Abstract
Lead acetate can cause testicular damage in males. In this study, we assessed the repairing effects of human umbilical cord mesenchymal stem cells (MSCs) on testicular injury caused by lead acetate in mice. MSCs were injected into mice with testicular injury by intraperitoneal injection, and the organ coefficient of reproductive organs, sperm motility, hormone level and antioxidant index of mice were tested. Compared with the normal group, the coefficient of reproductive organs and sperm motility were reduced in the model group, and histopathology showed obvious testicular injury, proving successful modeling. Compared with the model group, the reproductive organ coefficient and sperm motility were improved in the experimental group, and histopathology showed that the testicular injury could be significantly improved. Sex hormone secretion tends to be normal, and the antioxidant index increased. Sequencing results showed that there were 485 upregulated genes and 172 downregulated genes between the model group and the control group, and 210 upregulated genes and 482 downregulated genes between the experimental group and the model group. Differentially expressed genes are mainly concentrated in AMP-activated protein kinase (AMPK) signaling pathway, apoptosis signaling pathway, and arginine biosynthesis signaling pathway. Overall, MSCs can significantly improve the degree of damages to mice testis caused by lead acetate and have a certain repairing effect.
Introduction
In recent years, with the increasing environmental pollution, the accelerated pace of daily life, work pressure and unhealthy diet and lifestyle, the proportion of male infertility has gradually increased. According to the epidemiological survey in recent years, about 10%–15% of couples in the world are infertile, of which male infertility accounts for more than 40%. Without intervention, human fertility will continue to decline
1
. The factors affecting sperm motility include chemical factors, biological factors, physical factors, psychological behavior, and social factors. Chemical factors include heavy metals, toxic gases, plasticizers, pesticides, and antibiotic residues. Biological factors include all kinds of parasites, bacteria and viruses. Physical factors include high temperature and high pressure environment. Psychological factors include unhealthy living habits, bad mood, and social pressure
2
. At present, drugs commonly used in clinic to improve sperm motility include hormones, antioxidants,
Mesenchymal stem cells (MSCs) are widely welcomed in clinics because of their multiple functions of immune regulation, angiogenesis promotion, and anti-oxidative stress. Up to now, MSC therapy accounts for about 70% of stem cell clinical record projects 6 , and it has been proved to effectively treat lupus erythematosus, liver cirrhosis, pulmonary dysplasia of premature infants, and diabetic foot 7 . In this experiment, we established a mouse testicular injury model by gavage with lead acetate, and used intraperitoneal injection of human umbilical cord mesenchymal stem cells (UC-MSCs) to verify whether it can improve sperm activity and repair testicular injury, and analyzed the genes that changed significantly in this process by transcriptome sequencing technology, so as to select the targets for later biological treatment.
Materials and Methods
Experimental Animals
Thirty-six SPF BALB/c mice aged 6–7 weeks, weighing 18–20 g, were provided by SPF (Beijing) Biotechnology Co., Ltd. (SCXK [Beijing] 2019-0010). The mice were kept in the Animal Center of the Laboratory of Cryotherapy, Qilu Hospital of Shandong University. The temperature ranged from 20°C to 26°C, day and night were alternated (12/12), and the mice were free to take food and water. The umbilical cord has been approved by the Qilu Hospital Umbilical Cord Collection Ethics Committee (Approval Number: KYLL-2021[KS]-0172).
Main Instruments and Reagents
Automatic sperm quality analyzer (Weili Company, WLJY-900; Dynamic analysis system Ver7.1), upright microscope (OLYMPUS, CKX 53), gene sequencer (Illumina nova seq 6000), lead acetate (analytical pure, Sinopharm Group), superoxide dismutase (SOD), nitric oxide (NO), nitric oxide synthase (NOS), gonadotropin (luteinizing hormone [LH]), and gonadotropin-releasing hormone (GnRH) detection kits (all purchased from Nanjing Jiancheng Institute of Bioengineering with batch numbers of 20200109, 20200930, 20200623, 20200516, respectively).
Cultivation and Identification of MSC
The umbilical cord of full-term Cesarean section fetus was collected, the epidermis and vascular tissue were removed and the umbilical cord Wharton’s jelly was cut into tissue blocks of about 1 mm3, and the tissue blocks were cultured with the medium containing 10% fetal bovine serum by volume. One week later, the adherent cells can be seen growing at the bottom of the bottle, and subculture was carried out when the saturation rate reached about 85%.
When the cultured MSCs were subcultured to P3, flow cytometry phenotype detection was performed, with indicators, including HLA-DR, CD34, CD45, CD73, CD44, CD105, and CD90.
Experimental Grouping and Cell Intervention
Thirty-six SPF male BALB/c mice were randomly divided into normal control group, model group and model + MSC therapy group, with 12 mice in each group. Except the normal group, all groups were gavaged with lead acetate aqueous solution at a concentration of 40 mg/kg for 7 days, while the normal control group was gavaged with the same dose of ultrapure water. From the second day of modeling, the model + MSC therapy group was intraperitoneally injected with 5 × 105 cells, once every 5 days, for six consecutive times. Five days after the last administration, all mice were weighed and blood was taken from their eyeballs; 3.5% chloral hydrate was injected into the abdominal cavity for anesthesia at the rate of 1 ml/100 mg; and then, they were killed by neck removal. Both testicles and epididymis were quickly taken for later experiments.
Testicular Quality Coefficient Determination
Testicular weight and testicular coefficient (TW/BW, mg/g) was calculated according to testis mass/body mass.
Detection of Sperm Motility
The epididymis was peeled off and placed in normal saline at 37°C, and the epididymis was cut to free the sperm. Take the diluted sperm and filter it with four layers of lens cleaning paper, then make sperm smear to detect sperm motility. The sperms of mice in each group were stained by improved Pap staining method and recorded and photographed.
Through the morphological observation of sperm in each group of mice, 500 sperms in each group were randomly observed under the microscope. When collecting epididymal sperm, we cut it into several segments and gently squeeze the epididymis and vas deferens with ophthalmic forceps. Squeeze the sperm into the culture medium, and remove the epididymis and vas deferens. Then, the sample is incubated at 37°C, 5% CO2, and saturated humidity for 20 min to allow the sperm to self-disperse. We use a dropper to suck semen, take one small drop to store semen on a glass slide, and make a smear by pulling the sample. The sample is stained with 0.5% gentian violet alcohol for 3 min, dried naturally, washed with water, and sent to microscopic examination. Under the microscope, it can be observed that most of the sperm are structurally normal, with some being abnormal sperm. Under the microscope, normal and abnormal sperm are counted, with 500 sperms in each group.
