Abstract
The enteric nervous system (ENS) is an extensive network of neurons and glia within the wall of the gastrointestinal (GI) tract that regulates many essential GI functions. Consequently, disorders of the ENS due to developmental defects, inflammation, infection, or age-associated neurodegeneration lead to serious neurointestinal diseases. Despite the prevalence and severity of these diseases, effective treatments are lacking as they fail to directly address the underlying pathology. Neuronal stem cell therapy represents a promising approach to treating diseases of the ENS by replacing the absent or injured neurons, and an autologous source of stem cells would be optimal by obviating the need for immunosuppression. We utilized the swine model to address key questions concerning cell isolation, delivery, engraftment, and fate in a large animal relevant to human therapy. We successfully isolated neural stem cells from a segment of small intestine resected from 1-month-old swine. Enteric neuronal stem cells (ENSCs) were expanded as neurospheres that grew optimally in low-oxygen (5%) culture conditions. Enteric neuronal stem cells were labeled by lentiviral green fluorescent protein (GFP) transduction, then transplanted into the same swine from which they had been harvested. Endoscopic ultrasound was then utilized to deliver the ENSCs (10,000–30,000 neurospheres per animal) into the rectal wall. At 10 and 28 days following injection, autologously derived ENSCs were found to have engrafted within rectal wall, with neuroglial differentiation and no evidence of ectopic spreading. These findings strongly support the feasibility of autologous cell isolation and delivery using a clinically useful and minimally invasive technique, bringing us closer to first-in-human ENSC therapy for neurointestinal diseases.
Introduction
Enteric neuropathies result from congenital or acquired abnormalities of the enteric nervous system (ENS) and include conditions, such as congenital aganglionosis (Hirschsprung’s disease; HSCR), esophageal achalasia, chronic intestinal pseudo-obstruction, and gastroparesis. Despite the prevalence and severity of these diseases, current medical and surgical are associated with suboptimal outcomes rather aim to relieve the symptoms of the disease instead of directly addressing the underlying pathophysiology. Cell therapy offers the potential to treat the underlying pathophysiology by replacing the missing or damaged ENS by transplanting neural progenitors to restore the ENS and improve gut function. A number of studies have demonstrated successful isolation, expansion, and transplantation of enteric neuronal stem cells (ENSCs) to animal models of enteric neuropathies1–4 with functional recovery5,6 and even improved survival in mice with HSCR7,8. These promising findings support the feasibility of cell-based therapy for enteric neuropathies. However, several issues have to be taken into consideration for its future clinical application.
Optimization of cell delivery methods is critically important. Favorable features include a minimally invasive approach and the ability to target accurately the appropriate layer of the gut wall. Endoscopic cell delivery under ultrasound guidance can accomplish this. Endoscopy is commonly used for the diagnosis and treatment of gastrointestinal (GI) diseases. We and others have reported successful and efficient delivery of cells to the gut wall using colonoscopy in mice9–11, but utilization of endoscopic ultrasound (EUS) to improve targeting within the gut wall has not been previously reported. Endoscopic ultrasound is commonly used to perform preoperative staging of GI tumors12,13 based on its ability to allow visualization of the layers of the intestinal wall, leading us to propose EUS for targeting ENSC delivery to the muscularis propria of rectum.
To test the feasibility of this approach, we used the porcine model because of the size equivalency to human infants as an initial proof-of-concept for eventual application of this technique for delivery of ENSCs to infants with HSCR, in which the rectosigmoid colon is missing enteric ganglia. Furthermore, the experimental model we established allows us to harvest a segment of small intestine from the same swine thus permits testing of the feasibility of autologous cell transplantation. Since immunological and ethical difficulties are challenging hurdles for the use of other cell sources, autologously derived ENSCs are advantageous for clinical application. In this study, we demonstrate the successful isolation, expansion, and labeling of autologous swine ENSCs, with successful transplantation via EUS-guided fine-needle injection to the rectal wall, where engraftment and neuronal differentiation without ectopic spreading were achieved.
Methods
Animals
All animal protocols were approved by the Institutional Animal Care and Use Committee at Massachusetts General Hospital (MGH; Protocol #2018N000027). Female Yorkshire swine (age, 4–6 weeks; body weight, ~15 kg) were purchased from Tufts Comparative Medicine Services (Tufts University, MA) and housed in the animal facility at MGH.
