Abstract
Because there is a shortage of donor kidneys, researchers are exploring the possibility of using genetically modified pig kidneys for transplantation. Approaches involving knockout of carbohydrate genes or knockin of protective proteins have been attempted to determine the best gene modifications. In this study, we utilized GalT−/−;hCD39;hCD55 and GalT−/−;hCD39;hCD46;hCD55;thrombomodulin (TBM) pigs for transplantation in nonhuman primates (NHPs). The NHPs survived for 4 weeks after kidney transplantation (4 WAT) from the GalT−/−;hCD39;hCD55 pig and for 6 WAT from the GalT−/−;hCD39;hCD46;hCD55;TBM pig. However, messenger RNA (mRNA) sequencing and immunohistochemistry analysis revealed that the 6 WAT kidney exhibited more severe apoptosis, inflammation, loss of renal function, and renal fibrosis than the 4 WAT kidney. These results indicate that additional knockin of complement regulator (hCD46) and coagulation regulator (TBM) is not enough to prevent renal damage, suggesting that improved immune suppression is needed for more prolonged survival.
Introduction
The kidney plays a crucial role in maintaining the balance of water, salts, and electrolytes in the body, and removing waste products from the blood. Renal failure, a condition where less than 15% of kidney function remains, affects 0.3% of the US population. Transplantation is a curative therapy, but some patients die on the waiting list due to the limited availability of donors. Pigs are considered good candidates for kidney transplants due to their similar organ size, renal physiology, and immunity, compared with humans 1,2. Over the years, pig-to-nonhuman primate (NHP) transplants have been conducted. After discovering the significant xenoantigen, alpha-1,3-galactose, which causes hyperacute rejection 3 , various genetically engineered pigs have been suggested 4 .
Several strategies are being suggested to select the best genetically engineered pig for xenograft. One approach involves knocking out carbohydrate antigens (Gal, Neu5Gc, and Sda)3,5–8, knocking in protective proteins (complement-, coagulation-, and phagocytosis-regulatory proteins)4,9,10, and knocking out zoonotic risk (porcine endogenous retroviruses) 11 . Although gene editing can prevent antibody-mediated rejection by removing xenoantigens, immunosuppressive therapy is still necessary to block T-cell-mediated immune response. Many researchers use monoclonal antibodies to block the cluster of differentiation (CD)40:CD154 pathways12–14. This therapy is typically combined with anti-inflammatory and anticoagulative agents. To reduce systemic inflammation, tumor necrosis factor (TNF) inhibitor or interleukin-6 inhibitor is included15,16. Anticoagulation medicines are administered to prevent thrombotic microangiopathy17,18.
The data on kidney xenotransplantation in NHPs appear to be quite complex and difficult to compare. With so many different combinations of therapy and genetics at play, it is no wonder that researchers still need to fully understand this process. In this study, we compared the results of pig-to-NHP kidney xenotransplantation using GalT−/−;hCD39;hCD55 and GalT−/−;hCD39;hCD46;hCD55; thrombomodulin (TBM) donor pigs, with the same immunosuppressive, anti-inflammatory, anticoagulation agents. We hypothesized that two additional genes (hCD46 and TBM) impact extended survival. To explore the mechanism of renal damage, intracellular molecular [messenger RNA (mRNA) sequencing, immunohistochemistry, and immunoblot] and histopathological analyses were conducted.
Materials and Methods
Animals
Two kinds of genetically modified pigs, GalT−/−;hCD39;hCD55 and GalT−/−;hCD39;hCD46;hCD55;TBM were obtained from Optipharm Inc (Cheongju, Korea). The GalT−/−;hCD39;hCD55 pig was generated according to our previous study 19 . The GalT−/−;hCD39;hCD46;hCD55;TBM pig was generated as follows. CRISPR-Cas9 vectors to target exon4 of GGTA1, and pCAGGS-hCD46-2A-hTBM vector containing a 2A-linked human CD46 and human TBM transgene under controlled cytomegalovirus (CMV) early enhancer/chicken β actin promoter (pCAGGS) were produced. The expression of CD46 and TBM was confirmed by fluorescence-activated cell sorting and immunohistochemistry (IHC; data not shown). These vectors were transfected into Yucatan miniature pig fibroblasts by electroporation. Verified transgenic cells were then used for somatic-cell nuclear transfer to produce GalT−/−;hCD46;TBM pigs. The GalT−/−;hCD39;hCD46;hCD55;TBM pigs were produced by breeding of GalT−/−;hCD39;hCD55 pig and GalT−/−;hCD46;TBM pig. Information on donor pigs is in Table 1. Two male cynomolgus monkeys (Macaca fascicularis), with a body weight of 3.605 kg (age: 3 years and 9 months) and 3.166 kg (age: 3 years and 10 months) were selected. The NHP’s medical condition determined kidney collection after transplantation under humane euthanasia.
Information of Donor Pigs.