Determination of SOD Activity, NO Content, and NOS Activity in Testis
Take mouse testis tissue and make it into 10% homogenate, centrifuge at 4°C and 300 × g for 10 min, and take supernatant to determine SOD activity according to kit instructions. Take 1% mouse testis tissue homogenate, and determine NO content and NOS activity according to the kit instructions.
Enzyme-linked immunosorbent assay (ELISA) detection method: The reagent kit uses a competitive method to detect the content of LH and GnRH in the sample. We add samples to the enzyme labeled wells pre-coated with antibodies, and then add biotin labeled recognition antigens. It is incubated at 37°C for 30 min. The two compete with solid-phase antibodies to form immune complex. Then, we wash with phosphate-buffered saline with Tween 20 (PBST) to remove unbound biotin antigens and add avidin horse radish peroxidase (HRP) to incubate at 37°C for 30 min. The avidin HRP binds to biotin antigens, and the bound HRP catalyzes TMB (tetramethylbenzidine) to turn blue. Subsequently, it is converted into yellow under the action of acid, and there is an absorption peak at a wavelength of 450 nm. The final concentration is calculated based on the absorption value.
Determination of Reproductive Hormones in Mice Serum
Take 500 μl blood from eyeball of mice, centrifuge at 4°C and 300 × g for 10 min, separate serum, and determine serum LH and GnRH levels according to the instructions of ELISA kit.
Preparation and Observation of Mouse Testicular Tissue Section
Take the testicular tissue of mice in each group and fix it with testicular tissue fixative, 4 μM paraffin section and hematoxylin and eosin (HE) staining were carried out, observing the morphological changes of seminiferous tubules and germ cells in testicular tissue, and recording the photos.
Screening of Differentially Expressed Genes
Total RNA was extracted from the testes of mice in each group using the Direct-Zol RNA micro-preparation kit. Use the Illumina HiSeq sequencer (Illumina HiSeq 6000 platform) for mRNA sequencing.
Functional Enrichment Analysis
Through the analysis of GO (Gene Ontology) and KEGG (Kyoto Encyclopedia of Genes and Genomes), the potential functions and participating pathways of DEGs were obtained, and the screening criteria was adjusted P < 0.05.
Statistical Analysis
All data were analyzed with GraphPad Prism8.0.1 software, and the mean ± standard deviation (x ± s) was used. T test was used to compare the differences between the groups, and P < 0.05 was the significant difference.
Results
Cultivation and Identification of MSC
After 7 days of primary MSC cultivation, the cell morphology became regular, showing a long spindle or elliptical shape with strong refractive index. The MSC after subculture exhibits a long spindle shape with clear contours, and strong refractive index with rapid growth, as shown in Fig. 1A–C. The phenotype identification showed negative expression of HLA-DR, CD34, and CD45; while CD73, CD44, CD105, and CD90 were positively expressed, as shown in Fig. 1D.
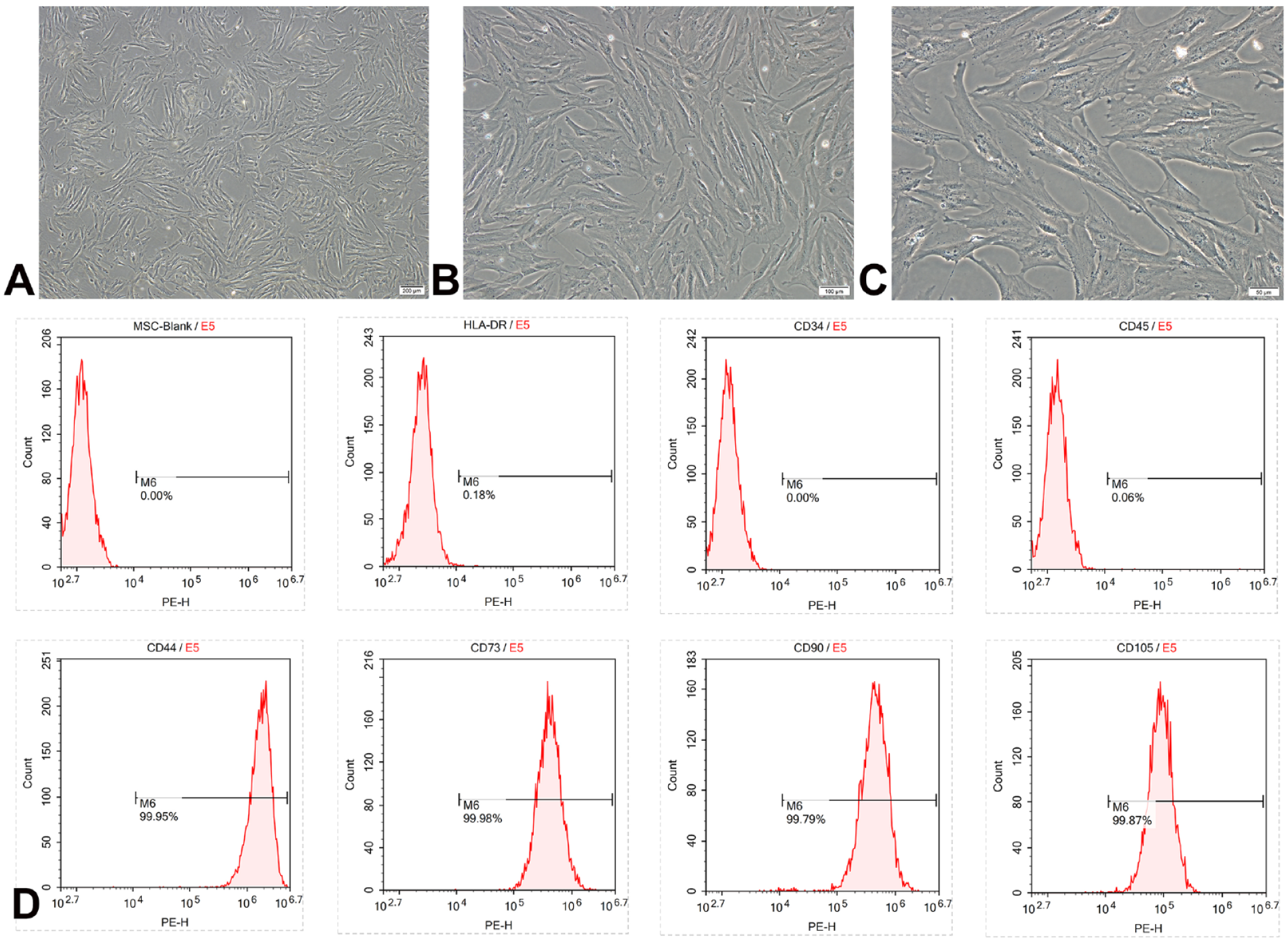
Identification of mesenchymal stem cells. (A–C) Morphological observation of MSC. (D) Identification of phenotype of MSC by flow cytometry. MSC: mesenchymal stem cell.