Tissue Preparation and Immunohistochemistry
Tissue preparation and immunohistochemistry were performed as previously described 14 . Cells, preparation of longitudinal muscle and myenteric plexus (LMMP), and full-thickness gut samples were fixed in 4% paraformaldehyde. For cryosections, full-thickness samples were embedded in 15% sucrose at 4°C overnight, and then in 15% sucrose plus 7.5% gelatin at 37°C for 1 h. The tissue was rapidly frozen at −50°C in nitrogen. Frozen sections were collected on glass slides at 12–14 µm thickness with a Leica CM3050 S cryostat (Leica, Buffalo Grove, IL). For immunohistochemical staining, samples were permeabilized with 0.1% Triton X-100 and blocked with 10% donkey serum for 30 min. Primary antibodies (Table 1) were diluted in 2% donkey serum, 0.01% Triton X-100, and incubated with tissues at 4°C overnight. For wholemount staining of swine LMMP, 10% donkey serum or bovine serum albumin (BSA) in phosphate-buffered saline Tween-20 (PBST; 0.1% Triton X-100 in phosphate-buffered saline [PBS]) was used for blocking, primary antibodies were incubated for 3 days on a shaker at 4°C, followed by addition of secondary antibodies for 2 days.
Primary Antibodies Used in This Study.
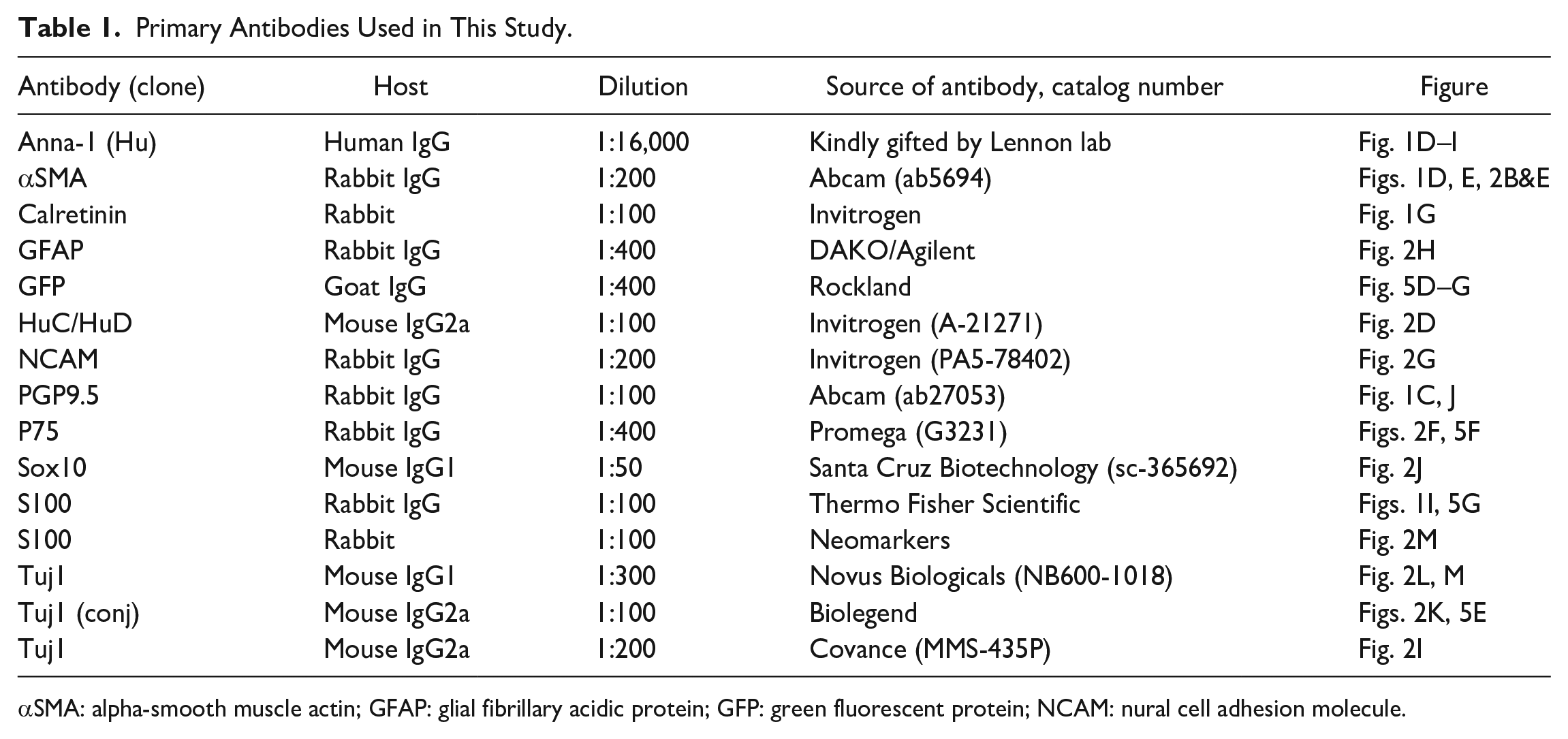
αSMA: alpha-smooth muscle actin; GFAP: glial fibrillary acidic protein; GFP: green fluorescent protein; NCAM: nural cell adhesion molecule.
