducted preclinical experiments in IVDD rats to evaluate the potency of anti-inflammation and tissue regeneration of the PBMCs. The clinical trials recruited discogenic CLBP patients and evaluated the effects of intradiscal PBMC injection on pain relief, disability improvement, and MRI differences, compared with those of the non-treated control and PRP group.
Materials and Methods
The PRP and Mononuclear Cell Preparations
For leukocyte-poor PRP preparation, we collected 10 ml blood from peripheral veins and the PRP was concentrated to 2 ml using an authorized centrifuge tube with separating gel (class II medical device; CPKit, Aeon Biotherapeutics, Taipei, Taiwan). For the PBMC preparation (peripheral blood cell purification [PCP]), 100 ml peripheral blood samples were taken and stored in blood bags with citrate phosphate dextrose adenine (CPDA; Japan Medical System, JMS, Singapore). We mixed 100 ml peripheral blood and 80 ml good manufacturing practice (GMP)-grade MoFi medium (comprising a chemical-defined medium for ex vivo human cells; class II medical device; Duogenic StemCells, Taichung, Taiwan) for 30 min. The mononuclear cells separation from mixed blood sample was automatically processed in a closed-tubing set (CS. 900.2, Cytiva, Marlborough, MA, USA) with the Sepax II machine (Cytiva) and Ficoll-Paque premium solution (Cytiva). The MoFi medium and Ficoll-Paque were removed by three times washing with normal saline. The final PBMC solution was adjusted to 2 ml, consisting of 6–9 × 107 PBMCs. The PRP and PCP processes were carried out at the point of care. The cell numbers of engrafted cells were analyzed by manual counting using a hemocytometer.
The Characterization of the PBMCs
We analyzed the percentage of monocytes in the PBMCs by flow cytometry (Accuri, Becton-Dickinson, USA) using forward scatter (FSC)/side scatter (SSC) or anti-CD14 fluorescein isothiocyanate (FITC)-conjugated antibody (Ab) (BioLegend, USA). To examine the M2 fate differentiation, we cultured the isolated PBMCs (about 2 × 106 cells) in 3 ml of Iscove’s modified Dulbecco’s medium (IMDM) containing 1% autologous plasma for 5 days culture at 37°C under 5% CO2. The ratios of M2 macrophages were identified at indicated times by measuring their specific surface markers with specific Abs, including anti-CD14 FITC Ab (a pan-monocyte and macrophage marker, BioLegend, clone M5E2, mouse IgG2a), and anti-CD206 PE Ab (an M2 marker, BD Pharmingen, clone 19.2, mouse IgG1)20,21. We complied with the manufacturer’s suggestions to perform the immunocytostaining protocols.
Animal Experiments
The experiment of transplanting human PBMCs in IVDD rats was approved by the Institutional Animal Care and Use Committee (IACUC) of National Chung Hsing University (NCHU; NCHU 109-159) in Taichung, Taiwan, and conducted in the Department of Life Sciences NCHU, Taichung, Taiwan.
Adult male Sprague–Dawley (SD) rats (BioLASCO, Taiwan) weighing 200–250 g were housed in a room with constant temperature (24–26°C) and humidity (40–60%) and had free access to food and water under a typical light cycle environment. The SD rats were divided into non-treated control (observation only, n = 10), IVDD (n = 10), and IVDD + PBMC (n = 10). Rats were individually sacrificed by isofluorane at 2 weeks (n = 5, for each group) and 4 weeks (n = 5, for each group) post-treatment.
The rats were anesthetized by 2%–3% isoflurane (Panion & BF Biotech, Taiwan) via intratracheal inhalation. The tail of the rats was first sterilized with povidone iodine and 70% alcohol. Three IVD of the coccygeal bones (Co 7/8, 8/9, and 9/10) of a rat tail were marked on the skin and disrupted with needle punctures on day 1. We used an 18G needle to completely penetrate the IVD vertically and rotated the needle 360° once to establish the IVDD and the endplate injury 22 . The enriched human PBMCs (5 × 105 cells) in 50 µl were transplanted into each damaged disk on day 2 post-injury. The disks were dissected aseptically at the two examination points (2 weeks and 4 weeks post-treatment; total disk number per group, n = 15). We allocated the 15 disks of each group for histology examination, including both histochemical (n = 7) and immunochemical staining (n = 5), and three disks for reverse transcription polymerase chain reaction (RT-PCR) examination.
Decalcification and Fixation
The tails were cut and fixed with 4% paraformaldehyde for 1 day. After rinsing in running tap water for 1 day, the tissues were decalcified for 5 days in the solution of Leica Decalcifier II (Leica Biosystems, IL, USA). The tail tissue became easily penetrated by a needle. We rinsed the tissue with tap water for 1 day, followed by standard dehydration and paraffin embedding. The disk explants were processed into 10 µm coronal plane sections.
Safranin O Staining and Alcian Blue Staining
For safranin O staining, the tissue slides were first stained with hematoxylin solution for 5 min and then washed with distilled water for 5 min. The samples were stained with 0.05% fast green solution (Sigma-Aldrich, USA) for 5 min and washed with 1% acetic acid for 15 s. Finally, the tissue will be stained with safranin O solution (TMS-009-C, Merck Millipore) for 5 min, followed by a wash with distilled water for 1 min.
The Alcian blue staining protocol is similar to the safranin O staining. After deparaffinization with xylene and gradual rehydration, 10 µm coronal plane sections were treated with 3% acetic acid for 5 min and stained with Alcian blue solution (TMS-010-C, Merck Millipore) for 30 min. The samples were rinsed with distilled water for 5 min and then ready for mounting.
Immunohistochemical Staining
The tissues were first incubated with antigen retrieval solution (immunohistological cytostaining [IHC] antigen retrieval solution, eBioscience, USA) for 30 min after deparaffinized with xylene and hydrated with gradient ethanol solutions (100%–70%). The tissue slides were rinsed with distilled water and incubated with a blocking buffer (Visual Protein, Taiwan) for 60 min. We treated the slides with the primary Ab in cell staining buffer (BioLegend, USA) overnight at 4°C. The primary Ab included mouse anti-human collagen II (Col II) mAb (1:100, Invitrogen, USA), rabbit anti-interleukin (IL)-6 Ab (1:100, Sigma-Aldrich, USA), and rabbit anti-tumor necrosis factor-alpha (TNF-α) Ab (1:100, Sigma-Aldrich, USA), Consequently, the tissue slides were washed with phosphate-buffered saline (PBS) and incubated with the corresponding secondary Ab for 30 min at room temperature. The secondary Abs against mouse or rabbit immunoglobulins were detected by the conjugated Alexa-488 or Alexa-647 fluorophores, respectively. The working secondary Abs (Thermo-Fisher, USA) were diluted 1,000 fold. Fluorescent images were captured using an upright microscope with a charge-coupled device (CCD) camera (80i, Nikon, Tokyo, Japan) and processed with the Nikon Elements software (Nikon) and ImageJ software. The relative fluorescent unit (RFU) was estimated from 10 regions of interest (ROI) in annulus fibrosus (AF) region of the disk and processed by ImageJ software 23 . The histopathological and RFU scoring of the IVDD was conducted blindly for analyzing the histology and fluorescent intensity, respectively, by a well-experienced veterinary pathologist according to the standards in a recent published literature 22 .
The Real-Time RT-PCR
The IVD tissues, including both nucleus pulposus (NP) and AF, were first homogenized and dissolved with 1.4 ml REzol C&T solution (KP200CT, Protech, Taiwan). After adding 140 µl 1-bromo-3-chloropropane into the REzol solution, the upper fraction (400 µl) was collected by 12,000 rpm centrifugation for 15 min at 4°C. We mixed the upper fraction with 400 µl isopropanol at −20°C overnight. RNA was precipitated by centrifugation (12,000 rpm, 15 min, 4°C), and then the RNA pellets were washed with 70% ethanol. The water in the RNA sample was vacuum-evaporated, and the residue pellets were dissolved in 50 µl diethyl pyrocarbonate (DEPC) water. The quantity and purity were determined by an ultraviolet (UV)/visible (Vis) spectrophotometer (NanoDrop ND-1000, Thermo-Fisher, USA).