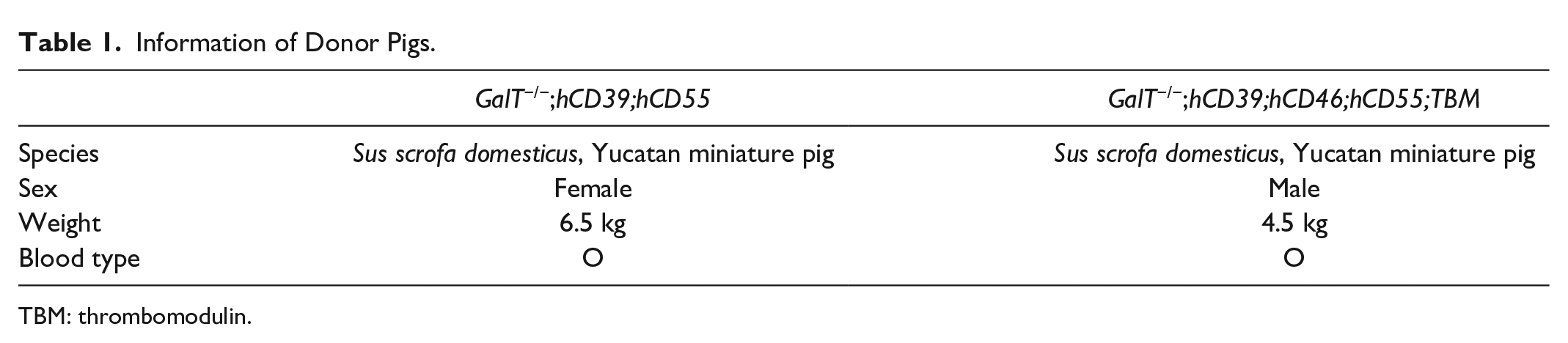
TBM: thrombomodulin.
Surgical Procedure
Kidney transplantation was performed on the recipient after 16 h fasting. After general anesthesia (20 mg/kg ketamine + 2.3 mg/kg xylazine, i.m. with isoflurane), the pig’s left kidney was collected for 0 week after transplantation (0 WAT) kidney and the pig’s right kidney was perfused for transplantation. The recipient monkey was anesthetized (5 mg/kg ketamine + 0.1 mg/kg medetomidine, i.m. with isoflurane) and the right kidney was removed before being replaced with the pig’s right kidney that was anastomosed to the aorta and inferior vena cava. Two weeks later, the monkey’s remaining left kidney was removed in a second operation. To control pain and bacterial infection, cephazolin (20 mg/kg, i.m., b.i.d. for 5 days) and tramadole (7 mg/head, i.m., for 3 days) were administered. The Institutional Animal Care and Use Committee (IACUC) at Korea Institute of Toxicology with approval no. 2012-0051 reviewed and approved the animal care and use protocol.
Immunosuppressive, Anti-Inflammatory, and Supportive Therapy
The monkeys in the study received various medications and treatments after kidney transplants. Rituximab [10 mg/kg/day intravenous (i.v.)] on day −7, antithymocyte globulin (5 mg/kg/day i.v.) on days −2 and −1, cobra venom factor (0.1 mg/kg/day i.v.) on day −1 to day 3, and anti-CD154 antibody (20 mg/kg i.v.) on days −1, 0, 3, 7, 10, and 14 followed once a week after day 21 were treated. Rapamycin [1 mg/head oral (p.o.)], mycophenolate mofetil (20 mg/kg/day p.o.), omeprazole (10 mg/head p.o.), aspirin (50 mg/head p.o.), enoxaparin (1 mg/kg/day subcutaneous), and solu-medrol [2 mg/kg/day intramuscular (i.m.) on day 0 to day 13, 1 mg/kg/day i.m. on day 14 to day 20, 0.5 mg/kg/day i.m. since day 21] were administered once a day after day 0 (transplantation day).
Histology
Terminal deoxynucleotidyl transferase biotin-dUTP nick end labeling assay
Terminal deoxynucleotidyl transferase biotin-dUTP nick end labeling (TUNEL) assay was carried out according to the manufacturer’s instructions (ApopTag Peroxidase In Situ Apoptosis Detection Kit, cat. no. S7100; Millipore, MA, USA). Deparaffinized slides were incubated with proteinase K (20 µg/ml) for 15 min at room temperature (RT) and washed three times with distilled water. Tissues were quenched with 3% hydrogen peroxide for 5 min at RT, followed by washing with phosphate-buffered saline (PBS). Then, tissues were incubated with TdT-enzyme for 1 h at 37°C and stop buffer for 10 min at RT. Slides were washed with PBS, incubated with horseradish peroxidase (HRP)-conjugated anti-digoxigenin antibody, and reacted with 3,3′-diaminobenzidine. Methyl green (0.5% v/v) was used to counterstain.
Masson’s trichrome
Masson’s trichrome staining was carried out according to the instruction provided by the manufacturer (cat no. IFU-2; ScyTek, UT, USA). Deparaffinized slides were incubated with Weigert’s iron hematoxylin, solutions of Biebrich scarlet-acid fuchsin, phosphomolybdic-phosphotungstic acid, and aniline blue. Then, the slides were rinsed with 1% acetic acid solution. The collagen connective tissues were stained blue, the nuclei were stained dark red/purple, and the cytoplasm was stained red/pink. The samples were mounted and photographed by microscopy (Zeiss; AxioVision).
Sirius red
Sirius red staining was carried out according to the manufacturer’s instructions (cat. no. ab150681, Abcam, Cambridge, UK). Deparaffinized slides were incubated with picro sirius red for 1 h at RT. The slides were rinsed with 1% acetic acid solution, mounted, and photographed by microscopy (Zeiss; AxioVision). The collagen connective tissues were stained red.
Immunohistochemistry
Kidney tissues were fixed overnight in 10% neutral-buffered formalin and embedded in paraffin. Then, the tissue samples were sectioned at 5 μm, deparaffinized, processed for antigen retrieval, blocked, incubated with the primary, and peroxidase-conjugated secondary antibodies. For the peroxidase-conjugated secondary antibody, 3,3′-diaminobenzidine substrate was used followed by hematoxylin for nuclear counterstaining. Primary antibodies against CD3ε (cat. no. 85061; 1:100 dilution; cell signaling), CD4 (cat. no. ab133616; 1:100 dilution; Abcam), sodium-glucose linked cotransporter (SGLT1; cat. no. 07-1417; 1:2000 dilution; Millipore), SGLT2 (cat. no. NBP1-92384; 1:500 dilution; Novus, CO, USA), vimentin (cat. no. 5743; 1:250 dilution; Cell signaling, MA, USA), alpha-smooth muscle actin (cat. no. 19245; 1:250 dilution; Cell signaling), and S100A4 (cat. no. ab868; 1:100 dilution; Abcam) were used. The samples were mounted on slides and photographed with an AxioCam microscope camera, and the images were analyzed with AxioVision software (Zeiss, Oberkochen, Germany).