Secondary antibodies included the following: donkey anti-rabbit IgG (1:500; Alexa Fluor 488 and 546; Thermo Fisher Scientific, Waltham, MA), donkey anti-goat IgG (1:500; Alexa Fluor 488; Thermo Fisher Scientific), goat anti-mouse IgG (1:500; Alexa Fluor 488; Thermo Fisher Scientific), and donkey anti-human IgG (1:200, Alexa Fluor 488 and 647; Thermo Fisher Scientific). Cell nuclei were identified with 4',6-diamidino-2-phenylindole (DAPI) solution (Vector Labs, Burlingame, CA) and sections were mounted with aqua-poly/mount (Fisher Scientific). Images were taken using a Nikon A1R laser scanning confocal microscope (Nikon Instruments, Melville, NY) or a KeyenceBZX-700All-In-OneMicroscopy (Keyence, IL).
Harvest Intestinal Tissue From Swine
Animals were fasted from the night before the operation. Anesthesia induction and maintenance was with isoflurane after endotracheal intubation. Carprofen (Rimadyl, 2 mg/kg), buprenorphine (Buprenex, 0.01 mg/kg), and cefazolin 40 mg/kg were administered intramuscularly (IM) prior to the operation. A laparotomy was created via a 5 cm vertical midline incision was made below the umbilicus. A segment (5–10 cm) of small bowel was resected following ligation of mesenteric vessels. The resected specimen was brought to the laboratory for cell harvesting. End-to-end bowel anastomosis was performed using two layers of interrupted 4-0 Vicryl sutures. The abdominal wall was closed in two layers with 0 polydioxanone suture (PDS) and the skin closed with staples. Local analgesia was given intradermally using 10 ml of 0.5% bupivacaine (Marcaine). Fentanyl patch (25 mcg/h) was applied and kept in place for 72 hours postoperatively. Animals were recovered from anesthesia at the animal facility under careful monitoring. Regular diet was commenced on post-operative Day 1.
Tissue Processing, Cell Isolation, and Culture
Intestinal tissues were kept in Hanks’ balanced salt solution (HBSS, Thermo Fisher Scientific) on ice during transfer. Tissue dissection and dissociation commenced within 1 h after the animal had been euthanized. Small intestine was used for cell isolation. Muscularis propria, including the longitudinal muscle, circular muscle, and myenteric plexus, was separated from the underlying submucosa using forceps. Tissues were enzymatically dissociated with 0.75 mg/ml Liberase Thermolysin High formulation (Roche, Indianapolis, IN) and DNAse 1 (1 mg/ml, Sigma Aldrich, St Louis, MO) at 37°C without mincing. Overnight (up to 18 h) digestion was typically required for full tissue dissociation. Pellets were obtained and washed with prewarmed complete medium, followed by resuspension in Dulbecco’s modified Eagle medium: nutrient mixture F-12 (DMEM/F12, Gibco, Life Technologies) supplemented with 1:100 glutamax (Gibco), 1:100 N2 (STEMCELL Technologies, Vancouver, BC), 1:50 B27 without vitamin A (Fisher Scientific), 20 ng/ml basic fibroblast growth factor (bFGF; STEMCELL Technologies), 20 ng/ml EGF (Fisher Scientific), 1:1,000 beta-mercaptoethanol (Sigma Aldrich), 1:1,000 heparin (STEMCELL Technologies), 1:500 primocin (Fisher Scientific), and 50 µg/ml metrodinazole (Sigma Aldrich). Cells were counted and plated at a density of 100,000 live cells/ml of complete media in T75 flasks and cultured to form enteric neurospheres for 10–14 days. A 1:1 media change was performed every 3 days.
Viral Production and Transduction
Lentivirus vector expressing fluc (firefly luciferase) and green fluorescent protein (GFP), separated by an internal ribosomal entry site, under the control of the cytomegalovirus (CMV) promotor 15 with a titer of 2.3 × 108 – 1.0 × 109 IU/ml was prepared by the MGH Viral Vector Core Facility (MGH Neuroscience Center, Charlestown, MA). Enteric neurospheres formed as above were gently triturated to single cell suspensions using Accutase (STEMCELL Technologies). Lentivirus was added at multiplicity of infection (MOI) in the range of 5–100. After 48 h, successful transduction was confirmed by GFP expression. To determine transduction efficiency, enteric neurospheres were generated from lentiviral transduced ENSCs, dissociated and plated on fibronectin-coated cover slips, and the presence of GFP fluorescence quantitatively measured using ImageJ software.
EUS-Guided Injection of Autologously Derived Swine ENSCs to the Rectum
Recipient swine were anesthetized as above and fecal material removed from the rectum digitally and with irrigation. A suspension of neurospheres was prepared (10,000–30,000 neurospheres in 0.5–1.0 ml saline containing 1:100 India ink). Endoscopy was performed to visualize the rectal lumen followed by (GF-UCT180, Olympus America, PA, USA) guided cell injection through a 22G endoscopic needle. For preliminary experiments, three different injection materials with different colors (Table 2) were used to optimize targeting of the injection site.