Effect of MSC on Testicular Coefficient of Mice With Testicular Injury Induced by Lead Acetate
Compared with normal mice, the testicular weight and testicular coefficient of model mice decreased significantly (P < 0.05). Compared with the model group, the testicular weight and testicular coefficient of mice in the model + MSC treatment group increased significantly (P < 0.05), as shown in Tables 1 and 2 and Fig. 2.
The Effect of MSCs on Mouse Testis Weight (n = 12, x ± s).
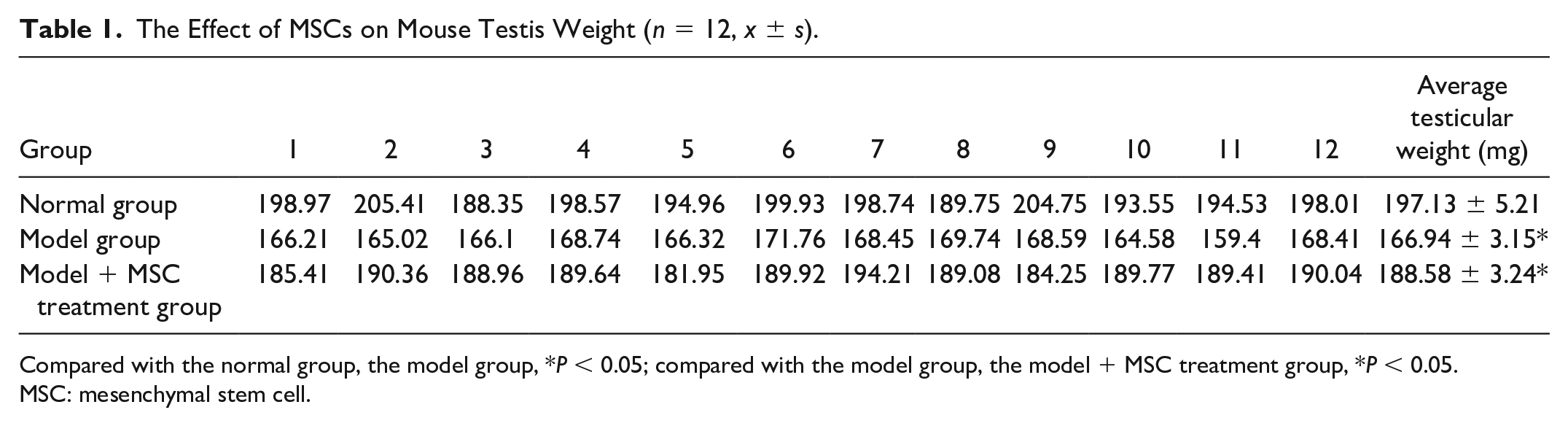
Compared with the normal group, the model group, *P < 0.05; compared with the model group, the model + MSC treatment group, *P < 0.05.
MSC: mesenchymal stem cell.
The Effect of MSCs on Mouse Testis Coefficient (n = 12, x ± s).
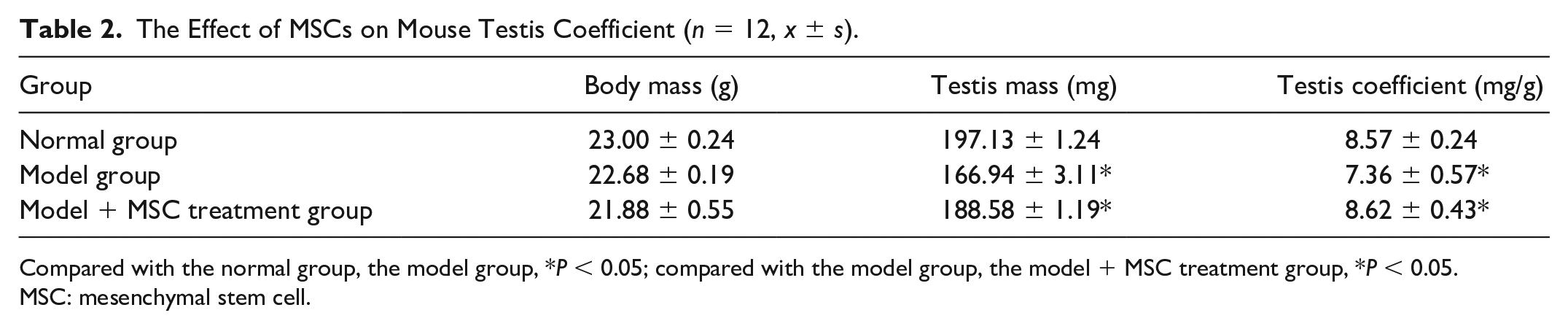
Compared with the normal group, the model group, *P < 0.05; compared with the model group, the model + MSC treatment group, *P < 0.05.
MSC: mesenchymal stem cell.
Observation on the whole mouse testis. Conventional morphological observation of normal testicles, lead acetate damaged testicles, and overall testicular morphology after MSC treatment. MSC: mesenchymal stem cell.
Effect of MSC on Sperm Viability in Mice With Testicular Injury Induced by Lead Acetate
It can be seen that the abnormal sperms in the normal group were relatively less, while the microcephalic sperms, decapitated sperms and tailless sperms in the model group increased significantly. After the intervention of MSCs, the abnormal sperm ratio decreased significantly, as shown in Table 3. In Fig. 3A–C, microcephalic, decapitated and tailless sperms were in the red circle.