Hematology and Serum Chemistry
For hematology, blood was collected into EDTA-2K tube and hematology parameters were measured using ADVIA2120i hematology analyzer (Siemens, USA). For serum chemistry, blood was collected, incubated for 30 min at RT, and centrifuged at 3,000 rpm for 10 min. The supernatant was isolated as serum, and serum chemistry was measured using TBA 120 FR chemistry analyzer (Toshiba Co, Japan).
MRNA Sequencing
Thereafter, 2 μg total RNA was extracted from normal pig kidney (untransplanted) and transplanted kidney using the Phenol/Chloroform extraction method. The integrity of RNA was assessed by Agilent 2100 BIoanalyzer (Agilent, CA, USA). A cDNA library was constructed using Quantseq 3′ mRNA-seq library prep kit (Lexogen, Vienna, Austria). Manufacturer’s instructions followed the process, including sequencing, mapping, and normalization. Differential expression gene (DEGs) analysis between groups and visualization were performed using Excel-based Differentially Expressed Gene Analysis (ExDEGA, E-biogen, Inc, Seoul, Korea). The DEGs were determined with the genes whose expression levels were changed as |log2 fold| ≥1. Raw data for mRNA sequencing was uploaded to Gene Expression Omnibus (GEO accession: GSE240012).
Immunoblot
Kidney tissues were dissolved in radioimmunoprecipitation assay buffer with protease/phosphatase inhibitor for 30 min. After centrifugation at 13,000 rpm for 30 min, the supernatant was collected as total protein lysate. Protein lysates (10 μg) were resolved by 4% to 20% gradient sodium dodecyl sulfate (SDS)-polyacrylamide gel electrophoresis and transferred to polyvinylidene fluoride membranes. Each membrane was incubated with 1% bovine serum albumin and probed with the primary and HRP-conjugated secondary antibodies. The bands were visualized by enhanced chemiluminescence, and images were acquired for the quantitative analysis with ChemiDoc XRS+ System (Bio-Rad, CA, USA). Raw data of immunoblot is in Supplemental Fig. S3.
Donor Pig–Specific Antibody Assay
Plasma IgG/IgM binding to pig endothelial cells (ECs) or pig peripheral blood mononuclear cells (PBMCs) was measured by flow cytometry (FACSCalibur; BD Biosciences). The assay was performed by using the modified methods of a previous study 20 . The ECs or PBMCs from each donor pig (GalT−/−;hCD39;hCD55 and GalT−/−;hCD39;hCD46;hCD55;TBM) were used to measure donor-specific anti-pig IgG and IgM antibodies in the plasma of each recipient monkey at multiple time points. The ECs (1 × 105 cells/test) or PBMCs (1 × 106 cells/test) were incubated with 50 μL of monkey plasma diluted 1:10 in PBS containing 1% human albumin with 30 mM edetic acid for 1 h at 4°C. The cells were washed with PBS and reacted with fluorescein isothiocyanate (FITC)-conjugated F(ab)2 fragments of rabbit immunoglobulin specific for human IgG and IgM antibodies, each diluted 1:100 for 30 min at 4°C (Dako, CA, USA). The concentration of donor-specific antibodies was determined as mean fluorescence intensities (MFIs) and each measurement was performed in duplicate. Corrected net MFIs (nMFIs) were determined by subtracting donor serum MFIs (negative control) from each sample MFIs. Serial postoperative nMFIs of IgG and IgM antibodies were compared with preoperative nMFIs.
Results
Renal Damage After Kidney Xenotransplantation
The NHPs survived for 4 weeks after receiving a kidney transplant (4 WAT) from a GalT−/−;hCD39;hCD55 pig and 6 weeks after receiving a kidney transplant (6 WAT) from a GalT−/−;hCD39;hCD46;hCD55;TBM pig. The only difference between the pigs is the modified genes, as they were all given equal doses of immunosuppressive, anti-inflammatory, and anticoagulative agents. To distinguish the injury mechanism and its severity, we analyzed both untransplanted and transplanted kidneys from both the GalT−/−;hCD39;hCD55 and GalT−/−;hCD39;hCD46;hCD55;TBM pigs. In histopathological analysis, the transplanted kidney showed decreased glomeruli and loss of intact tubular structure (Fig. 1A). These renal damages are mediated by apoptosis, proven by TUNEL assay and mRNA sequencing (Fig. 1B, C). Significant upregulation of apoptosis-related genes [BCL2 Binding Component 3 (BBC3)] in transplanted kidney was supported by the activation of apoptosis signal pathways (cleaved caspase 3, cleaved caspase 7, and cleaved caspase 9) in transplanted kidneys (Fig. 1D). When comparing the 4 WAT with 6 WAT kidneys, 6 WAT showed a higher number of apoptotic cells, additional upregulated apoptosis-related genes (5 vs 8 genes; Log2 fold value of over 2 with P < 0.05), and increased expression of apoptosis signal pathways than the 4 WAT. The C-reactive protein also showed a higher level in 6 WAT NHP (Fig. 1E). Based on the abovementioned findings, the longer surviving graft sustained greater immune injury despite the additional genetic modification.