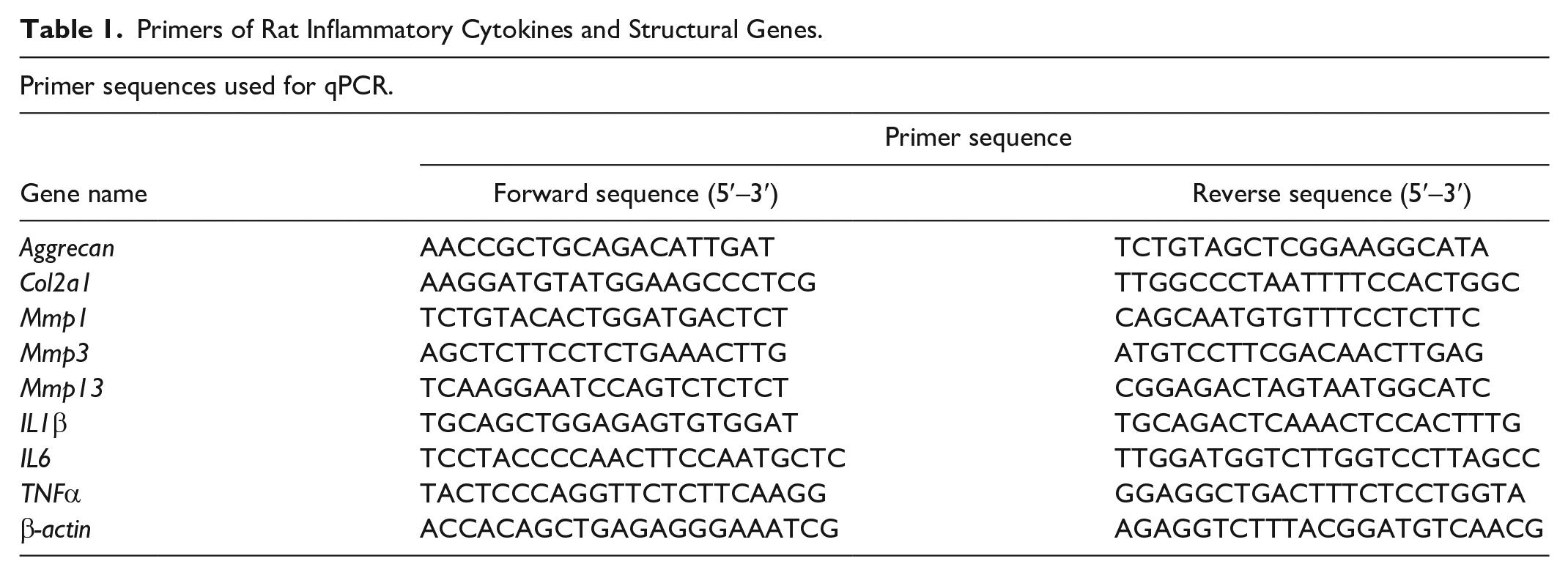
We detected the gene expression of the rat disks with real-time RT-PCR. The cDNA was synthesized from 3 µg total RNA using Moloney murine leukemia virus (MMLV) reverse transcription kit (RT-KIT, Protech, Taiwan). We mixed 20 ng of the cDNA with the specific primers (0.4 μM for each primer, listed in Table 1) and determined the gene expressions using PowerUp SYBR master mix (Applied Biosystems, USA) with a quantitative PCR (qPCR) machine (QuantStudio 6 Pro, A43180, Applied Biosystems, USA). The programs of the gene amplification were 1 cycle at 95°C 5 min, and the cycling setting was 40 cycles at 95°C 20 s plus 60°C 60 s. The final melt curve analysis condition is 95°C 15 s, 60°C 60 s, and 95°C 15 s. The concentrations of the qPCR amplicons were defined by melting curve, and the relative amount of mRNA in collected tissues was calculated by the comparative Cp method following normalization against the expression of β-actin, a housekeeping gene.
Primers of Rat Inflammatory Cytokines and Structural Genes.
Clinical Trials
We initiated the pilot clinical trials of intradiscal PBMC injection in two hospitals in Taiwan between April 2021 and December 2022. The protocols were approved by the Institutional Review Board (IRB), Ethic Committee of China Medical University (IRB No. CMUH 110-REC2-189), and Show Chwan Memorial Hospital (IRB No. 1050206 and 1091210). These trials were three arms, open-labeled pilot studies, including the conservative care without intradiscal injection (control), leukocyte-poor PRP and autologous PBMC treatments. The platelet concentration in the PRP was four to seven fold higher than that in blood, and the cell number of the PBMCs was 6–9 × 107 cells. We enrolled the patients with discogenic LBP on most days for at least 4 months and modified Pfirrmann grades III–VIII. A provocative discogram was applied to determine the pain origin if more than one IVD was degenerative under MRI examination (GE Signa Explorer 1.5T; GE Healthcare, Chicago, Illinois, USA). The exclusion criteria for the participation were facet joint syndrome, spondylitis, or vertebral fractures. Patients with a history of systemic diseases, such as cancer, autoimmune disease, or kidney failure, were also excluded from this study.
The sequence of MRI included both T1-weighted and T2-weighted scans, and the voxel size of the resolution setting was 5.0 × 0.59 × 0.59 mm. We analyzed the MRI scans utilizing PACS software (GE Healthcare Volume Viewer Plus 5.11). The height and volume were measured using T2-weighted sequences from the patient’s MRI scan. We assessed the height of the disk at its center in the midsagittal plane and determined the disk volume by multiplying the total of measured disk areas across each sagittal MRI layer by the 5.0 mm scan thickness24,25.
The enrolled participants all agreed with informed consent and were aware of the off-label use of medical devices and the risks of medical treatment. They took 2 ml intradiscal injection of PRP or PBMCs into an affected disk and were followed up at 3 and 6 months for efficacy and safety evaluations after a single intradiscal injection. Pain Visual Analog Scale (pain-VAS, scale 0-10) and Oswestry Disability Index (ODI, scale 0–50) scores were applied to assess pain relief, motor recovery, and life quality. Radiographs and MRI of the vertebrates obtained at the pre-treatment stage were scored by modified Pfirrmann grades I–VIII levels.
Adverse events of the PBMC treatment were recorded at each visit during the study. Any serious adverse event (SAE) was reported and treated within 24 h. Principle investigators (PIs) closely followed the patients with SAE during the trial period.
The Cell Transplantation Into the Human IVD
The PIs transplanted the isolated PBMCs into the IVD, and the CLBP patients had kept conscious during the operation. The skin was first disinfected with chlorhexidine, and sterile sheets were applied to create a safe field for the injection. The operative procedure included three steps; local anesthesia, precise needle insertion, and cell injection. Initially, a 23-gauge short spinal needle was inserted, and 0.5% lidocaine was injected into the skin, fascia, and muscle toward the target disk. Consequently, PIs inserted a 22-gauge needle at an angle of 25–35 degrees toward the target disk under fluoroscopy guidance. Once the needle tip passed the medial pedicle line, the fluoroscopy was changed to the lateral direction to ensure that the tip was aligned with the posterior vertebral body line. These two views were necessary to confirm the correct needle route through Kambin’s triangle without penetrating the dura medially or the nerve root laterally. We applied lidocaine again when the needle tip reached the AF. Once the injection site was confirmed, we slowly injected 2 ml of PRP or PBMCs (6–9 × 107 cells) into the affected disk. Some patients felt pain, soreness, or fullness. After the injection, the patient was closely monitored in the post-operative care unit for 1 h and then discharged with oral analgesics for 3 days.
Statistical Analysis
We used one-way or two-way analysis of variance (ANOVA) with Tukey’s post hoc to determine the significance of differences (P < 0.05) among the experimental groups, except the histological grading of the IVDD in rats and the clinical pilot studies. The statistical analyses of the histological IVDD grading and the pilot human trials were performed by nonparametric Kruskal–Wallis test or Wilcoxon signed-rank test 22 . The data were presented as mean ± standard deviation (SD). This study’s graphic creation and statistical analysis were conducted using Microsoft Excel (version 2019) or GraphPad Prism 9 (GraphPad, La Jolla, CA, USA).
Results
Enriched Monocytes From Peripheral Blood
We have established a PCP method for preparing mononuclear cells (MNCs)-enriched concentrate from peripheral blood using a GMP-grade MoFi medium and a density-gradient separation solution 19 . We collected 100 ml of peripheral blood from the patients and separated the PBMCs automatically with a single-use, closed-tubing device (Sepax II, Cytiva).
The average of collected PBMC numbers from 100 ml peripheral blood was 7.7 ± 1.5 × 107 cells (n = 20). MoFi treatment enriched the ratios of monocytes from 5.4 ± 1.5%–12.4 ± 2.5%, similar to our previous report (Fig. 1A) 19 . To examine the M2 macrophage differentiation, we cultured the PBMCs in IMDM with 1.0% auto-plasma in low-binding culture dishes. The PBMCs were maintained and analyzed at indicated times. Flow cytometry analysis in Fig. 1B revealed that freshly harvested monocytes in both groups (less than 4% on day 0) expressed CD14 but barely expressed CD206, a specific mature M2 macrophage marker. Treating monocytes (CD14+ cells) with M-CSF plus IL-4 steered the polarization toward CD206+ M2 macrophages at a ratio of 19.0 ± 5.7% on day 1 (n = 6), 59.5 ± 7.2% on day 3 and 81.0 ± 6.4 % on day 5, respectively (n = 6, Fig. 1B). The other nine patients in PBMCs’ group showed the similar results of macrophage differentiation. The M2 differentiation potency of collected monocytes was consistently recapitulated in recruited donors (Fig. 1C).
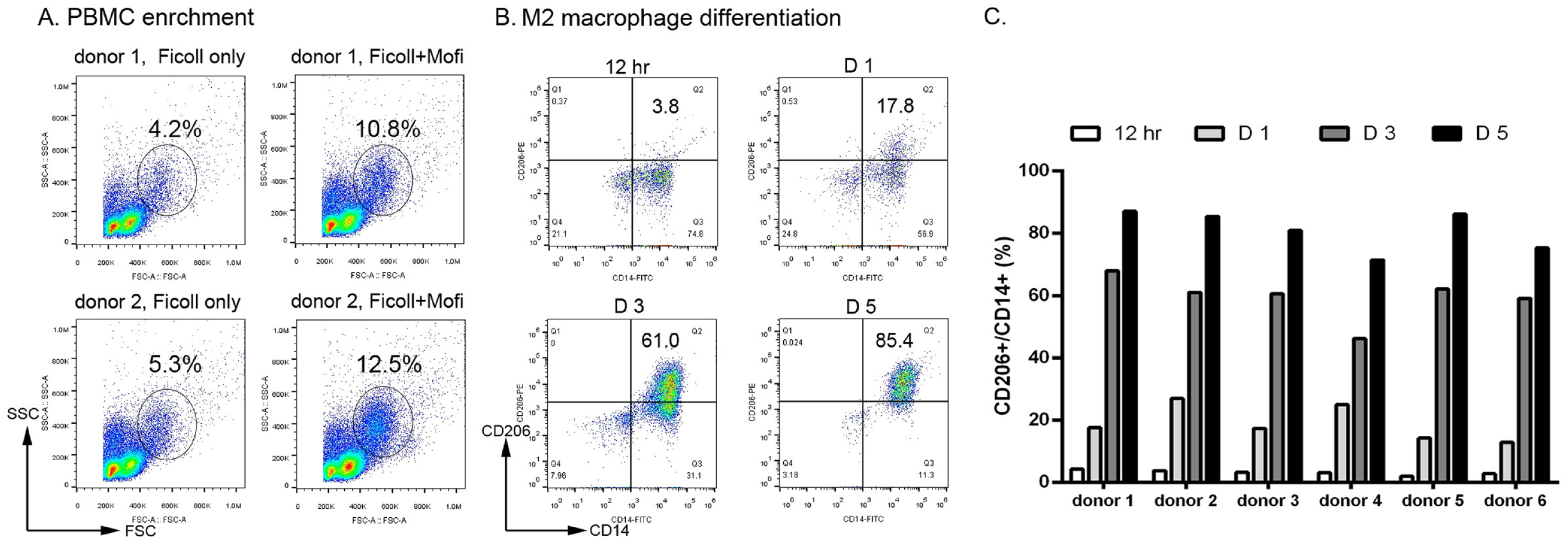
The MoFi-enriched monocytes and the M2 macrophage differentiation in collected PBMCs. (A) Flow cytometry marked the monocyte population by the cell size (FSC) and granule intensity (SSC). Pre-treating the PBMC with MoFi medium enriched the monocyte populations in two donors’ PBMCs. (B) After the in vitro cell culture, the potency of the M2 macrophage differentiation was analyzed by the cytostaining with CD14 (X-axis) and CD206 (Y-axis) with flow cytometry. The ratios of CD14/CD206 double-positive cells represented the percentages of M2 cells in the collected monocytes. (C) The trend of M2 differentiation was consistent in six collected PBMCs of donors.