Sperm Ratio of Mice in Each Group.
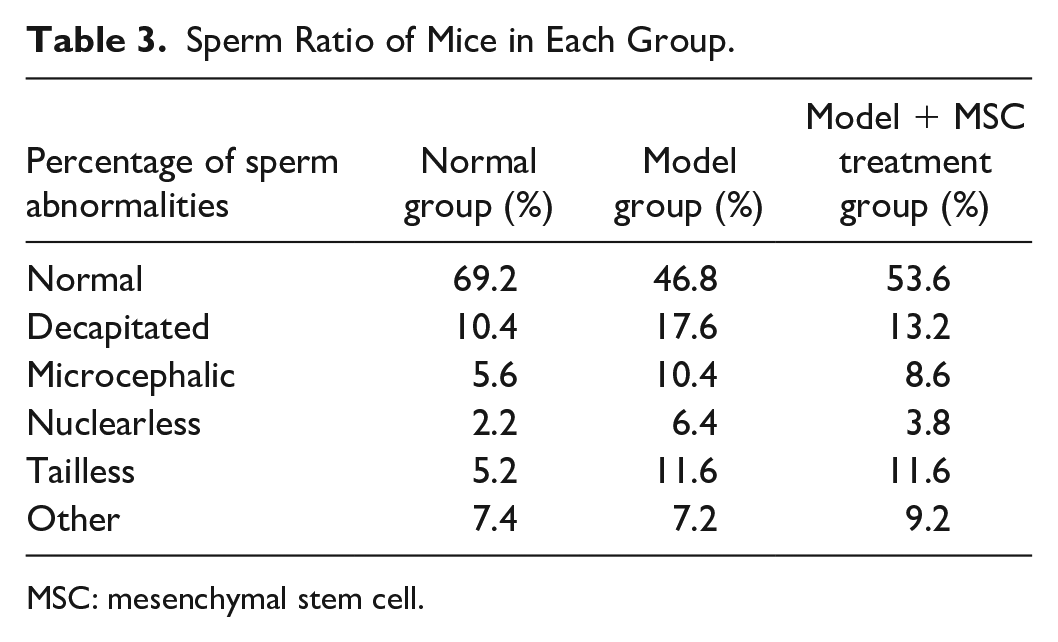
MSC: mesenchymal stem cell.
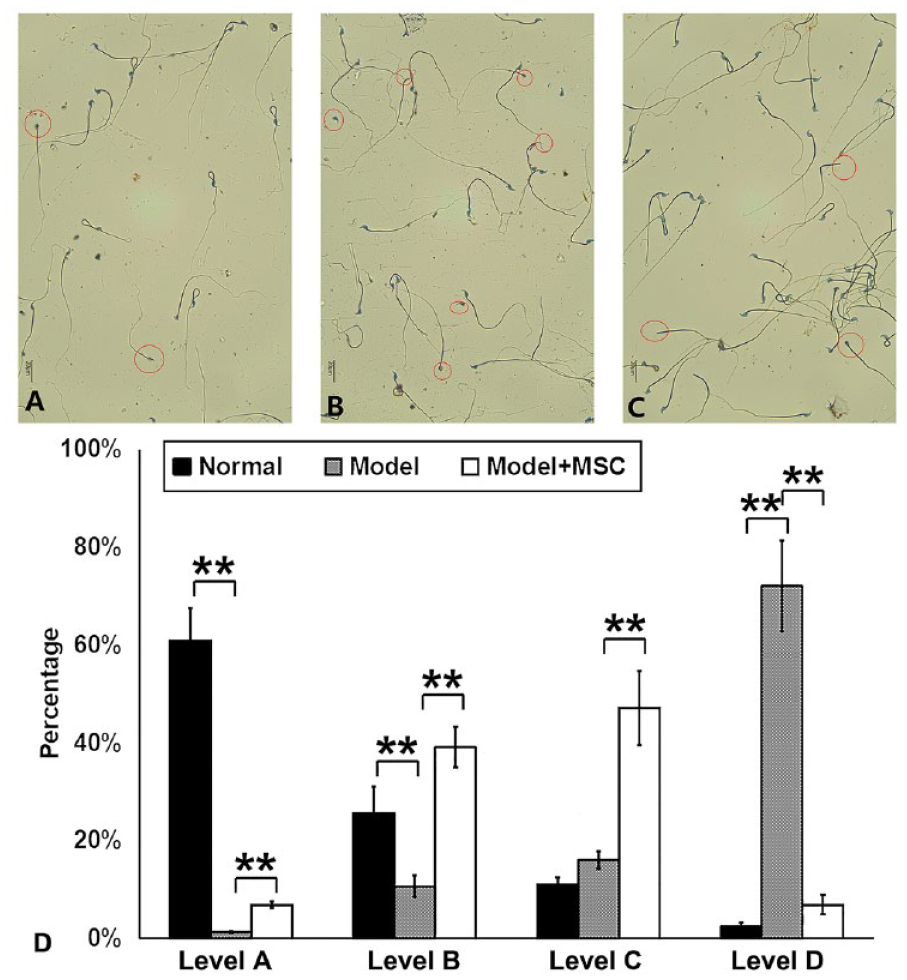
The effect of lead acetate modeling and MSC treatment on sperm quality. Morphological observation of each component of mouse sperm. (A) Normal mouse sperm. (B) Model mouse sperm. (C) Model + MSC mouse sperm (abnormal sperm is shown in the red circle). (D) Percentage of mouse sperm motility. Observing the percentage of sperm at all levels in normal testes, lead acetate damaged testes, and testes treated with MSC under an optical microscope. MSC: mesenchymal stem cell. **This indicates a significant difference between the two groups.
A-level sperm is fast forward moving sperm, B-level sperm is slow and sluggish forward moving sperm, C-level sperm is non-forward moving sperm, and D-level sperm is spermatium. As can be seen from Fig. 3D, compared with normal mice, the number of A-level sperm and B-level sperm in model mice decreased significantly (**P < 0.01), indicating that sperm motility decreased. Compared with the model mice, in the model + MSC treatment group, the number of A-level sperm and B-level sperm increased, while the number of C-level sperm and D-level sperm decreased (**P < 0.01), which indicated that MSC had a certain repairing effect on the testicular tissue of mice, and the sperm motility was improved.