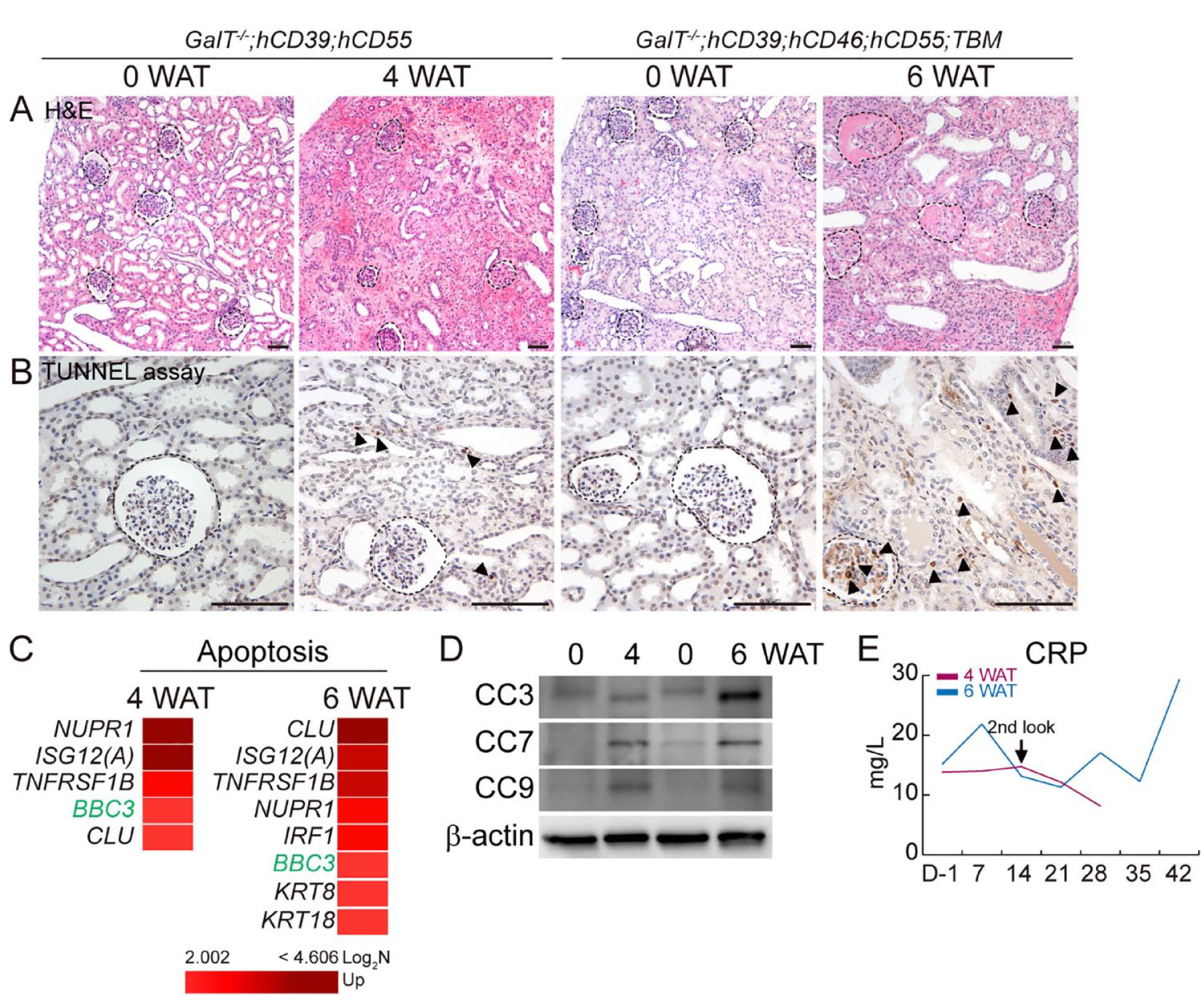
Renal damage after kidney xenotransplantation. Untransplanted [0 week after transplantation (WAT)] versus transplanted pig kidneys from GalT−/−;hCD39;hCD55 (4 WAT) and untransplanted (0 WAT) versus transplanted pig kidneys from GalT−/−;hCD39;hCD46;hCD55;TBM (6 WAT) were analyzed. Histopathological analysis of H&E staining (A) and TUNEL assay (B). Scale bar = 100 µm. (C) mRNA sequencing data of upregulated apoptosis-related genes (Log2 fold value of above 2; P < 0.05). (D) Immunoblot of apoptosis signal pathways [cleaved caspase 3 (CC3), CC7, and CC9]. (E) C-reactive protein level in serum. TBM: thrombomodulin; CRP: C-reactive protein; mRNA: messenger RNA; WAT: week after transplantation; TUNEL: terminal deoxynucleotidyl transferase biotin-dUTP nick end labeling; H&E: hematoxylin and eosin.
Monkey T Cell-Mediated Immune Response After Kidney Xenotransplantation
The NHPs were administered immunosuppressive agents, as mentioned in “Materials and Methods” section. The lymphocyte population was decreased after rituximab (day −7) and antithymocyte globulin (day−2) induction in monkey blood before transplant (Supplemental Fig. S2). However, we discovered the presence of monkey CD3 and CD4 cells in the renal interstitial (Fig. 2A, B). Among 10 toll-like receptor (TLR) subtypes, TLR3 and TLR5 were highly upregulated, and TLR9 was slightly upregulated in both 4 WAT and 6 WAT kidneys (Fig. 2C). As a downstream signal pathway of TLRs, the expression of MyD88 and Toll/IL-1 receptor domain-containing adaptor (TRIF) and the phosphorylation of NF-kB were increased (Fig. 2D). The level of anti-pig antibodies (IgG and IgM) did not differ before and after transplantation. Development of anti-donor antibodies was not observed after transplantation (Fig. 2E). These results indicate that the cellular- and antibody-mediated immune responses were maintained at a low level after transplantation. However, CD3+ or CD4+ lymphocyte infiltration and TLR-dependent signaling activation suggest the remaining immune response, even under the immunosuppressive agents.