Injection Materials.

GFP: green fluorescent protein.
Results
Small intestinal resection was performed on 4- to 6-week-old female Yorkshire swine and the tissue processed for immunohistochemistry to characterize the ENS and identify antibodies that can be used in this species (Table 1). Staining with the neuronal marker, PGP9.5, shows nerve fibers projecting to the intestinal villi (Fig. 1C, arrows). Hu+ neuronal cell bodies (Fig. 1D, arrows) and P75+ glial cells (Fig. 1F) are present in the myenteric ganglia located between the SMA+ circular and longitudinal muscle layers (Fig. 1D). Submucosal enteric ganglia are also visualized with Hu (Fig. 1E). Wholemount preparations of the muscularis propria from 4- to 6-week-old swine small intestine were also stained immunohistochemically (Fig. 1G–J). Enteric ganglia with interconnecting fibers label with the neuronal markers Hu and calretinin (Fig. 1G). High-power image reveals Hu+ neurons surrounded by S100β+ glia in a myenteric ganglion (Fig. 1H, I). The interconnected myenteric neuroglial network is visualized with PGP9.5 staining of neuronal fibers, Hu+ neuronal cell bodies, and P75+ enteric glia (Fig. 1J)

Immunofluorescent characterization of swine small intestine ENS. (A) Segment of swine small intestine was harvested and processed for H&E (B) and immunohistochemistry, including PGP9.5 (C), Hu/SMA (D, E), and Hu/P75 (F). Wholemount immunostaining of the myenteric plexus with Hu and calretinin reveals the myenteric ganglia with interconnecting nerve fibers (G). High-power images of myenteric ganglia show enteric neurons (PGP9.5, Hu) and glia (S100β, P75) (I, J–J’’’). Scale bars: A, 1 cm; G, 1 mm; H, 500 µm; E, 200 µm; C, D, F, I, and J, 100 µm. CM: circular muscle; ENS: enteric nervous system; GFP: green fluorescent protein; LM: longitudinal muscle; SMA: smooth muscle actin.
To isolate swine ENSCs, the muscularis propria was mechanically separated from a segment of small intestine (Fig. 2A,
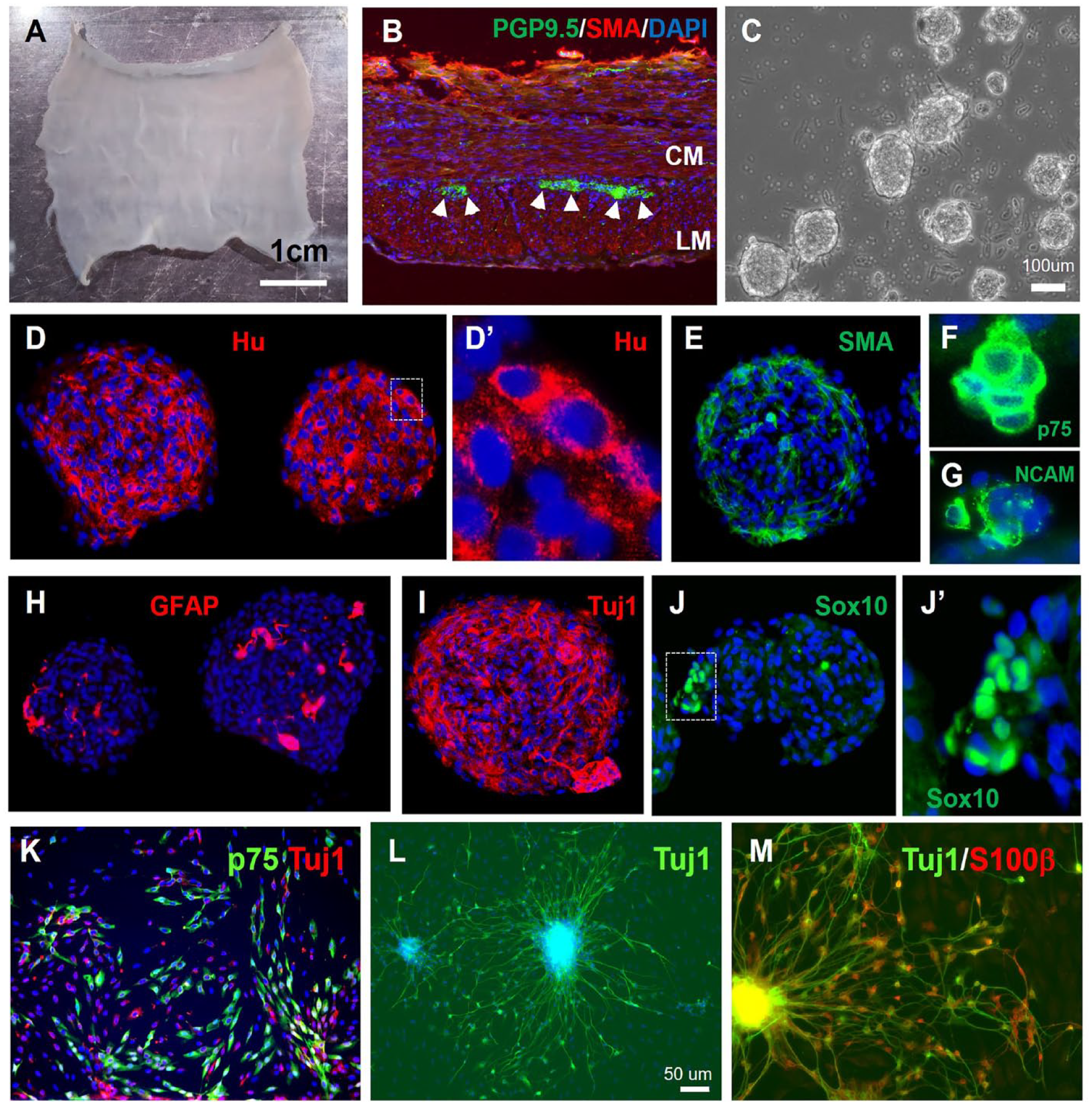