Determination Results of SOD Activity, NO Content, and NOS Activity in Mouse Testis
Compared with the normal group, the NO content, NOS activity, and SOD activity in the testis of the model group decreased significantly (P < 0.05), and the NO content, NOS content, and SOD activity increased significantly after the intervention of MSCs, as shown in Table 4.
Effect of MSCs on the Oxidation Coefficient of Mice (n = 12, x− ± s).
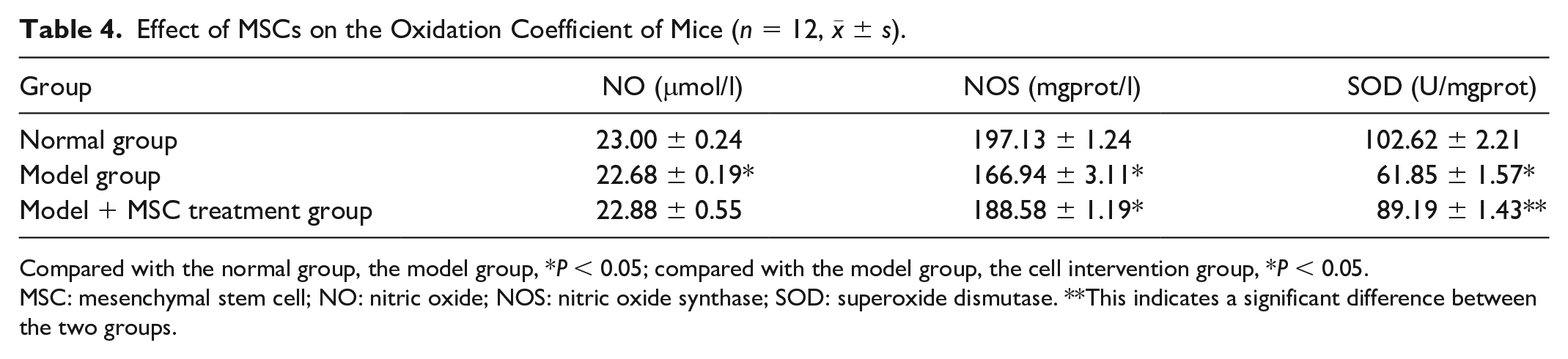
Compared with the normal group, the model group, *P < 0.05; compared with the model group, the cell intervention group, *P < 0.05.
MSC: mesenchymal stem cell; NO: nitric oxide; NOS: nitric oxide synthase; SOD: superoxide dismutase. **This indicates a significant difference between the two groups.
Determination Results of LH Content and GnRH Content in Mouse Testis
Compared with the normal group, the contents of LH and GnRH in the testis of the model group were significantly increased (P < 0.05), indicating that gonadal hormone secretion was disordered. After the intervention of MSC, LH content and GnRH content decreased, and gonadal hormone secretion tended to a normal level, as shown in Table 5.
The Effect of MSCs on Mouse Gonadal Hormones (n = 12, x− ± s).

Compared with the normal group, the model group, *P < 0.05; compared with the model group, the cell intervention group, *P < 0.05.
MSC: mesenchymal stem cell; LH: luteinizing hormone; GnRH: gonadotropin-releasing hormone.
Effect of MSC on the Histopathology of Testis in Mice With Lead Acetate-Induced Testicular Injury
After HE staining, it can be seen under the microscope that the spermatogenic tubules in the testis of normal mice are arranged closely and orderly, and the number of cell layers is clear and complete. There are more spermatogonia, spermatocytes, and sperm at various stages of differentiation in the spermatogenic epithelium, and there are more formed sperms in the uterine cavity. Compared with the normal control group, the arrangement of cells in the model group is disordered, the spermatogonia and spermatocytes in the seminiferous epithelium are disordered, there are obvious vacuoles and atrophy, and the number of formed sperm in the lumen is small. Compared with the model group, after the intervention of MSCs, it can be seen under the microscope that the levels of seminiferous tubules are clearly increased, the vacuole phenomenon is improved, the spermatogenic cells are densely distributed, arranged neatly, and there are more sperms. MSCs have a certain repairing effect on the testicular damage caused by lead acetate in mice (Fig. 4).