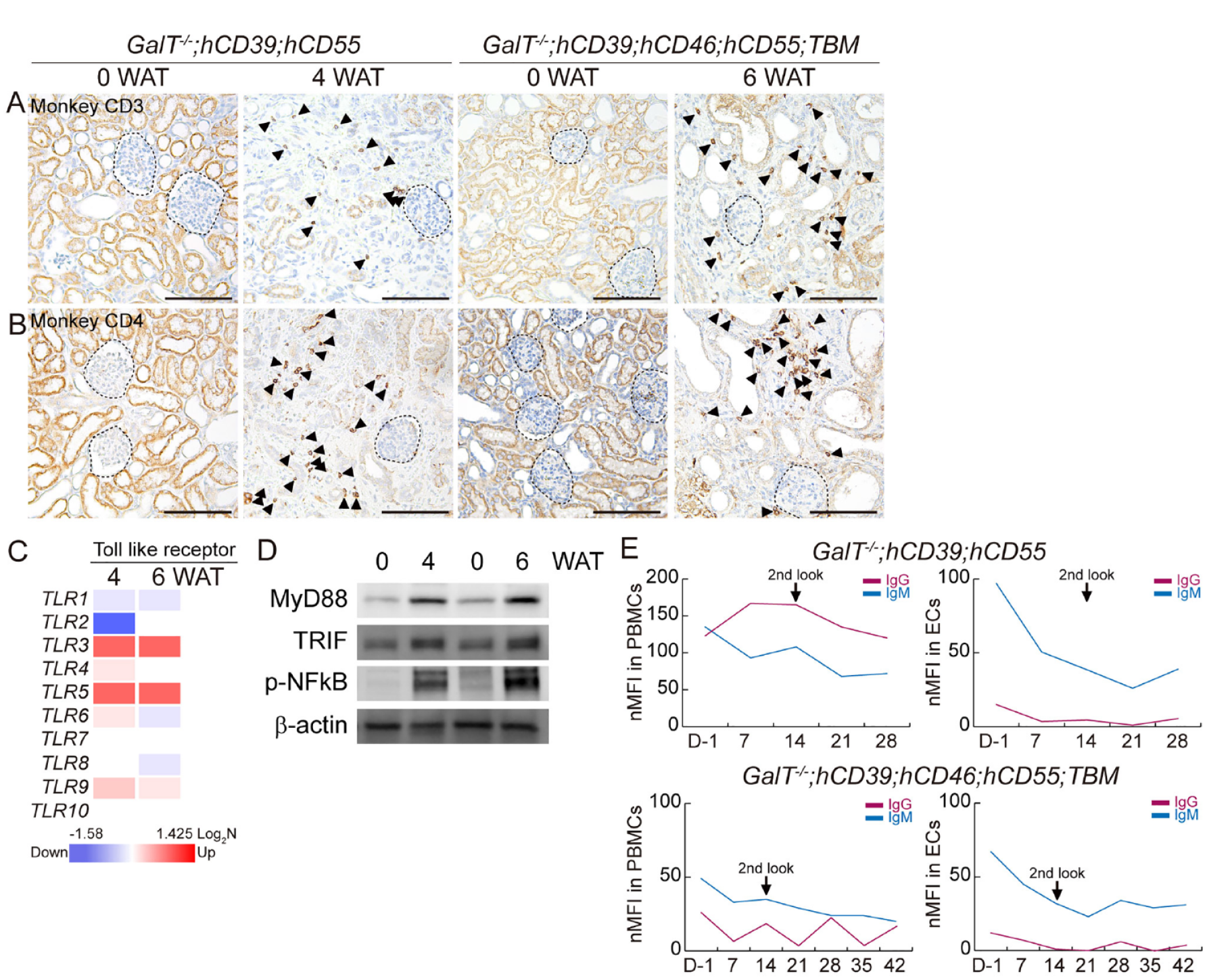
Monkey T cell-mediated immune response after kidney xenotransplantation. Untransplanted (0 WAT) versus transplanted pig kidneys from GalT−/−;hCD39;hCD55 (4 WAT) and untransplanted (0 WAT) versus transplanted pig kidneys from GalT−/−;hCD39;hCD46;hCD55;TBM (6 WAT) were analyzed. Immunohistochemistry of Monkey CD3 (A) and Monkey CD4 (B) Scale bar = 100 µm. (C) mRNA sequencing data of toll-like receptors (TLR1−TLR10). (Log2 fold value; P < 0.05) (D) Immunoblot of MyD88, TRIF, and phospho-NF-kB. (E) Anti-donor IgG and IgM antibodies pretransplantation and posttransplantation. Antibody binding to donor pig endothelial cells (ECs) or peripheral blood mononuclear cells (PBMCs) was measured by FACS. WAT: week after transplantation; TBM: thrombomodulin; mRNA: messenger RNA; TRIF: Toll/IL-1 receptor domain-containing adaptor.
Renal Functional Disorder After Kidney Xenotransplantation
When muscles or livers produce nitrogenous end products filtered through the glomeruli, high blood urea nitrogen (BUN) and creatinine levels can indicate kidney malfunction. Upon studying kidney transplants, we observed a steady increase of BUN and creatinine levels (Fig. 3A, B). The renal tubule cells are responsible for reabsorbing glucose filtered through the SGLT. In 4 WAT and 6 WAT kidneys, we noted a decrease in expression of SGLT1 and SGLT2, and a decrease in the number of tubular cells (Fig. 3C, D). These findings suggest that kidney function gradually declines until it is no longer viable.