Isolation, characterization, and differentiation of swine ENSCs. (A) Small intestinal muscularis propria was isolated by microdissection. (B) Cross-sections show PGP9.5+ myenteric neurons between the SMA-expressing circular muscle (CM) and longitudinal muscle (LM). Following tissue digestion and culture for 10 days, enteric neurospheres are formed (C). Immunohistochemical characterization confirms presence of neurons expressing Hu (D, D’), smooth muscle (E); and glial cells expressing P75 (F), NCAM (G), GFAP (H), Tuj1 (I), and Sox10 (J, J’). After neurosphere dissociation, cells were cultured on a fibronectin-coated surface, where Tuj1+ neurons (K, L) and P75+/S100B+ (K, M) glia are seen. Scale bars: A, 1 cm; C, 100 µm; L, 50 µm. ENSC: enteric neuronal stem cell; GFP: green fluorescent protein; SMA: smooth muscle actin.
To develop ENSCs as a potential regenerative cell therapy, we first aimed to identify potential growth conditions to maximize cell numbers and the proportion of neuronal progenitors. Compounds tested included a 5HT4 agonist (RS67506), N2 supplement (commonly used for neural stem cell expansion), and retinoic acid, but these did not lead to an improvement in the number of neurospheres generated (Fig. 3A). In contrast, culturing neurospheres in 5% oxygen dramatically enhanced the number of neurospheres (Fig. 3A, 313.3 ± 50.5 in Control vs 1492.5 ± 242.3 in 5% O2, ** p < 0.01). No change was observed in their size (Fig. 3B). qPCR was performed on RNA extracted from the neurospheres to determine whether low-oxygen conditions altered gene expression as an indicator of whether growth in these conditions expanded particular cell types. Gene expression in the neurospheres was compared to that in primary ENSCs just after harvesting and prior to culture. Results show a two-fold increase in the expression of Nestin and a significantly lower expression of SMA in neurospheres (Fig. 3C), suggesting that growth in hypoxia conditions supports expansion of the neural stem cell population while also potentially inhibiting the growth of myofibroblasts.