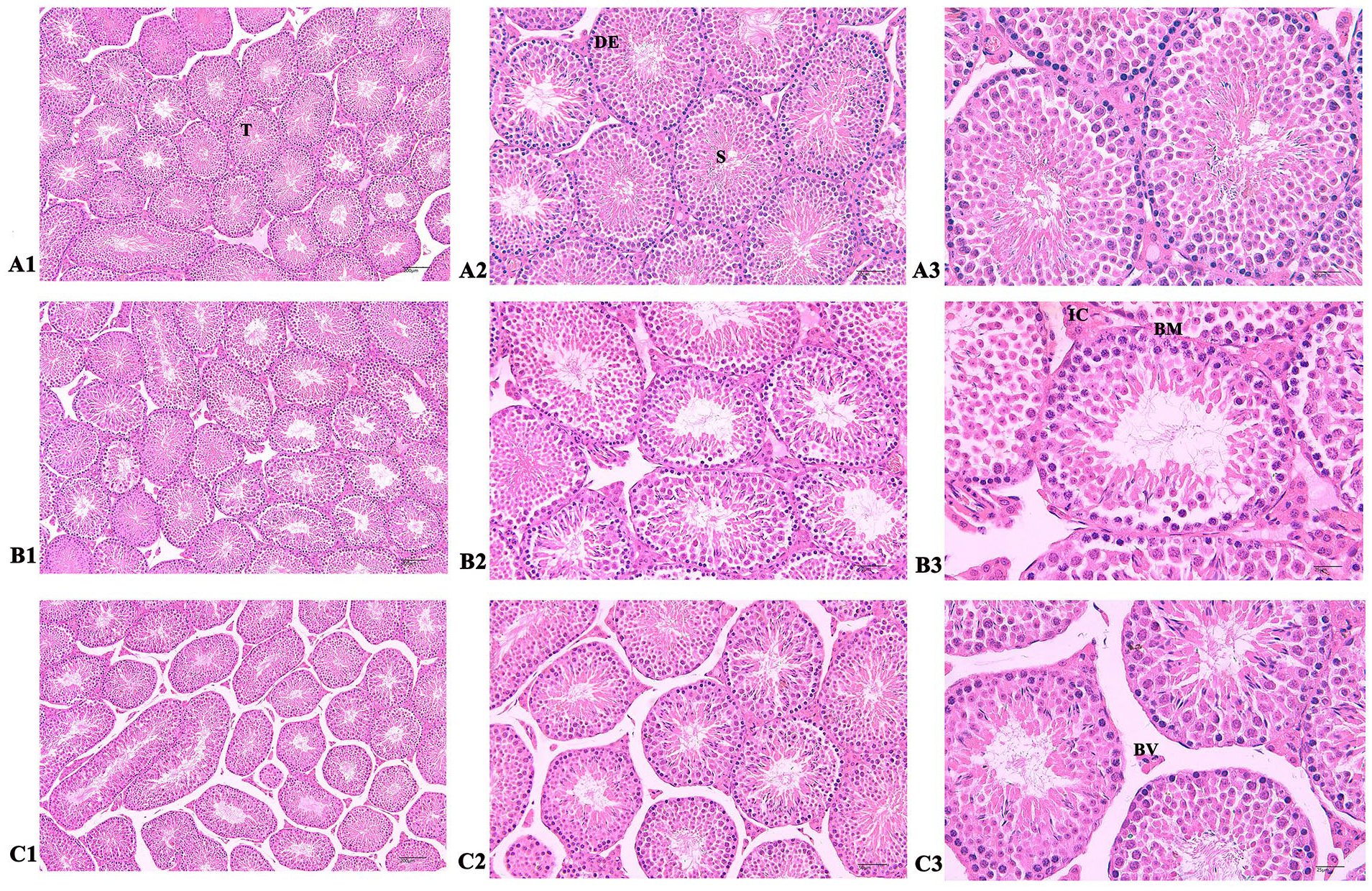
Pathological observation of each component of mouse by HE staining. (A1–A3) Normal mouse testis (100×, 200×, and 400×). The control group showed that the seminiferous tubules (T) were rich in spermatogonia, spermatocytes, and sperm cell populations (S), while testicular tissue showed a large number of interstitial tissue cells (BV). (B1–B3) Model mouse testis (100×, 200×, and 400×). The interstitial cells in the model group were significantly damaged and almost invisible. The seminiferous tubules were filled with IC, and the number of spermatogonia, spermatocytes, and sperm cells in the seminiferous tubules was significantly reduced. The interstitium and subcapsular BV were severely congested and edematous, with thickening of the BM and disordered arrangement of epithelial cells (DE). (C1–C3) Model + MSC mouse testis (100×, 200×, and 400×). After MSC treatment, there were a large number of spermatogonia, spermatocytes, and sperm cells in the interstitial tissue cells and seminiferous tubules. Interstitial cells reappear and the basement membrane returned to normal levels. BV: blood vessel; IC: inflammatory cell; BM: basement membrane; DE: epithelial cells; HE: hematoxylin and eosin; MSC: mesenchymal stem cell.
PPI Analysis of Differential Genes, Screening, and Enrichment Analysis of Key Genes
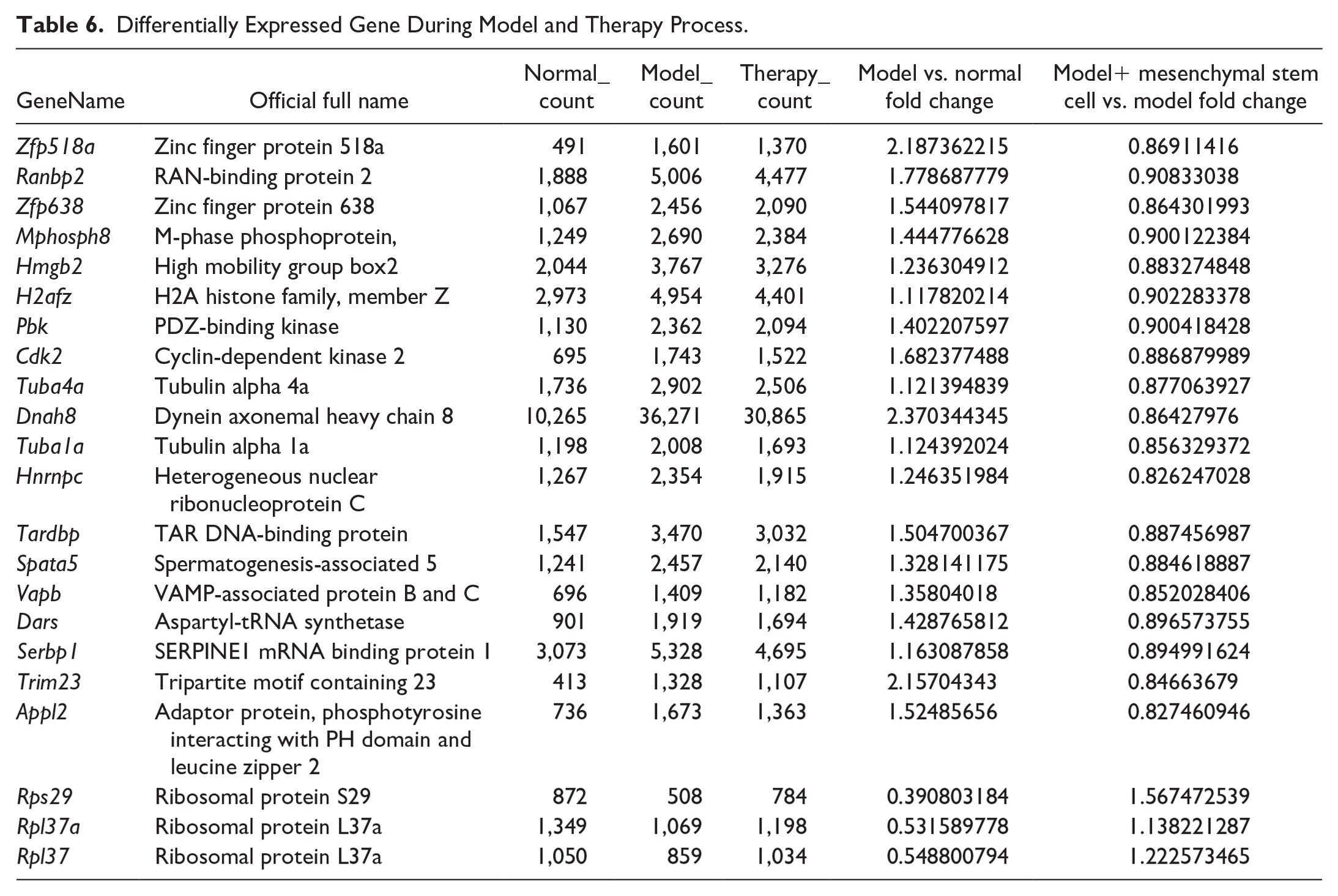
By screening differentially expressed genes, 22 key genes were screened according to the size of nodes, among which 19 genes in the testis of the model group increased compared with the normal group, and the expression decreased after MSCs intervened. The expression levels of three genes in the model group were lower than those in the normal group, but increased after the intervention of MSCs. These 22 key genes were mainly concentrated in AMPK (AMP-activated protein kinase) signaling pathway and apoptosis pathway (Fig. 5 and Table 6).