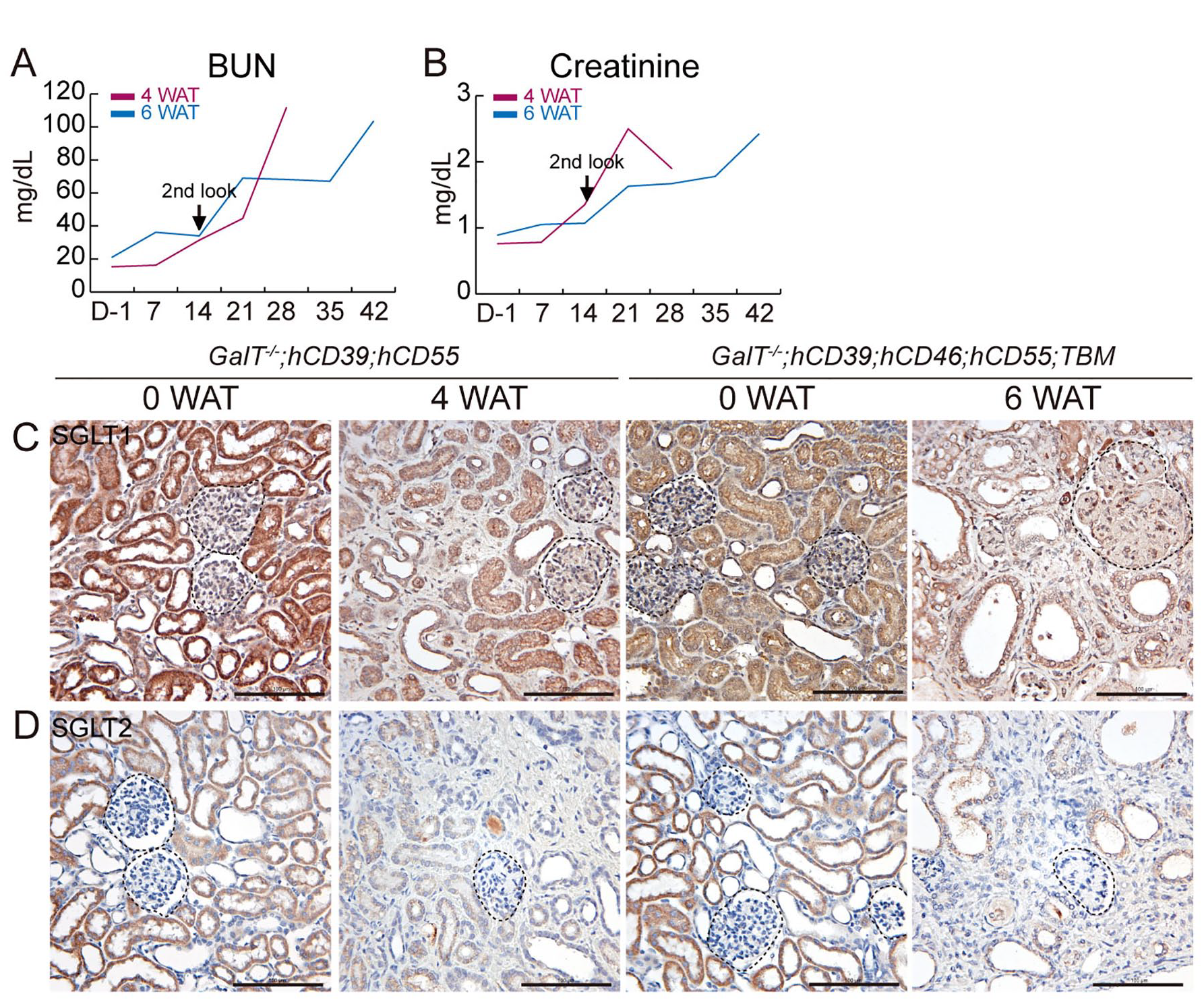
Renal functional disorder after kidney xenotransplantation. The serum chemistry of NHPs was analyzed. Serum chemistry of blood urea nitrogen (BUN) (A) and creatinine (B). Untransplanted (0 WAT) versus transplanted pig kidneys from GalT−/−;hCD39;hCD55 (4 WAT) and untransplanted (0 WAT) versus transplanted pig kidneys from GalT−/−;hCD39;hCD46;hCD55;TBM (6 WAT) were analyzed. Immunohistochemistry of sodium-glucose cotransporter 1 (SGLT1) (C) and SGLT2 (D). Scale bar = 100 µm. NHP: nonhuman primate; WAT: week after transplantation; TBM: thrombomodulin; SGLT: sodium-glucose linked cotransporter.
Renal Fibrosis After Kidney Xenotransplantation
We investigated whether renal fibrosis occurs after kidney transplantation. We noticed a significant increase in collagen accumulation in the 4 WAT and 6 WAT kidneys, as seen in the Masson’s trichrome and Sirius red staining (blue and red, respectively), indicating excessive fibrosis (Fig. 4A, B). The expression of vimentin, an epithelial-mesenchymal transition (EMT) marker, was also increased around the tubule cells (Fig. 4C). In addition, the number of alpha-smooth muscle actin and S100A4 positive cells was increased after transplantation (Fig. 4D, E), which indicates an increase in fibroblast activity. Furthermore, the extracellular matrix-related genes were upregulated in 4 WAT and 6 WAT kidneys (Fig. 4F). These findings suggest severe renal fibrosis after kidney transplantation, mediated by EMT, fibroblast activity, and extracellular component deposition.