The Tissue Repair Activities of the PBMCs in the IVDD Rat Model
A rat IVDD model was established by needle penetration into the IVD of the tail. Severe and irreversible disk disruption was observed till 4 weeks post-operation 22 . The animals were all alive. No infection or severe surgical complication was recorded during the experiment period. Both safranin O (Fig. 2A) and Alcian blue staining (Fig. 2B) revealed that in the IVDD tissues, the NP matrix was lost, and the morphology of the NP was completely disorganized, compared with the non-operative control group (control). The AF was also ruptured and intact ring-shaped collagen lamellae were not reserved (Fig. 2A, B). To evaluate the therapeutic potential of PBMCs on IVDD, we injected 5 × 105 cells of human PBMCs into the lesioned disk on day 2 post-injury. In contrast to the non-treated IVDD group, the PBMC injection (IVDD + PBMCs) moderately, but not significantly, attenuated the inflammation and tissue damage after 2 and 4 weeks of engraftment (Fig. 2C, P = 0.17 and 0.26, respectively). We illustrated that PBMC transplantation could partially attenuate the disk degeneration caused by mechanical disruption and moderately maintain the disk integrity in the IVDD rat model (Fig. 2C).
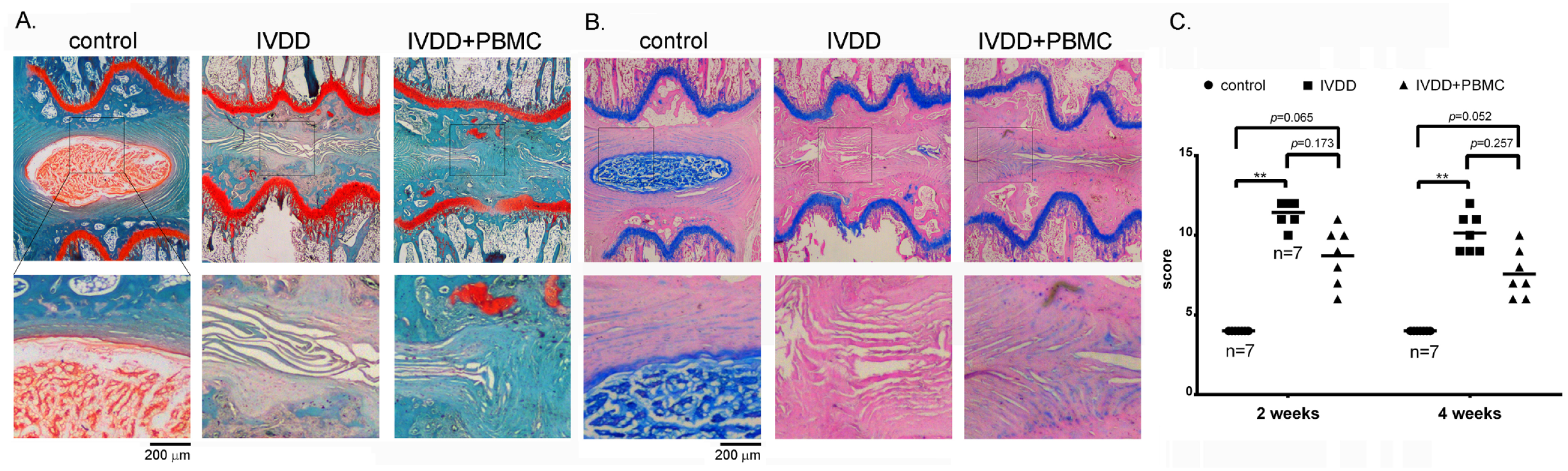
Transplantation of human PBMCs attenuated IVDD in the rat model. (A, B) The disk and endplate cartilage of rat tails were stained with Safranin O (A) and Alcian blue (B), showing the distributions of glycosaminoglycans (GAG). Lower panels were enlarged views of the squares in the upper panels, illustrating the border of the nucleus pulposus and annulus fibrosus. The IVDD model in rats was established by needle acupuncture, and 5 × 105 human-enriched PBMCs in 50 µl were transplanted into the damaged disk at day 2 post-injury (IVDD + PBMC). (C) Histopathological scoring of the disks showed that providing PBMCs moderately, but not significantly, alleviated the tissue disruption and degradation by mechanical acupuncture at 2 weeks (P = 0.17) and 4 weeks (P = 0.26) post-injury (NS, Kruskal–Wallis test). IVDD + PBMC vs IVDD, NS, no significance, P > 0.05. Disk number, n = 7, for each group.
We next detected the expression of rat Col II, IL-6, and TNF-α to examine the degree of tissue damage and inflammation (Fig. 2). In contrast to those of the control group, the AF cells of the disrupted disk exhibited low Col II expression, but elevated inflammatory IL-6 and TNF-α protein expressions (Fig. 3A). Transplanting PBMCs partially rescued the Col II expression and ameliorated the IL-6 and TNF-α expressions (Fig. 3A). The relative fluorescent intensity of the tissues was quantified, shown as fold changes of the staining results in Fig. 3A, B.
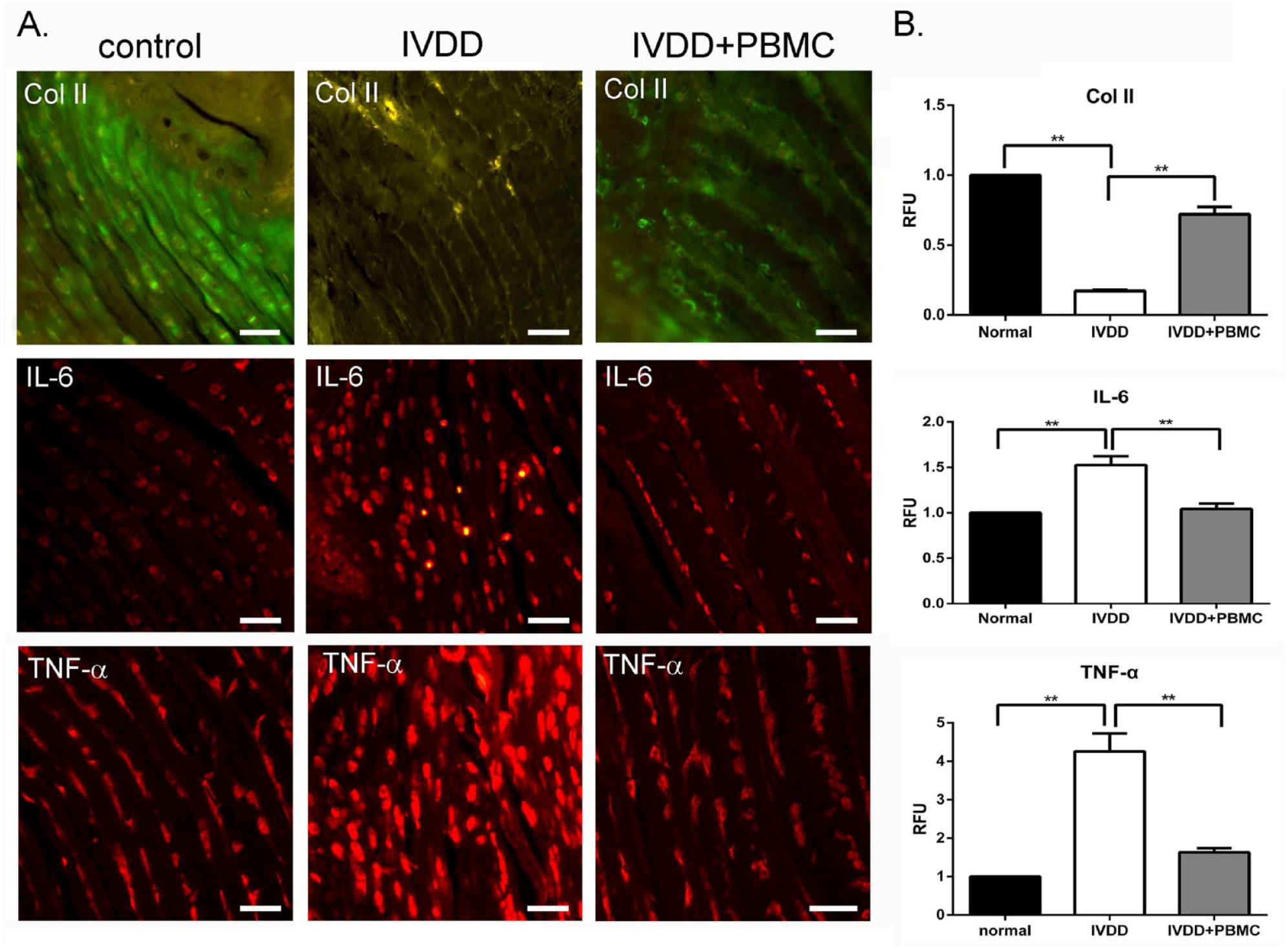
Transplantation of human PBMCs attenuated the inflammation of IVDD tissues. (A) Immunohistological cytostaining (IHC) showed the endogenous rat type II collagen (Col II), interleukin-6 (IL-6), and tumor necrosis factor-α (TNF-α) in control, damaged disks (IVDD), and the damaged disks with engrafted PBMCs (IVDD + PBMCs). Scale bar, 30 µm. (B) The fluorescent intensities (relative fluorescence unit, RFU) of the IHC in Fig. 3A were semi-quantified by Image J software. The positive signal of control in Fig. 3A was set as 1.0 RFU. **P < 0.01. Disk number, n = 5, for each group.