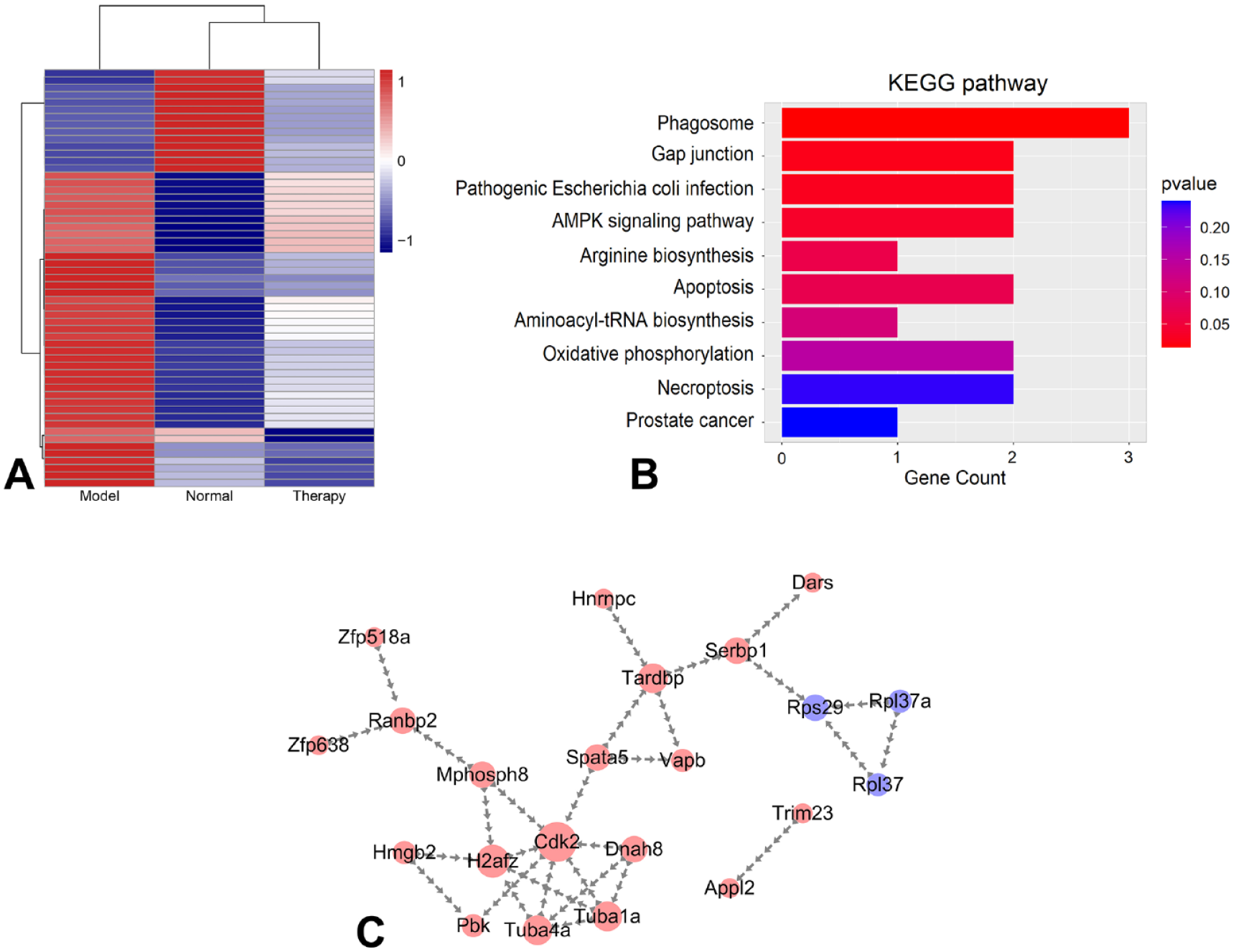
RNA-seq analysis of mouse testis. (A) Differential gene heat map. (B) GO analysis of differentially expressed genes. (C) Protein interaction mapping of 22 key genes. AMPK: AMP-activated protein kinase; KEGG: Kyoto Encyclopedia of Genes and Genomes.
Differentially Expressed Gene During Model and Therapy Process.
Discussion
In this research, a mouse testicular injury model was established by gavage with lead acetate for 7 days. From the day after the first gavage, the mice in the model + MSC treatment group were treated by intraperitoneal injection of MSC for six times in a row. The pathological section results showed that the testis of the model group was seriously damaged, and the situation was ameliorated after the intervention of MSC. The sperm motility test showed that the sperm motility of the model + MSC treatment group was significantly higher than that of the model group, and the sex hormones and antioxidant indexes were significantly improved. We measured the NO content and NOS activity, which can effectively reflect the antioxidant capacity of testicular tissue, as well as the quality, damage, and recovery degree of testicles. The significant comparison between the model group and the MSC group proves that MSC has a good repair effect on testicular injury.