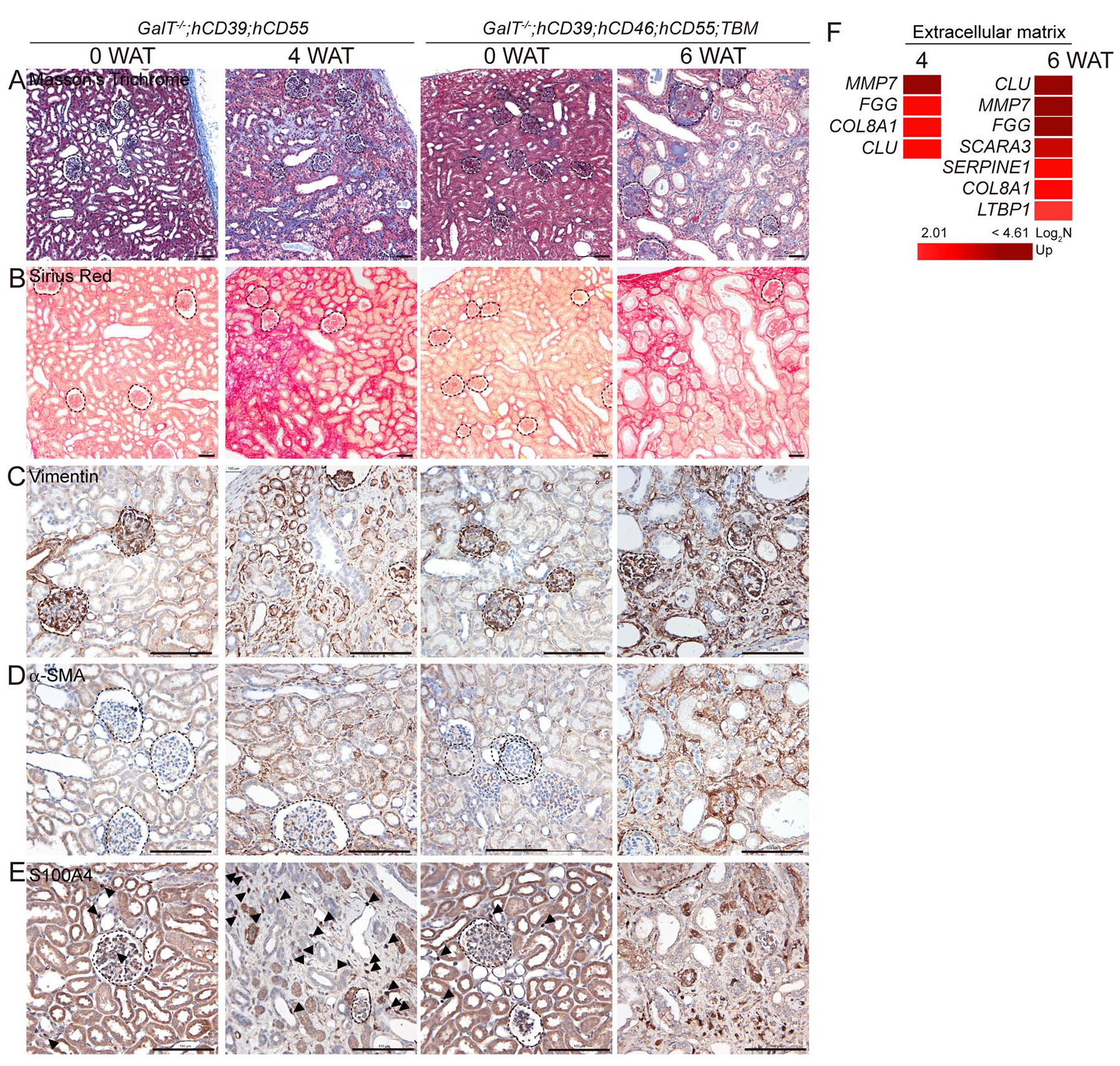
Renal fibrosis after kidney xenotransplantation. Untransplanted (0 WAT) versus transplanted pig kidneys from GalT−/−;hCD39;hCD55 (4 WAT) and untransplanted (0 WAT) versus transplanted pig kidneys from GalT−/−;hCD39;hCD46;hCD55;TBM (6 WAT) were analyzed. Histopathological analysis of Masson’s trichrome staining (A) and Sirius red (B). Immunohistochemistry of vimentin (C), alpha-smooth muscle actin (α-SMA) (D), S100A4 (E); scale bar = 100 µm. (F) mRNA sequencing data of upregulated extracellular matrix-related genes in transplanted kidney compared with normal kidney (Log2 fold value of above 2; P < 0.05). WAT: week after transplantation; TBM: thrombomodulin; mRNA: messenger RNA.
Discussion
In this study, we have compared the untransplanted and transplanted kidneys from genetically modified pigs: GalT−/−;hCD39;hCD55 and GalT−/−;hCD39;hCD46;hCD55;TBM. Both pigs removed the xenoantigen, α 1,3-galactosyltransferase, and inserted the CD39 and CD55 genes. The kidney sustained for 6 weeks had an added gene insertion for regulating complement, CD46, and coagulation, TBM. In our study, the NHPs survived for a shorter period of time (≤6 weeks) compared with the previous study21,22. According to a study by Kim and colleagues, they transplanted a GGTA knockout/hDAF pig kidney to Rhesus macaques after pretreating with αCD4 monoclonal antibody. The result showed that the median survival time was 238 days. Adams et al 21 found that pig kidneys with Gal and Sda double knockout could survive for more than a year after pretreatment with αCD4. Seventeen recipient monkeys were successfully maintained with pig kidneys from triple knockout (GGTA;CMAH;B4GALNT2), expressing multiple human proteins for an average of 238 days 23 . Based on these studies, GalT knockout may not be enough to fully inhibit the humoral immune response even if rituximab suppresses the development of anti-pig antibodies effectively (Fig. 2E).