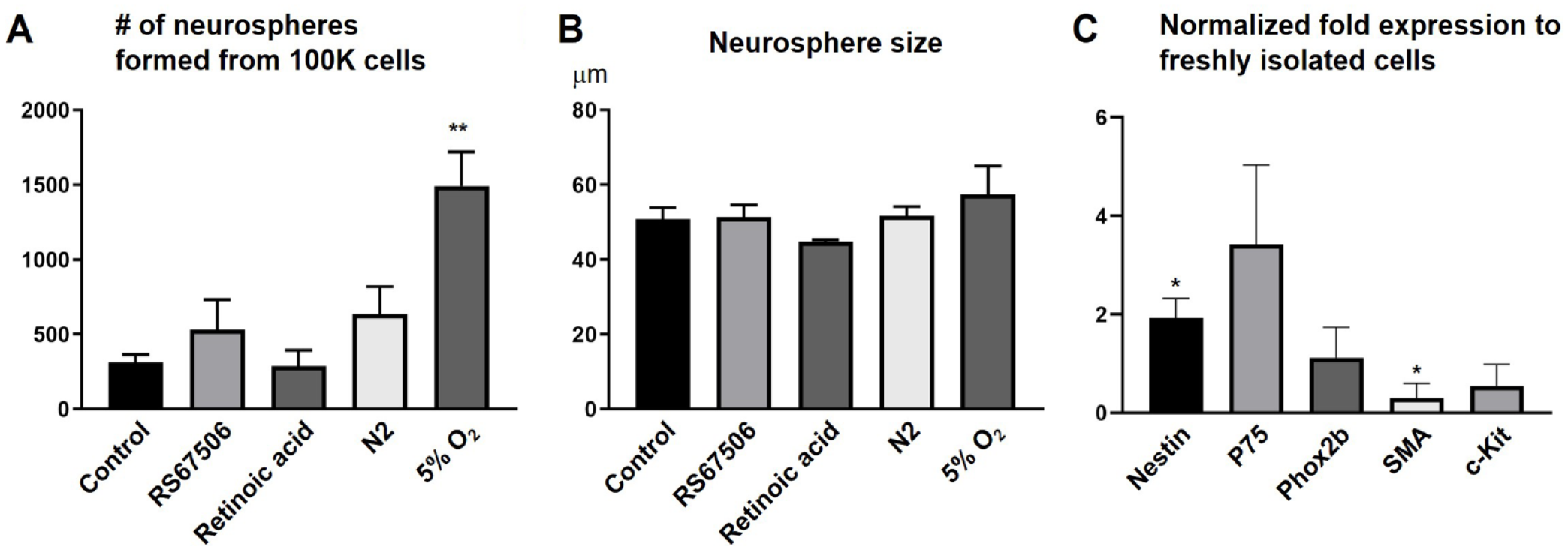
Optimizing culture conditions for swine ENSCs. (A) ENSCs cultured in 5% O2 conditions formed significantly more enteric neurospheres than those grown in control conditions (20% O2) or in the presence of various other putative growth factors. No change was observed in neurosphere size (B). Neurospheres grown in 5% O2 exhibit significantly increased levels of Nestin transcript and decreased expression of SMA, relative to transcript levels in freshly isolated cells (C, *
We developed a protocol for isolation and transplantation of autologous ENSCs to the swine rectum, as shown in Fig. 4A (
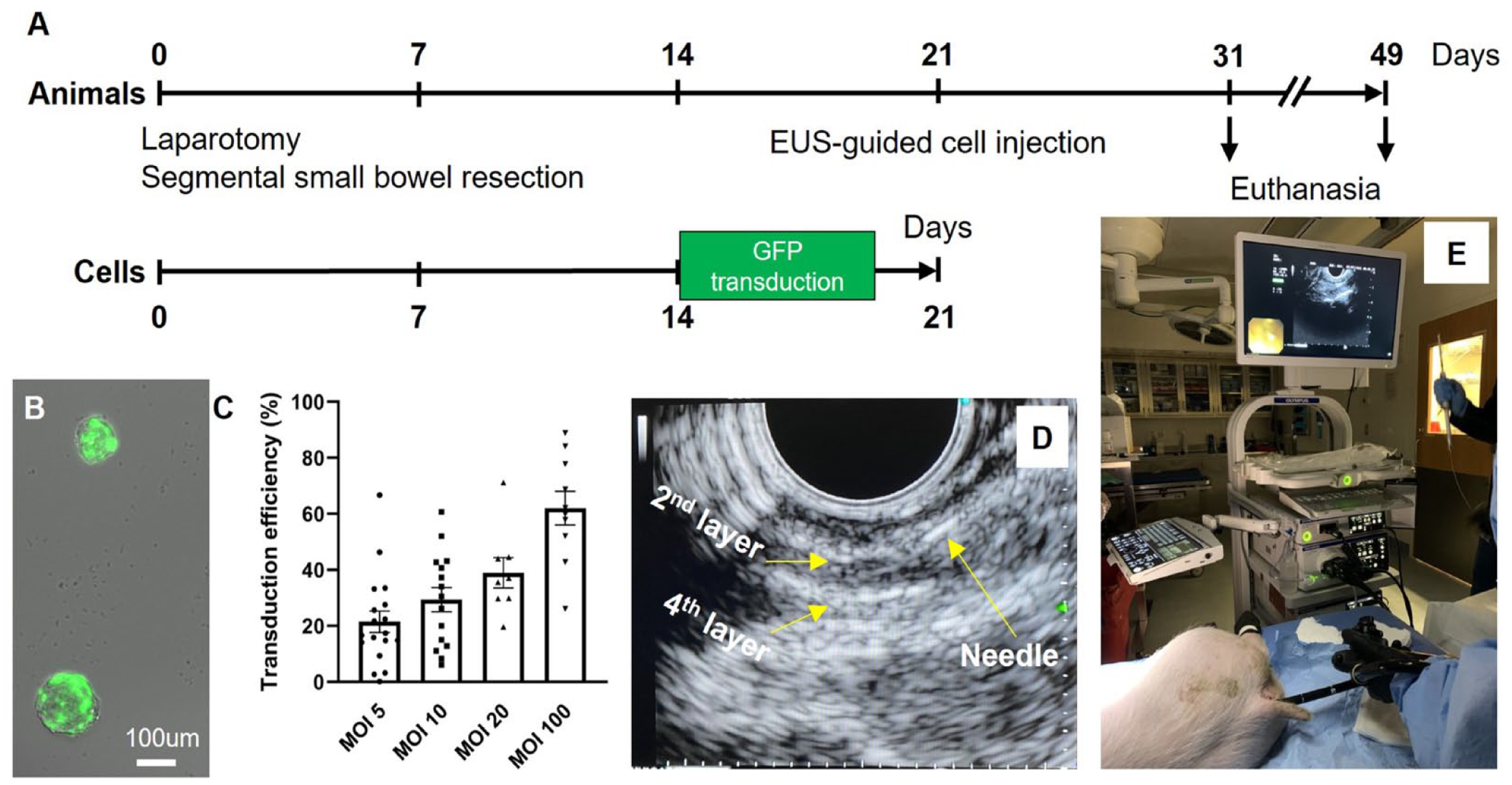
Isolation, expansion, and labeling of autologous ENSCs followed by
Lenti-GFP transduction was performed on dissociated cells after 14 days of neurosphere culture, and the cells allowed to re-form neurospheres over an additional 7 days (Fig. 4A). On Day 21, swine were transplanted with their own autologously derived GFP+ ENSCs using EUS to target the cells to the appropriate layer of the rectal wall. Animals were anesthetized and placed in the lateral position. Fecal material was removed manually and by irrigation from the anorectum. Endoscopic ultrasound (GF-UCT180, Olympus America, PA, USA) was used to visualize the layers of the rectal wall (Fig. 4D). A single injection of 0.5–1.0 ml cell suspension (containing 10–30,000 neurospheres) was performed using a 22G endoscopic needle advanced through the scope (Fig. 4E). Animals also were injected with 1.0 × 106 red fluorescent protein (RFP) beads, India ink, as well as yellow and green dye to determine the accuracy of targeting specific layers and to facilitate later identification of the cell injection site. At 10 days (

Identification of transplanted ENSCs and their differentiation within the rectum. (A) Rectum was explanted immediately following EUS-guided colonoscopic injection of multiple colored dyes into the rectal wall: green dye (green arrows), yellow dye (yellow arrows), and black ink (white arrows and circle). High-power imaging identified the injected RFP beads (B). Sections of recipient gut reveal successful targeting of the submucosa and muscularis layers with dye (C), and identification of GFP+ cells (D, green arrows) and RFP beads (D, white arrows) within the submucosal layer. Immunohistochemistry shows transplanted GFP+ ENSCs give rise to neurons (Tuj1; E, G, and H, arrows; arrowheads indicate endogenous plexus) and glial cells (F, S100β). High-power images of white dotted boxes in E are shown in (G and H). ENSC: enteric neuronal stem cell; EUS: endoscopic ultrasound; RFP: red fluorescent protein.