Aggrecan, Col I, and Col II are the dominant proteoglycan and proteins in the annulus of IVD, respectively 26 . Compared with the control of the disks, applied acupuncture to the disk triggered the endogenous aggrecan RNA expression (P < 0.05) but moderately reduced the Col II mRNA expression (Fig. 4A). These findings were similar to the RNA expression profiles in previous studies27,28. Injection of PBMCs augmented the structural Col II and aggrecan mRNAs, partially accounting for the tissue restoration by PBMCs injection in Figs. 3 and 4A.
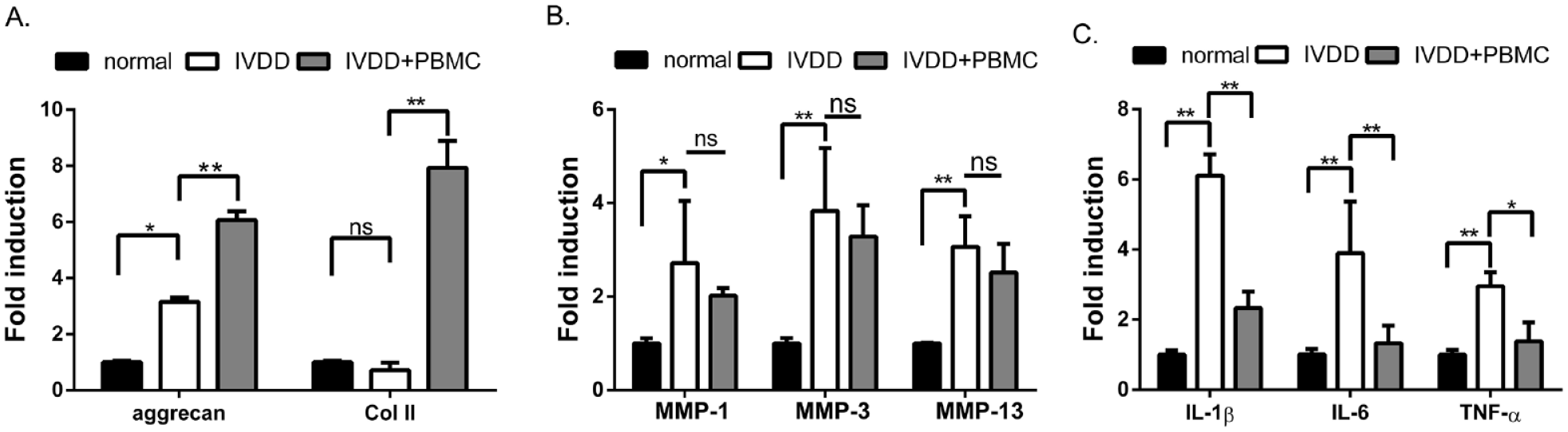
PBMC transplantation ameliorated the inflammatory cytokines in IVDD tissues. The total RNAs were extracted from the control disk tissues (control), the degenerative disks (IVDD), and PBMC-engrafted degenerative disks (IVDD + PBMC). The basal expression of the endogenous genes in control disks was set as 1.0. The relative induction of the structural genes (aggrecan and type II collagen) (A), matrix metalloproteinases (MMP-1, MMP-3, and MMP-13) (B), and pro-inflammatory cytokines (IL-1β, IL-6, and TNF-α) (C) were estimated by real-time RT-PCR. *P<0.05. **P<0.01. ns, no significance.
The mechanical disruption of the disk also brought the gene induction of inflammatory proteins, such as matrix metalloproteinases (MMPs) (Fig. 4B), IL-1β, IL-6, and TNF-α in the disk cells (Fig. 4C). Transplanting PBMCs inhibited the inflammatory cytokines (P < 0.05) and moderately attenuated the MMPs’ expressions (P > 0.05) (Fig. 4B, C). These results suggest that the engrafted PBMCs may attenuate the rapid degradation of the injured disk through their anti-inflammatory and tissue regenerative activities.
A Human Pilot Study of MoFi-Treated PBMCs for Controlling IVDD
Based on the immuno-modulatory potency of the PBMCs, we organized investigator-initiated trials (IITs) in two medical institutes to evaluate the cells’ safety and efficacy in controlling IVDD with modified Pfirrmann grades III–VIII. The flowchart of the human trial is illustrated in Fig. 5.
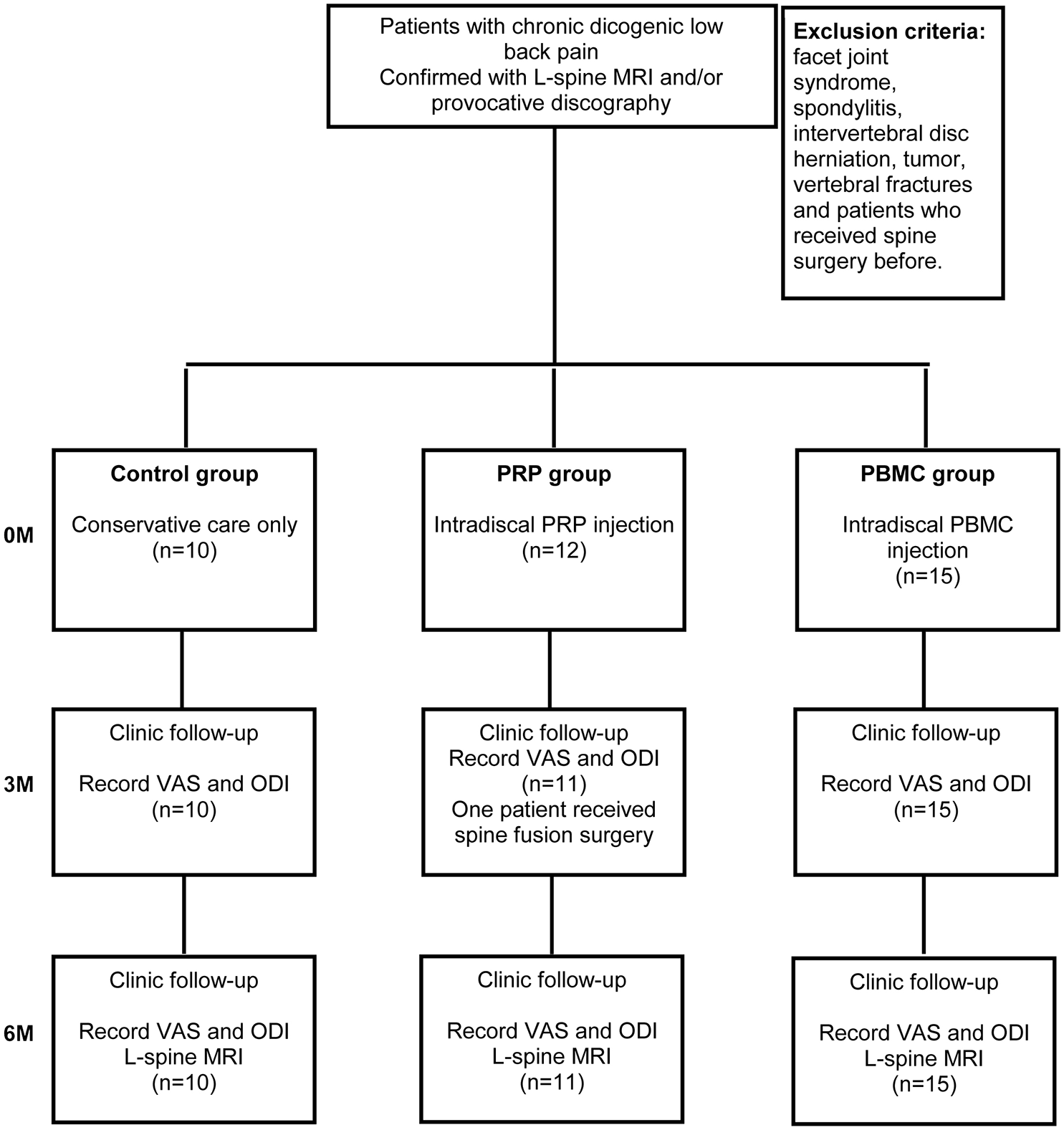
The flowchart of the clinical trial design.
The baseline profile of the recruited patients is summarized in Table 2. Thirty-six per-protocol patients were assigned to non-treated control (n = 10), PRP-treated (n = 11), and PBMCs-treated group (n = 15) (Fig. 5). PRP and PBMCs were injected into one level of the affected disk according to the MRI examination. Provocative discography examination was applied to confirm the level of pain origin when multiple injured disks were discovered (n = 5, in PBMC group). Patients were treated once with autologous PRP or PBMCs in 2 ml (total 6–9 × 107 cells in 2 ml). All patients were followed for 6 months of observation and examined with MRI at 6 months post-treatment. The averaged values and the statistical analyses of the demographic characteristics, basal VAS, and ODI in this trial were summarized in Table 2.