The recipient monkey showed similar hematological features, such as platelet increase and lymphocyte decline, and similar serum chemistry, such as BUN and creatinine elevation. However, the 6 WAT kidney displayed more severe apoptosis, inflammation, loss of function, and renal fibrosis than the 4 WAT kidney. We have discovered that a combination of tubule cell apoptosis, EMT, interstitial fibrosis, and immune cell infiltration causes renal damage. Renal tubule cells have specific functions maintained by their polarity, including the apical (lumen-facing), lateral, and basal (attached to the basement membrane) surface. If the tubule is injured, epithelial cells may express mesenchymal markers, such as vimentin and alpha-smooth muscle actin, transforming into myofibroblast12,24. This is closely linked with renal fibrosis 25 . When a kidney is transplanted, there is a decrease in SGLT expression (Fig. 3) and an increase in the number of fibroblasts in the tubular interstitium, collagen deposition, and gene expression related to the extracellular matrix (Fig. 4). These changes suggest that the kidney may not be functioning correctly. Determining whether the immune response impacts renal fibrosis or vice versa is challenging. However, the changes in both the phenotype and genotype after xenograft are certainly part of a complicated process. Therefore, a multifactorial graft design needs to be taken into consideration. In this study, we have found that tubular apoptosis, immune cell infiltration, and renal fibrosis induce renal damage in xenografted kidneys from GalT−/−;hCD39;hCD55 and GalT−/−;hCD39;hCD46;hCD55;TBM pigs.
Although the number of animals tested in this study was limited, various analytical methods (histopathology, serum chemistry, mRNA sequencing, immunoblot, and immunohistochemistry) provided direct evidence of severe renal damage. This study would benefit researchers focusing on xenotransplantation. We have been collecting more cases of kidney xenotransplantation to determine the best combination of gene editing and agents for further study, which can provide a scientific conclusion. We found a steady level of anti-pig IgG and IgM after transplantation (Fig. 2E) and an increase in the platelet population (Supplemental Fig. S1), suggesting a distant correlation between humoral rejection and our results. However, to conclude this, direct detection of antibody and complement in kidney tissue is required. We also found a decrease in the lymphocyte population (Supplemental Fig. S2) during CD3+ or CD4+ cell infiltration in the kidney, which suggests the partial involvement of cell-mediated rejection. Moreover, we found the histopathological phenotype of thrombotic microangiopathy in transplanted kidneys (Fig. 4A). Therefore, it is difficult to isolate the principal rejection mechanism responsible for renal damage, and we cannot rule out all possibilities.
Conclusion
In conclusion, the short-term survival of NHPs and severe renal damage in both donors suggests that improved immune suppression is needed for more prolonged survival.
Supplemental Material
sj-jpg-1-cll-10.1177_09636897231217382 – Supplemental material for A Comparison Between GalT−/−;hCD39;hCD55 and GalT−/−;hCD39;hCD46;hCD55;TBM Pig Kidneys Transplanted in Nonhuman Primates
Supplemental material, sj-jpg-1-cll-10.1177_09636897231217382 for A Comparison Between GalT−/−;hCD39;hCD55 and GalT−/−;hCD39;hCD46;hCD55;TBM Pig Kidneys Transplanted in Nonhuman Primates by Han Na Suh, Ju Young Lee, Hee Jung Kang, Eun Mi Park, Ik Jin Yun, Wan Seop Kim, Kimyung Choi and Jeong Ho Hwang in Cell Transplantation
Supplemental Material
sj-jpg-2-cll-10.1177_09636897231217382 – Supplemental material for A Comparison Between GalT−/−;hCD39;hCD55 and GalT−/−;hCD39;hCD46;hCD55;TBM Pig Kidneys Transplanted in Nonhuman Primates
Supplemental material, sj-jpg-2-cll-10.1177_09636897231217382 for A Comparison Between GalT−/−;hCD39;hCD55 and GalT−/−;hCD39;hCD46;hCD55;TBM Pig Kidneys Transplanted in Nonhuman Primates by Han Na Suh, Ju Young Lee, Hee Jung Kang, Eun Mi Park, Ik Jin Yun, Wan Seop Kim, Kimyung Choi and Jeong Ho Hwang in Cell Transplantation
Supplemental Material
sj-jpg-3-cll-10.1177_09636897231217382 – Supplemental material for A Comparison Between GalT−/−;hCD39;hCD55 and GalT−/−;hCD39;hCD46;hCD55;TBM Pig Kidneys Transplanted in Nonhuman Primates
Supplemental material, sj-jpg-3-cll-10.1177_09636897231217382 for A Comparison Between GalT−/−;hCD39;hCD55 and GalT−/−;hCD39;hCD46;hCD55;TBM Pig Kidneys Transplanted in Nonhuman Primates by Han Na Suh, Ju Young Lee, Hee Jung Kang, Eun Mi Park, Ik Jin Yun, Wan Seop Kim, Kimyung Choi and Jeong Ho Hwang in Cell Transplantation
Footnotes
Author Contributions
H.N.S. conceived experiments, performed experiments, conducted data analysis, and wrote a manuscript. J.Y.L. analyzed the mRNA sequencing data. H.J.K., E.M.P., and W.S.K. analyzed the hematological and histological data. I.J.Y. conducted the surgical procedure (xenograft). K.C. generated the genetically engineered pig. J.H.H. supported the funding.
Availability of Data and Materials
Raw data for mRNA sequencing is available at Gene Expression Omnibus (GEO accession: GSE240012). Other data are included in the “Results” section.
Ethical Approval
The Institutional Animal Care and Use Committee (IACUC) at Korea Institute of Toxicology with approval no. 2012-0051 reviewed and approved the animal care and use protocol.
Statement of Human and Animal Rights
This article contain studies with animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Korea Institute of Toxicology under grant no. 1711195889 and Rural Development Administration under grant no. PJ015610022021, Republic of Korea.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