The reagents commonly used in animal testicular injury models are ethanol, cyclophosphamide, adenine, lead acetate, and so on. The testicular injury caused by lead acetate is mainly caused by the action of lead ions. Lead is an endocrine disruptor with male reproductive toxicity, which can affect male reproductive health. Testis is the target of lead toxicity and can cause testicular degenerative diseases in male animals. In addition, lead can directly damage the spermatogenic epithelium, thus reducing the level, looseness and even shedding of spermatogenic cells 8 . Lead also hinders and interferes with the development and maturation of sperm, and at the same time directly poisons sperm. Therefore, lead acetate is selected as the modeling reagent. After lead acetate is administered to the stomach in this research, Lead acetate is absorbed through the digestive tract and enters the bloodstream, spermatogonia, and spermatocytes in the spermatogenic epithelium of mouse testis are disordered, vacuoles and atrophy are obvious, and abnormal sperm is significantly increased. The disorder of serum levels of hormones and testicular antioxidant index levels is consistent with the characteristics of testicular injury. These phenomena are consistent with the modeling phenomena published by Zhao et al. 9 and Abdrabou et al. 10 , proving that our modeling is successful.
MSCs have been proved to be effective in lung injury, liver injury and kidney injury, and their mechanisms include the following aspects. First, they secrete nutritional repair factors, play an anti-apoptosis and promote tissue repair. Hepatocyte growth factor (HGF) and transforming growth factor (TGF-β) have repair effects on liver cirrhosis and liver injury11–13. Nrf2 (NF-E2-related factor 2) and Keap1 have positive effects on lung injury.14–16 Insulin-like growth factor (IGF)) and epidermal growth factor (EGF) have significant effects on the repair of acute kidney injury17–19. Second, exosomes can be produced, which contain many small molecular nucleic acids, protein, and so on, and cooperate with cytokines. Moreover, it can promote the expression of heme oxygenase system, especially SOD, eliminate free radicals, reduce the oxidation rate. The organ damages of the body are largely due to collagen accumulation and fibrosis caused by repeated repair, while MSC can promote the expression of MMP9 and eliminate collagen. Finally, MSC can also promote the expression of Ang-1, Ang-2, and vascular endothelial growth factor (VEGF), improve local microcirculation, increase the expression of CD31 and vWF, and improve the overall quality of the body 20 . We measured the content of NO and the activity of NOS, which can effectively reflect the antioxidant capacity of testicular tissue, as well as the quality, damage, and recovery degree of testicles.
In this experiment, we screened a total of 22 genes with abnormal expression for analysis. These 22 genes interact with each other, and the products formed through gene transcription and translation have impacts on biological individuals. Among them, Mphosph8, Hmgb2, H2afz, Pbk, Cdk2, Hnrnpc, Dars, and Serbp1 genes are mainly expressed in cancer cells and play crucial roles in primary tumors and tumor metastasis. They mediate testicular damage through cycle regulation, cell adhesion, and P53 signaling pathways, resulting in adverse consequences. Tuba4a, Tuba1a, Tardbp, and Vapb mainly mediate neurodegenerative changes, which can increase the probability of sperm abnormalities and lead to phenomena, such as dead sperm and weak sperm. Dnah8 and Spata5 are biallelic genes, and genetic mutations can lead to abnormal sperm development, causing male infertility. Appl2 is an apoptosis-related gene that mainly mediates cell apoptosis and plays an important role in the cell apoptosis pathway. Rps29 is a mutated gene in the Diamond Blackfan anemia family, and its reduced expression can lead to anemia, affecting blood supply to the reproductive system, and ultimately affecting fertility. Through the analysis of gene expression, it was found that the genes contained in AMPK signaling pathway and apoptosis signaling pathway were significantly up-regulated and down-regulated in the model group and the model + MSC treatment group, which preliminarily indicated that they played their roles through these channels. Spermatogenesis is regulated by many genes and proteins. Testosterone plays an important role in gonadal differentiation, spermatogenesis, sexual function maintenance, and so on. Testosterone is secreted by Leydig cells of testis, and the content of zinc finger protein (Zfp) in testis is higher than that in other tissues, which is related to spermatogenesis. As an important adipocyte factor, Zfp can regulate the key enzymes in fat, thus affecting lipid metabolism and spermatogenesis. Low expression can promote the rapid proliferation of immature sperm Sertoli cells21,22. In this experiment, the gene expression of Zfp in model group was significantly higher than that in control group, while decreased in model + MSC group, indicating that the spermatogenic ability of mice decreased in model group, and was improved after the intervention of MSCs.
Apoptosis mitochondrial pathway plays an important role in the process of germ cell apoptosis, mainly to maintain the proportion balance between different cells. The way of action is to change its membrane structure through signals, thus affecting the opening of membrane permeability conversion pores, releasing a series of apoptosis-promoting substances, and regulating downstream factors through such substances, causing the apoptosis of germ cells 23 .
To sum up, our research proves that UC-MSCs can repair the testicular injury caused by lead acetate, and their effects mainly focus on AMPK signal pathway, apoptosis signal pathway, arginine biosynthesis signal pathway, and so on, which can be regulated to repair DNA damage, regulate apoptosis and promote arginine synthesis, providing a good foundation for subsequent researches.
Footnotes
Author Contributions
Shasha Zhao and Li Zhaozhi performed all the experiments and participated in writing the manuscript. Xiaoyu Dai, Lei Pan, Zhe Xu, Li Li, Huanhuan Wang, Xiaodun Liu, and Dong Li assisted with the experiments. Xiaodun Liu and Dong Li conceived the project, analyzed the data, and actively participated in manuscript writing.
Ethical Approval
This study was approved by the Ethics Committee of Qilu Hospital of Shandong University, China (approval no. KYLL-2021[KS]-0172).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Ethical Committee on Animal Experiment of Shandong University Qilu Hospital (approval no. DWLL-2021-036) approved protocols.
Statement of Informed Consent
Written informed consent was obtained from each umbilical cord donor.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Department of Ji’nan Science & Technology Bureau (2022TSGC2001) and Department of Industry and Information Technology of Shandong Province (202020100416). Shandong Province Science and Technology Small and Medium Enterprises Innovation Ability Enhancement Project (2022TSGC2001), Shandong Province Technology Innovation Project (202020100416), and Natural Science Foundation of Shandong Province (ZR2020MH063).