Baseline Characteristics of the Patients.
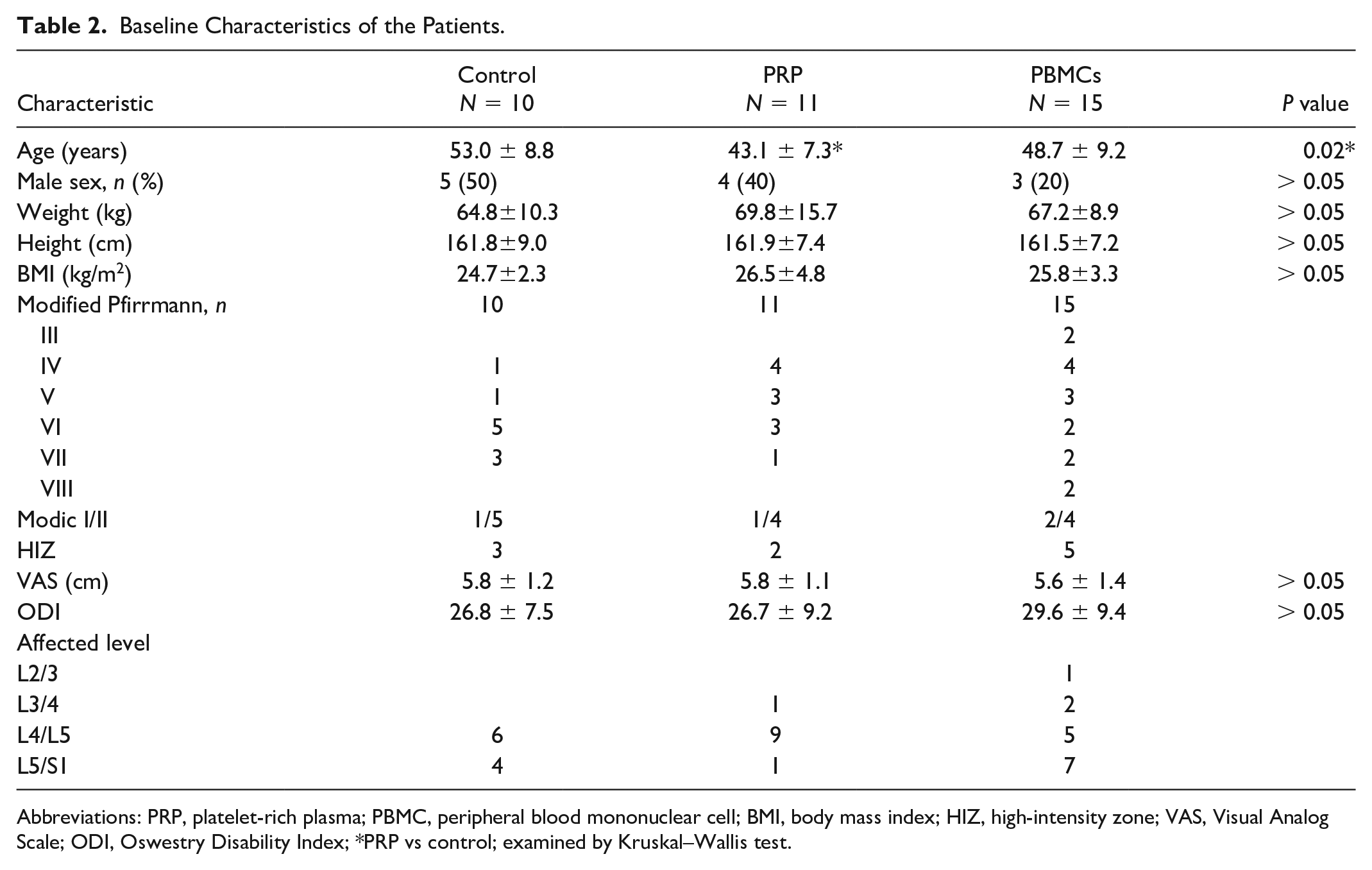
Abbreviations: PRP, platelet-rich plasma; PBMC, peripheral blood mononuclear cell; BMI, body mass index; HIZ, high-intensity zone; VAS, Visual Analog Scale; ODI, Oswestry Disability Index; *PRP vs control; examined by Kruskal–Wallis test.
Our results revealed that compared with the control at 3 and 6 months, only the PBMCs-treated patients, but not the PRP group, reported significant improvement in their pain (Fig. 6A, B, Table 3) and ODI scores (Fig. 6C, D, and Table 3). Statistically, the PBMC treatment exhibited superior improvements in ODI scores than the PRP treatment (P < 0.05, Fig. 6C, D). Two control patients, but none of the treated groups, took lumber interbody fusion operation after 6 months of enrollment. Interestingly, we found that 40% of control (4/10) and 18% of PRP-treated patients (2/11) were below 50% pain reduction (less than 50% improvement, Y-axis in Fig. 6B) at 6 months post-treatment (Fig. 6B). ODI investigation revealed that none of the control showed 50% improvement at 3 months, and 60% (6/10) were below 50% recovery at 6 months (Fig. 5D, control). Surprisingly, all the PBMC-treated patients achieved over 50% VAS and ODI improvements, suggesting that the injection of PBMCs might provide persistent and stable therapeutic effects on controlling the clinical symptoms of IVDD.
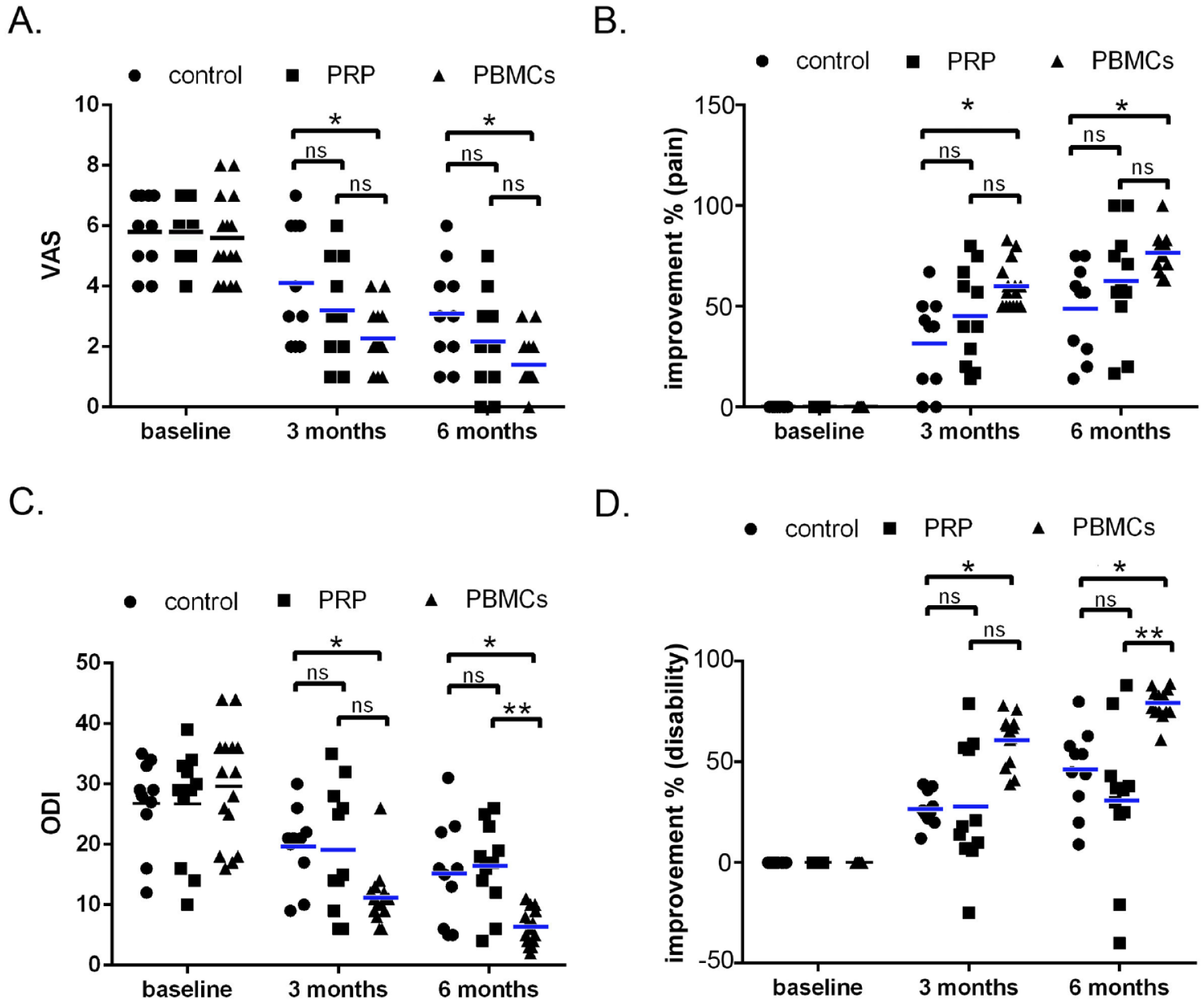
The pain relief and the improved life quality in treated IVDD patients with PBMCs. IVDD patients (n = 36) were divided into non-treated control (n = 10), PRP-treated group (n = 11), and PBMC-treated group (n = 15). We injected 2 ml PRP or PBMCs intradiscally in a level under a fluoroscope. The pain-VAS scores (A) and the ODI scores (C) were recorded at pre-treatment (baseline), 3 and 6 months post-treatment. The percentages of reduced pain-VAS and ODI scores, compared with baseline, were shown as improvement percentages (Y-axis in panels B and D). *P<0.05. **P<0.01. ns, no significance. VAS, Visual Analog Scale; ODI, Oswestry Disability Index.