Sections of transplanted rectum were immunostained with neuronal and glial markers to assess ENSC differentiation following transplantation. A subpopulation of GFP+ ENSCs were immunoreactive for Tuj1 (Fig. 5E, G, and H, arrows), suggesting neuronal differentiation. Immunostaining for S100β confirmed that GFP+ transplanted ENSCs cells underwent glial differentiation (Fig. 5F). To determine whether any ectopic distribution of ENSCs to remote sites occurred, several recipient animals (
Discussion
In this study, we utilized a large animal model to test the feasibility of isolating, expanding, and delivering autologously derived ENSCs for the treatment of enteric neuropathies. Importantly, we found that culturing swine ENSCs in reduced oxygen conditions led to a marked increase in the number and purity of ENSCs. Following successful labeling of ENSCs using lentiviral GFP vector, we were able to deliver autologous ENSCs to the rectal wall using EUS. This allowed accurate targeting of the gut layer with successful engraftment and differentiation of autologous ENSCs. No remote ectopic cell spread was observed at 4 weeks following injection. These observations are highly supportive of the potential for utilizing autologous cells for the treatment of neurointestinal diseases.
The porcine gut has been increasingly proposed as a useful model for human translational studies because of its many similarities to human intestine17,18. The swine ENS has been characterized extensively and shares commonalities with the human ENS, including architecture and chemical coding19–22, physiological properties23,24 and transcriptomic signatures 25 . There are known anatomical differences between pig and human colon, such as the spiral orientation and absence of an appendix in the former 17 . However, the size and anatomy of the anorectal region in neonatal pigs is almost identical to that of human infants, and their small intestines are long enough to tolerate segmental small bowel resection 26 for harvesting autologous ENSCs. We therefore chose the swine as the best translational model for us to test the feasibility of autologous ENSCs isolation and transplantation.