Outcome Measures at Baseline and 3, 6 Months.
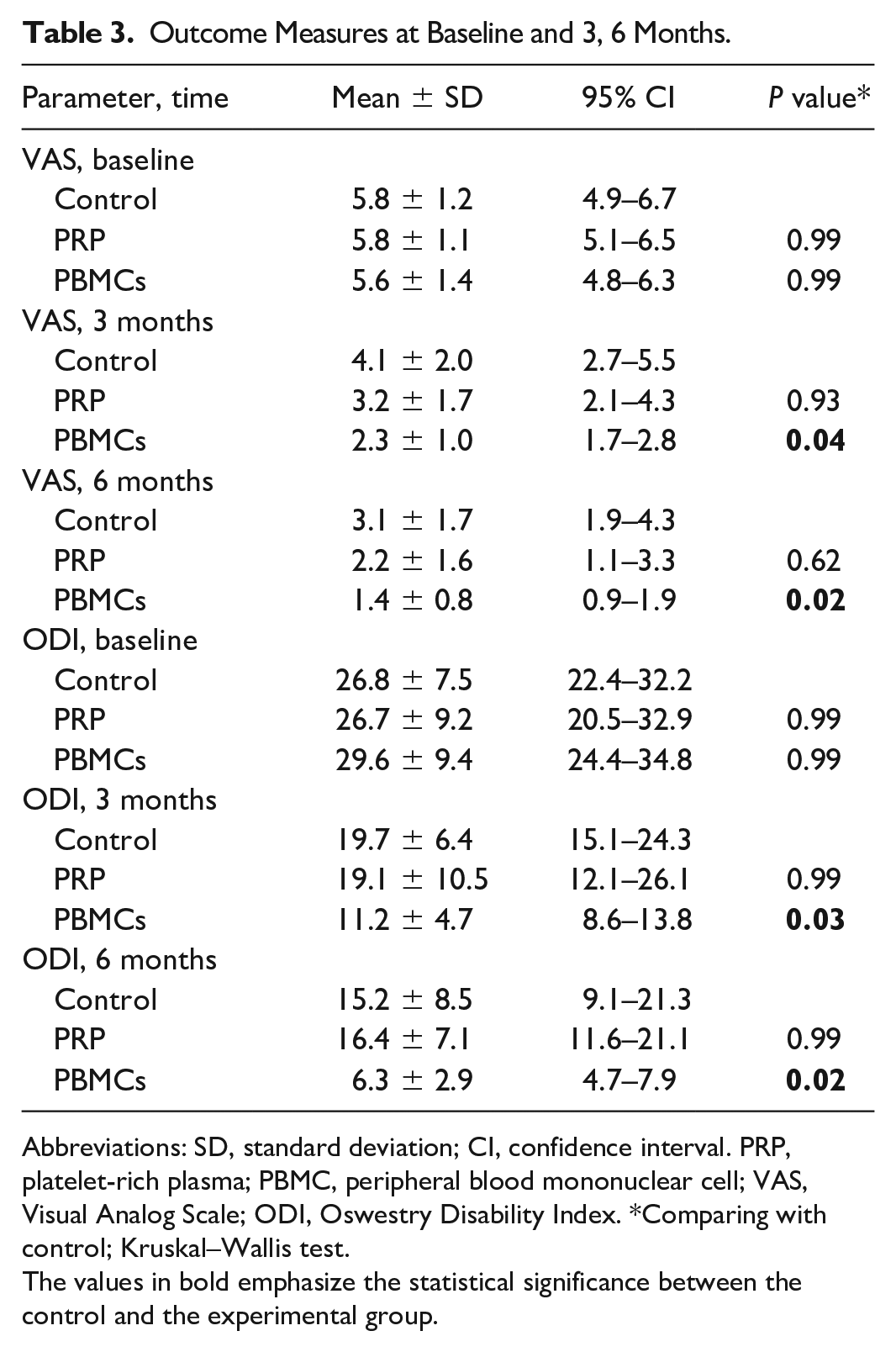
Abbreviations: SD, standard deviation; CI, confidence interval. PRP, platelet-rich plasma; PBMC, peripheral blood mononuclear cell; VAS, Visual Analog Scale; ODI, Oswestry Disability Index. *Comparing with control; Kruskal–Wallis test.
The values in bold emphasize the statistical significance between the control and the experimental group.
Notably, one patient with classical modic change type 2 (MC2) was examined with the discography (Fig. 7A), and the affected L4/L5 was treated with the PBMCs intradiscal injection (Fig. 7B). At 6 months of PBMC treatment, the T1 signal of MRI showed no difference, suggesting that the infiltrated adipose tissue in both L4 and L5 spinal segments were not altered (Fig. 7A, B, left two panels). In contrast, the T2 weight images showed that the clear “claw-sign” signals in both L4 and L5 bones (Fig. 7A, right panels) were obviously attenuated (Fig. 7B, right panels), suggesting that the bone edema and inflammation were effectively reduced by PBMC injection.
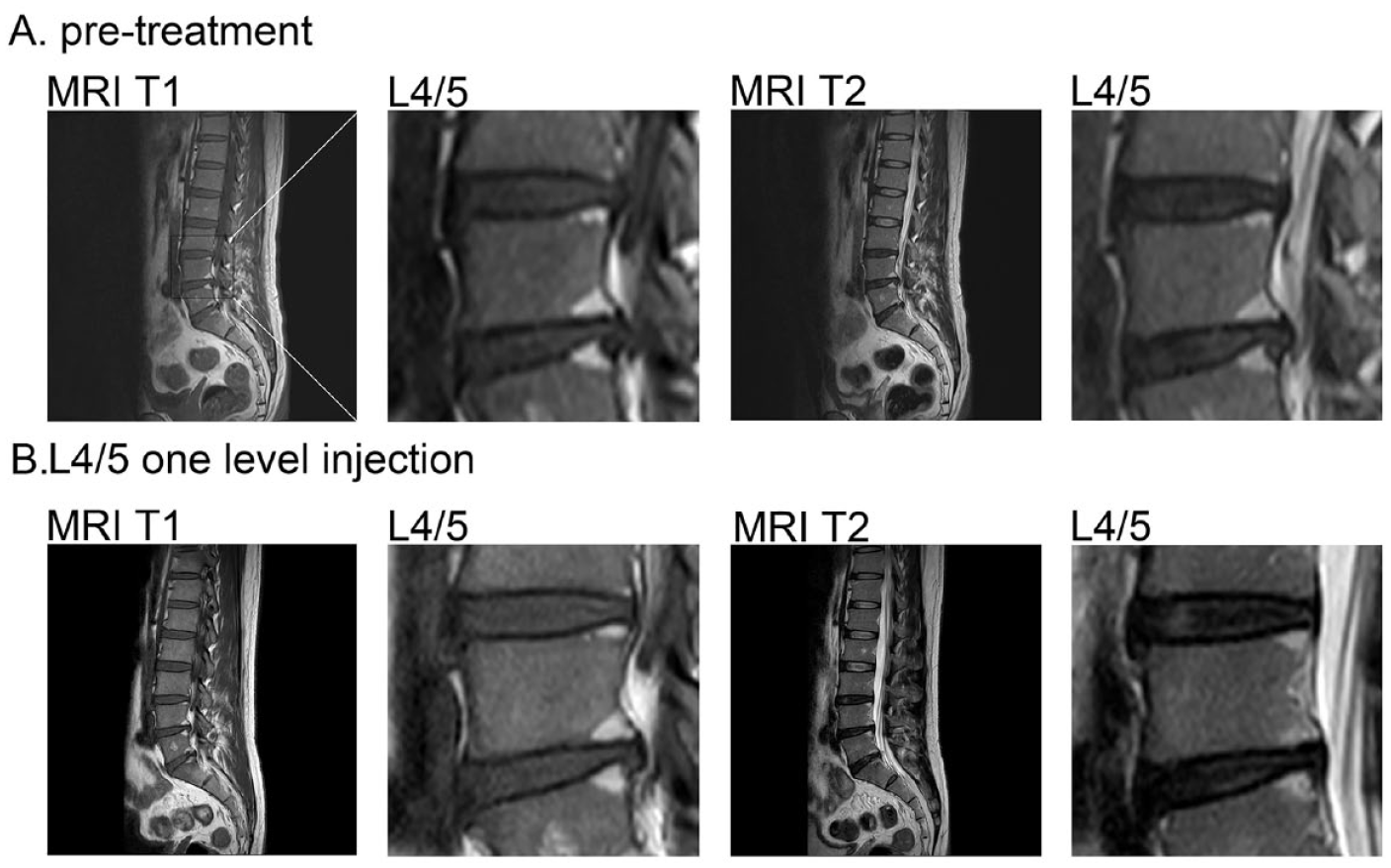
Bone edema reduction in a PBMC-treated patient. A recruited patient has severe pain and showed classic modic change type 2 (MC2) features under MRI examination (A). Both T1 FSE and T2 FrFSE of MRI at dorsal L4 and L5 segments exhibited clear white claw-sign signals, indicating vertebra degeneration with classical MCII phenotype. (B) After intradiscal PBMC injection into the disk between L4 and L5 (L4–L5), the T2 MC signals were reduced (right panels) but the T1 MC signals were still intact (left panels) at 6 months post-treatment. MRI, magnetic resonance imaging. FSE, fast-spin echo. FrFSE, fast recovery fast-spin echo. L4, the fourth lumbar spine vertebra.
MRI examinations revealed that the affected disks were degenerated continuously, evidenced by the increased Pfirrmann grading, and reduced disk height and disk volume in the control group within the experimental period (Fig. 8A and Table 4). We found that neither PRP nor PBMC treatments significantly reversed disk degeneration but sustained the integrity of the disk architecture (Fig. 8B, C, Table 4). In terms of the modified Pfirrmann grading, two patients showed one-grade improvement in the PBMC group (both from grades 4 to 3, Fig. 8C, red arrow). Representative MRI scans were provided to illustrate the shifting of the grading (Fig. 8D–F). These MRI findings indicate that the primary pain-relief effects of the PBMCs might result from their immune-modulatory activities on reducing edema-associated bone lesions, rather than disk reconstruction.
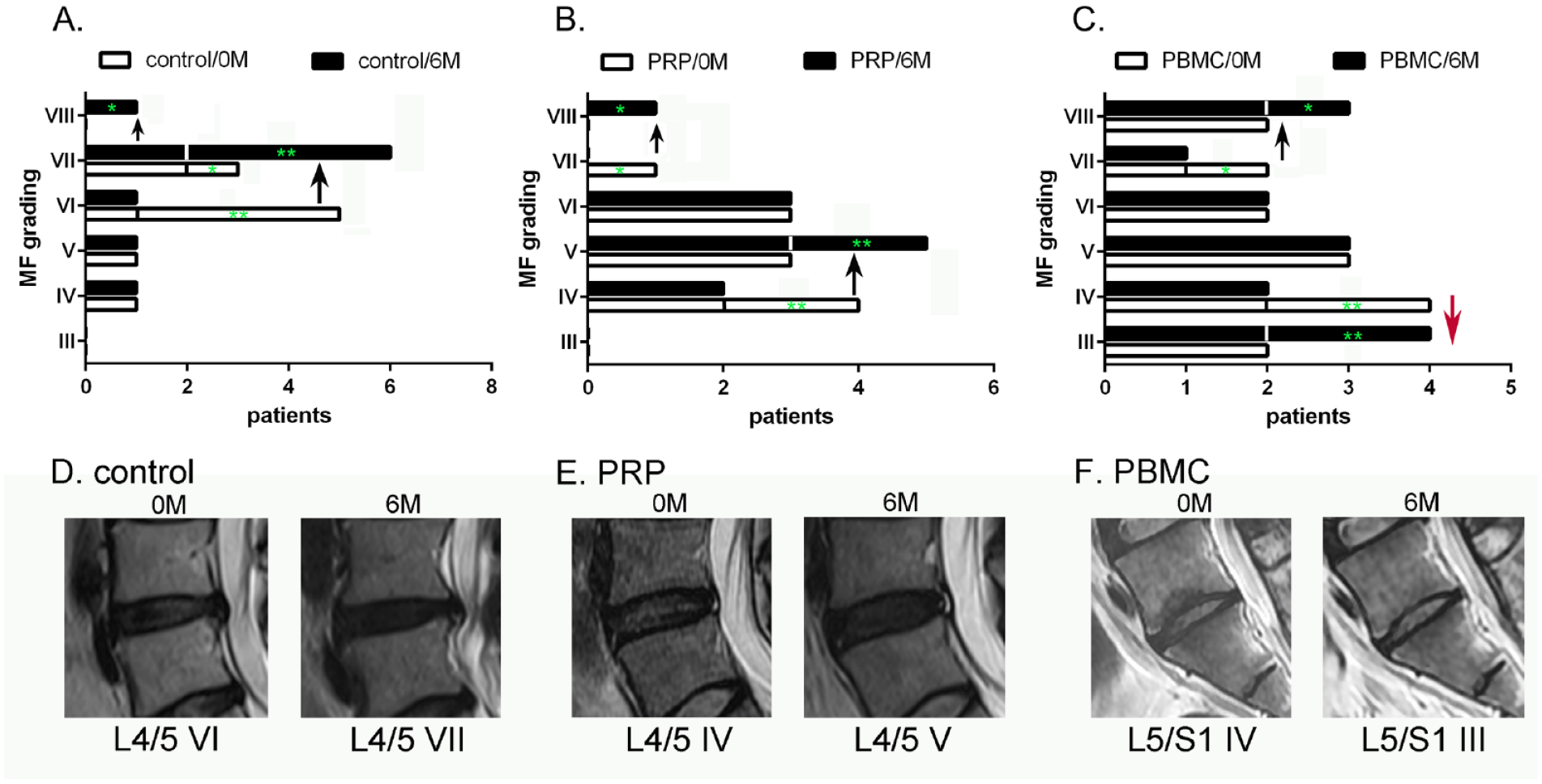
The measures of the modified Pfirrmann (MF) grade in control and treated patients. We list the numbers of patients with individual MF grading at pre-treatment (0M) and 6 months (6M) post-treatment in control (A), PRP (B), and PBMCs (C) groups. The MF upgrading and down-grading are indicated by black and red arrows, respectively. (A) In the control group, four-grade VI patients became grade VII (**), and one-grade VII patient became grade VIII (*) at 6M post-treatment. (B) In the PRP group, two-grade IV patients became grade V (**), and one-grade VII patient became grade VIII (*). (C) In the PBMC group, one-grade VII patient became grade VIII (*), and two-grade IV patients converted to be grade III (**, red arrow). The representative T2 disk images of the control, PRP and PBMCs were illustrated (D, E, F), respectively.
MRI Measures at Baseline and 6 Months Post-Treatment.
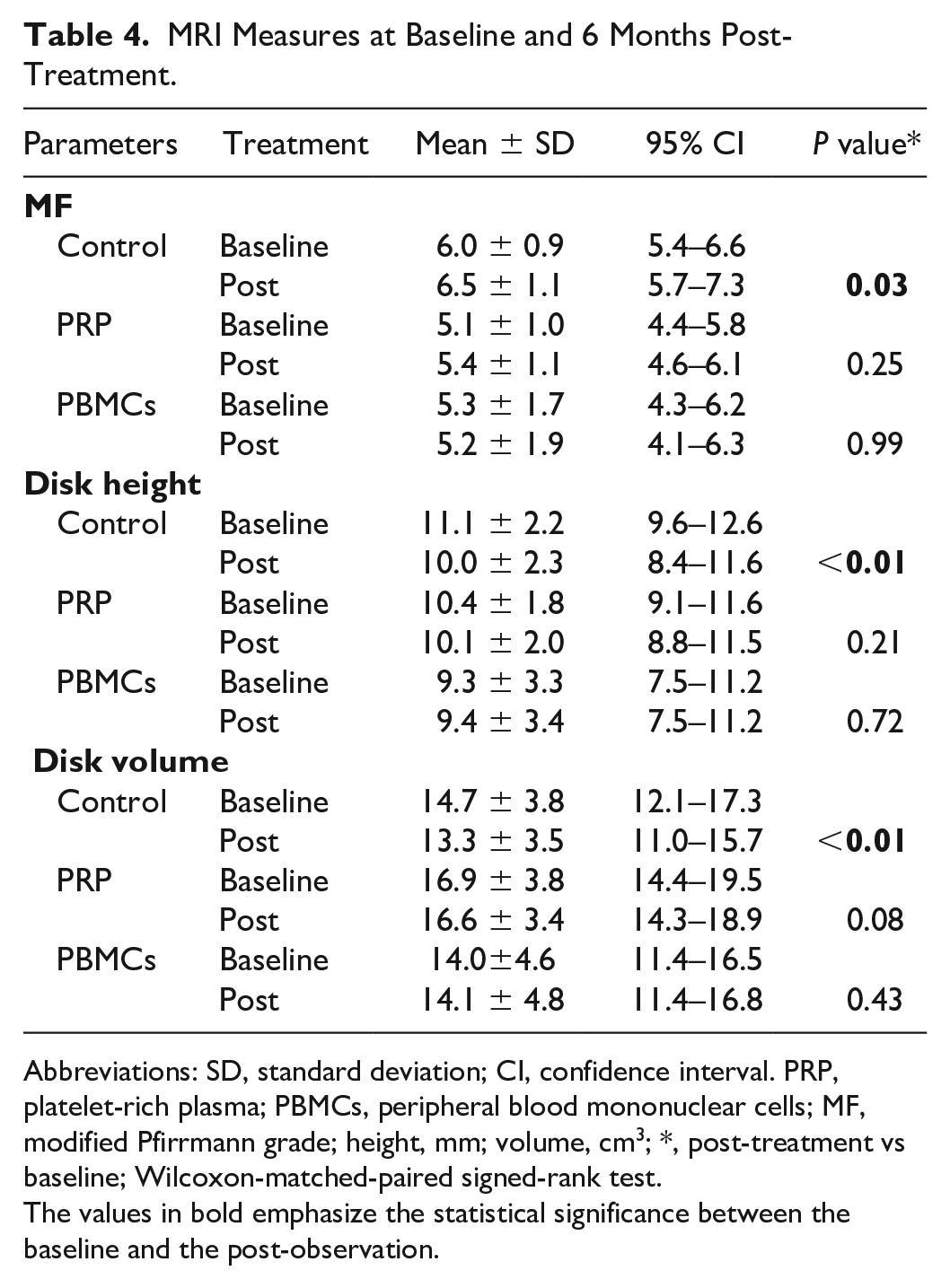
Abbreviations: SD, standard deviation; CI, confidence interval. PRP, platelet-rich plasma; PBMCs, peripheral blood mononuclear cells; MF, modified Pfirrmann grade; height, mm; volume, cm3; *, post-treatment vs baseline; Wilcoxon-matched-paired signed-rank test.
The values in bold emphasize the statistical significance between the baseline and the post-observation.