Cell therapy offers the potential to replace the ENS in patients with neurointestinal disorders. We and others have demonstrated successful isolation and expansion of neural progenitors from embryonic and postnatal rodents27–30, and from patients with neurointestinal disorders5,31,32. These cells have been shown to possess the ability to engraft, migrate, and differentiate into functioning neurons, with restoration of gut function
To date, three potential autologous sources of neural progenitors have been described in the field of neurogastroenterology. Rollo et al
37
isolated ENSCs from human colonic tissues surgically resected from patients with HSCR. Following their expansion in culture in the presence of GSK3 inhibitor, those human autologous ENSCs were transplanted into explants of aganglionic colonic muscle from the same patient from whom the ENSCs had been harvested
37
and showed successful engraftment and neuroglial differentiation
Endoscopic ultrasound-guided injection offers a safe, efficient, and minimally invasive approach to deliver cells to the wall of the GI tract. Several studies including ours have shown that endoscopy allows effective delivery of cancer cells
10
or neural progenitors
9
to the wall of murine colon. It is well known in developmental biology that environmental cues provided by the gut mesenchyme are important for progenitor cells to differentiate into appropriate phenotypes and acquire normal function39–41. Therefore, placing ENSCs in the right layer can be important to maximize the effect of cell transplantation to treat neurointestinal diseases. Endoscopic ultrasound has evolved from a purely diagnostic imaging modality to an interventional procedure that provides a minimally invasive alternative to interventional radiologic and surgical techniques
42
. Endoscopic ultrasound-guided fine-needle aspiration has been established as a tissue acquisition method43,44, for drainage, ablation, or even necrosectomy in pancreatic and hepatobiliary disease45–48, and for vascular access49,50, celiac plexus block51,52, or injection of antitumoral agents53,54. In the current study, EUS successfully visualized the layers of the swine rectum and allowed effective cell delivery. Although histological examination of recipient colon revealed their successful engraftment and neuroglial differentiation of, it is unknown how important the appropriate location is and even which layer is optimal. In fact, previous reports have demonstrated that neural progenitors injected to intraperitoneal cavity can home to the gut wall, engraft, and differentiate into neurons within the wall of the gut
This work has several limitations. As a proof-of-concept study demonstrating the feasibility of harvesting and transplanting autologous ENSCs into the rectal wall in a large animal model, we did not assess the function of the cells following transplantation or their contributions to colonic motility. To date, only a few reports have described a porcine model of colonic aganglionosis created by focal ablation of the ENS using topical application of benzalkonium chloride to the serosa of rectosigmoid colon38,59. Although none of the studies include functional characterization of aganglionic porcine colon, this model can be useful to determine if the aganglionic gut environment is permissive for transplanted autologous ENSCs in a large animal setting. We recently observed that the area engrafted by ENSCs correlates with the degree of functional improvement in colonic motility in a mouse model of enteric neuropathy. Endoscopic cell delivery would allow multiple injections of cells to maximize the cell coverage over the recipient colon. Future studies will be warranted to determine how efficiently multiple cell injections can optimize cell coverage in the colorectum of large animals and whether this correlates with colonic motor function.
Footnotes
Acknowledgements
The authors thank Dr Vanda Lennon (Mayo Clinic) for providing human anti-Anna-1 (Hu) antibody. The lentiviral GFP virus was prepared at the MGH Vector Core Facility, Massachusetts General Hospital Neuroscience Center. The authors wish to acknowledge the support and expertise of the Knight Surgical Research Core staff in support of this research.
Authors’ Contributions
Conceptualization, R.H., R.M., L.R.B., A.J.B., and A.M.G.; methodology, R.H., W.P., S.B., T.O., and K.K.; formal analysis, S.B., W.P., R.S. N.N., and T.O.; investigation, R.H., S.B., W.P., and R.S.; resources, R.M. A.J.B., and A.M.G.; data curation, R.H., W.P., R.S., S.B., N.N., and O.T.; writing original draft preparation, R.H. and S.B.; writing review and editing, G.C., L.R.B., R.M., A.J.B., and A.M.G.; visualization, R.H., S.B., W.P., R.S., and T.O.; supervision, R.M., L.R.B, A.J.B., and A.M.G.; funding acquisition, A.M.G. All authors have read and agreed to the published version of the manuscript.
Availability of Data and Materials
All data are available upon request to the corresponding author.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Research Ethics
This study was reviewed and approved by the Institutional Animal Care and Use Committee at Massachusetts General Hospital (Protocol #2018N000027, “Enteric neuronal cell therapy for neurointestinal disease,” approved on 4/27/2021).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: G.C., L.R.B., R.M., and A.J.B. are employees of Takeda Development Center Americas and hold stock and/or stock options in Takeda. R.H., W.P., S.B., N.N., R.S., T.O., K.K., and A.M.G. do not possess any competing interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Takeda Development Center Americas.