Finally, we found no severe adverse effects of PRP or PBMC injection during the trial period. The minor adverse effects in PRP and PBMCs cohorts were back pain and soreness, lasting 1–3 days after intradiscal injection (8/11, 72.7%, in PRP; 10/15, 66.7%, in PBMCs). A similar frequency of the AE was found between the PRP and PBMC groups (P>0.05). Persistent pain/swelling over 1 week, burning sensation, arthralgia, muscle spasms, infection, or extra-care of hospitalization caused by the PRP or cell transplantation were not observed.
Discussion
M2 macrophages are well-documented for their anti-inflammatory activities and tissue regeneration29,30. M2 cells can attenuate the production of T cells-mediated pro-inflammatory cytokines, such as IL-6, IL-13, interferon gamma (IFN-γ), and TNF-α31,32. We have demonstrated that intra-articular injection of the M2 macrophages effectively controlled the adjuvant-and collagen-induced arthritis17,19. We also provide pilot clinical evidence showing that the MC injection might be a feasible and stable treatment for controlling pain and motor disability in patients with knee osteoarthritis 17 . Here, we further illustrate that the enriched PBMCs exhibited anti-inflammatory activities and therapeutic potency for attenuating IVDD.
Acupuncture-triggered disk degeneration is accompanied by disrupted disk architecture, degraded disk matrix, and elevated inflammatory cytokines 33 . Fibrotic transformation of the affected rat disk was initiated by the aggrecan and collagen I induction within the first 2–4 weeks27,28,34. We discovered that PBMC injection promoted tissue regeneration by enhancing aggrecan and Col II mRNA, and ameliorated the inflammatory cytokines expression, such as IL-1β, IL-6, and TNF-α. According to our previous finding for the characterization of the enriched PBMCs 17 , we postulate that the engrafted monocytes may differentiate into M2 macrophages in the disk, and the secreted endogenous indoleamine 2,3-dioxygenase 1 (IDO-1) may play one of the key factors involving in the immuno-modulatory activities.
NP cells and MSC coculture experiments revealed that transplanted MSCs might release transforming growth factor-β (TGF-β) to enhance the NP cells proliferation and the production of extracellular matrices (ECMs), such as aggrecan and collagens 35 . It is intriguing to investigate whether TGF-β also be a critical mediator for the PBMC-enhanced ECM production from the affected NP cells. Moreover, cell fate tracing of engrafted PBMCs, knocking down the expression of Col II or other candidate molecules will provide insight for the detailed underlying mechanism of action (MOA) for tissue regeneration. The MOA investigation in preclinical studies will substantially benefit the designing of cell manufacturing and clinical protocols in humans.
Inflammatory cytokines are highly associated with IVDD and pain generation 33 . Several clinical trials demonstrated that anti-cytokine agents, such as infliximab, a TNF-α Ab, mitigated the pain score of IVDD patients in a short-term treatment window. However, randomized-controlled trials (RCTs) demonstrated that one-shot intravenous infusion of infliximab did not significantly improve lumber radicular pain in 3 months and 1 year of follow-up36,37. In contrast to chemical and protein drugs, one postulated advantage of cell therapy is the long-term and stable therapeutic effects on the degenerative tissues, given by the surviving cells and their persistent secretomes. In our IVDD rat model, auto-recovery of IVDD and immune rejection for the xenogenic blood cells limit the long-term observation of cellular therapeutic effects. We, therefore, conducted a pilot clinical study with 6 months of follow-up to provide the first hint that the enriched PBMCs into the disk might be a candidate solution for the persistent pain relief of IVDD.
In the pilot human clinical trial, we demonstrated that the PBMCs effectively alleviated pain and disability in IVDD patients. Previous studies have shown that the administration of the PBMCs might prohibit the inflammatory response of synovial tissues by suppressing IL-1, TNF-α, and C-reactive protein (CRP) expression and decreasing bone marrow lesions or bone marrow edema (BML/BME)38,39. We also validated the anti-inflammatory effects of PBMC injection in the patients with MC2 at 6 months post-treatment (Fig. 7).
As the high tolerance of MSC and BMAC transplantation, PBMC treatment was safe and did not cause severe adverse effects after 6 months of follow-up. We will further address the tolerance of PBMC numbers and the injection frequency in the affected disk by measuring the clinical signs and tissue integrity under MRI examination.
A recent systematic review comprehensively searched the clinical trials in 2018–2020 using PRP, BMAC, and MSCs and evaluated the effectiveness of intradiscal biologics for treating LBP 40 . They emphasize that the affected disk of patients concordant with discogenic LBP should be confirmed by discography or MRI. A qualified prospective PRP treatment trial showed no statistically significant difference in Numeric Rating Scale (NRS) current pain score, SF-36 pain, and SF-36 physical function scores, between the control and treated groups 41 . However, at 8 weeks of visiting, the quality of life (QOL) score by North American Spine Society (NASS) measurement revealed a significant improvement, showing that 56% (15/27) of PRP-treated patients were satisfied with the intervention, compared with 18% (3/17) controls (P = 0.01) 41 . In our study, we also found that only the PBMCs treatment, but not the PRP, reported significant improvement in their pain and ODI scores, compared with the non-treating control (Fig. 6A, C, and Table 3).
An updated review also summarized the human trials of cell transplantation for chronic LBP. Our approach using PBMCs is similar to the autologous BMAC (or named as BMC or BMA), which may be an alternative cell source for controlling IVDD. For instance, Pettine et al. conducted a prospective observational trial, including 26 patients with LBP and modified Pfirrmann gradings IV–VIII9,10. The authors found that the average percentage of VAS reduction was 64.2%, after 6 months and the average percentage of ODI reduction was 55.5% after 6-month treatments. The pain relief appeared durable as the VAS baseline of 82 ± 2.6 mm improved to 21 ± 4.4 mm at 3 years of follow-up 9 . Our study found that at 6 months post-treatment, the average percentage of VAS reduction was 75.0%, and the average percentage of ODI reduction was 79.1% after 6 months of treatment. The better outcome of the PBMCs treatment than that of BMAC might be attributed to the eliminating of inflammatory granulocytes from the total nucleated cells. Nevertheless, the detailed mechanisms have to be validated and long-term results of the PBMCs still have to be followed up.
The HSCs and MSCs are supposed therapeutic factors in the BMAC13,42. However, both numbers of HSCs and MSCs are limited in the BMAC populations. Peripheral blood stem cells (PBSCs) have shown the safety and potential for controlling osteoarthritis43–48. The concentrated PBSCs are generally collected from granulocyte colony-stimulating factor (G-CSF)-stimulated patients by leukapheresis43,46. Compared with the BMAC and PBSC preparations, the advantages of the PCP are short-term production, point-of-care, and no concern of surgical operation or G-CSF-associated adverse effects43,46. Moreover, unlike the apheresis of PBSCs, the Ficoll gradient centrifugation effectively excludes the RBCs and granulocytes from harvested PBMCs. This low contamination of RBC and granulocytes might potentiate the anti-inflammatory and regenerative effects of the grafted PBMCs in this study.
Here, we showed that the collected monocytes could consistently differentiate into M2 macrophage lineages. The MoFi-enriched PBMCs significantly alleviated acupuncture-triggered IVDD in rats. The pilot clinical trial further illustrated that intradiscal injection of MoFi-processed PBMCs relieved pain symptoms and disability in CLBP patients. Consolidating the conclusion still needs an RCT at multi-centers, a larger sample size, and an extended observation period. Assessing structural changes in patients with MC will be informative to demonstrate the anti-inflammatory potency of the PBMCs.
Footnotes
Acknowledgements
The authors are grateful to Professor Hong-Lin Su’s lab members for their technical support in this study. They also thank Ms. Syuan-Ru Chen, the staff of the animal center of NCHU, and the study nurses of the Department of Neurosurgery, Asia University Hospital, Taichung, Taiwan, for assisting with the experiments.
Author Contributions
Conceptualization: Y-HC, M-HH, H-LS, and C-LL. In vitro and in vivo experiments and analysis: S-CK, Y-HK, F-HW, C-YH, C-IS, and H-LS. Clinical experiments and analysis: Y-HC, M-HH, C-HC, D-WCC, H-LS, and C-LL. Resources: Y-HC, M-HH, C-HC, D-WCC, H-LS, and C-LL. Writing the original draft: Y-HC, H-LS, and C-LL. Writing, review & editing: Y-HC, C-CK, H-LS, and C-LL.
Ethical Approval
Ethical issues of the study were approved by the Research Ethics Committee of China Medical University (IRB No. CMUH 110-REC2-189), and Show Chwan Memorial Hospital (IRB No. 1050206 and 1091210).
Statement of Human and Animal Rights
All procedures involving human subjects were conducted in accordance with the clinical trials act and the tenets set down in the Declaration of Helsinki, and with the ethical guidelines for medical and health research involving human subjects of Taiwan policies, adopted by the Research Ethics Committee of China Medical University (CMUH 110-REC 2-189) and Show Chwan Memorial Hospital (1050206 and 1091210). All procedures involving animal subjects were conducted following the animal protection policies of Taiwan, adopted by the Institutional Animal Care and Use Committee (IACUC) of National Chung Hsing University (NCHU) (NCHU 109-159).
Statement of Informed Consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information following the Declaration of Helsinki.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors have the following competing interests. The technology transfer office of National Chung Hsing University has received consultancy, speaker fees, and research grants on behalf of H-LS from Duogenic StemCells Corporation and Hualien Tzu Chi Medical Center. F-HW, C-YH, and C-IS are employees of Duogenic StemCells Corporation. F-HW, C-YH, C-IS, H-LS, and C-LL are shareholders of Duogenic StemCells Corporation. Y-HC, M-HH, S-CK, Y-HK, C-HC, and D-WCC have no conflict of interest with respect to the research, authorship, and publication of this article. Patents, products in development, and marketing products are associated with this research.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Duogenic StemCells Corporation, Taiwan. This work was also supported by the Industry-Academic cooperation project (108-D-595, 109-D-525) between the Duogenic StemCells Corporation and National Chung Hsing University.
